Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista colombiana de Gastroenterología
Print version ISSN 0120-9957
Rev Col Gastroenterol vol.33 no.2 Bogotá Apr./June 2018
https://doi.org/10.22516/25007440.255
Review articles
Are self-expanding metal prostheses useful for controlling refractory variceal digestive hemorrhaging?
1Médico internista y gastroenterólogo, profesor de Medicina Interna y Gastroenterología, Pontificia Universidad Javeriana. Gastroenterólogo, Hospital Universitario San Ignacio-Fundación Clínica Shaio. Bogotá D. C., Colombia. Correo: ramrs@yahoo.com
Variceal bleeding is a medical emergency which has high rates of morbidity and mortality. Up to 10% of these patients treated in hospitals do not respond to conventional hemostatic measures. For this reason, the use of self-expanding metal stents (SEMS) has been proposed for management of these patients. It has had promising results.
This paper discusses the literature available internationally about experience with the use of SEMS, their efficacy for acute control, long-term patient prognoses, types of stents, alternatives, complications, and challenges for the use of SEMS for patient management that should be considered in our environment.
Keywords: Refractory variceal bleeding; rescue therapy; self-expanding metal stents; endoscopic management.
El sangrado varicoso es una urgencia médica con altas tasas de morbimortalidad. De los pacientes que llegan para recibir manejo hospitalario, hasta un 10% no responden a medidas hemostáticas convencionales, por lo que se ha planteado el uso de prótesis metálicas autoexpandibles para el manejo con resultados prometedores de dichos pacientes.
En el presente documento se discute la literatura mundial disponible acerca de la experiencia en el uso de dichos dispositivos; la eficacia en el control agudo; el pronóstico a largo plazo; el tipo de prótesis utilizadas y las alternativas, complicaciones y retos del manejo de pacientes con esta técnica, que puede considerarse emergente en nuestro medio.
Palabras clave: Sangrado varicoso refractario; terapia de rescate; prótesis metálicas autoexpandibles; manejo endoscópico
Introduction
Gastrointestinal bleeding due to ruptured esophageal varices is one of the most frequent and dramatic complications associated with liver cirrhosis. The incidence of this complication is as high as 25% in patients with portal hypertension, and the average mortality at six weeks is over 20%. It occurs more frequently as the liver deteriorates: it occurs in 10% of Child- Pugh A patients but in up to 70% of Child-Pugh C patients. Another characteristic of this complication is a high rate of recurrence in the first two to three months after the initial hemorrhage. 1,2.
Taking into account its dramatic panorama, it is of vital importance to develop strategies aimed at preventing the first episode of varicose bleeding and also preventing recurrence. One of the most important issues is identification of at-risk patients. Risk factors associated with varicose ruptures include portal and varicose pressure; endoscopic characteristics of the varices, especially the size and presence of alarm signal; and the severity of liver disease and its etiology.
Unfortunately, in many cases failures to control risk factors trigger episodes of varicose digestive bleeding. Fifteen percent are located in the stomach or duodenum while 85% are esophageal. Due to anatomical circumstances, bleeding occurs more frequently in the distal 5 cm of the esophagus. Under normal conditions, these veins are directed longitudinally by the lamina propria of the esophageal mucosa. In portal hypertension, increased blood pressure and flow increases the caliber of blood vessels which also results in moving them to the surface so that they lose the protection of the surrounding tissues. When portal venous pressure exceeds 12 mm Hg, the risk of rupturing varices and consequent digestive bleeding increases. 2
At the time of admission, these patients require appropriate evaluation, respiratory and hemodynamic stabilization (taking into account the start of intravenous fluids and protection of the airway), and even orotracheal intubation in case of deterioration of the state of consciousness that increases the risk of bronchial aspiration of blood materials. The requirement for blood products to maintain hemoglobin between 7 and 8 g/dL should also be considered.
Similarly, the use of antibiotics can be important for cirrhotic patients with digestive bleeding since infectious complications in this group of patients is a cause of significant morbidity and mortality. The consensus recommendation of Baveno is to use 1 g/day of ceftriaxone in populations with high resistance to fluoroquinolones antibiotics and in those who have previously used this group of antibiotics for prophylaxis. 1
Gastrointestinal endoscopy with therapeutic intent should be performed as soon as possible since a direct correlation has been identified between delays of over 15 hours from admission and hospital deaths. 3
However, in at least 10% of cases there is no response, so that active bleeding continues despite conventional hemostatic measures such as band ligation and injection of sclerosing solutions used simultaneously with splanchnic vasodilators such as terlipressin, somatostatin and octreotide. 4
Although there is consensus that transjugular portosystemic shunts (TIPS) are useful tools for treatment of patients with Child-Pugh C cirrhosis of more than 13 points who have bleeding varices, the consensus does not extend to all patients. TIPS is still a therapy with difficulties for those with Child-Pugh B who have active bleeding at the time of the first endoscopy and for those patients for whom medical and endoscopic treatment has failed. These include clinical worsening of patients with severe hepatic insufficiency (which would limit its use to a small group of patients in whom it would otherwise be indicated) and availability of the resource for use within 24 hours after the episode of digestive bleeding. A significant number of patients continue to bleed, a situation that causes death in a matter of hours. 5,6
There has been an increase in the use of temporary devices such as the Sengstaken-Blakemore balloon and fully coated self-expanding metal stents (SEMS) in hemostatic methods that seek to control refractory bleeding by compression of the bleeding varicose vein. Although as the Sengstaken-Blakemore balloon has been used for approximately 25 years, there have been serious difficulties with proper placement since placement cannot be easily verified. Patient discomfort also requires a traction system to ensure compression on the gastric fundus. Moreover, the 24 hour time of therapy is often too short to establish definitive treatment.7,8
On the other hand, SEMS are relatively simple and available and do not require fluoroscopic guidance. They also haves advantages such as up to 100% bleeding control without significant associated morbidity and mortality. Bridge therapy includes early reopening of the oral feeding route within 7 to 14 days as a temporary solution to allow for measures to reduce portal pressure and administer antibiotics. 8 Subsequently, patients can undergo longer-term treatments such as placement of TIPS, liver transplantation, or even band-ligation for patients who initially could not undergo this treatment due to limited visualization or hemodynamic instability. 4 If these measures do not achieve effective control of bleeding, there would be no time to implement others resulting in many cases in the death of the patient. 9
In the vast majority of cases, stents are removed without complications after definitive treatment has been established. 9 However, it is clear that the risk of early rebleeding after removal is high if the definitive treatment has not yet been implemented. Possible contraindications for such treatments include severe impairment of liver functioning prior to bleeding in the case of TIPS. This is indicated by bilirubin over 3, alteration in thromboplastin time, and/or development of hepatic encephalopathy). Another contraindication is a central hepatic neoplasm. In the case of transplantation, consumption of alcohol in the six months prior to the procedure and cardiac, pulmonary, infectious or neoplastic comorbidities are all contraindications. 4,10,11
Randomized, blinded studies would be the model for determining whether intervention is adequate, but none have been performed for this specific condition. The methodological difficulties involved make this type of study practically impossible to carry out considering that this device cannot be blinded, the short term prognosis of the patient with or without intervention is bad, and there is no satisfactory control group. 6 For this reason, experience captured in case series is the main tool for reporting this intervention everywhere in the world, and its conclusions have come to be included in the Baveno VI Consensus of 2015 as salvage therapy for patients with refractory bleeding varices. 1
Justification
A large percentage of patients with bleeding varices that are refractory to conventional treatment die or have serious complications from invasive treatments such as emergency surgery. Management with self-expanding metal stents could be an effective measure to control bleeding efficiently, increase life expectancy of these patients, and lead some of them to more definitive therapy such as the placement of TIPS or liver transplantation.
Until now, there have been experiences reported of adjustment of this technique to our resources and needs.
Reflection
In many countries, self-expanding metal stents have had convincing results as rescue treatment for patients with digestive bleeding secondary to esophageal varices refractory to conventional endoscopic measures such as band ligation and sclerotherapy. Immediate control of bleeding reaches up to 96%, 12 and increased life expectancy allows patients to undergo bridge treatments while undergoing therapies with curative intent such as endoscopic therapy for the eradication of varices or - in the best case - liver transplantation. Patients for whom immediate control of bleeding is not achieved are mainly patients with gastric or gastric junction varices that cannot be covered by the body of the stent. These cases require complementary management such as injection of cyanoacrylate. 13
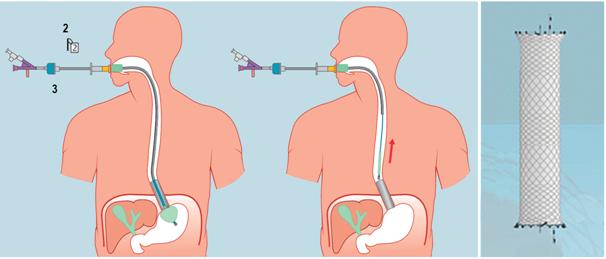
Most published studies were performed with the SX-ELLA Stent Danis (Hradec Kralove, Czech Republic) (Figure 1) which was developed specifically for placement in patients with varicose bleeding without the need for endoscopic or fluoroscopic assistance. Its positioning system has a distal balloon that is inflated in the gastric chamber. This allows it to be located immediately subcardial when pulled, ensuring placement of the stent immediately next to the balloon in the distal esophagus. It is a 13.5 cm long, fully coated nitinol prosthesis whose diameter is 25 mm. Cup sizes range from 28 mm to 30 mm. In addition, it has 2 metal removal tapes located in the extreme proximal and distal ends of the stnet that allow repositioning and extraction (Figure 2). 4,13,14
In studies with this device, placement times of between 4 and 10 minutes have been reported with bleeding control in 77% of the cases. The rates of complications was less than 1% due to ulceration of the distal esophagus without clinically significant consequences. 15.
Nevertheless, institutions that have the endoscopic resources available for management of patients with gastrointestinal bleeding have the possibility of placing self-expanding stents with a conventional placement system. This is supported by studies published by Hubmann using Choo Stent NES-T18-080-070 and by Dahlerup using the Shim-Hanaro antireflux stent which had results similar to those found with the Czech stent previously described. 16,17
After measuring the position of the gastroesophageal junction and the site of the bleeding varice, this type of fully covered self-expanding stent is placed. Ideally, they have a body diameter of more than 25 mm and cups from 28 to 30 mm. They achieve adequate control of bleeding in more than 90% of cases without the need to transfer the patient to fluoroscopy rooms. They have the advantages of being available in most endoscopy services although the use of this type of prosthesis should be ruled out in bleeding due to gastric varices.
Among the most frequent complications is migration of the prosthesis to the stomach. This occurs in between 21% to 36% of cases although there is no correlation between migration and increased incidence of rebleeding. 13 Zakaria et al. have suggested that performing an endoscopy to confirm adequate positioning of the prosthesis five minutes after placement favors complete expansion and decreases the risk of migration. 12 Another relatively frequent complication is superficial ulceration, especially at the extreme proximal end of the stent. This can be seen at the time of extraction and has no significant pathological outcomes. 12 Another complication described in the literature is obstruction of the airway secondary to extrinsic compression in patients with a history of lung volume reduction surgery due to emphysema or congenital airway changes. 18
In November 2015, Marot and colleagues published the first systematic review and metaanalysis of treatment of bleeding varices with SEMS. It included 13 studies from around the world that met the inclusion criteria. These studies covered a total of 146 patients from whom relevant data were obtained within the weighting of the application of the intervention. It was concluded that the 30-day mortality rate was 36%, although there was no control group for comparison. A significant decrease of mortality with respect to that in patients not treated with SEMS was assumed (Untreated patient mortality could be higher than 60%). The migration rate was 28% and utility as bridge therapy was 10% for transplantation and 26% for placement of TIPS. These figures are much higher than those documented in patients with refractory bleeding varices. 6
Conclusions
Self-expanding esophageal stents are tools that are available to health professionals in many institutions. They are useful for management of patients with bleeding due to esophageal varices that are refractory to conventional pharmacological and endoscopic measures.
Reports of complications, mainly migration and rebleeding, have generated development of placement devices and stents designed specifically for this application. These could reduce some limitations and could be useful for services that do not always have endoscopic treatment available which must refer patients to other institutions offering higher levels of care after stabilization.
Referencias
1. de Franchis R; Baveno VI Faculty. Expanding consensus in portal hypertension: Report of the Baveno VI Consensus Workshop: Stratifying risk and individualizing care for portal hypertension. J Hepatol. 2015;63(3):743-52. doi: 10.1016/j.jhep.2015.05.022. [ Links ]
2. Arakawa M, Masuzaki T, Okuda K. Pathology of fundic varices of the stomach and rupture. J Gastroenterol Hepatol. 2002;17(10):1064-9. [ Links ]
3. Hsu YC, Chung CS, Tseng CH, et al. Delayed endoscopy as a risk factor for in-hospital mortality in cirrhotic patients with acute variceal hemorrhage. J Gastroenterol Hepatol. 2009;24(7):1294-9. doi: 10.1111/j.1440-1746.2009.05903.x. [ Links ]
4. Dechêne A, El Fouly AH, Bechmann LP, et al. Acute management of refractory variceal bleeding in liver cirrhosis by self-expanding metal stents. Digestion. 2012;85(3):185-91. doi: 10.1159/000335081. [ Links ]
5. Escorsell A, Bosch J. Self-expandable metal stents in the treatment of acute esophageal variceal bleeding. Gastroenterol Res Pract. 2011;2011:910986. doi: 10.1155/2011/910986. [ Links ]
6. Marot A, Trépo E, Doerig C, et al. Systematic review with meta-analysis: self-expanding metal stents in patients with cirrhosis and severe or refractory oesophageal variceal bleeding. Aliment Pharmacol Ther. 2015;42(11-12):1250-60. doi: 10.1111/apt.13424. [ Links ]
7. Maufa F, Al-Kawas FH. Role of self-expandable metal stents in acute variceal bleeding. Int J Hepatol. 2012;2012:418369. doi: 10.1155/2012/418369. [ Links ]
8. El Sayed G, Tarff S, O’Beirne J, et al. Endoscopy management algorithms: role of cyanoacrylate glue injection and self-expanding metal stents in acute variceal haemorrhage. Frontline Gastroenterol. 2015;6(3):208-216. doi: 10.1136/flgastro-2013-100428. [ Links ]
9. Müller M, Seufferlein T, Perkhofer L, et al. Self-expandable metal stents for persisting esophageal variceal bleeding after band ligation or injection-therapy: A retrospective study. PLoS One. 2015;10(6):e0126525. doi: 10.1371/journal.pone.0126525. [ Links ]
10. García-Pagán JC, Caca K, Bureau C, et al. Early use of TIPS in patients with cirrhosis and variceal bleeding. N Engl J Med. 2010;362(25):2370-9. doi: 10.1056/NEJMoa0910102. [ Links ]
11. Deltenre P, Trépo E, Rudler M, et al. Early transjugular intrahepatic portosystemic shunt in cirrhotic patients with acute variceal bleeding: a systematic review and meta-analysis of controlled trials. Eur J Gastroenterol Hepatol. 2015;27(9):e1-9. doi: 10.1097/MEG.0000000000000403. [ Links ]
12. Zakaria MS, Hamza IM, Mohey MA, et al. The first Egyptian experience using new self-expandable metal stents in acute esophageal variceal bleeding: pilot study. Saudi J Gastroenterol. 2013;19(4):177-81. doi: 10.4103/1319-3767.114516. [ Links ]
13. Changela K, Ona MA, Anand S, et al. Self-Expanding Metal Stent (SEMS): an innovative rescue therapy for refractory acute variceal bleeding. Endosc Int Open. 2014;2(4):E244-51. doi: 10.1055/s-0034-1377980. [ Links ]
14. Hogan B, Patch D, Burroughs A, et al. Use of the SX-Ella self-expanding mesh metal stent in the management of complex variceal haemorrhage: initial experience in a single centre. J Hepatol. 2009;50(Suppl 1):s86-7. doi: 10.1016/S0168-8278(09)60214-2. [ Links ]
15. Pontone S, Giusto M, Filippini A, et al. Hemostasis in uncontrolled esophageal variceal bleeding by self-expanding metal stents: a systematic review. Gastroenterol Hepatol Bed Bench. 2016;9(1):6-11. [ Links ]
16. Hubmann R, Bodlaj G, Czompo M, et al. The use of self-expanding metal stents to treat acute esophageal variceal bleeding. Endoscopy. 2006;38(9):896-901. doi: 10.1055/s-2006-944662. [ Links ]
17. Dahlerup JF, Kruse A, Grønbaek H, et al. Therapy of continuously bleeding oesophageal varices by self expanding metal stents. Ugeskr Laeger. 2007;169(34):2784-5. [ Links ]
18. Dechene A, Adamzik M, Gerken G, et al. Acute bronchial obstruction following esophageal stent implantation for variceal bleeding. Endoscopy. 2009;41 Suppl 2:E146-7. doi: 10.1055/s-0028-1119725. [ Links ]
Received: August 28, 2017; Accepted: April 13, 2018











 text in
text in