Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista colombiana de Gastroenterología
Print version ISSN 0120-9957
Rev Col Gastroenterol vol.32 no.1 Bogotá Jan./Mar. 2017
https://doi.org/https://doi.org.10.22516/25007440.130
Literature Review and Case Report of Post-Inflammatory Polyps and Inflammatory Bowel Disease
Hernando Marulanda MD (1), Â William Otero MD (2), Martín Gómez MD.(3)
(1) Internist and Gastroenterology Fellow at the National University of Colombia and the National University Hospital of Colombia in Bogotá, Colombia
(2) Professor in the Gastroenterology Unit of the National University and the National University Hospital of Colombia and Gastroenterologist at Clínica Fundadores in Bogotá Colombia. Mail: waoteror@gmail.com
(3) Professor in the Gastroenterology Unit of the National University and the National University Hospital of Colombia and Gastroenterologist at El Tunal Hospital and Kennedy Hospital in Bogotá, Colombia
Received:Â Â Â 11-07-16Â Accepted:Â Â Â 16-12-16
Abstract
Post-inflammatory polyps or âpseudopolypsâ may occur in patients who have long-term inflammatory bowel disease (IBD). They occur at sites where severe inflammation had previously occurred. We describe the case of a patient who had suffered from ulcerative colitis for five years when generalized post-inflammatory polyposis was discovered during a follow-upl colonoscopy. We review the meaning of this condition as well as its classification and treatment.
Key words: Pseudopolyps, post inflammatory polyps, ulcerative colitis, inflammation.
INTRODUCTION
Ulcerative colitis (UC) is an autoinflammatory disease that, together with Crohn's disease (CD), accounts for the vast majority of patients with inflammatory bowel disease (IBD). (1) The current objectives of treatment are remission of clinical symptoms and mucosal healing visible to endoscopy. (2) With these new treatment target, it is more common to find endoscopically visible residual structural alterations resulting from healing and repair of previously affected mucosa. (3) One of these is pseudopolyposis. (4) Pseudopolyps are polypoid neoplasias formed from scar tissue at sites of previous inflammatory activity. They reflect the severity the inflammation that had affected the site. (4) The name âpseudopolyposisâ is today considered obsolete, since both histologically and macroscopically these are true polyps. (5) The current recommendation is that they be called postinflammatory polyps which may be either inflammatory polyps or filiform polyps. (6, 7) They are twice as frequent in UC as in CD, (8) and they generally do not produce symptoms and are almost always incidental findings in routine and follow-up radiological and endoscopic studies. (9) Nevertheless, they may produce intestinal bleeding or obstruction when they reach large sizes. (10) In these latter cases, the treatment is surgical. Morphologically they can mimic a neoplasm.
This article presents a patient who had extensive ulcerative colitis for five years but who is currently in clinical and endoscopic remission. Generalized postinflammatory polyposis was found in a colonoscopy study of recurrent diarrhea control.
Clinical Case
The patient is a 42-year-old man who had had extensive ulcerative colitis for five years but is currently in clinical and endoscopic remission with a Truelove and Witts severity index of 6 points. (11) When the initial diagnosis was made, the patient weighed 45 kg and was 1.90 m tall. He currently weighs 100 kg and has been under treatment with 3 g/day of mesalazine (MSLZ) in granules and 1 mg/kg/day of azathioprine (AZA) for the past year. The usual recommended doses of AZA from 2.5 to 3 mg/kg are not being administered because MSLZ can increase AZA levels resulting in higher risks of toxicity. (12) Due to the severity of the patient's clinical picture, biological therapy with anti-tumor necrosis factor (anti-TNF) was proposed in first year following diagnosis,. However, the treatment using MSLZ was optimized by adding one enema/day of MSLZ and 40 mg/day of prednisone. Prednisone does were progressively diminished after the first month until it was suspended. He has had several recurrences including two documented clostridium difficile infections which were treated with oral metronidazole, and on the second occasion with oral 125 mg of vancomycin four times a day for 10 days.
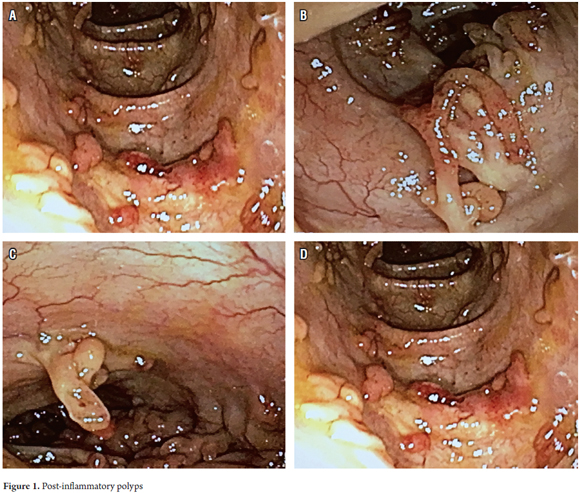
The patient is currently in clinical remission except for recurrent diarrhea secondary to irritable bowel syndrome. Fecal calprotectin is normal. The hematic picture and liver profile are normal. Follow-up total colonoscopy and ileoscopy showed only postinflammatory polyps. The evaluation of the colonic mucosa showed May scores of 0 or 1 in the various segments and disbursed postinflammatory polyps which, more frequent in the sigmoid and transverse regions as shown in Figure 1 (A-D). (13) Multiple biopsies of the colonic mucosa and postinflammatory polyps were taken. The pathologist reported focal hyperplasia of crypts without distortion of their structure or inflammatory infiltrates. We concluded that the alterations are residual reactive epithelial changes associated with chronic quiescent colitis which correlates with the residual underlying inflammatory process. The findings are characteristic of scarring of the gastrointestinal mucosa. There were no documented alterations suggesting compromise by premalignant or neoplastic lesions which correlates with the majority of the reports in the world literature.
Endoscopic follow-up was the standard for care of inflammatory bowel disease.
DISCUSSION
The term Filiform polyposis (FP) was first coined in 1974 by Appelman et al. to describe a radiological clinical syndrome that most frequently affected patients with IBD. The associated imaging findings mainly affected the sigmoid colon with numerous filiform defects similar to worms and a pattern of normal haustra. (14, 15)
Other terms such as giant pseudopolyposis and giant inflammatory polyposis and have been used in the literature to describe this type of polyposis. (16)
Postinflammatory polyps are polypoid neoplasms that develop during mucosal healing processes after severe long-term inflammation. (17)
Although the pathogenesis of these structures is not known with accuracy, (18) it is thought that they may represent foci of cell proliferation in response to a previous episode of severe inflammation. (19) Mechanical traction and peristalsis progressively produce elongation and coalescence of several proliferation foci could determine polyps' final sizes. (20)
Postinflammatory polyps occur more frequently UC than in CD. It is presumed that this is due to greater mucosal inflammation in UC in which prevalence ranges from 12.5% ââto 75%. (22) It is more often found in colectomy pieces than in endoscopic examinations. (21, 22)
The presence of postinflammatory polyps increases with the extent and severity of the disease. Although they are most frequently described within the limits of the mucosa of the colon, they have been described in the esophagus, stomach and small intestine in patients with Crohn's disease. (23)
Although UC and CD are the inflammatory diseases most frequently associated with postinflammatory polyps, they have also been described in infectious, ischemic, and necrotizing colitis. (24)
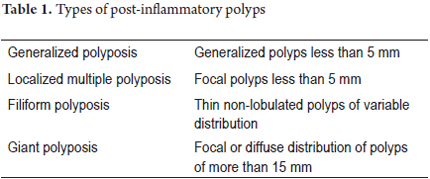
Based on macroscopic features and extent of lesions, these postinflammatory polyps are classified as shown in Table 1. (25)
Giant lesions have been subclassified in the following categories (26):
- Localized multiple pseudopolyposis
- Localized giant pseudopolyposis
- Generalized pseudopolyposis
- Finger-like pseudopolyposis.
Clinical presentation may not vary from the specific symptoms of IBD such as diarrhea, bleeding, anemia, weight loss and abdominal pain. (27) However, in some cases, there is a sensation of the presence of an abdominal mass sensation or the appearance of complications such as intussusception or mechanical intestinal obstruction due to the presence and size of the polyps. (28)
Long-term treatment with MSLZ, steroids and AZA has been reported to decrease the size of these lesions, even if they are gigantic. (29)
Recent reports highlight the value of anti-TNF for controlling outbreaks of extra-colonic proliferation, mainly in the esophagus, for those patients whose initial therapy has failed and who have persistent symptoms. (23-30)
Polyps are lined with normal edematous or superficially ulcerated colonic mucosa. The stalk is often more fibrous and vascular but may also contain smooth muscle fascicles. Segmental cases and cases associated with stenosis can be confused with neoplasia. (31)
The vast majority of microscopic examinations of biopsies show marked fibrosis with relatively well conserved histological architecture. (32) It is common to find crypt hyperplasia and nonspecific focal inflammation, and to find inflammatory infiltrate on the lamina propria mucosae associated with lymphoid hyperplasia. These findings are generally considered to be benign. (33) The most severe cases are related to erosions, transmural extension and lymphatic hyperplasia which are related to intense inflammatory involvement. (34) Non-polypoid segments are of great importance since this is where the precursors of neoplastic lesions have frequently been documented. (35)
Immunohistochemical analysis using anti-Ki67 and anti-p53 antibodies in tissue fragments can be a complementary tool for establishing abnormal patterns of cell proliferation and thereby identifying the substrate to tumor pathology. (36)
The importance of postinflammatory polyps is that they may be correlated with increased risk of colon cancer associated with IBD or may be premalignant lesions. (37) Rutter et al. found that the odds ratio (OR) of these lesions for ACEI is 2.29 (95% CI: 1.28 to 4.11), and Velayos et al. found a similar risk with an OR of 2.29 (95% CI: 1.28 to 4.11). (38, 39) In addition, findings of giant postinflammatory polyps with dysplasia around them in patients with long-standing UC may be confused with mass-associated dysplasia. (38, 39)
In addition, the relationship between polyp size and susceptibility to histological alterations has been established in case series, and large polyps are often found to be adenomas. (40, 41)
In view of all of this, it seems that more frequent follow-ups are necessary. Although there is no indication for prophylactic colectomy to manage these lesions, surgery must be performed in order to obtain a complete histopathological study when endoscopic, radiological or clinical findings point to neoplasia. (42)
Surgical management is indicated when the lesions are giant and produce mechanical complications and when there is dysplasia in the areas adjacent to the lesions. (43)
There is currently no agreement about how to monitor these lesions. At the moment, the standard of care for patients with UC is to monitor them with great care by colonoscopy and biopsies taken from both the polyps and the mucosa adjacent to them plus random polypectomy of some of the lesions. (44, 45)
Some authors recommend intensifying the endoscopic follow-up of patients with postinflammatory polyps especially when there are large focal lesions (greater than 15 mm). The guideline calls for at least one annual endoscopic control. (46, 47)
Conclusion
Postinflammatory polyps are frequently found in patients with inflammatory bowel disease (IBD) who are in clinical and endoscopic remission.
They are considered a consequence of the severe underlying inflammatory activity. At present, they are not considered to be premalignant lesions, but recent reports call for closer endoscopic follow-up including a histopathological study of the lesion and adjacent healthy mucosal areas.
REFERENCES
1. Chen JH, Andrews JM, Kariyawasam V, et al. Review article: acute severe ulcerative colitis - evidence-based consensus statements. Aliment Pharmacol Ther. 2016;44 (2):127-44. [ Links ]
2. Peyrin-Biroulet L, Sandborn W, Sands BE, et al. Selecting therapeutic targets in inflammatory bowel disease (STRIDE): determining therapeutic goals for treat-to-target. Am J Gastroenterol. 2015;110:1324-38. [ Links ]
3. Christensen KR, Steenholdt C, Buhl SS, et al. Systematic information to health-care professionals about vaccination guidelines improves adherence in patients with inflammatory bowel disease in anti-TNFα therapy. Am J Gastroenterol. 2015;110(11):1526-32. [ Links ]
4. Sheikholeslami MR, Schaefer RF, Mukunyadzi P. Diffuse giant inflammatory polyposis: a challenging clinicopathologic diagnosis. Arch Pathol Lab Med. 2004;128(11):1286-8. [ Links ]
5. Goldglaber MB. Pseudopolyposis in ulcerative colitis. Dis Colon Rectum. 1965:8;355-63. [ Links ]
6. Suzuki Y, Nakao K, Hishikawa E, et al. Filiform polyposis: inflammatory polyposis in ulcerative colitis, report of rare case. Stomach & Intestine. 2001;36:585-9. [ Links ]
7. Renison DM, Forouhar FA, Levine JB, et al. Filiform polyposis of the colon presenting as massive hemorrhage: an uncommon complication of Crohns disease. Am J Gastroenterol. 1983;78:413-6. [ Links ]
8. Balazs M. Giant inflammatory polyps associated with idiopathic inflammatory bowel disease. An ultrastructural study of five cases. Dis Colon Rectum. 1990;33:773-7. [ Links ]
9. Teague RH, Read AE. Polyposis in ulcerative colitis. Gut. 1975;16:792-5. [ Links ]
10. Schneider R, Dickersin GR, Patterson JF. Localized giant pseudopolyposis. A complication of granulomatous colitis. Am J Dig Dis. 1973;18:265-70. [ Links ]
11. Goenka MK, Nag S, Kumar A, et al. Diagnosis of acute severe colitis. Trop Gastroenterol. 2014;35(Suppl 1):S1-8. [ Links ]
12. Gao X, Zhang FB, Ding L, et al. The potential influence of 5-aminosalicylic acid on the induction of myelotoxicity during thiopurine therapy in inflammatory bowel disease patients. Eur J Gastroenterol Hepatol. 2012;24(8):958-64. [ Links ]
13. Schroeder KW, Tremaine WJ, Ilstrup DM. Coated oral 5-aminosalicylic acid therapy for mildly to moderately active ulcerative colitis: a randomized study. N Engl J Med. 1987;317:1625-9. [ Links ]
14. Appelman HD, Threatt BA, Ernst C, et al. Filiform polyposis of the colon: an unusual sequel of ulcerative colitis. Am J Clin Pathol. 1974;62:145-6. [ Links ]
15. Samach M, Train J. Demonstration of mucosal bridging in Crohns colitis. Am J Gastroenterol. 1980;74:50-4. [ Links ]
16. Peh WC. Filiform polyposis in tuberculosis of the colon. Clin Radiol. 1988;39:534-6. [ Links ]
17. Dellinger GW, Lynch CA, Mikas AA. Colonic ganglioneuroma presenting as filiform polyposis. J Clin Gastroenterol. 1996;22:66-70. [ Links ]
18. Kelly JK, Gabos S. The pathogenesis of inflammatory polyps. Dis Colon Rectum. 1987;30:251-4. [ Links ]
19. Buck JL, Dachman AH, Sobin LH. Polypoid and pseudopolypoid manifestation of inflammatory bowel disease. Radiographics. 1991;11:293-304. [ Links ]
20. Loddo I, Romano C. Inflammatory bowel disease: genetics, epigenetics, and pathogenesis. Front Immunol. 2015;6:551. [ Links ]
21. Choi YS, Suh JP, Lee IT, et al. Regression of giant pseudopolyps in inflammatory bowel disease. J Crohns Colitis 2012;6:240-3. [ Links ]
22. Dombal FT, Watts JM, Watkinson G, et al. Local complications of ulcerative colitis: stricture, pseudopolyposis, and carcinoma of colon and rectum. Br Med J. 1966;1:1442-7. [ Links ]
23. Soriani P, Tontini G, Neumann H, et al. Esophageal post-inflammatory polyposis in extensive and severe Crohns disease treated with anti-tumor necrosis factor alpha. Endoscopy. 2016;48(S 01):E261-2. [ Links ]
24. Hinrichs HR, Goldman H. Localized giant pseudopolyps of the colon. JAMA. 1968;205:248-9. [ Links ]
25. Tzkowitz SH, Yio X. Inflammation and cancer IV. Colorectal cancer in inflammatory bowel disease: the role of inflammation. Am J Physiol Gastrointest Liver Physiol. 2004;287:G7-17. [ Links ]
26. Freeman AH, Berridge FR, Dick AP, et al. Pseudopolyposis in Crohns disease. Br J Radiol. 1978;51:782-7. [ Links ]
27. Mavrogenis G, Ngendahayo P, Kisoka P, et al. Intestinal obstruction caused by giant filiform polyposis in a patient with normal colon. Endoscopy. 2013;45(Suppl 2):UCTN: E80-1. [ Links ]
28. Lee CG, Lim YJ, Choi JS, et al. Filiform polyposis in the sigmoid colon: a case series. World J. Gastroenterol. 2010;16:2443-7. [ Links ]
29. Levine DS, Surawicz CM, Spencer GD, et al. Inflammatory polyposis two years after ischemic colon injury. Dig Dis Sci. 1986;31:1159-67. [ Links ]
30. Vainer B, Jess T, Andersen PS. Rapid tumour-like growth of giant filiform polyposis in a patient without a history of chronic bowel inflammation. APMIS. 2007;115(11):1306-10. [ Links ]
31. Oakley G, Schraut WH, Peel R, et al. Diffuse filiform polyposis with unique histology mimicking familial adenomatous polyposis in a patient without inflammatory bowel disease. Arch Pathol Lab Med. 2007;131:1821-4. [ Links ]
32. Raila FA, Athar M, Balaa MA. Colonic filiform polyposis and a malignant adenomatous polyp. South Med J. 1989;82(11):1448-9. [ Links ]
33. Sheikholeslami MR, Schaefer RF, Mukunyadzi P. Diffuse giant inflammatory polyposis: a challenging clinicopathologic diagnosis. Arch Pathol Lab Med. 2004;128:1286-8. [ Links ]
34. Macaigne G, Boivin JF, Cheaib S, et al. Polype filiforme unique développé dans un côlon normal et révélé par une hémorragie digestive sévère, Gastroenterol Clin Biol. 2006;30:913-5. [ Links ]
35. Yantiss RK, Oh KY, Chen YT, et al. Filiform serrated adenomas: a clinicopathologic and immunophenotypic study of 18 cases. Am J Surg Pathol. 2007;31:1238-45. [ Links ]
36. Rozenbajgier C, Ruck P, Jenss H, et al. Filiform polyposis: a case report describing clinical, morphological, and immunohistochemical findings. Clin Investig. 1992;70:520-8. [ Links ]
37. Rutter MD, Saunders BP, Wilkinson KH, et al. Thirty-year analysis of a colonoscopic surveillance program for neoplasia in ulcerative colitis. Gastroenterology. 2006;130:1030-8. [ Links ]
38. Rutter M, Saunders B, Wilkinson K, et al. Cancer surveillance in longstanding ulcerative colitis: endoscopic appearances help predict cancer risk. Gut. 2004;53:1813-6. [ Links ]
39. Velayos F, Loftus E, Jess T, et al. Predictive and protective factors associated with colorectal cancer in ulcerative colitis: a case-control study. Gastroenterology. 2006;130:1941-9. [ Links ]
40. Rutter M, Saunders B, Wilkinson K, et al. Severity of inflammation is a risk factor for colorectal neoplasia in ulcerative colitis. Gastroenterology. 2004;126:451-9. [ Links ]
41. Kusunoki M, Nishigami T, Yanagi H, et al. Occult cancer in localized giant pseudopolyposis. Am J Gastroenterol. 1992;87:379-81. [ Links ]
42. Okayama N, Itoh M, Yokoyama Y, et al. Total obliteration of colonic lumen by localized giant inflammatory polyposis in ulcerative colitis; report of a Japanese case. Int Med. 1996;35:24-9. [ Links ]
43. Katz S, Rosenberg RF, Katzca I. Giant pseudopolyps in Crohn´s colitis. Am J Gastroenterol. 1981;76:267-71. [ Links ]
44. Esaki M, Matsumoto T, Fuyuno Y, et al. Giant inflammatory polyposis of the cecum with repeated intussusception in ulcerative colitis: report of a case. Am J Gastroenterol. 2009;104:2873-4. [ Links ]
45. Maldonado TS, Firozzi B, Stone D, et al. Colocolonic intusscesption of a giant pseudopolyp in a patient with ulcerative colitis. Inflamm Bowel Dis. 2004;10(1):41-4. [ Links ]
46. Maggs JRL, Browning LC, Warren BF, et al. Obstructing giant post inflammatory polyposis in ulcerative colitis: Case report and review of the literature. J Crohns Colitis. 2008;2(2):170-80. [ Links ]
47. Boulagnon C, Jarezon JF, Diaz-Cives A, et al. Filiform polyposis: a benign entity? Case report and literature review. Pathol Res Pract. 2014;210:189-93. [ Links ]











 text in
text in