Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista colombiana de Gastroenterología
Print version ISSN 0120-9957
Rev Col Gastroenterol vol.33 no.3 Bogotá July/Sept. 2018
https://doi.org/10.22516/25007440.165
Case report
Two cases of Menétrier’s disease in children
1Hospital Universitario San Vicente Fundación. Medellín, Colombia
We present two cases of Menétrier’s Disease (MS) referred to our institution due to edema. The prevalence of this disease is low. It is a hypertrophic gastropathy which causes loss of proteins and which has unknown causes in the majority of cases although it has been associated with infectious processes. It is characterized by edema, hypoproteinemia, hypoalbuminemia. In childhood it is benign and self-limited.
Keywords: Menétrier’s disease (ME); protein-losing gastroenteropathy; edematous syndrome; hypoalbuminemia; hypertrophic gastritis
Se presentan 2 casos de enfermedad de Ménétrier (EM) remitidos a nuestra institución por síndrome edematoso. Esta enfermedad de poca prevalencia es una gastropatía hipertrófica perdedora de proteínas que en la mayoría de los casos es de causa desconocida, aunque se ha asociado con procesos infecciosos. Se caracteriza por edema, hipoproteinemia, hipoalbuminemia y, en la infancia, es de carácter benigno y autolimitado.
Palabras clave: Enfermedad de Ménétrier (EM); gastropatía perdedora de proteínas; síndrome edematoso; hipoalbuminemia; gastritis hipertrófica
Introduction
Ménétrier’s disease was first described in adult patients in 1888 by the French pathologist Pierre Eugene Ménétrier. The presentation of the disease in children is rare, with less than 100 cases published in the literature. 1 It is characterized by a thickening of gastric folds with hypoalbuminemia secondary to the loss of proteins through the gastrointestinal mucosa. The majority of pediatric cases are associated with viral infections, among which cytomegalovirus (CMV) is the most frequently mentioned etiological agent. Unlike the adult disease, this is an acute disorder with a benign and self-limiting course.
This disease should be suspected in patients with nonspecific gastrointestinal symptoms, hypoproteinemia and edema for whom causes related to the liver, kidney and heart have been excluded. 2
Clinical case 1
The first patient was a 23-month-old male with a personal history of asthma and allergic rhinitis who was referred to our hospital because of hypoalbuminemia. Two weeks before admission, he had developed a flu and fever which soon become associated with vomiting, bloating and diarrhea. He went to other health centers which treated his symptoms. Then, on the tenth day, he suffered clinical deterioration including asthenia, adynamia, loss of appetite, generalized edema, and an objective weight gain of 4 kilos.
Upon admission, general conditions were observed to be below normal. His vital signs included arterial pressure of 95/41/59, heart rate of 140 beats per minute, respiratory rate of 24 breaths per minute, 98% oxygen saturation, 15 kg weight and 92 cm length. He was pallid with signs of mild dehydration, his breathing was rapid, and he had runny nose with clear mucus. The generalized edema was striking, and he was hospitalized in an intermediate care unit with a diagnostic impression of nephrotic syndrome.
A complete blood count had normal red cell and platelet series. The white cell series showed a leukocyte count of 18,300 with 27% reactive lymphocytes, and erythrocyte sedimentation rate (ESR) of two, and a C-reactive protein (CRP) level of 0.83. The level of triglycerides was 220, the coagulation study was normal, kidney function was normal (creatinine: 0.83 mg/dL), liver functions were normal (alanine aminotransferase [ALT]: 10 IU/L, aspartate aminotransferase [AST]: 31 IU/L), and the patient had hypoproteinemia (total proteins: 3.1 g/dL) and hypoalbuminemia (1.9 g/dL). The urine study was normal without evidence of proteinuria or hematuria.
The patient’s blood also tested negative for hepatitis B (HBV), hepatitis C (HCV), Epstein-Barr virus (EBV), human immunodeficiency virus (HIV), leptospira, mycoplasma, dengue and toxoplasma but tested positive for cytomegalovirus (CMV) immunoglobulin M (IgM) and immunoglobulin G (IgG).
Abdominal ultrasound showed bilateral pleural effusion and free fluid in the perirenal and perisplenic spaces.
The patient was evaluated by a pediatric nephrologist and a hepatologist who ruled out renal causes and acute portal thrombosis as explanations for the patient’s condition. An infectious disease specialist, consulted because of the finding of a CMV infection, considered that the patient did not merit antiviral treatment despite the evidence of infection (CMV viral load: 9932 copies/mL) since no involvement of the target organ was demonstrated and the risk of toxicity due to the medication was great.
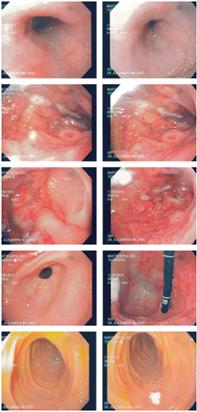
Finally, on the sixth day of hospitalization, a pediatric gastroenterologist performed an upper gastrointestinal endoscopy due to suspicion of gastropathy and/or protein losing enteropathy. Eroded hypertrophic folds with friable mucosa were found in the corpus, the antral mucosa’s appearance was normal, and the fundic mucosa had superficial erosions, so Ménétrier’s disease was suspected (Figure 1). With these findings, a colonoscopy was postponed indefinitely.
The pathological study found mild esophagitis, hypertrophic gastropathy (foveolar hyperplasia, dilatation and atrophy of glands, increased muscle fibers in the lamina propria, but no Helicobacter pylori) and signs of chronic duodenitis. These findings confirmed the diagnosis of Ménétrier’s disease (Figure 2).
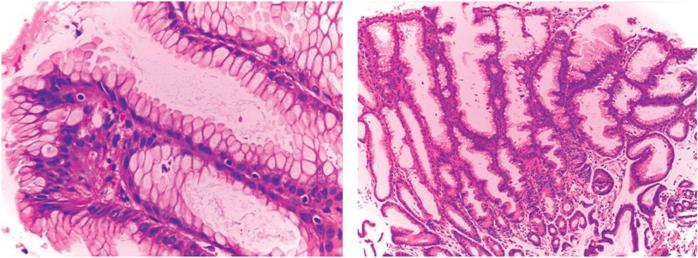
Figure 2 Elongation and hyperplasia of foveolar cells, increased muscle fibers in the lamina propria and predominantly mononuclear inflammatory infiltrate (200x and 100x).
The patient’s evolution was favorable. Initially, he was treated with infusions of albumin and diuretic to improve the edema, and he also received nutritional support and antisecretory treatment with esomeprazole. He was discharged on day 17 of hospitalization with outpatient follow-up 15 days later. At that time he was asymptomatic and with a serum albumin level of 3.6 mg/dL. He was scheduled for a follow-up upper endoscopy, but his parents decided not to have it performed. Periodic follow-ups have continued without relapse.
Clinical case 2
The second patient was a 5-year-old male who came to a second level hospital following 15 days of abdominal pain associated with multiple episodes of vomiting and diarrhea with mucus but without blood three to four times per day usually after meals. Subsequently, he developed edema of his eyelids, oliguria and foamy urine. While hospitalized there, doctors ruled out nephrotic syndrome and liver causes as explanations for the edema. They performed a CT scan of the abdomen which found multiple mesenteric adenopathies and thickening of the jejunal and ileal loops, so they referred him to our third level center for studies.
His vital signs at admission included a heart rate of 130 bpm, a respiration rate of 24 breathes per minute, oxygen saturation of 97%, arterial pressure of 69/45/56, 19.7 kg weight, and 112 cm height. He had generalized edema with basal right crepitus, a positive ascitic wave and collateral circulation in the upper abdomen.
Results of a complete blood count included normal red cell and platelet series and a leukocyte count of 15,100 (A peripheral blood smear showed 2% reactive lymphocytes). A urinalysis was normal as were kidney functions (creatinine: 0.53 mg/dL, blood urea nitrogen 18 mg/dL) and liver functions. Coagulation tests were normal, his triglyceride level was 135 mg/dL, and he had hypoproteinemia (The total serum protein level were 2.7 g/dL and the albumin level was 1.6 g/dL). Similarly, a chest x-ray showed moderate right pleural effusion, and an abdominal ultrasound showed no alterations.
He was evaluated by a pediatric gastroenterologist who suspected protein-losing gastropathy related to inflammatory bowel disease (due to abdominal CT findings). Upper endoscopy and colonoscopy revealed that the folds of the corpus were very thick and friable, the corporeal mucosa had pseudopolyps and erosions, the antral mucosa had a follicular appearance, the mucosa of the duodenal bulb and second portion appeared to be normal, and the colonic mucosa appeared normal throughout (Figure 3).
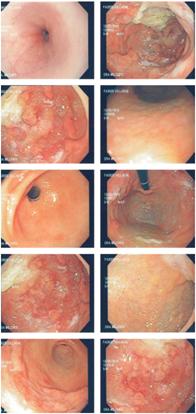
Figure 3 Hypertrophic gastric folds, with pseudopolyps and friability, and follicular antral mucosa. Thick, viscous gastric discharge.
Based on these findings, Ménétrier’s disease was suspected, and antibodies were requested for CMV even though the patient’s test for CMV IgG and IgM were negative and his CMV viral load was also negative (77 DNA/mL).
The patient evolved favorably after diuretic treatment, replacement with intravenous albumin, antisecretory management and nutritional management. Biopsy results showed hypertrophic gastropathy compatible with Ménétrier’s disease, 60 eosinophils per field and H. pylori negative (Figure 4). Colonic mucosal biopsies showed non-specific chronic inflammation.
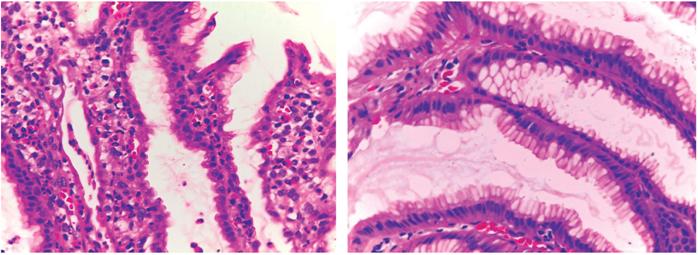
Figure 4 Architecture altered by marked elongation and dilatation of foveolar cells coated by the hyperplastic cylindrical epithelium with monomorphic nuclear and polymorphonuclear inflammatory infiltrate, neutrophils and eosinophils (100x and 200x).
After 9 days of hospitalization, he was discharged with prescriptions for outpatient management with esomeprazole and nutritional recommendations. A follow-up one month later found him to be completely asymptomatic.
Continued antisecretory treatment and a follow-up endoscopy were prescribed but the patient did not return for the follow-up examination.
Discussion
Ménétrier’s disease is an acquired gastric disorder characterized by giant hyperplastic folds, excess mucus secretion and hypoproteinemia due to the selective loss of serum proteins through the gastric mucosa. The clinical course of the disease in adults is insidious and progressive and is associated with malignancy, but in children, the disorder is characterized by abrupt onset and spontaneous resolution in two to ten weeks with only supportive treatment. 3
Although its cause is unknown, the literature has described possible associations with chemical irritants, toxins, dietary factors, neuro-emotional factors, endocrinological factors, allergic processes or immunological alterations.
The association with infectious processes has been strongly related to CMV (up to 70% of cases), 4,5,6 but there are also descriptions of association with H. pylori 7, Herpes simplex, Giardia intestinalis, Mycoplasma pneumoniae, and others. 3
Recent research suggests that the pathophysiology may be explained by the stimulus produced by triggers on the epidermal growth factor receptor (EGFR), a transmembrane receptor with tyrosine kinase activity. This in turn stimulates the transforming growth factor alpha (TGF-α) signaling pathway which eventually generates overgrowth of mucosal epithelial cells, especially in the gastric corpus and fundus. 6,7,8
Infantile Ménétrier’s disease usually affects children under six years of age and usually has an acute onset. The initial symptoms are nonspecific and are similar to many viral pictures with fatigue, nausea, vomiting and abdominal pain. When hypoalbuminemia occurs, peripheral edema occurs and subsequently ascites, pleural effusion and even pericardial effusion. Patients usually present to the emergency service with anasarca, as was evident in the two cases presented here. 6 Analytically, hypoproteinemia and hypoalbuminemia are characteristic and make it necessary to rule out renal and hepatic involvement. 9
A diagnosis of Ménétrier’s disease is made by an endoscopic study in which thickened folds are observed in the gastric fundus and corpus, and the gastric mucosa is slightly nodular with small areas of ulcerated or necrotic tissue. In addition, an increase in thick, viscous, transparent gastric secretions can be seen. 10 During upper endoscopy, samples must be taken to correlate with the histopathological study. The findings are similar to those of adult Ménétrier’s disease and are characterized by hypertrophy of gastric folds and foveolar hyperplasia, thickening of the mucosa due to proliferation, elongation and cystic dilation of gastric glands, hypersecretion, edema and inflammatory infiltrate. 11,12
Associations between Ménétrier’s disease and acute CMV infections are complex. Evidence of a relationship with CMV infection is established by a finding of infected cells in the gastric mucosa by PCR, detection of viral antigens by immunohistochemistry, viral cultures of the gastric mucosa or by viewing typical viral inclusions. The cytomegalovirus (CMV) antigenemia test, PCR of blood, and blood tests are less sensitive for detection of gastrointestinal involvement, but they can detect the infection and establish diagnostic suspicion. In-situ hybridization and immunohistochemistry have been proposed as the most sensitive laboratory techniques, but there are cases in which the presence of the virus has not been identified. The problem is that CMV infections may be a primary infection, a reactivation or a reinfection. Viral excretion in the urine and positive IgM serology can be maintained for months, so an isolated finding does not confirm that a CMV infection is the cause of Ménétrier’s disease. 2
Because of the benign, self-limiting course of Ménétrier’s disease in children, it has been determined that only antiviral treatment with ganciclovir should be given to those patients in whom the presence of CMV can be demonstrated in the gastric mucosa when they do not demonstrate clinical and paraclinical improvement with support measures that include hydration, infusion of albumin and diuretics, and associated antisecretory and dietary measures. 13 Experimentally, monoclonal antibodies against epidermal growth factor receptor (EGFR) have been tested in adults whose cases are refractory. The literature eve mentions an atypical case refractory and in a child for which octreotide was successfully used. 14
Other causes of gastropathy and protein-losing enteropathies to be taken into account in the differential diagnosis are celiac disease, gastric lymphoma, eosinophilic gastroenteritis, hypertrophic gastropathy associated with H. pylori and Crohn’s disease.
Eosinophilic gastroenteritis may affect the gastric antrum and small intestine, but there is often a family history of allergic disease. Gastric lymphoma is very rare in children and is not related to edema and hypoproteinemia. Crohn’s disease and Peutz-Jeghers syndrome have different signs and symptoms. Other rare diseases that have radiological signs similar to those of Ménétrier’s disease are Zollinger-Ellison syndrome, hypertrophic secretory gastritis, gastric varices and intestinal lymphangiectasia. 15
Conclusions
We have presented two typical clinical cases of childhood Ménétrier’s disease. These patients arrived at our health center with behavior and clinical courses similar to that described in the world literature, so we can conclude that Ménétrier’s disease should be suspected if a child has edema and hypoalbuminemia without renal involvement or hepatic or cardiac dysfunction and that is associated with hypertrophy of gastric folds and foveolar hyperplasia.
REFERENCES
1. Hillman M, Meinarde L. Gastroenteropatía perdedora de proteínas y posible relación con una infección por citomegalovirus. Presentación de un caso de enfermedad de Ménétrier infantil. Arch Argent Pediatr. 2013;111(5):446-9. doi: 10.5546/aap.2013.e121. [ Links ]
2. Fernández Caamaño B, Ramos Boluda E, Martínez-Ojinaga Nodal E, Molina Arias M, Sarría Osés J, Prieto Bozano G. Enfermedad de Ménétrier asociada a infección por citomegalovirus. An Pediatr (Barc). 2015;82(1):e113-6. doi: 10.1016/j.anpedi.2014.02.019. [ Links ]
3. Son KH, Kwak JJ, Park JO. A case of cytomegalovirus-negative Ménétrier’s disease with eosinophilia in a child. Korean J Pediatr. 2012;55(8):293-6. doi: 10.3345/kjp.2012.55.8.293. [ Links ]
4. Volonaki E, Haliotis F, Van Vliet-constantinidou C, Avlonitis S, Roma E. Cytomegalovirus infection in a child with Menetrier’s disease: a case report. Ann Gastroenterol. 2007;20(3):223-5. [ Links ]
5. Canan O, Ozçay F, Bilezikçi B. Ménétrier’s disease and severe gastric ulcers associated with cytomegalovirus infection in an immunocompetent child: a case report. Turk J Pediatr. 2008;50(3):291-5. [ Links ]
6. De Vivero-Camacho R, Muñoz-Álvarez N, Redondo-Bermúdez C, Alvis-Guzmán N, Tovar-Correa LE. Enfermedad de menetrier asociada a infección por citomegalovirus. Rev Cienc Biomed. 2015;6(2):354-9. [ Links ]
7. Tokuhara D, Okano Y, Asou K, Tamamori A, Yamano T. Cytomegalovirus and Helicobacter pylori co-infection in a child with Ménétrier disease. Eur J Pediatr. 2007;166(1):63-5. doi: 10.1007/s00431-006-0183-3. [ Links ]
8. Sánchez TL, González Hernández OJ. El receptor del factor de crecimiento epidérmico y su papel en el desarrollo tumoral. Rev Haban Cienc Méd. 2010;9(2):172-80. [ Links ]
9. Gómez Santos E, Balboa Vega MJ, Díaz Torrado N, Mora Navarro D, Pérez Serralvo C. Edemas generalizados en infección por citomegalovirus. Vox Paediatrica 2012;19(1):50-2. [ Links ]
10. Kirberg B A, Rodríguez V B, Donoso V F, Kirhman T M, Noriel V M. Gastropatía hipertrófica perdedora de proteínas. enfermedad de Ménétrier. caso clínico. Rev Chil Pediatr. 2014;85(1):80-5. doi: 10.4067/S0370-41062014000100011. [ Links ]
11. Williamson K, Park HK, Schacht R, Kaistha A. A case of Ménétrier disease in a child. Pediatr Emerg Care. 2012;28(3):277-9. doi: 10.1097/PEC.0b013e31824957b8. [ Links ]
12. Chung M, Pittenger J, Flomenhoft D, Bennett J, Lee EY, Shashidhar H. Atypical Clinical and Diagnostic Features in Ménétrier’s Disease in a Child. Case Rep Gastrointest Med. 2011;2011:480610. doi: 10.1155/2011/480610. [ Links ]
13. Alfares FA, Raddaoui EM, El-Mouzan MI, Alangari AA. Menetrier`s disease in a Saudi child. Saudi Med J. 2013;34(11):1192-4. [ Links ]
14. Di Nardo G, Oliva S, Aloi M, Ferrari F, Frediani S, Marcheggiano A, et al. A pediatric non-protein losing Menetrier’s disease successfully treated with octreotide long acting release. World J Gastroenterol. 2012;18(21):2727-9. doi: 10.3748/wjg.v18.i21.2727. [ Links ]
15. Velmishi V, Elezi B, Cekodhima G. Menetrier disease in a five year old girl - a case report and review of literature. J Gastrointest Dig Syst. 2016;6(2):2-4. doi: 10.4172/2161-069X.1000395. [ Links ]
academic meetingThese cases were presented at an of the Department of Pediatrics at the University of Antioquia in Medellín on July 26, 2016
Received: September 28, 2017; Accepted: November 23, 2017











 text in
text in