Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista colombiana de Gastroenterología
Print version ISSN 0120-9957
Rev Col Gastroenterol vol.35 no.1 Bogotá Jan./Mar. 2020
https://doi.org/10.22516/25007440.268
Original articles
Warthin-Starry stain identification of Helicobacter pylori in biopsies of patients who previously tested negative in hematoxylin-eosin staining for follicular gastritis
1Médico patólogo. Dinámica IPS. Medellín, Antioquia, Colombia
2Médica general. Corporación Universitaria Remington. Medellín, Antioquia, Colombia
3Médica general. Corporación Universitaria Remington. Medellín, Antioquia, Colombia
4Estudiante de medicina. Universidad Pontificia Bolivariana. Medellín, Antioquia, Colombia
5Microbiólogo y bioanalista, MSc. PhD. Grupo de Investigación en Neurociencias y Envejecimiento. Corporación Universitaria Remington. Medellín, Antioquia, Colombia
Non-invasive and invasive techniques can be used for detection of Helicobacter pylori. An invasive technique identifies the bacteria through routine hematoxylin-eosin staining. Warthin-Starry stain is rarely used.
Objective:
Our objective was to identify H. pylori by Warthin-Starry staining of patient’s biopsies with chronic follicular gastritis who had previously tested negative in hematoxylin-eosin staining.
Materials and methods:
This is a descriptive, cross-sectional descriptive study that was carried out over a period of 12 months. The study examined paraffin blocks of samples taken from the gastric mucosa of patients diagnosed with chronic gastritis and follicular hyperplasia. A histological section was extracted from a block and tested with hematoxylin-eosin staining for the presence or absence of H. pylori. If absent, an additional cut was taken from the same block and Warthin-Starry staining was used to retest for the presence of the bacteria.
Results:
Of the 314 samples collected, 209 tested negative, and 105 tested positive for H. pylori when hematoxylin-eosin staining was used. Of the 209 negative samples, 45% (94) tested positive when Warthin Starry stain was used, and 55% (115) still tested negative.
Conclusion:
Findings of H. pylori are significantly higher when Warthin Starry stain was used to test samples whose previous histological study had evidenced an absence of the bacillus, especially in samples with a small amount of bacteria.
Keywords: Chronic gastritis; follicular hyperplasia; Helicobacter pylori; Hematoxylin-Eosin; Warthin Starry
Las técnicas empleadas para la detección del Helicobacter pylori (H. pylori) son no invasivas e invasivas. En estas últimas, la presencia del H. pylori se determina a partir de la tinción de hematoxilina-eosina (HE), prueba rutinaria, mientras que en pocas ocasiones se aplica la tinción de Warthin-Starry (WS) como coloración especial.
Objetivo:
identificar la presencia de H. pylori por medio de la coloración especial de la WS en biopsias de pacientes con gastritis crónica folicular, previamente negativas en la tinción HE.
Materiales y métodos:
se desarrolló un estudio de tipo descriptivo transversal, en un período de 12 meses. Se tomaron los bloques de parafina de las muestras de la mucosa gástrica de pacientes con diagnóstico de gastritis crónica e hiperplasia folicular. Además, se extrajo un corte histológico del mismo bloque, al cual se le aplicó HE y se determinó la presencia o ausencia de H. pylori. Así, de estar ausente, se tomó del mismo bloque un corte adicional y se aplicó WS. Esto se evaluó con el fin de identificar la existencia o no del bacilo.
Resultados:
se recolectaron 314 muestras; 209 fueron negativas y 105 fueron positivas para HE. El 45 % (94) de estas muestras fueron positivas respecto a la presencia del bacilo, al aplicar la segunda coloración, y el 55 % (115) de las muestras persistieron negativas.
Conclusión:
el hallazgo de H. pylori es significativamente alto al aplicar la coloración de WS a muestras cuyo estudio histológico evidenció la ausencia del bacilo en biopsias de la mucosa gástrica, especialmente en muestras con escasa cantidad de bacterias.
Palabras claves: Gastritis crónica; hiperplasia folicular; Helicobacter pylori; hematoxilina-eosina; Warthin-Starry
Introduction
Peptic acid disease is one of the main reasons for medical consultation in Colombia: 80% of these cases occur due to gastritis defined as inflammation of the gastric mucosa due to an imbalance between protective and promoter factors of inflammation. 1,2 Helicobacter pylori (H. pylori) is one of the most important risk factors, especially for the pathogenesis of chronic gastritis with follicular hyperplasia. 3
This type of gastritis produces a strong inflammatory reaction characterized by a dense infiltrate of mononuclear cells, primarily monocytes, and the formation of aggregates or lymphoid follicles with a germinal center. 4 Recent studies suggest that persistent inflammation caused by this type of gastritis may increase the risk of developing low-grade mucosa-associated lymphoid tissue (MALT), a type of gastric neoplasia, and adenocarcinoma. 5
About 60% of the world’s population is expected to become infected with H. pylori which will make it the world’s most common chronic bacterial infection. 6 In Colombia, the prevalence of H. pylori infections in adults is high and is associated with unhealthy conditions and unhealthy lifestyles. 7 Person-to-person contact, including fecal-oral, oral-oral and gastric-oral, is considered to be the most common transmission route of this bacteria. 8
The most widely used method for diagnosing H. pylori gastritis is to stain a tissue sample and then examine it using optical microscopy. This involves heat dehydration and routine hematoxylin-eosin (HE) staining. Bacteria within the mucus and on the surface of the epithelium can be observed with HE staining, but those that adhere closely to neutrophil phagocytized cells are generally indistinguishable from adjacent cells. 9
HE’s high sensitivity for detecting H. pylori in biopsies of infected patients is offset by factors such as previous treatment of patients, small amounts of H. pylori in a sample, and technical conditions such as stain quality and observe expertise. Consequently, auxiliary stains such as Warthin-Starry (WS) stain are often used in diagnosis of chronic follicular gastritis. 10
WS has the ability to highlight bacteria hidden in gastric mucus due to the contrast of shades in its brownish-yellow background within which the bacilli stand out unlike in HE’s purple-pink coloration. 11
Timely diagnosis is important for starting triple antimicrobial drug regimens early to prevent complications related to the persistence of the bacillus. 12 This study’s purpose is to determine presence of H. pylori using WS staining of biopsies of patients with chronic follicular gastritis who had previously been diagnosed as H. pylori negative through HE staining.
Materials and methods
This cross-sectional descriptive study was conducted between August 2016 and July 2017. A convenience sample from biopsy samples from a referral medical center in Medellín was taken. Biopsies were included from male and female patients who had been diagnosed with chronic gastritis and follicular hyperplasia but which were negative for H. pylori using routine HE staining. Pathology samples that had been collected and/or stored incorrectly were excluded.
The pathology laboratory at the Medellín headquarters of Dinámica IPS took paraffin blocks from samples of gastric mucosa that had been obtained endoscopically. Histological sections extracted from each block underwent routine HE staining following the protocol of the laboratory of the United States Armed Forces. Commercially available Instant Hematoxylin from Thermo scientific®) was used.
After HE staining, the sample was examined by optical microscopy to determine whether H. pylori was present. If absent, an additional slice was taken from the same block and stained with WS according to the protocol of the United States Armed Forces. 11
Warthin-Starry Staining Procedure
Paraffin is removed from the tissue.
Two flotation baths are prepared: one at 43 ºC for impregnation and another at 54 ºC for revelation.
One percent silver nitrate is added to the impregnation bath. When hot, slides are placed in the bath and left for 30 minutes.
Silver nitrate is placed in the revelation bath for 30 minutes together with 5% gelatin, 0.15% hydroquinone or pyrocatechol, and distilled water.
After 30 min, 1.5 mL of 2% silver nitrate; 3.75 mL of gelatin and 2 mL of pyrocatechol were mixed.
Next, the slides are removed from the 1% silver nitrate impregnation bath, placed on the grid and the solution is applied until a yellow or golden brown color is obtained.
Slides are washed with hot distilled water and dehydrated with heat.
Slides are evaluated with optical microscopy to determine whether H. pylori is present.
Data from pathology reports were recorded only from cases of patients whose HE stains were negative for H. Pylori. Data extracted for analysis was used to build a Microsoft Excel database. Variables included whether WS stains were positive for H. Pylori, the amount of H. pylori found by WS staining, inflammation, degree inflammation, inflammatory activity, and hyperplasia. Demographic characteristics such as age and gender obtained from patients’ admission clinical records were also included.
Descriptive measurements including relative and absolute frequency as well as measurements of central tendency and dispersion were considered in the statistical analysis which was done with IBM SPSS for Windows version 24.0.
Results
A total of 314 samples were analyzed. Of these, 105 HE stains were positive for H. pylori, and 209 were negative. Only samples that were negative were included: those that were positive were excluded. Of the 209 negative samples, WS staining found that 94 (45%) were positive for H. Pylori, and 115 (55%) samples remained negative (Figure 1).
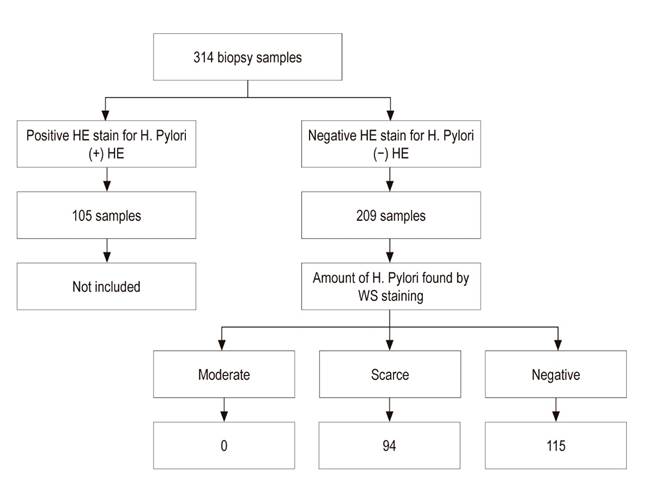
Figure 1 Distribution of samples analyzed by HE and WS. Of the 314 samples from the gastric mucosa, HE staining found 105 to be positive and 209 to be negative. HE: hematoxylin-eosin.
WS staining detected very small amounts of the bacillus in biopsies that had previously tested negative with HE (Figure 2). Of the 209 samples in the study, 140 (66.9%) were female and 69 (33%) were male. Sixty-two of the 140 samples from female patients were positive and 78 were negative with WS staining, while of the 69 samples from male patients, 32 were positive and 37 were negative with WS.
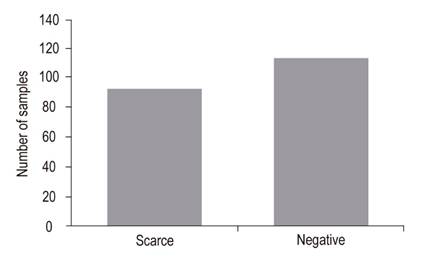
Figure 2 Amount of H. pylori in WS staining. WS detected very small scarce amounts of the bacillus in biopsies that had previously tested negative with HE. WS: Warthin-Starry staining
The average age for female patients with chronic follicular gastritis and H. pylori was 50 years, while for male patients it was 42. In the 209 samples that tested negative for H. Pylori with HE, patients ages ranged from 1 to 90 years. In the most frequently affected age group, between 41 and 60 years, 46 patients were positive for H. pylori and 47 were negative with WS.
The least affected population, between 81 and 100 years, had only one case that tested positive for H. pylori with WS. The 0-20 year old group had 8 positive and 21 negative cases with WS (Figure 3).
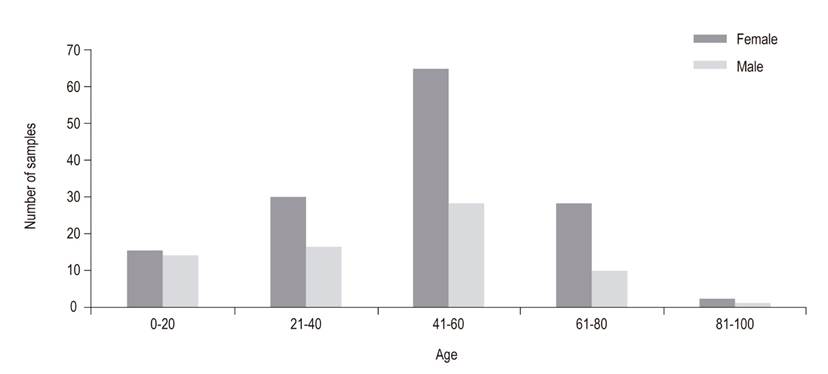
Figure 3 Age groups affected by sex. The average age for female patients with chronic follicular gastritis and H. pylori was 50 years, while for male patients it was 42. The most frequently affected age group was between 41 and 60 years.
We found that 94 (100%) of patients who tested positive for H. Pylori with WS had very small amounts of H. pylori, 79 (84%) had moderate inflammation, 9 (9.6%) had severe inflammation and 6 (6.4%) had mild inflammation. Among those who tested negative with WS, 91 had moderate inflammation, 14 had severe inflammation, and 10 had mild inflammation (Figure 4).
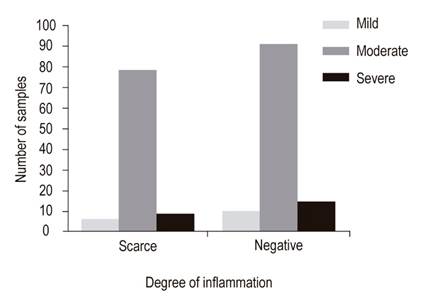
Figure 4 Relationship of degree of inflammation with amount of H. pylori found by WS staining. Of the total of 94 samples that were positive with WS, 79 (84%) presented a moderate degree of inflammation, 9 (9.6%) showed severe inflammation and 6 (6.4%) had mild inflammation.
Eighty four (91%) of the WS positive samples had mild inflammatory activity, compared to 107 of those who tested negative (Figure 5).
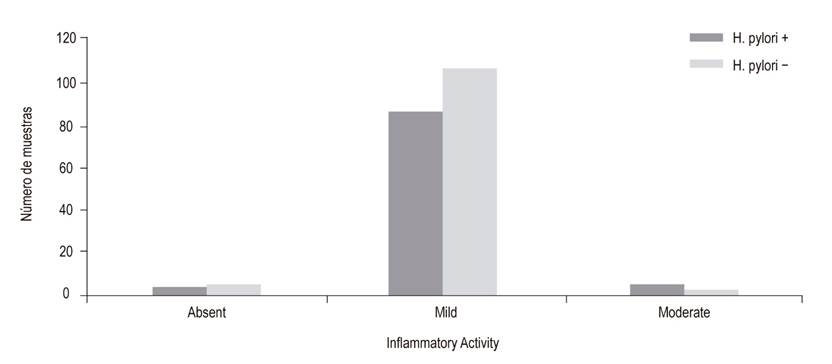
Figure 5 H. pylori in WS staining according to inflammatory activity. 84 (91%) of WS positive samples were identified as having mild inflammatory activity, compared to 107 of the WS negative samples.
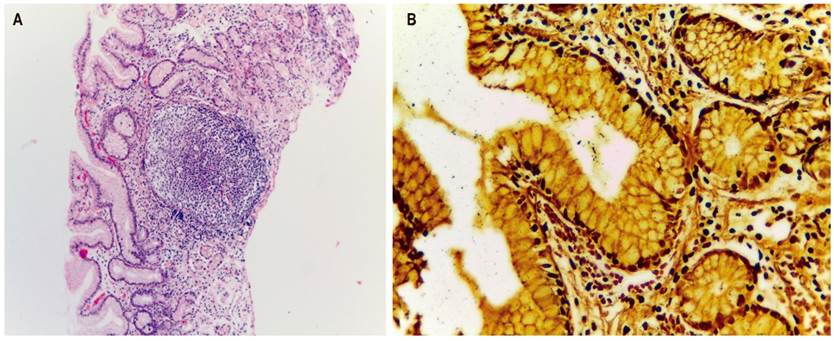
A comparison of the two staining methods shows marked differences. Lymphoid aggregates with germinal centers are clearly present in HE stained slides, but it is not possible to clearly identify H. pylori (Figure 6A). In contrast, on WS stained slides even a small number of bacilli H. pylori bacilli stand out (Figure 6B).
Discussion
This study compares two histological staining methods to determine the diagnostic value of WS staining for identification of H. pylori in gastric biopsies of patients with chronic follicular gastritis who had previously tested negative with HE staining.
WS is a special stain used for histological detection of H. pylori and spirochetes in gastric tissue samples. 13 Its main component is silver nitrate which precipitates onto the microorganism’s membrane making it look thicker so that it is easier to identify with an optical microscope. 14 WS staining must be done by an expert in staining since it requires great care especially since excessive silver precipitation and excessive discoloration of samples can alter the result of the microscope reading. 15
WS stain adheres better to bacilli in the presence of mild inflammatory activity than in the presence of moderate inflammatory activity. When there is moderate inflammatory activity, inflammatory cell infiltrates overlap with the bacillus and decrease WS stain’s capacity to binding with this bacillus. Most of the histological diagnoses reported in this study had mild inflammatory activity.
For this reason, WS staining is ideal for detecting the presence of the bacillus. Similar to the findings of Velásquez et al., our study found that WS has the ability to identify very small amounts of the bacteria. In contrast, HE staining only identifies large amounts of bacteria on histological slides which is why routine HE staining often generates false negatives. 15,16
Our results confirm that special WS staining detects the presence of H. pylori more frequently in cases of chronic follicular gastritis than does HE staining, as has been documented in other studies. 13-17
Similarly, our study concludes that WS is more efficient than HE for confirming the presence of the bacterium in follicular gastritis, so it should be considered for inclusion in diagnostic protocols in local pathology laboratories as the initial staining method. Since WS staining practice is currently not used by the majority of pathologists, the bacillus is often not identified initially.
WS costs more than HE and requires specialized technical personnel. In addition, preparation of the reagents requires more time since they are not commercially available in our environment. Nevertheless, WS staining would make diagnosis of H. pylori more cost-effective because early diagnosis and treatment help prevent sequelae of this bacterium, especially gastric cancer. 18
REFERENCES
1. Correa S, Cardona AF, Correa T, Correa LA, García HI, Estrada S. Prevalencia de Helicobacter pylori y características histopatológicas en biopsias gástricas de pacientes con síntomas dispépticos en un centro de referencia de Medellín. Rev Col Gastroenterol. 2016;31(1):9-15. https://doi.org/10.22516/25007440.67 [ Links ]
2. Buriticá VJ, Becerra LF, Salazar A. Enfermedad ácido péptica. Arch Med Univ Manizales Fac Med. 2005;11:46-55. https://doi.org/10.30554/archmed.11.0.1534.2005 [ Links ]
3. Makola D, Peura DA, Crowe SE. Helicobacter pylori infection and related gastrointestinal diseases. J Clin Gastroenterol. 2007;41(6):548-58. http://dx.doi.org/10.1097/MCG.0b013e318030e3c3 [ Links ]
4. Garg B, Sandhu V, Sood N, Sood A, Malhotra V. Histopathological analysis of chronic gastritis and correlation of pathological features with each other and with endoscopic findings. Pol J Pathol. 2012;63(3):172-8. https://doi.org/10.5114/pjp.2012.31501 [ Links ]
5. Ramírez A, Sánchez R. Helicobacter Pylori y Cáncer Gástrico. Rev Gastroenterol Perú. 2008;28:258-266. [ Links ]
6. Martínez JD, Riveros SC. Hiperplasia linfoide folicular gástrica e infección por Helicobacter pylori en adultos colombianos. Rev Colomb Gastroenterol 2009;24(2):148-156. [ Links ]
7. Camargo MC, Yepez MC, Cerón C, Guerrero N, Bravo LE, Correa P, et al. Age at acquisition of Helicobacter pylori infection: comparison of two areas with contrasting risk of gastric cancer. Helicobacter. 2004;9(3):262-70. https://doi.org/10.1111/j.1083-4389.2004.00221.x [ Links ]
8. Alba RS, Toledo RA, Viana ML. Helicobacter pylori: Clínica, diagnóstico y tratamiento. Rev Posgrado VIa Cátedra Med. 2006;158:9-12. [ Links ]
9. Marín R, Salas P, Mena F, Sierra R. Identificación histológica de Helicobacter pylori por los métodos de tinción de Warthins-Stary y Giménez en biopsias gástricas. Rev Med Costa Rica Centroam. 1996;53(537):147-151. [ Links ]
10. Fallone CA, Loo VG, Lough J, Barkun AN. Hematoxylin and eosin staining of gastric tissue for the detection of Helicobacter pylori. Helicobacter . 1997;2(1):32-5. https://doi.org/10.1111/j.1523-5378.1997.tb00054.x [ Links ]
11. Prophet EB, Mills B, Arrington J, Sobón L. Métodos histotecnológicos. Washington: Registro de Patología de los Estados Unidos de América (ARP) e Instituto de Patología de las Fuerzas Armadas de los Estados Unidos de América (AFIP);1995. [ Links ]
12. Campuzano G. Helicobacter Pylori: de la gastritis al cáncer gástrico. Medellín: Editora Médica Colombiana S.A., 8ª edición; 2017. [ Links ]
13. Kit de plateado según Warthin-Starry modificado para la detección de Helicobacter pylori y Spirochaetes en cortes parafínicos. Burlington: EMD Millipore Corporation; 2019. [ Links ]
14. Giugni MC, Benmelej A, Graciani G, Gómez JM, Roldán JN, Costamagna A, et al. Comparación de coloraciones histológicas para el diagnóstico de Helicobacter Pylori. Rev FABICIB. 1998;2(1):151-155. https://doi.org/10.14409/fabicib.v2i1.611 [ Links ]
15. Joachín ID, Mendoza AG, Salazar JL. Valor diagnóstico de la tinción de Warthin Starry para la identificación de H. pylori en biopsias gástricas. Guatemala: Universidad de San Carlos de Guatemala, Facultad de Ciencias Químicas y Farmacia; 2016. [ Links ]
16. Amores J, Arredondo A, Martínez B, Estrada Y, Pereira L, Potente A. Correlación histologicamicrobiológica en el diagnóstico de Helicobacter pylori. Rev Mex Patol Clin. 2010;57(3):135-142. [ Links ]
17. Ortiz-Martínez MA, Salazar-Valdez OR, Brito-Zurita OR, Abundis-Castro L, García-Bajeca C, Gutiérrez-López SJ, et al. Detección de Helicobacter pylori en niños con los métodos de Gram, Giemsa y Warthing-Starry, inicialmente negativos con otras técnicas histológicas. Rev Gastroenterol Mex. 2005;70(2):143-145. [ Links ]
18. Vela-Velásquez CT. Comparación entre las biopsias gástricas sin fijar 24 horas frente a la biopsia convencional para el diagnóstico de Helicobacter pylori en un hospital de referencia de Perú. Rev Perú Med Exp Salud Pública. 2011;28(1):42-46. https://doi.org/10.17843/rpmesp.2011.281.454 [ Links ]
Received: July 26, 2018; Accepted: December 20, 2019











 text in
text in