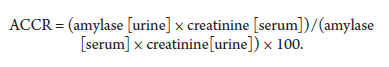Introduction
Macroamylasemia diagnosis is challenging when it is associated with abdominal pain, as it can lead to a false diagnosis of acute pancreatitis. Acute pancreatitis is characterized by pancreatitis-like abdominal pain, imaging findings of pancreatitis, and a three-fold increase in the plasma upper normal limit of the catalytic concentration of α-amylase and lipase enzymes1-4.
However, said increase may also be related to pancreatic and extrapancreatic metabolic diseases and pancreatic duct anomalies5. Non-pancreatic hyperamylasemia is mainly caused by salivary gland diseases (mumps, lithiasis), intestinal diseases (bowerl infarction, celiac disease, ulcerative colitis and peritonitis), chronic liver diseases, neoplasms, diabetic ketoacidosis, renal failure, ectopic pregnancy, polycystic ovary syndrome, head injury, and macroamylasemia6-8. The existence of macroamylase was first described by Wilding in 19646. The term was proposed by Berk in 1967 and published in several case reports9.
Macroamylasemia is a rare benign condition characterized by the presence of hyperamylasemia or elevated serum α-amylase levels without elevation of α-amylase levels in urine, provided that kidney function is preserved10. Macroamylasemia occurs in 0.1%-1.5% of the general population (nonalcoholic); amylase isoforms are encoded in chromosome 1p2111. It occurs more frequently in males, but the reasons of this predominance are unknown. Macroamylasemia is a condition in which serum amylase protein binds to other serum proteins such as immunoglobulins A (IgA) (up to 92%) and G (IgG) (<30%), among others (<5%), forming large macromolecule complexes that exceed 400 KDa (amylase in its active cytoplasmic form contains 496 amino acids with a molecular weight of 54 KDa)11-13.
Macroamylasemia can be diagnosed indirectly by measuring amylase activity in urine; specifically, the amylase to creatinine clearance ratio (ACCR) can contribute to differentiating hyperamylasemia. This ratio is calculated using the following equation:
An ACCR >5% is suggestive of acute pancreatitis.
An increased ACCR may also occur in diabetic ketoacidosis, surgery, and renal disease. An ACCR <1% is suggestive of macroamylasemia11. Chromatography and polyethylene glycol (PEG) precipitation are the methods of choice for detecting macromolecules that contain macroamylase6,11.
Clinical case presentation
This is the case of a 53-year-old women with a history of high blood pressure (treated with losartan 50 mg every 12 hours) and cholecystectomy who was admitted due acute pancreatic abdominal pain. Epigastric pain on palpation was evidenced on physical examination. The following laboratory and imaging studies were performed: amylase: 1407 U/L; ultrasound: normal pancreas, 7.4 mm common bile duct, and mild left hydronephrosis. The patient was diagnosed with mild acute pancreatitis, for which she was hospitalized. Five days after hospital admission, a control amylase test was performed (1419 U/L; associated with absence of pain). She was then discharged and referred to the outpatient department to conduct further tests aimed at determining the causes of hyperamylasemia. Table 1 summarizes the results of these tests on hospital admission and at 5 days, 3 months, 8 months, 10 months and 1 year in the outpatient service.
Table 1 Evolution of laboratory test results.
| On hospital admission | At 5 days | At 3 months | At 8 months | At 10 months | At 1 year | |
|---|---|---|---|---|---|---|
| Amylase α (IU/L) | 1407 | 1419 | 2445 | 2454 | 2495 | 1174 |
| Lipase (IU/L) | 32 | --- | ||||
| Creatinine (mg/dL) | 0.48 | --- | 0.54 | --- | --- | --- |
| Urea (mg/dL) | 23 | --- | 22 | --- | --- | --- |
| Creatinine clearance (mL/min) | 120 | --- | 107 | --- | --- | --- |
| Glucose (mg/dL) | 106 | --- | 88 | 90 | --- | --- |
| AST/GOT (IU/L) | 17 | --- | 18 | 22 | --- | --- |
| ALT/GPT (IU/L) | 13 | --- | 10 | 27 | --- | --- |
| Albumin (g/dL) | 4.50 | --- | 4.54 | 4.43 | --- | --- |
| Total protein (g/dL) | 7.32 | --- | 7.30 | 7.45 | --- | --- |
| Globulin (g/dL) | 2.82 | --- | 2.76 | 3.02 | --- | --- |
| Alkaline phosphatase | 200 | --- | 205 | 244 | --- | --- |
| Gamma-glutamyl transpeptidase | 40 | --- | 14 | --- | --- | |
| Total bilirubin (mg/dL) | 0.8 | --- | 0.57 | 0.53 | --- | --- |
| Triglycerides | 156 | --- | 66 | --- | --- | |
| Hemoglobin (g/dL) | 12.8 | --- | 12.9 | --- | --- | |
| Platelets mL/mm3 | 350.000 | --- | 380.000 | --- | --- | |
| TSH (mIU/dL) | --- | --- | --- | 3.1 | --- | --- |
| Free T4 (ng/dL) | --- | --- | --- | 1.83 | --- | --- |
| ANA | --- | --- | --- | --- | Negative | --- |
| VRDL | --- | --- | --- | --- | Negative | --- |
| Rheumatoid factor | --- | --- | --- | --- | 28.18 | --- |
| Total hepatitis B core antibody | --- | --- | --- | --- | Non-reactive | --- |
| Hepatitis B, Australia antigen | --- | --- | --- | --- | Non-reactive | --- |
| Hepatitis C Virus | --- | --- | --- | --- | Non-reactive | --- |
| Elisa for HIV | --- | --- | --- | --- | Non-reactive | --- |
| AMA | --- | --- | --- | Negative | --- |
Source: Sistema de Atención Historia Clínica Hospital Lazarte Echegaray - Essalud (Medical Record Health Care System Hospital Lazarte Echegaray - Essalud). GOT: glutamic-oxalacetic transaminase (NV: 0-35 IU); GPT: glutamic-pyruvic transaminase (NV: 0-35 IU); TB: total bilirubin (NV: 0.3-1.2 mg/dL); DB: direct bilirubin (NV: 0-0.3 mg/dL); TP: total proteins (NV: 6-8 g/dL); ALB: albumin (NV: 3.5-5.5 g/dL); ALP: alkaline phosphatase (36-129 IU); GGT: gamma-glutamyl transpeptidase (NV: 8-78 IU); serum amylase (NV:0-100IU/L); T4 (NV: 0.8 to 1.9 ng/dL); TSH (NV: 0. 5-4.5 mU/dL); platelets (NV: 150-450 ml/mm3); hemoglobin (NV: 12-14 g/dL); serum creatinine (NV: 0.4 to 1.4 mg/dL); VRDL: The Viral and Rickettsial Disease Laboratory; NV: normal values.
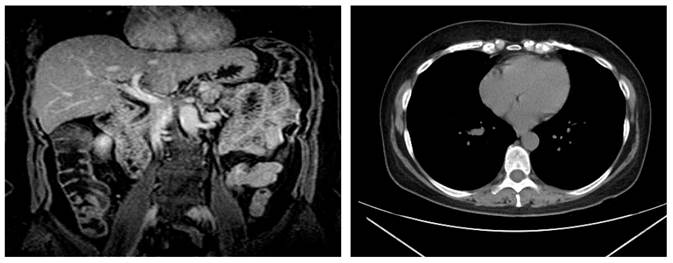
During the patient’s follow-up, laboratory tests were performed to rule out associated diseases that could cause hyperamylasemia (Table 1). She also underwent an endoscopy in which a duodenal biopsy was performed, which in turn showed glandular architecture associated with chronic nonspecific infiltrate, intraepithelial lymphocyte count <40/100 epithelial cells, without atrophy. Besides, during colonoscopy diverticula were observed in the sigmoid colon. Regarding imaging studies, no lymphadenopathies were evidenced on a CT of the neck (soft tissue), and evidence of cancer was observed in the CT scan of the chest, abdomen and pelvis (Figure 1); likewise, a normal biliary tract and no signs of choledocholithiasis were evidenced during magnetic resonance cholangiopancreatography (Figure 2). Further tests included ACCR (0.02%) and a confirmation test for macroamylasemia, which was positive (Table 2). The patient was asymptomatic during her evaluation at the outpatient department.
Table 2 Macroamylasemia study
| Result | |
|---|---|
| Amylase clearance | 0.02 |
| Amylase in urine | 19 IU/L |
| Serum amylase | 1174 IU/L |
| Creatinine | 0.5 mg/dL |
| Urine creatinine (24-hour urine collection) | 43.9 mg/dL |
| Macroamylasemia* | Positive |
*Chromatography; precipitation with polyethylene glycol (PEG). Source: Sistema de Atención Historia Clínica Hospital Lazarte Echegaray - Essalud (Medical Record Health Care System Hospital Lazarte Echegaray - Essalud). Amylase clearance (NV: <5); amylase in urine (NV: <460 U/L); serum amylase (NV: 0-100 U/L); serum creatinine (NV: 0.4-1.4 mg/dL).
Discussion
It should be noted that our patient was a woman, as it has been described macroamylasemia occurs more frequently in males11. In this case, the patient’s abdominal pain on admission was associated with hyperamylasemia, which led to a wrong diagnosis of acute pancreatitis; 6% to 9.6% cases of hyperamylasemia are caused by macroamylasemia14,15. The highlight of our case is that no abdominal pain was experienced by the patient during the course of hyperamylasemia, so further testing was required to rule out other pancreatic and extrapancreatic causes, as well as associations with other diseases. Celiac disease is the condition most frequently associated with macroamylasemia development; in this regard, Rabsztyn et al.16, in a case-control study, concluded that a significant proportion of patients recently diagnosed with celiac disease have macroamylasemia; therefore, if a patient has macroamylasemia, celiac disease must be also considered. In our case, 6 biopsy samples were taken by means of an endoscopy, and in which a MARSH 0 score was reported, without criteria to carry out more tests to confirm or rule out the presence of celiac disease. Other macroamylasemia-associated diseases include chronic liver diseases17, diabetes mellitus18, rheumatoid arthritis19, systemic lupus erythematosus20, T-cell lymphoma21, inflammatory bowel disease22, asymptomatic HIV23 and renal cell and thyroid carcinoma16. All these conditions were ruled out in our patient.
Levitt et al. proposed the existence of two forms of macroamylasemia24: the first one was characterized by the presence of malabsorption with an 11 S serum amylase resulting from the binding of normal-sized amylase by IgA immunoglobulin. In the second type, patients did not suffer from malabsorption, the sedimentation coefficient was 7 S and the complex was not the result of amylase binding to an immunoglobulin. Currently, 3 types of macroamylasemia have been described (Table 3)25.
Table 3 Types of macroamylasemia
| Typo | 1 | 2 | 3 |
|---|---|---|---|
| Serum amylase | Persistently elevated amylase levels | Increased | Normal |
| Amylase in urine Decreased | Decreased or Normal | Decreased or Normal | Normal |
| ACCR | Very decreased | Disminuido | Decreased |
| Macroamylase concentration | Relatively high | Lower than type 1 | Low |
| Time to result | Short | Long | Long |
Source: taken from25.
A very low ACCR (0.02%) and a short time to the positive result were observed in our patient; based on these findings, type 1 macroamylasemia was suspected, which was confirmed by means of chromatography and PEG precipitation. Thus, it can be concluded that this is a case of a type 1 macroamylasemia. Macroamylasemia must be considered in cases of persistent hyperamylasemia, even if the initial manifestation is abdominal pain, also ruling out other etiologies and associated diseases of importance.











 texto en
texto en