Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista colombiana de Gastroenterología
Print version ISSN 0120-9957
Rev Col Gastroenterol vol.33 no.2 Bogotá Apr./June 2018
https://doi.org/10.22516/25007440.196
Original articles
Phenotypic characteristics and treatment of inflammatory bowel disease at a university hospital in Bogotá, Colombia
1Departamento de Gastroenterología y Endoscopia Digestiva de la Clínica Universitaria Colombia, Fundación Universitaria Sánitas. Bogotá D. C., Colombia
2Jefe del servicio de Gastroenterología y Endoscopia Digestiva de la Clínica Reina Sofía. Bogotá D. C., Colombia
Inflammatory bowel disease (IBD) includes ulcerative colitis and Crohn’s disease which are chronic and recurrent inflammatory pathologies of the digestive tract whose causes are still uncertain. IBD’s incidence and prevalence are increasing worldwide, especially in developing countries.
Objectives:
The objective of this study was to describe phenotypic characteristics and types of treatment of 165 patients diagnosed with IBD treated between July 5, 2013 and December 31, 2016 at a university hospital in Bogotá, Colombia.
Methods:
This is a descriptive study based on frequencies and proportions of patients diagnosed with IBD at the University Clinic Colombia in Bogotá between July 5, 2013 and December 31, 2016.
Results:
UC accounted for 75.8% of the cases while CD accounted for 24.2% of these cases. Women accounted for a slightly larger proportion of patients than did men: 56% of UC cases and 55% of CD cases. UC’s most frequent form of presentation was pancolitis which was found 46.4% of the patients. Frequent forms of CD presentation were ileocolonic (52.5%) and inflammatory non-stenosing or fistulizing (60%). CD patients had a greater percentage of extraintestinal manifestations (35%), greater use of biological medications (35%) and required a greater number of surgeries (27.5%) than did UC patients.
Keywords: Inflammatory bowel disease; ulcerative colitis; Crohn’s disease
Introducción:
la enfermedad inflamatoria intestinal (EII) (colitis ulcerativa [CU], enfermedad de Crohn [EC]) es una patología inflamatoria crónica y recurrente del tracto digestivo, de causa aún incierta. La incidencia y prevalencia de esta enfermedad están en aumento a nivel mundial, especialmente en los países en vías de desarrollo.
Objetivo:
describir las características del fenotipo y el tipo de tratamiento administrado a 165 pacientes con diagnóstico de EII que consultaron durante el período comprendido entre el 5 de julio del 2013 y el 31 de diciembre del 2016 en un hospital universitario de la ciudad de Bogotá, Colombia.
Materiales y métodos:
se realizó un estudio descriptivo basado en frecuencias y proporciones de los pacientes con diagnóstico de EII atendidos en la Clínica Universitaria Colombia (Bogotá) entre el 5 de julio del 2013 y el 31 de diciembre del 2016.
Resultados:
el 75,8% de los casos correspondió a CU y el 24,2% a EC, con un leve predominio del sexo femenino en ambas (56% y 55%, respectivamente); la forma de presentación más frecuente de la CU fue la pancolitis en el 46,4%. Los pacientes con EC tuvieron un compromiso más frecuentemente ileocolónico (52,5%) e inflamatorio no estenosante ni fistulizante (60%), un mayor porcentaje de manifestaciones extraintestinales (35%), de uso de medicamentos biológicos (35%) y requirieron un mayor número de cirugías (27,5%) en comparación con los pacientes con CU.
Palabras clave: Enfermedad inflamatoria intestinal; colitis ulcerativa; enfermedad de Crohn
Introduction
Inflammatory bowel disease (IBD) is a chronic and recurrent inflammatory pathology of the digestive tract of uncertain etiology. In some cases it involves other organs. It is considered that a combination of genetic and environmental factors causes an alteration in the immune response that affects the gastrointestinal tract. Ulcerative colitis (UC) and Crohn’s disease (CD) are the main types of IBD.
Although the incidence and prevalence of IBD is highest in industrialized countries, there is a global trend toward increased incidence and prevalence of this disease throughout the world including non-industrialized countries. In 1991, a study published in Colombia described 108 IBD (98 UC, 10 CD) cases in two medical centers in Bogotá in the period between 1968 and 1990. 1 In 2010, 26 patients were described in Cartagena (20 UC, 6 CD), and a general prevalence of 29/100,000 was estimated for this city. 2 In 2010, a study was published describing the phenotype and natural history of IBD in 202 patients at a referral center in Medellin between 2001 and 2009 (80.7% UC, 15.8% CD). 3
This study describes phenotypic characteristics and type of treatment administered to 165 patients with IBD who were seen at a university hospital in Bogotá, Colombia between July 5, 2013 and December 31, 2016.
Materials and Methods
This is a descriptive study based on frequencies and proportions of patients diagnosed with IBD treated at the Clínica Universitaria Colombia in Bogotá between July 5, 2013 and December 31, 2016. Only patients with confirmed diagnoses of CD and UC were included. Diagnoses were based on the recommendations of the European guidelines for diagnosis of IBD and were only made symptoms had lasted at least 3 months. 4,5
Results
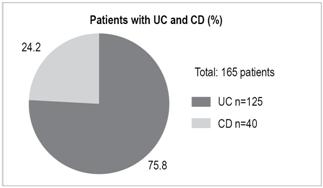
One hundred sixty-five patients were evaluated: 125 with UC (75.8%) and 40 with CD (24.2%) (Figure 1).
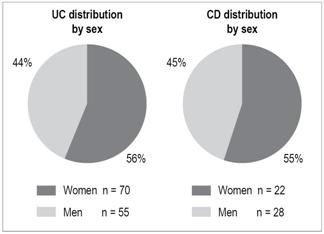
Seventy women (56%) and 55 men (44%) were found to have UC while 22 women (55%) and 18 men (45%) were found to have CD (Figure 2).
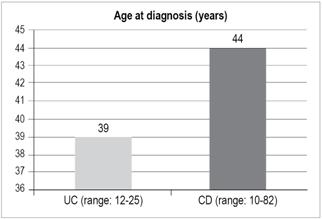
The average age at diagnosis was 39 years (12-75 years) for UC and 44 years (10-82 years) for CD (Figure 3).
Montreal Classification according to age
UC (Figure 4):
A1 (<17 years): n = 5 (4%)
A2 (between 17 and 40 years): n = 67 (53.6%)
A3 (> 40 years): n = 53 (42.4%)
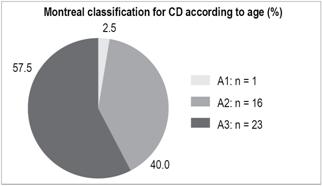
CD (Figure 5):
A1 (<17 years): n = 1 (2.5%)
A2 (between 17 and 40 years): n = 16 (40%)
A3 (> 40 years): n = 23 (57.5%).
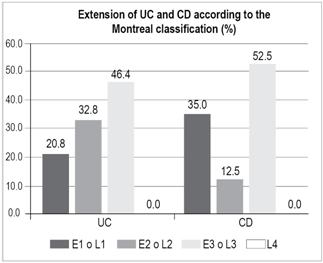
Disease extension (Montreal classification) (Figure 6)
UC:
E1 (proctitis): n = 26 (20.8%)
E2 (left colitis): n = 41 (32.8%)
E3 (pancolitis): n = 58 (46.4%)
CD:
L1 (ileum): n = 14 (35%)
L2 (colon): n = 5 (12.5%)
L3 (ileocolonic): n = 21 (52.5%)
L4 (isolated upper digestive tract): n = 0.
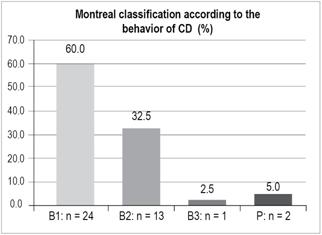
Behavior of CD (Figure 7)
B1 (inflammatory): n = 24 (60%)
B2 (stenosing): n = 13 (32.5%)
B3 (fistulizing/penetrating other than perianal): n = 1 (2.5%)
P (perianal): n = 2 (5%)
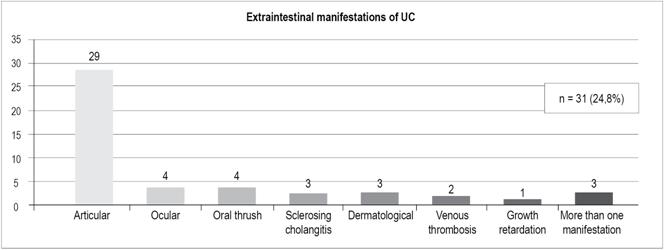
Extraintestinal manifestations
In patients with UC (Figure 8), there were 31 cases (24.8%) distributed as follows:
Articular: 29 cases
Ocular: 4
Oral thrush: 4
Sclerosing cholangitis: 3
Dermatological (erythema nodosum, pyoderma gangrenosum): 3
Venous thrombosis: 2
Growth retardation: 1. Three patients had more than one extraintestinal manifestation
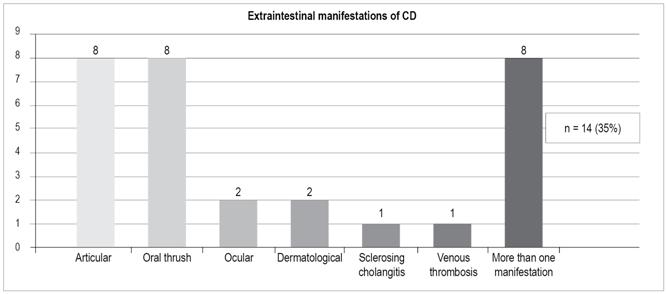
In patients with CD (Figure 9) there were 14 cases (35%) distributed as follows:
Articular: 8
Oral thrush: 8
Ocular: 2
Dermatological: 2 (dermatitis herpetiformis, pyoderma gangrenosum)
Sclerosing cholangitis: 1
Venous thrombosis: 1. Eight patients had more than 1 extraintestinal manifestation.
Treatment for patients with UC (Figure 10)
No treatment: 16 (12.8%)
Aminosalicylates: 104 (83.2%)
Oral only: 53 (42.4%)
Oral and rectal: 51 (40.8%)
Immunomodulators
Azathioprine, mercaptopurine: 29 (23.2%)
Steroid exposure: 88 (70.4%)
Tacrolimus: 1 (0.8%)
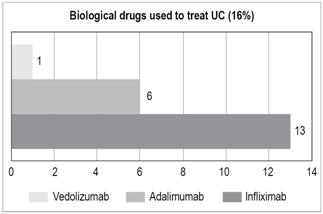
Biological: 20 (16%):
Infliximab: 13
Adalimumab: 6
Vedolizumab: 1. Forty-four percent of patients with biologics received combination therapy with an immunomodulator.
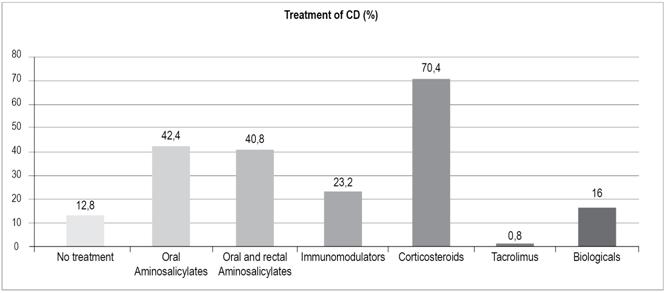
Treatment for patients with CD (Figure 11)
Aminosalicylates: 12 (30%)
Immunomodulators: 14 (35%)
Exposure to corticosteroids: 14 (35%)
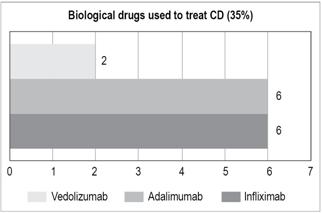
Biological: 14 patients (35%):
Adalimumab: 6
Infliximab: 6
Vedolizumab: 2. Of patients treatment with biologicals, four (28.5%) were under combined therapy with an immunomodulator. One patient received tacrolimus (2.5%).
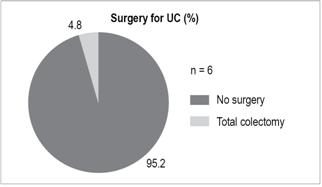
Surgery
Six UC patients (4.8%) required total colectomies (Figure 12).
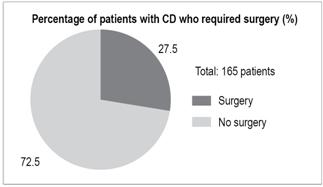
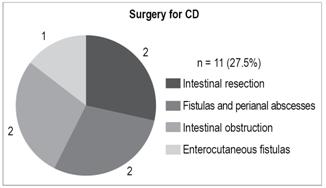
Eleven CD patients (27.5%) required surgery:
Intestinal resections: 2
Abscesses and perianal fistulas: 2
Intestinal obstruction: 2
Enterocutaneous fistulas: 1 (Figures 13 and 14).
Discussion
IBD is more common in developed countries, especially in North America and Western Europe. 6. The annual incidence of UC in North America is 19 cases/100,000 inhabitants and its prevalence is 37-248 cases/100,000 inhabitants. European incidence is 24 cases per 100,000 inhabitants with a prevalence of 4.9-505 cases/100,000 inhabitants. The incidence of CD is similar: 20 cases/100,000 inhabitants in North America and 12.7 cases/100,000 inhabitants in Europe 7. The incidence of IBD has increased worldwide. In Europe, the incidence of UC increased from 6 cases/100,000 people/year in 1962 to 9.8 cases/100,000 people/year in 2010 while the incidence of CD increased from 1 case/100,000 people/year to 6.3 case/100,000 people/year during the same period. 8 Similar results have been observed in the United States. 9 IBD was considered to be infrequent in Eastern countries, but epidemiological data in Japan, Korea and Hong Kong have shown increased incidences between 1980 and 2003. 10 In these populations the incidence of UC is greater than that of CD, and increases in incidence of UC almost always precede increases in the incidence of CD by a decade. (7 This may be similar to what is happening in the Colombian population. It is considered that westernization of life habits is a factor that may influence the increasing incidence of IBD. 6
This study describes 165 patients with IBD covered by a healthcare provider that manages obligatory healthcare and prepaid medicine. Patients were evaluated at the Clínica Universitaria Colombia in Bogotá. UC accounted for 75.8% of the cases, and CD accounted for 24.2%. This agrees with other studies conducted in Colombia which have observed that UC is more frequent than CD. 1,2,3
Women accounted for 56% of the UC patients and 55% of the CD patients. In other studies, the female to male ration has been 1.3 to 1.0 for CD with no differences between men and women for UC. 11,12,13
UC can start at any age, although it is uncommon before 5 years of age and after 75 years. Peak incidence occurs in the second and third decades of life with a second peak between 60 and 70 years. CD occurs most frequently between 15 and 30 years with an average age of 30 years. Nevertheless, an increase in the diagnosis of CD has been observed in patients 60 years and older. 14,15 In this study, the average age at diagnosis was 39 years (12 to 75 years) for UC and 44 years (10 to 82 years) for CD. The majority of patients with UC were in group A2 (53%), and the majority of CD patients were in group A3 (57.5%). CD’s more frequent occurrence in the older age group is similar to the findings of Juliao. 3 Because this study was based on a population served by an adult gastroenterology service, it is to be expected that A1 group patients (under 17 years of age) are underrepresented for both UC and CD.
The phenotype of IBD has changed in recent years 16,17. Although the extent of UC is relatively evenly divided between proctitis, left colitis and pancolitis, the proportion of patients presenting pancolitis has increased in recent decades in countries such as Denmark where pancolitis accounted for 18% of the cases between 1962-1987 but increased to 27% between 2003-2004. 18 In Australia, pancolitis predominates (41%). 19 Left colitis is the most common initial manifestation in Hungary (50%) and Holland (52%),20,21 while proctitis is most frequent in Asia (37%) (Figure 15).19
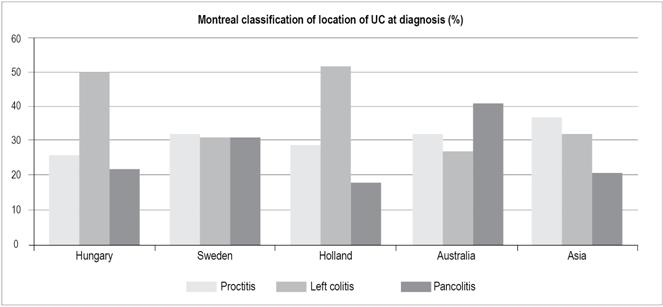
Figure 15 Montreal classification of location of UC at diagnosis. Modified from: Moran C et al. Gastroenterol Res Pract. 2016; 2016:1-9.
Previous studies in Colombia have found a predominance of left colitis (45%) whereas this study found that pancolitis (46.4%) predominated while proctitis was the least frequent (20.8%). 3
CD most frequently affected the ileum and colon (52.5%) which is similar to findings in other studies from Hungary, the Netherlands, Australia-Asia (Figure 16) and Colombia (50%).3,19,20,21 Isolated ileal compromises were found in 35% of the case while isolated colonic compromise was found in 12.5%. There were no cases of CD found in the upper digestive tract. CD was mainly inflammatory (60%), followed by stenosing (32.5%), perianal (5%) and non-perianal fistulas (2.5%). This behavior is similar to that found in a study in the Netherlands where the majority of cases were of the luminal inflammatory type (68%), followed by the stenosing phenotype (19%) with a lower proportion of fistulizing and perianal manifestations (11% and 9%, respectively). Similar findings were obtained by a European multicenter study and a study in Norway which reported predominance of the inflammatory phenotype in 73.7% and 62%, respectively. In both studies, this was followed by the stenosing phenotype (15.9% and 27%, respectively).22,23 In contrast, the study by Dr. Juliao in Colombia found a lower percentage of the inflammatory phenotype (34.4%) which was probably due to the prolonged period between the onset of symptoms and consultation. 3
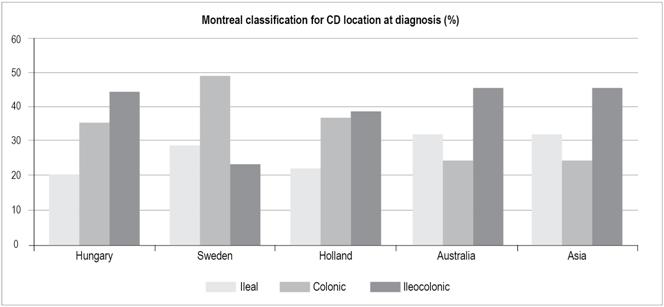
Figure 16 Montreal classification for CD location at diagnosis. Modified from: Moran C et al. Gastroenterol Res Pract. 2016; 2016: 1-9.
Fifty percent of patients with IBD have at least one extraintestinal manifestation although they are more frequent in CD patients, especially those with colonic involvement, than in UC patients. 24 This study found that a greater percentage of patients with CD (35%) presented extraintestinal manifestations than did patients with UC (24.8%). The most frequent locations joints in both groups (23% in UC and 20% in CD) and oral aphthae (3.2% in UC and 20% in CD). These results are similar to those found in the Colombian population and in other groups for which joint involvement has been reported in 16% to 35% of patients.3,25,26,27. A greater percentage of patients with CD (20%) had more than one extraintestinal manifestation. This happened in three patients with UC (2.4%).
Treatment of UC depends on several factors,28 but first line therapy is aminosalicylates. In this study, 100% of patients received aminosalicylates at some point. Because it was a descriptive study, at the time the information was collected, 83.2% of the patients had received aminosalicylates (42.4% orally and 40.8% orally and rectally). Only 12.8% had not received aminosalicylates, mainly due to the lack of efficacy of the drug or histories of total colectomies. Immunomodulators, azathioprine or 6-mercaptopurine, had been administered to 23.2% of the patients and 70.4% of the patients had been exposed to corticosteroids although in no case were they used chronically. For patients who were refractory to initial therapy using aminosalicylates and immunomodulators, patients who were dependent on, or refractory to, corticosteroids, and patients who had been hospitalized with severe active colitis but had not responded to intravenous corticosteroids within 3 to 5 days, biological therapy was used. 28 Sixteen percent of the patients with UC received biological treatment. Most of these had pancolitis (75%), 20% were categorized as E2 and 5% as E1. The most frequently administered biologic was infliximab (13 patients). Six patients received adalimumab, and one patient received vedolizumab (Figure 17). Forty-four percent of patients who received biologics simultaneously received an immunomodulator: 61.5% of the patients with infliximab and 16.6% of the patients with adalimumab. The primary reasons patients receiving infliximab did not receive an immunomodulator were adverse effects including abdominal pain, elevated liver enzymes and hematological toxicity. One patient (0.8%) with pancolitis received tacrolimus because of failure to respond to infliximab, and one other patient received vedolizumab due to lack of response to anti-tumor necrosis factor (anti-TNF) and the impossibility of a colectomy.
Alternatives for treating CD include corticosteroids, immunomodulators and biologics. 29 Mesalazine has shown very marginal benefits for CD patients. One study found that mesalazine was equivalent to budesonide in mild ileocecal disease which might suggest a role for aminosalicylates in cases of mild CD. Nevertheless, a metaanalysis has confirmed that budesonide should be preferred in this clinical scenario. No clear evidence has been found that mesalazine is better than placebos for treatment of CD. 29 In this study, 30% of patients with CD received aminosalicylates, 35% were exposed to corticosteroids (21% budesonide and 79% systemic corticosteroids), and 35% received immunomodulators (azathioprine or 6-mercaptopurine). In comparison with patients with UC, a greater percentage of patients with CD (35%) required treatment with biologics. The frequency of administration of infliximab and adalimumab was the same, six patients for each medication (Figure 18). Two patients received vedolizumab, one patient who was intolerant to infliximab because of severe leukopenia who was also a primary non-responder to adalimumab, and a patient whose response to infliximab diminished who was also a primary non-responder to adalimumab. Anti-TNF therapy combined with an immunomodulator was administered to 28.5% of the patients. One patient (2.5%) received tacrolimus.
Between 4% and 9% of patients with UC require proctocolectomies as definitive treatment during the first year after diagnosis. The risk of requiring surgery thereafter is 1% per year. Indications for surgery are sometimes urgent as in cases of colon perforation, toxic megacolon, fulminant colitis refractory to medical management and uncontrolled bleeding. Surgery may also be elective when patients are refractory to medical management or have adenocarcinoma of the colon and sometimes when dysplasia is found in a biopsy. 30 In this study, 4.8% of patients with UC required total colectomies, a figure which is similar to data published in other countries and here in Colombia (6% in Juliao’s study). 3
Despite advances in the medical treatment of CD, a significant percentage of patients still need surgery. Eighty percent of patients require at least one surgical procedure during their lifetimes, and they have high risks of postoperative recurrence: 30% at 3 years and 60% at 10 years. Seventy percent of patients may need a second intestinal resection. 31 Indications for surgery in CD vary from obstructive disease without significant inflammatory activity to abscesses, fistulas, bowel narrowing due to scar tissue, and perianal disease.31,32 In this study, 27.5% of patients with CD required some type of surgery including intestinal resections due to stenosis and masses (n = 2), abscesses and perianal fistulas (n = 2), intestinal obstructions that did not require intestinal resection (n = 2), and an enterocutaneous fistula (n = 1).
Referencias
1. Argüello M, Archila PE, Sierra F, et al. Enfermedad inflamatoria intestinal. Rev Col Gastroenterol. 1991;6(4):237-72. [ Links ]
2. Yepes I, Carmona R, Díaz F, et al. Prevalencia y características demográficas de la enfermedad inflamatoria intestinal en Cartagena, Colombia. Rev Col Gastroenterol. 2010;25(2):107-11. [ Links ]
3. Juliao F, Ruiz M, Flórez J, et al. Fenotipo e historia natural de la enfermedad inflamatoria intestinal en un centro de referencia en Medellín, Colombia. Rev Col Gastroenterol. 2010;25(3):240-51. [ Links ]
4. Van Assche G, Dignass A, Panés J, et al. The second European evidence-based Consensus on the diagnosis and management of Crohn’s disease: Definitions and diagnosis. J Crohns Colitis. 2010;4(1):63-101. doi: 10.1016/j.crohns.2009.09.009. [ Links ]
5. Dignass A, Eliakim R, Magro F, et al. Second European evidence-based consensus on the diagnosis and management of ulcerative colitis part 1: definitions and diagnosis. J Crohns Colitis. 2012;6(10):965-90. doi: 10.1016/j.crohns.2012.09.003. [ Links ]
6. Hanauer S. Inflammatory bowel disease: epidemiology, pathogenesis and therapeutic opportunities. Inflamm Bowel Dis. 2006;12 Suppl 1:S3-9. [ Links ]
7. Ananthakrishnan AN. Epidemiology and risk factors for IBD. Nat Rev Gastroenterol Hepatol. 2015;12(4):205-17. doi: 10.1038/nrgastro.2015.34. [ Links ]
8. Burisch J, Munkholm P. The epidemiology of inflammatory bowel disease. Scand J Gastroenterol. 2015;50(8):942-5. doi: 10.3109/00365521.2015.1014407. [ Links ]
9. Loftus EV Jr, Silverstein MD, Sandborn WJ, et al. Ulcerative colitis in Olmsted County, Minnesota, 1940-1993: incidence, prevalence, and survival. Gut. 2000;46(3):336-43. doi: 10.1136/gut.46.3.336. [ Links ]
10. Thia KT, Loftus EV Jr, Sandborn WJ, et al. An update on the epidemiology of inflammatory bowel disease in Asia. Am J Gastroenterol. 2008;103(12):3167-82. doi: 10.1111/j.1572-0241.2008.02158.x. [ Links ]
11. Kappelman MD, Rifas-Shiman SL, Kleinman K, et al. The prevalence and geographic distribution of Crohn’s disease and ulcerative colitis in the United States. Clin Gastroenterol Hepatol. 2007;5(12):1424-9. doi: 10.1016/j.cgh.2007.07.012. [ Links ]
12. Jacobsen BA, Fallingborg J, Rasmussen HH, et al. Increase in incidence and prevalence of inflammatory bowel disease in northern Denmark: a population-based study, 1978-2002. Eur J Gastroenterol Hepatol. 2006;18(6):601-6. [ Links ]
13. Osterman M, Lichtenstein G. Ulcerative colitis. En: Feldman M, Friedman LS, Brandt LJ (editores). Sleisenger and Fordtran’s gastrointestinal and liver diseases. 10.a edición. Elsevier; 2015. pp. 2023-61. [ Links ]
14. Loftus CG, Loftus EV Jr, Harmsen WS, et al. Update on the incidence and prevalence of Crohn’s disease and ulcerative colitis in Olmsted County, Minnesota, 1940-2000. Inflamm Bowel Dis. 2007;13(3):254-61. doi: 10.1002/ibd.20029. [ Links ]
15. Bernstein CN, Wajda A, Svenson LW, et al. The epidemiology of inflammatory bowel disease in Canada: a population-based study. Am J Gastroenterol. 2006;101(7):1559-68. doi: 10.1111/j.1572-0241.2006.00603.x. [ Links ]
16. Langholz E, Munkholm P, Davidsen M, et al. Course of ulcerative colitis: Analysis of changes in disease activity over years. Gastroenterology. 1994;107(1):3-11. doi: 10.1016/0016-5085(94)90054-X. [ Links ]
17. Moran C, Sheehan D, Shanahan F. The changing phenotype of inflammatory bowel disease. Gastroenterol Res Pract. 2016;2016:1-9. doi: 10.1155/2016/1619053. [ Links ]
18. Jess T, Riis L, Vind I, et al. Changes in clinical characteristics, course, and prognosis of inflammatory bowel disease during the last 5 decades: a population-based study from Copenhagen, Denmark. Inflamm Bowel Dis. 2007;13(4):481-9. doi: 10.1002/ibd.20036. [ Links ]
19. Ng SC, Tang W, Ching JY, et al. Incidence and phenotype of inflammatory bowel disease based on results from the Asia-Pacific Crohn’s and colitis epidemiology study. Gastroenterology. 2013;145(1):158-65.e2. doi: 10.1053/j.gastro.2013.04.007. [ Links ]
20. Lakatos L, Kiss LS, David G, et al. Incidence, disease phenotype at diagnosis, and early disease course in inflammatory bowel diseases in Western Hungary, 2002-2006. Inflamm Bowel Dis. 2011;17(12):2558-65. doi: 10.1002/ibd.21607. [ Links ]
21. Nuij VJ, Zelinkova Z, Rijk MC, et al. Phenotype of inflammatory bowel disease at diagnosis in the Netherlands: a population-based inception cohort study (the Delta Cohort) Inflamm Bowel Dis. 2013;19(10):2215-22. doi: 10.1097/MIB.0b013e3182961626. [ Links ]
22. Wolters FL, Russel MG, Sijbrandij J, et al. Phenotype at diagnosis predicts recurrence rates in Crohn’s disease. Gut 2006;55(8):1124-30. doi: 10.1136/gut.2005.084061. [ Links ]
23. Solberg IC, Vatn MH, Høie O, et al. Clinical course in Crohn’s disease: results of a Norwegian population-based ten-year follow-up study. Clin Gastroenterol Hepatol. 2007;5(12):1430-8. doi: 10.1016/j.cgh.2007.09.002. [ Links ]
24. Harbord M, Annese V, Vavricka S, et al. The first european evidence-based consensus on extra-intestinal manifestations in inflammatory bowel disease. J Crohns Colitis. 2016;10(3):239-54. doi: 10.1093/ecco-jcc/jjv213. [ Links ]
25. Orchard TR, Wordsworth BP, Jewell DP. Peripheral arthropathies in inflammatory bowel disease: their articular distribution and natural history. Gut. 1998;42(3):387-91. doi: 10.1136/gut.42.3.387. [ Links ]
26. Kethu S. Extraintestinal manifestations of inflammatory bowel diseases. J Clin Gastroenterol, 2006;40(6):467-75. doi: 10.1097/00004836-200607000-00003. [ Links ]
27. Danese S, Semeraro S, Para A, et al. Extraintestinal manifestations in inflammatory bowel disease. World J Gastroenterol. 2005;11(46):7227-36. doi: 10.3748/wjg.v11.i46.7227. [ Links ]
28. Dignass A, Lindsay J, Sturm A, et al. Second European evidence-based consensus on the diagnosis and management of ulcerative colitis Part 2: Current management. J Crohns Colitis. 2012;6(10):991-1030. doi: 10.1016/j.crohns.2012.09.002. [ Links ]
29. Gomollón F, Dignass A, Annese V, et al. 3rd European evidence-based consensus on the diagnosis and management of Crohn’s disease 2016: Part 1: Diagnosis and medical management. J Crohns Colitis. 2017;11(1):3-25. doi: 10.1093/ecco-jcc/jjw168. [ Links ]
30. Bohl J, Soba K. Indications and options for surgery in ulcerative colitis. Surg Clin N Am. 2015;95(6):1211-32. doi: 10.1016/j.suc.2015.07.003. [ Links ]
31. Bailay H, Glasgow S. Challenges in the medical and surgical management of chronic inflammatory bowel disease. Surg Clin N Am. 2015;95(6):1233-44. doi: 10.1016/j.suc.2015.08.003. [ Links ]
32. Gionchetti P, Dignass A, Danese S, et al. 3rd European evidence-based consensus on the diagnosis and management of Crohn’s disease 2016: Part 2: Surgical management and special situations. J Crohns Colitis. 2017;11(2):135-49. doi: 10.1093/ecco-jcc/jjw169. [ Links ]
Received: November 23, 2017; Accepted: April 13, 2018











 text in
text in