Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista colombiana de Gastroenterología
Print version ISSN 0120-9957
Rev Col Gastroenterol vol.33 no.4 Bogotá Oct./Dec. 2018
https://doi.org/10.22516/25007440.312
Original articles
Comparison of Flower Petal Stenting, a new prototype of covered biliary stent, with conventional biliary stenting for prevention of pancreatitis and post-stent cholecystitis
1 Cirugía Gastrointestinal y Endoscopia. Profesor titular Grupo de Gastrohepatología U de A. Instituto de Cancerología, Clínica las Américas. Medellín, Colombia
2 Radiólogo, Internista, Gastroenterólogo. Profesor Universidad de Texas, Campo Regional San Antonio. Director Gastroenterología VA Texas Valley Coastal Bend. Harlingen. Texas, EE.UU
3 Professor Department of Gastroenterology, Sungkyunkwan University School of Medicine. Samsung Medical Center, Seoul, Korea Office
4 Profesor de Radiología. UT Health San AntonioTexas, Estados Unidos
5 Epidemiólogo, Grupo Alergología clínica y experimental. Universidad de Antioquia. Medellín, Colombia.
Background and objectives:
Pancreatitis and cholecystitis are major complications following stenting with covered metallic stents to manage malignant distal biliary obstructions. We compare the use of a conventional covered biliary stent with a new prototype of a covered biliary stent with a flower-pedal structure that uses with low axial force and radial force in order to evaluate impacts on cholecystitis and pancreatitis after stenting.
Methods:
This is a non-randomized prospective comparison of a group of 18 patients stented with a flower-like biliary stent (FS group) with a group of 36 patients who received conventional biliary stents (CS group) ratio for management of malignant distal biliary obstructions. Demographic data and data about technical and clinical success including the appearance of cholecystitis and/or pancreatitis, migration rates and/or occlusion were evaluated.
Results:
The 18 FS group patients included 7 men (39%) and had a median age of 72 years. The 36 CS group patients included 16 men (44%) and had a median age of 71 years. There were three cases of cholecystitis and four cases of pancreatitis, all of which were in the CS group. Multivariate analysis showed statistically significant differences between the two groups in four clinical areas: hemoglobin, length of stenosis, etiology and occlusion of stents. No significant differences were found in survival time functions (Log Rank, p = 0.399). In Cox’s proportional hazards model, the only significant difference with respect to survival times was found on the issue of whether or not metastasis was present.
Conclusions:
The use of the new prototype flower-like biliary stent with lateral grooves and lower axial force and radial force is related to a lower rate of complications such as cholecystitis and pancreatitis than the use of conventional stents for palliation of distal malignant biliary stenoses. The new stent has no detriments in terms of permeability or migration and did not affect the survival of these patients.
Keywords: Metallic biliary stent; biliary obstruction; cholecystitis; pancreatitis
Antecedentes y objetivos:
la pancreatitis y la colecistitis son complicaciones mayores después de la postura de un stent metálico cubierto en el manejo de la obstrucción biliar distal maligna. Se pretende comparar el uso de un stent biliar cubierto convencional con un nuevo prototipo de stent biliar cubierto de morfología en flor con baja fuerza axial (FA) y fuerza radial (FR), y evaluar el impacto de las cifras de colecistitis y pancreatitis poststent.
Métodos:
se realizó un estudio prospectivo no aleatorizado en el que se comparó un grupo de 18 pacientes con stent biliar tipo flor (SF) con un grupo de pacientes que recibieron un stent biliar convencional (SC) con una relación 1:2, para el manejo de la obstrucción biliar distal maligna. Se evaluaron datos demográficos; el éxito técnico y clínico; así como la aparición de colecistitis y/o pancreatitis, tasas de migración y/u oclusión.
Resultados:
se evaluaron 18 pacientes en el grupo con SF (7 hombres, 39 %) con una edad mediana de 72 años y 36 pacientes (16 hombres, 44 %) en el grupo de SC con una edad mediana de 71 años. Se presentaron 3 casos de colecistitis y 4 casos de pancreatitis, todos ellos en el grupo de SC. Al explorar la asociación de los aspectos clínicos con respecto al tipo de stent, el análisis multivariado mostró diferencias estadísticamente significativas en 4 aspectos: la hemoglobina, la longitud de estenosis, la etiología y la oclusión del stent. Al evaluar los tiempos de supervivencia entre los pacientes con SC y SF, no se hallaron diferencias significativas en las funciones de supervivencia (log rank, p = 0,399). En el modelo de riesgos proporcionales de Cox, solo un aspecto mostró diferencias significativas frente a los tiempos de supervivencia: la presencia de metástasis.
Conclusiones:
el uso de un nuevo prototipo de stent SF (con surcos laterales) con menor FA y FR se relaciona con un menor índice de complicaciones como colecistitis y pancreatitis en comparación con el SC, en la paliación de las estenosis biliares malignas distales sin detrimento de su permeabilidad o migración y sin afectar la supervivencia de estos pacientes.
Palabras clave: Stent biliar metálico; obstrucción biliar; colecistitis; pancreatitis
Introduction
In endoscopic retrograde cholangiopancreatography (ERCP) to manage malignant biliary obstructions (MBOs), metal stents (MSs) have greater patency than do plastic stents. 1,2,3 It has also been suggested that they are more cost-effective although several complications other than stent occlusion may be associated with MSs and may affect patient quality of life of patients who have poor prognoses. 4 Covered stents prevent tumor growth inside the stent but may have a higher migration rate than other stents. 5 In addition, covered biliary stents can occlude the drainage orifice of the cystic or pancreatic duct and induce cholecystitis or pancreatitis. 6,7,8,9 Although there are conflicting data, two recent metaanalyses that compared covered and open stents found that cholecystitis is related more to tumor invasion of the cystic duct and to axial and radial expansive strengths of the stent than to the use of covered stents. 9,11
MSs have been grouped into covered stents, partially covered stents and uncovered stents, but several types of MS whose mechanical properties differ have had differing clinical outcomes that were unrelated to whether or not they were covered or uncovered. Two mechanical properties of stents are their axial force (AF) and the radial force (RF). AF, the force of recovery that straightens the stent out after it has been released, is related to the formability of the bile duct. RF is the force of expansion that guarantees the patency of the stent against the pressure of the stricture of the bile duct. 12,13 An MS with high AF tends to remain straight within the bile duct, compresses the bile duct, and can lead to angulations. 14 Previous studies have shown that MSs with high AFs are associated with high rates of pancreatitis probably due to compression of the pancreatic orifice by the pressure of the stent. 15
Various studies have shown that either a tumor invasion of a the cystic duct or the use of a covered stent can induce cholecystitis, but these studies have not shown how to prevent cholecystitis.7,8,16 The incidence of cholecystitis with the use of covered MSs to manage distal MBOs is 4.2% to 11.5%.11,17 Although sepsis of pancreatic origin has not been described, pancreatitis has been reported in 9.3% of cases in which covered stents were used to manage MBOs. These complications have created a need for a new type of endoscopic or surgical intervention which can delay or replace treatments such as chemotherapy or radiotherapy and improve patient quality of life.
Cholecystitis after placement of a biliary stent may occur due to bacterial colonization of the bile after endoscopic manipulation or because the gallbladder ceases to empty completely because of obstruction of the cystic duct by tumor invasion. This can allow the gallbladder to fill with non-sterile contrast medium. Cholecystitis that develops at the time of stenting can be associated with occlusion of the cystic duct due to the expansive force of the covered metal stent, a situation in which AF of the stent has a high degree of interference. Three studies have shown associations of cystic ducts compromised by tumors with the onset of cholecystitis but have not demonstrated measures to prevent its occurrence.7,8,16
An experimental study in a pig model has had promising results with the new morphologically different stent called the flower pedal stent (FPS). With the use of the new stent, the bile flow bile was greater, histological inflammatory changes were less frequent and less severe, and C-reactive protein (CRP) levels were lower. 18
None of the stents currently available are ideal for use in the bile duct, but the objective of an ideal stent would be to achieve maximum complication-free patency over time once the stent has been placed. From the mechanical point of view, an ideal biliary stent should provide low AF and high RF and have anti-migration systems. Many complications associated with stents could be resolved in the future with use of the ideal stent.
The new stent with flower petal morphology is intended to prevent complications such as cholecystitis and pancreatitis that are inherent in the use of covered MSs. Flower pedal stents (FPS) have circular morphology and lower AF and RF than do conventional stents (CSs). This article reports the development of the new stent and the results of a multicenter prospective follow-up study conducted to evaluate the technical feasibility and safety of FPSs by comparing their use to management with conventional covered MSs and conventional stents (CSs).
Materials and methods
Experimental Phase
Covered metal stents have the risk of obstructing the cystic duct or the main pancreatic duct due to their high RF and coverage which result in greater possibilities of cholecystitis and pancreatitis. A covered, flower-shaped, self-expanding metal stent resembles a 5-petal flower with lateral grooves. It has been designed to avoid occlusion of cystic ducts and was initially evaluated in porcine models of around 30 kilos.
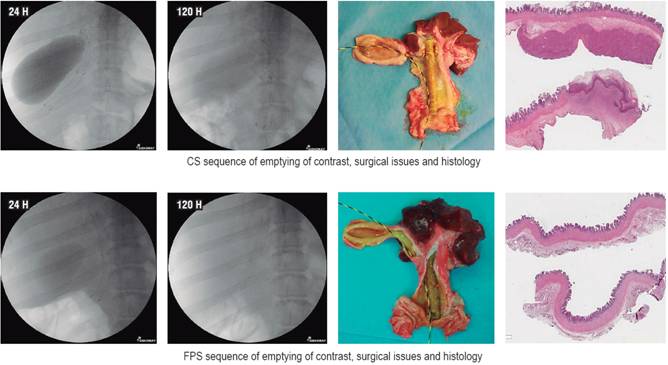
In Korea, Dr. Young Sik Woo 18 randomly placed FPSs or CS sin 14 pigs (7 per group). Stents were placed so as to occlude drainage through the cystic duct thus hindering drainage of the gallbladder. Gallbladder drainage was measured at 24, 48, 120 and 168 hours after implantation. Blood tests were taken on the second and seventh days after stent placement. The animals survived the seven days set as the deadline for euthanasia and corresponding histological evaluation (Figure 1).
After the administration of 25 mL of contrast into the gallbladder, stents were successfully placed in the required position in all cases, and there were no complications with the interventions. At 48 hours, the gallbladder’s drainage rate was higher in the group with the FPS than in the group with CS without a significant difference (71.4% versus 28.6%, p = 0.28). There was one early stent migration (within 7 days) in each group. The CSs were associated with higher CRP levels indicating greater inflammation (35.2 μg vs. 20.5 μg/dL, p = 0.03).
During laparotomies, the livers, gallbladders, bile ducts, and duodenums were removed en bloc. The gallbladders were opened longitudinally and samples were taken for histological study. There were no signs of infections or peritonitis in the hepatic, duodenal, bile duct or gallbladder tissue at the time of laparotomy. The histological study was reviewed blindly using a scoring scale that evaluates inflammation (1 = minimal, 2 = confined to the mucosa, 3 = compromised muscle), severity of neutrophil infiltrate (0 = absent, 1 = mild, 2 = moderate and 3 = severe) and the presence of ulcers on the mucosa (0 = absent, 1 = focal and 2 = diffuse).
At the time of euthanasia, three pigs in the CS group had distended gallbladders with smelly bilious material and with mild to moderate inflammatory histological changes. The inflammation scores and overall scores including neutrophil infiltrate and mucosal ulcers were significantly higher in the CS group than in the FPS group (p = 0.04 and p = 0.03).
Based on these preliminary experimental results, the clinical study described below was undertaken.
Study Population
Information on patients who had MBOs between March 2015 and February 2016 were collected in a prospective database. Patients were included if they had obstructive jaundice, an MBO demonstrated by abdominal computerized axial tomography (CAT) and/or magnetic resonance imaging, radiological appearance of MBO during ERCP, and her or his gallbladder was in situ, he or she was not a candidate for surgical resection due to tumor status or surgical risks, and he or she was over 18 years of age and had signed informed consent designed for this project. The exclusion criteria were any contraindication for performing ERCP; presence of a metal stent, prior surgery including cholecystectomy and percutaneous drainage procedures; hepatitis or other liver disease that causes jaundice; and suspicion of gallbladder cancer.
Information entered into the database included demographic data, comorbidities, tumor status, histories of chemotherapy and/or radiation therapy, details of ERCP, type and number of stents used and adverse events as recommended by the guidelines of the American Association of Endoscopy (ASGE). 19
Types of Stents Used
The new FPS is 10 mm in diameter by 80 cm in length, covered by polytetrafluoroethylene, has side grooves at each end, and has low AF and RF. It was compared with a Boston Scientific CS of the same diameter and length but which has high AF and RF. Both stents are mounted in 8 Fr. introducer mechanisms. The FPS is made of nitinol and shaped like a five petal flower at each end. Parallel grooves on the long axis at each end are designed specifically to prevent occlusion of the cystic or pancreatic duct and to facilitate drainage of the gallbladder and pancreatic duct (Figure 2).
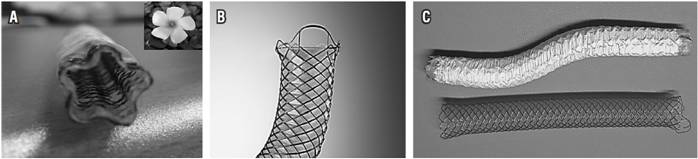
Figure 2 Types of covered stents used in the study. A. Covered pentagonal FPS with parallel grooves along its major axis. B. Standard cylindrical, fully covered metal stent (WALLSTENT™ Endoprosthesis from Boston Scientific) C. The FPS above has greater formability than the CS below. The FPS preserves its S shape and has lower RF and AF than does the CS.
In addition, FPSs have high levels of formability, defined as the ability of the stent to fully adapt to the anatomy of the region where it is released independent of any irregularity of the area due to a tumor. This means that the flower petal stent does not immediately return to its initial shape after it has been deformed along its axial axis.
AF and RF of the Stents Used
The metal stents used in this study are classified according to their AF and RF. AF is defined as the force necessary to return the stent to its original shape after being bent. Two issues related to RF are evaluated: the strength necessary to contract the stent and, the most clinically relevant, the strength of expansion of the stent once it has been collapsed. Measurements of the axial forces of FPS and CS showed that the AF of CSs was four times that of the FPSs (Figure 3).
Study Design
This is an open label study with subjects grouped by age with one FPS subject for every two CS subjects. It was carried out in two health care institutions in Medellín, Colombia. Once a patient met the inclusion criteria and the exclusion criteria were ruled out for that patient, informed consent was obtained and an FPS was placed. Control group data were obtained from the prospective databases of the institutions.
Findings from patients with malignant biliary stenoses who received the FPSs were compared with a control matched by age (± 2 years) who had also been diagnosed with a malignant biliary stenosis but had undergone ERCP and placement of a CS.
Endoscopic Technique
All procedures were performed using standardized protocols designed by endoscopists with more than ten years of experience in biliary endoscopy. The patients received broad-spectrum prophylactic antibiotics, standard technique was used to cannulate the bile duct, and contrast was injected to identify the position and length of the stenosis. Routine biliary sphincterotomies were was not performed in any of these patients. Fluoroscopy aided determination of stenosis length, tumor location, whether or not the tumor had compromised the cystic duct, and the degree to which the gallbladder had filled with contrast.
The newly designed flower pedal stent was placed and then released by pulling the introducer mechanism. The length of the stent was determined according to the length of the stenosis by positioning the center of the stent in the center of the stenosis and locating the duodenal end of the stent 10 to 15 mm outside of the ampulla.
Prophylactic pancreatic stents were not required for any of these patients, nor were prophylactic medications administered for the onset of acute pancreatitis.
Definition of Events and Follow-Up
To confirm whether biliary drainage was successful, liver function tests were performed a week prior to stenting and were repeated on the second, fifth and seventh days after placement with follow-ups at 30 days and monthly follow-ups thereafter. Patients were interviewed monthly after liver function tests either in person or by telephone. Patients who missed follow-ups were analyzed based on intention to treat and data were censored at the time of their last contact for follow-up.
Technical success was defined as successful insertion and placement of a stent based on radiological confirmation during ERCP. Clinical success was defined as more than 50% decrease of the patient’s bilirubin level within the first seven days after stent placement. All adverse effects related to procedures were recorded. Early adverse effects were defined as those that occurred in the first 30 days after stent placement, and late adverse effects were defined as those that occurred after this period. Stent dysfunction was considered to have occurred in patients with obstructive jaundice and dilation of the biliary tract with or without cholangitis confirmed by imaging (ultrasound, CT scans or MRI). Stent dysfunction motivated a second ERCP to evaluate and correct the cause of the dysfunction. If recurrent jaundice was accompanied by fever, an urgent ERCP was performed even when images did not show biliary dilation. Occlusion of the stent can occur due to tumor growth over or through the stent. Proximal or distal migration of the stent is defined as displacement of the stent above or below the MBO after its release.
Measurement of Outcomes
The primary results evaluated were cholecystitis and/or pancreatitis. After the release of the stent, cholecystitis was defined as the presence of upper abdominal pain accompanied by sensitivity in the upper right quadrant (Murphy’s sign) and by clinical examination or ultrasound findings of thickening of the gallbladder wall greater than 3 mm with or without perivesicular fluid collection.
Pancreatitis was defined on the basis of standard criteria as new or worsened abdominal pain after stent placement with amylase or lipase levels three times normal, with or without radiographic evidence of pancreatitis. 20 Severity was determined to be mild if the hospital stay lasted no more than three days longer than planned, moderate if it lasted from four to ten days, and severe, when the hospital stay was longer than 10 days or the patient was moved to intensive care or had to undergo surgery.
Secondary analyses included those for technical and clinical success documented by laboratory results, causes of stent dysfunction that warranted reoperation, adverse effects and survival. Survival was measured from the time of stenting until the date of death or end of follow-up.
Statistical Analysis
Absolute and relative distributions and summary indicators such as quartiles and interquartile range were used for descriptive analysis of sociodemographic and clinical information. Normality of continuous variables was established by means of the Shapiro-Wilk test and, based on those results, the U-Mann Whitney test was used to differentiate between the two types of stents.
Pearson’s chi-squared test (χ2) of independence or Fisher’s exact test was used establish relationships between clinical information and the type of stent. Similarly, the strengths of associations were assessed by means of relative risk (RR) and 95% confidence intervals (95% CI). Multivariate regression was used as an exploratory method to evaluate the influence of sociodemographic and clinical characteristics according to the type of stent. For this purpose, a generalized linear model (GML) of Poisson regression with robust variance to adjust the RR and its 95% CI for multiple variables was used.
The Kaplan-Meier method was used to evaluate overall patient survival and survival according to the duration of stent patency. This established the probability of a patient-s death within a determined amount of time from the moment of the stent placement. The event of interest for this analysis was death (a non-censorship condition). To determine whether there were differences in survival times related to sociodemographic and clinical characteristics, the logrank test, hazard ratio (HR), 95% CI were used. They were adjusted by means of an exploratory multivariate model of Cox proportional hazards. Values of p <0.05 were considered statistically significant. All statistical analyses were performed with the SPSS version 21.
Results
The stents were successfully released and crossed the implantation orifice of the cystic ducts in all 54 patients without evidence of immediate major complications. The basic characteristics of both groups were very similar. In 30 days the following stent placement, follow-up laboratory tests were performed on day 7 and day 30. Monthly laboratory follow-up tests continued to be conducted thereafter. There were no loss of follow-up in any of the groups. Causes of MBO were pancreatic cancer (n = 37), cholangiocarcinoma (n = 14) and periampullary tumors (n = 2). In total, 28 patients received adjuvant therapy after stent insertion: 10 (55.6%) in the FPS group and 18 in the CS group (50%) (Table 1).
Table 1 Basic characteristics of CS and FPS groups of patients
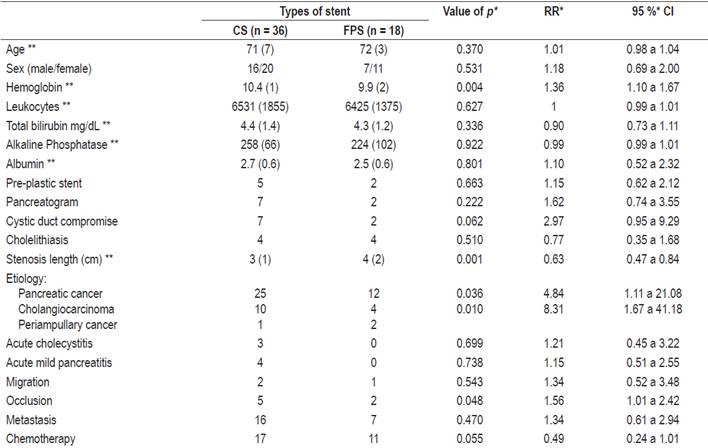
* Adjusted by the Poisson regression model (GLM). ** The values are medians of interquartile ranges.
Evaluation of sociodemographic and clinical factors with respect to the type of stent found statistically significant differences of hemoglobin levels (lowest in FPS), length of stenosis (longer in FPS), etiology (more cholangiocarcinoma in CS) and stent occlusion (less in FPS) (Table 1).
Acute Cholecystitis
Acute cholecystitis occurred in three patients in the CS group with at 6, 8 and 16 days after stent placement (mean: 10 days). Cholecystitis improved with conservative management with parenteral antibiotics in one patient, but two required cholecystostomy with radiological support. No cholecystectomies were performed in any of the patients. Characteristically, the three patients who presented acute cholecystitis had tumors which compromised the tops of the cystic duct implantation. Contrast injection in the gallbladder was not associated with onset of cholecystitis. Also, gall stones were not associated with greater probability of cholecystitis in these patients.
Acute Pancreatitis
Acute pancreatitis developed in four of the 36 patients (11.1%) with CS. Onset occurred in the first 24 hours after stent placement. All cases of pancreatitis were mild according to the consensus criteria cited. Conservative management without stent removal led to improvement of all patients. The average fasting time of these patients was five days (2 to 8 days). Two of the patients with pancreatitis had histories of cannulation and/or contrast injections into the pancreatic duct. The tumors in three out of the four patients whose cystic ducts had been invaded were extrapancreatic (cholangiocarcinoma).
Laboratory Results
Laboratory results for hemoglobin, leukocytes, total bilirubin, alkaline phosphatase and albumin prior to stent placement were compared, and only hemoglobin was found to have a significant difference. Patients with FPS had 0.5 g/dL less hemoglobin than did CS patients.
Other Complications
The stent migration rates were similar for both groups, but occlusion the occlusion occurrence rate was greater for the CS group, and this difference was statistically significant (p = 0.048, RR 1.56, 95% CI 1.01 to 2.42).
Survival Analysis
Time elapsed from the date of stent placement until the date of a patient’s death was considered here, and the event of interest, death due to basic cause related to malignant biliary pathology, was a non-censorship condition.
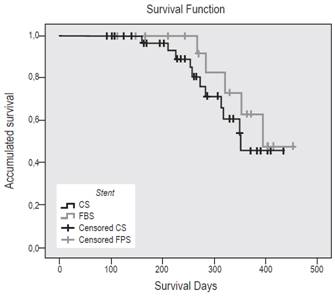
When survival times of the CS and FPS groups were compared. no significant differences were found in survival functions (log rank, p = 0.399). The median survival time of the FPS patients was 395 days with a probability of death of 0.471. The median survival time of the CS was 351 days with a probability of death of 0.460 (Figure 4).
Cox Proportional Hazards Analysis
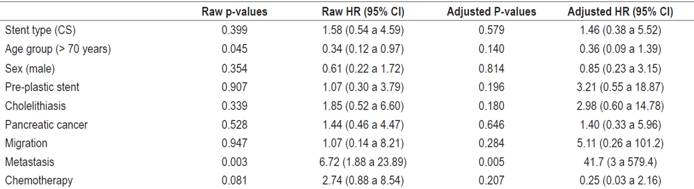
Based on logrank test results and the criteria, knowledge and experience of the researchers, information about the following variables was entered into the model: type of stent, age group, sex, previous plastic stents, cholelithiasis, pancreatic cancer, migration, metastases and chemotherapy. The model is adequate according to the maximum likelihood method (p = 0.017). Both the overall evaluation of the proportional hazards assumption and the evaluation of each covariate within the model using the Schoenfeld residuals are presented in Table 4.
According to the final model, only one characteristic, the presence of metastasis, demonstrated significant differences with respect to survival times. The risk of death for the group of patients with metastasis was 41.7 times the risk of death for patients without metastasis at all times and adjusted for the other variables (Table 2).
The overall evaluation of the proportional hazards assumptions (p = 0.1722) and of each model variable (p> 0.05) fulfills the assumption according to the Schoenfeld residuals.
Discussion
In general, endoscopic drainage with a covered MS is accepted as the first line of palliative therapy for a patient with an unresectable distal MBO. It is not very invasive and is superior to plastic stents due to the greater duration of the patency of MSs. However, covered MSs are associated with various complications that affect a patient’s quality of life which is already compromised by an underlying tumor. Acute pancreatitis and cholecystitis are two complications associated with MS management of biliary stenosis. Rates of pancreatitis associated with MS range between 1% and 8% and rates of cholecystitis range between 6% and 12%.8,15,20 In our study, these rates are adjusted to those reported in the literature.
Prevention of these complications is as important as long-term patency of the stent. This multicenter study and a prospective follow-up database reveals that compromise of the cystic duct by tumors and the use of stents with high AF (shown in other studies with shorter stents) are associated with increased risk of cholecystitis and pancreatitis.
Two retrospective studies suggest that the association of tumor invasion of the cystic duct and Wirsung’s duct with cholecystitis and pancreatitis is due to orifice stenosis or local obstruction with loss of elasticity due to the presence of a tumor and compression by the covered stent that displaces the tumor causing difficulty for the passage of bile flow from the gallbladder. 8,16 In contrast, in cases in which the cystic duct is not invaded by a tumor commitment, the elasticity of the duct allows for space for passage of bile from the gallbladder. Theoretically, a bare stent is less likely to result in cholecystitis. However, the incidences of cholecystitis and pancreatitis with bare stents do not differ significantly for those with covered stents. Therefore, AF rather than the stent covering is more likely to produce compression of a cystic ducts invaded by and prevent passage of bile or pancreatic juice with the risk of cholecystitis or pancreatitis. 11
Mechanical properties of stents are thought to affect clinical prognosis, and stents’ two most relevant mechanical properties are RF and AF. 12 Studies show that stents with high AF are associated with cholecystitis and pancreatitis. 15 A high RF has not been related to cholecystitis but high AF has. Stents with high AF can cause deviation of the opening of the cystic duct due to compression. In contrast, radial expansion is uniform along the axis of the bile duct (Figure 5). 11
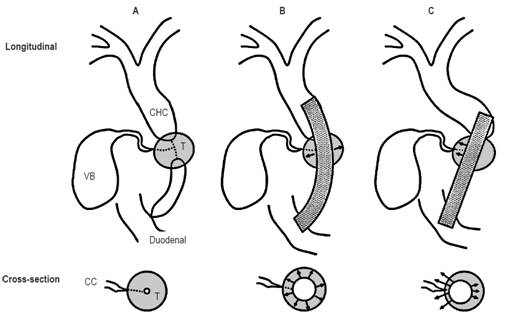
Figure 5 Longitudinal and transverse views of the effects of metal stent AF released in patients with cystic ducts compromised by tumors. 11 A. Before stent placement. B. After placement of stent with low AF. C. After placement of stent with high AF. CC: cystic duct; CHC: common hepatic duct; T: tumor; VB: gallbladder. Modified from: Nakai Y et al. J Gastroenterol Hepatol. 2014;29(7):1557-62.
Shimizu has investigated associations of AF and RF with the onset of pancreatitis and/or cholecystitis and did not find a direct relationship. 16 It is possible that the incidence of cholecystitis (n = 4) or pancreatitis (n = 8) in that study population (n = 72) was too small to evaluate the differences in the characteristics of covered stents since the four patients who suffered from cholecystitis did receive covered stents with high AF.
Although the length of the stent is selected according to the extent of stenosis, the longest stents used in this study were 10 cm in length. The advantage of using longer stents to reduce related complications has been reported to prevent cholecystitis and occlusion due to tumor overgrowth. Shorter stents have been associated with higher rates of cholecystitis. 12,21 If a short stent is placed, the AF will be greater because the distance between the stenosis and the end of the stent is shorter. Long stents have been reported to have advantages for reducing complications, 21 and longer stents can prevent the onset of cholecystitis by preventing cystic occlusion due to tumor overgrowth. 11
The diameter of the stent can also affect the incidence of cholecystitis. Stents with smaller diameters result in less cholecystitis. A cholecystitis rate of 2.9% and a 0% malignant obstruction rate has been described for 8 mm stents while the same rates were 7% and 18% in patients with 10 mm stents. 11 In our study, all stents were 10 mm in diameter.
Although our study suggests that covered stents with low AF are associated with low incidences of cholecystitis and pancreatitis, the main variable to be evaluated is the permeability of the stents. A covered stent which has low AF better accommodates the curvature of the common bile duct without compromising the permeability of the stent. Although analysis of stent patency is outside the final objective of this study, the mean time for reappearance of an obstruction was greater for stents with low AF than for stents with high AF (p = 0.048; RR: 1.56; 95% CI: 1.01 to 2.42). Stents with low AF can reduce complications such as cholecystitis and pancreatitis while maintaining greater patency.
One study has shown that cholelithiasis is a risk factor for cholecystitis, 8 but this was not reproduced in our study. This difference could be explained by the high prevalences in that study (18.1%) and this study (11.1%).
Empirical procedures such as transcystic washing of the gallbladder have been tried as ways to reduce the incidence of cholecystitis. 8 In addition, there are factors that predispose to sepsis such as inadequate disinfection of equipment, injection of contaminated contrast and incomplete drainage of the obstruction all of which can lead to cholecystitis. 22 In practice, another study has documented infection with pseudomonas after ERCP. 23 For this reason, prophylactic antibiotics should be administered to all patients so that there is no specific risk of infection. 24 A course of antibiotics, unlike a single prophylactic dose, may be more effective for the prevention of cholecystitis if patients have risk factors for it.
Percutaneous cholecystostomy has traditionally been the treatment of choice for patients with acute cholecystitis after a metal stent has migrated due to malignancy. 8,25,26,27 In this study three patients in the CS group developed cholecystitis. Two required percutaneous cholecystostomies with radiological support, and the other resolved spontaneously. Another management alternative, not used in this study, is to replace a covered metal stent with an uncovered metal stent or a plastic stent. 28
Percutaneous insertion of a biliary stent has been related to less possibility of developing cholecystitis. This has been attributed to the fact that there is no contamination of the bile duct which is particularly important in the stents that cross the papilla. 7
This study has several limitations. Since stent placement was not randomized, selection biases may be present. The number of patients in both groups is small. Also, clinical outcomes of the use of stents can vary according to properties such as types of covering material (silicone, polyurethane or polytetrafluoroethylene), structure (braided or laser cut) and the type of wire (steel or nitinol). A prospective, randomized controlled study is required to confirm the hypothesis that stents with low RF can reduce the incidence of cholecystitis and/or pancreatitis.
Conclusions
This study confirms that the main cause of the genesis of pancreatitis and cholecystitis is compromise of cystic or Wirsung’s duct by tumors and that this is frequently related to the use of high AF stents which also showed low levels of long-term patency. A large prospective and randomized study is necessary to evaluate the implications of covered stents in the genesis of cholecystitis and/or pancreatitis.
Acknowledgements
We express our gratitude to Dr. J. Y. Song and Kang S & G Biotech Inc. of Seoul, Korea for providing flower petal stents free of charge for this study. This study was carried out with the support of the sustainability project of the Vice-Rector for Research of the University of Antioquia
REFERENCES
1. Castaño R, Álvarez O, García A, Quintero V, Sanín E, Erebrie F, et al. Stent metálico versus plástico en la obstrucción biliar maligna distal. Rev Col Gastroenterol. 2009;24(3):248-55. [ Links ]
2. Sawas T, Al Halabi S, Parsi MA, Vargo JJ. Self-expandable metal stents versus plastic stents for malignant biliary obstruction: a meta-analysis. Gastrointest Endosc. 2015;82(2):256-267.e7. doi: 10.1016/j.gie.2015.03.1980. [ Links ]
3. Hong WD, Chen XW, Wu WZ, Zhu QH, Chen XR. Metal versus plastic stents for malignant biliary obstruction: an update meta-analysis. Clin Res Hepatol Gastroenterol. 2013;37(5):496-500. doi: 10.1016/j.clinre.2012.12.002. [ Links ]
4. Barkun AN, Adam V, Martel M, AlNaamani K, Moses PL. Partially covered self-expandable metal stents versus polyethylene stents for malignant biliary obstruction: a cost-effectiveness analysis. Can J Gastroenterol Hepatol. 2015;29(7):377-83. [ Links ]
5. Lee JH, Krishna SG, Singh A, Ladha HS, Slack RS, Ramireddy S, et al. Comparison of the utility of covered metal stents versus uncovered metal stents in the management of malignant biliary strictures in 749 patients. Gastrointest Endosc. 2013;78(2):312-24. doi: 10.1016/j.gie.2013.02.032. [ Links ]
6. Fumex F, Coumaros D, Napoleon B, Barthet M, Laugier R, Yzet T, et al. Similar performance but higher cholecystitis rate with covered biliary stents: results from a prospective multicenter evaluation. Endoscopy. 2006;38(8):787-92. doi: 10.1055/s-2006-944515. [ Links ]
7. Isayama H, Kawabe T, Nakai Y, Tsujino T, Sasahira N, Yamamoto N, et al. Cholecystitis after metallic stent placement in patients with malignant distal biliary obstruction. Clin Gastroenterol Hepatol. 2006;4(9):1148-53. doi: 10.1016/j.cgh.2006.06.004. [ Links ]
8. Suk KT, Kim HS, Kim JW, Baik SK, Kwon SO, Kim HG, et al. Risk factors for cholecystitis after metal stent placement in malignant biliary obstruction. Gastrointest Endosc. 2006;64(4):522-9. doi: 10.1016/j.gie.2006.06.022. [ Links ]
9. Almadi MA, Barkun AN, Martel M. No benefit of covered vs uncovered self-expandable metal stents in patients with malignant distal biliary obstruction: a meta-analysis. Clin Gastroenterol Hepatol. 2013;11(1):27-37.e1. doi: 10.1016/j.cgh.2012.10.019. [ Links ]
10. Saleem A, Leggett CL, Murad MH, Baron TH. Meta-analysis of randomized trials comparing the patency of covered and uncovered self-expandable metal stents for palliation of distal malignant bile duct obstruction. Gastrointest Endosc. 2011;74(2):321-327.e1-3. doi: 10.1016/j.gie.2011.03.1249. [ Links ]
11. Nakai Y, Isayama H, Kawakubo K, Kogure H, Hamada T, Togawa O, et al. Metallic stent with high axial force as a risk factor for cholecystitis in distal malignant biliary obstruction. J Gastroenterol Hepatol. 2014;29(7):1557-62. doi: 10.1111/jgh.12582. [ Links ]
12. Isayama H, Nakai Y, Toyokawa Y, Togawa O, Gon C, Ito Y, et al. Measurement of radial and axial forces of biliary self-expandable metallic stents. Gastrointest Endosc. 2009;70(1):37-44. doi: 10.1016/j.gie.2008.09.032. [ Links ]
13. Isayama H, Nakai Y, Hamada T, Matsubara S, Kogure H, Koike K. Understanding the Mechanical forces of Self-Expandable Metal Stents in the Biliary Ducts. Curr Gastroenterol Rep. 2016;18(12):64. doi: 10.1007/s11894-016-0538-5. [ Links ]
14. Mukai T, Yasuda I, Isayama H, Nakashima M, Doi S, Iwashita T et al. Comparison of axial force and cell width of self-expandable metallic stents: which type of stent is better suited for hilar biliary strictures? J Hepatobiliary Pancreat Sci. 2011;18(5):646-52. doi: 10.1007/s00534-011-0406-5. [ Links ]
15. Kawakubo K, Isayama H, Nakai Y, Togawa O, Sasahira N, Kogure H, et al. Risk factors for pancreatitis following transpapillary self-expandable metal stent placement. Surg Endosc. 2012;26(3):771-6. doi: 10.1007/s00464-011-1950-4. [ Links ]
16. Shimizu S, Naitoh I, Nakazawa T, Hayashi K, Miyabe K, Kondo H, et al. Predictive factors for pancreatitis and cholecystitis in endoscopic covered metal stenting for distal malignant biliary obstruction. J Gastroenterol Hepatol. 2013;28(1):68-72. doi: 10.1111/j.1440-1746.2012.07283.x. [ Links ]
17. Nishida H, Inoue H, Ueno K, Nagata Y, Kato T, Miyazono N, et al. Cholecystoduodenal fistula: a complication of inserted self-expandable metallic biliary stents. Cardiovasc Intervent Radiol. 1998;21(3):251-3. [ Links ]
18. Woo YS, Lee KH, Lee JK, Noh DH, Park JK, Lee KT, et al. Novel flower-type covered metal stent to prevent cholecystitis: experimental study in a pig model. Surg Endosc. 2016;30(3):1141-5. doi: 10.1007/s00464-015-4317-4. [ Links ]
19. Cotton PB, Eisen G, Romagnuolo J, Vargo J, Baron T, Tarnasky P, et al. Grading the complexity of endoscopic procedures: results of an ASGE working party. Gastrointest Endosc. 2011;73(5):868-74. doi: 10.1016/j.gie.2010.12.036. [ Links ]
20. Coté GA, Kumar N, Ansstas M, Edmundowicz SA, Jonnalagadda S, Mullady DK, et al. Risk of post-ERCP pancreatitis with placement of self-expandable metallic stents. Gastrointest Endosc. 2010;72(4):748-54. doi: 10.1016/j.gie.2010.05.023. [ Links ]
21. Nakai Y, Isayama H, Togawa O, Kogure H, Tsujino T, Yagioka H, et al. New method of covered wallstents for distal malignant biliary obstruction to reduce early stent-related complications based on characteristics. Dig Endosc. 2011;23(1):49-55. doi: 10.1111/j.1443-1661.2010.01043.x. [ Links ]
22. Devière J, Motte S, Dumonceau JM, Serruys E, Thys JP, Cremer M. Septicemia after endoscopic retrograde cholangiopancreatography. Endoscopy. 1990;22(2):72-5. [ Links ]
23. Allen JI, Allen MO, Olson MM, Gerding DN, Shanholtzer CJ, Meier PB, et al. Pseudomonas infection of the biliary system resulting from use of a contaminated endoscope. Gastroenterology. 1987;92(3):759-63. [ Links ]
24. Bari K, Aslanian HR, Pollak J, Reiner E, Salem RR, Taddei TH, et al. Emphysematous Cholecystitis Resulting in Secondary Biliary Cirrhosis: A Rare Complication of Endoscopic Retrograde Cholangiopancreatography. ACG Case Rep J. 2013;1(1):51-4. doi: 10.14309/crj.2013.18. [ Links ]
25. Ainley CC, Williams SJ, Smith AC, Hatfield AR, Russell RC, Lees WR. Gallbladder sepsis after stent insertion for bile duct obstruction: management by percutaneous cholecystostomy. Br J Surg. 1991;78(8):961-3. [ Links ]
26. Vingan HL, Wohlgemuth SD, Bell JS 3rd. Percutaneous cholecystostomy drainage for the treatment of acute emphysematous cholecystitis. AJR Am J Roentgenol. 1990;155(5):1013-4. doi: 10.2214/ajr.155.5.2120927. [ Links ]
27. Dolan R, Pinkas H, Brady PG. Acute cholecystitis after palliative stenting for malignant obstruction of the biliary tree. Gastrointest Endosc. 1993;39(3):447-9. [ Links ]
28. Saxena P, Singh VK, Lennon AM, Okolo PI, Kalloo AN, Khashab MA. Endoscopic management of acute cholecystitis after metal stent placement in patients with malignant biliary obstruction: a case series. Gastrointest Endosc. 2013;78(1):175-8. doi: 10.1016/j.gie.2013.02.038. [ Links ]
Received: April 02, 2018; Accepted: November 07, 2018











 text in
text in