Clinical case
We present the case of a 64-year-old female patient with a history of gastric adenocarcinoma surgically managed outside the institution (total gastrectomy and esophagoenteral anastomosis). She exhibited fibrotic stenosis of the esophagojejunal anastomosis with secondary esophageal dysphagia for solids. Extrainstitutional clinical follow-up was performed by gastroenterology, indicating endoscopic dilatation protocol with CRE balloon under general anesthesia. The patient underwent an esophagoscopy on 02/06/2019, noting a fibrous esophagojejunal anastomosis with a 3 mm residual lumen at 36 cm from the dental arches. An 8-10 mm CRE balloon was dilated on fluoroscopic and endoscopic views, leaving an 8 mm residual lumen.
A second esophageal dilation is performed two weeks later, and stricture of the anastomosis is observed again. This time, the biopsy is taken on a stenotic area with a 4-mm esophageal lumen, and then CRE balloon dilation is performed to 8-10 mm, leaving a 9-mm residual lumen. The patient was scheduled for a third endoscopic dilation on 03/04/2019 with a sequential dilatation protocol with a CRE balloon to 11 mm (5 ATM) on a 9 mm stenotic esophageal lumen. Its diameter was increased to 10 mm, with evidence of mucosal tear expected at this level (Figure 1). After intervention and during endoscope extraction, a deep laceration with a 4 cm long perforation was identified on the middle esophagus towards the left posterolateral wall, with minor bleeding in the layer dependent on the laceration edges. The characteristics and location of the deep laceration (8 cm proximal to the recently dilated anastomosis, 28-32 cm from the dental arches) are striking, considering the easy non-traumatic entry of the gastroscope at the beginning of the procedure (Figure 2).
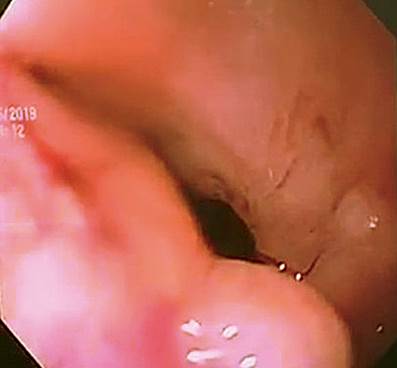
Figure 1 Fibrotic stenosis of the esophagojejunal anastomosis after CRE balloon dilation. A 10 mm residual lumen is observed after dilation in the presence of an expected mucosal tear.
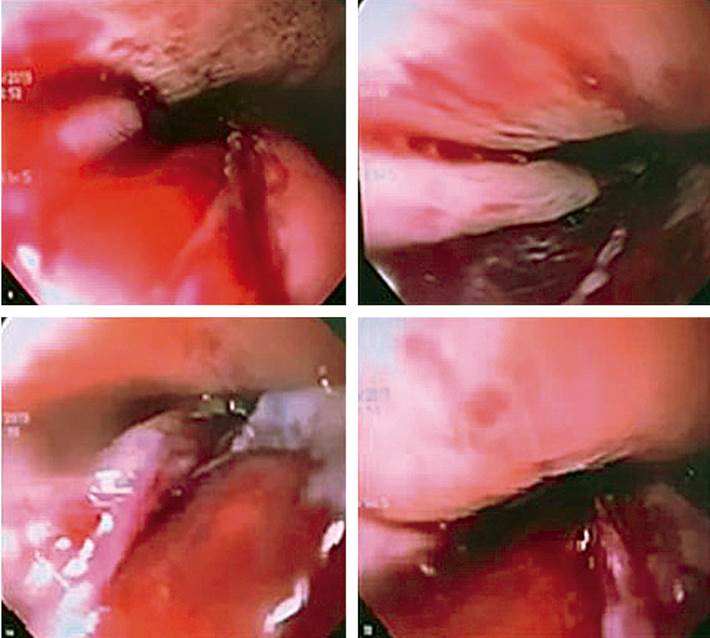
Figure 2 Spontaneous esophageal perforation is a 4-cm deep laceration, 8 cm above the newly dilated anastomosis.
Physical examination under anesthetic monitoring shows vital signs within normal limits, without hemodynamic instability or deterioration of ventilatory parameters. Supraclavicular subcutaneous emphysema with left predominance is regarded as positive. Findings related to a barogenic perforation of the esophagus are identified, considering the characteristics and location of the lesion and the chronology of events during the procedure. Thus, broad-spectrum empiric antimicrobial coverage is started immediately, and the gastrointestinal surgery service is called. Upon joint assessment, urgent transfer to the operating room for surgical and intraoperative endoscopic exploration is defined.
Thoracoscopy and endoscopic exploration are performed at the same surgical time. Through endoluminal transillumination with the gastroscope and on extraluminal view through the laparoscope, they detected a full-thickness esophageal perforation on the middle esophagus towards the left posterolateral wall, with 4 mm mucosal tear and 8 cm longitudinal muscle layer tear, and no evidence of contamination (Figure 3A).
After identifying the solution of continuity, a primary defect suture is performed with simple 2-0 PDS stitches, achieving adequate closure. Esophageal impermeability is verified at this level, an advanced feeding tube is passed under endoscopic guidance, and the left chest tube is inserted (Figures 3B and 3C). The patient is then transferred to the recovery room. During in-hospital surveillance, the patient evolved adequately, achieving pain control and a progressive decrease in analgesia, with scant serosanguineous drainage through the left chest tube. The control esophagogram seven days after the event found a small solution of continuity, which communicated with a contained saccular image of 45 x 9 mm proximal to the defect area, with no outflow of contrast into the thorax or the mediastinum (Figure 4A). A new control esophagogram seven days prior shows a small leak with a decrease in size compared to the first imaging control; so the chest tube is removed, enteral nutrition is performed through an advanced tube, and oral administration is started with adequate tolerance. Hospital discharge will continue with outpatient follow-up (Figure 4B).
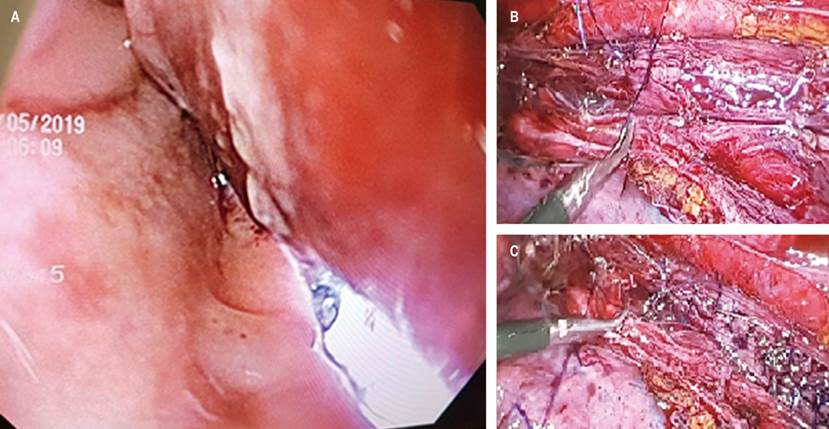
Figure 3 A. Endoscopic view of the esophageal perforation with external transillumination through the laparoscope. B and C. Thoracoscopy esophagography with simple stitches.
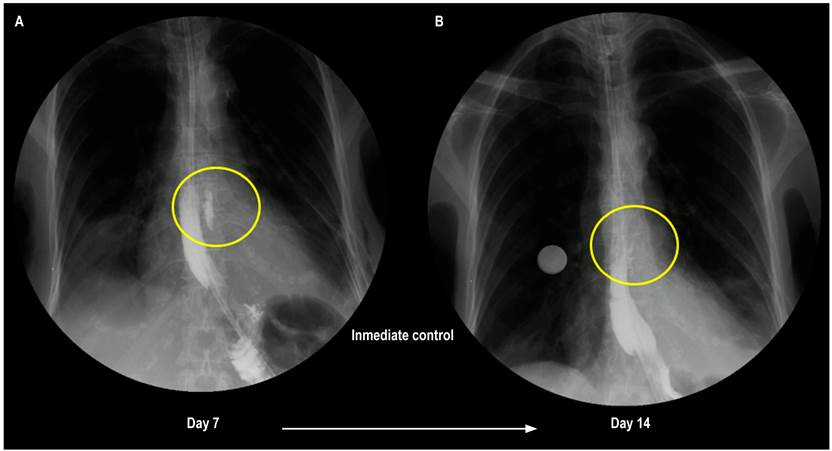
Figure 4 A. Control esophagogram seven days after the event: Minor solution of continuity, communicating with a contained saccular image of 45 x 9 mm proximal to the defect area. B. Control esophagogram 14 days after the event: Contained leak with a decrease in size compared to the initial imaging control (24 x 4 mm).
Discussion
Esophageal perforation is a rare but catastrophic gastrointestinal pathology (incidence 3.1/1,000,000 inhabitants/year)1,2, with high morbidity and mortality ranging between 10%-40%, being 10%-25% when treated in the first 24 hours and up to 40%-60% when management is delayed3,4. In their systematic review of the literature published in 2017, Sdralis et al reported an overall mortality of 13.5% based on the analysis of 39 studies (1,644 patients), with perforation of the thoracic esophagus being the most frequent location in 72.6%, followed by involvement of the cervical (15.2%) and abdominal (12.5%) esophagus5.
Its etiology is variable, with iatrogenic (46.5%) being the most frequent cause. It has a risk of occurring in diagnostic esophagogastroduodenoscopy of 0.03%, increasing when therapeutic endoscopy is performed (0.5% in esophageal dilation, 1.7% in dilatation for achalasia, 1%-6% in varicose vein sclerotherapy, 4.6% in photodynamic thermal therapy, 5% in laser therapy, 5%-25% in stent insertion)4. It is followed in frequency by spontaneous perforation (37.8%), this being the most common cause of non-iatrogenic perforation, esophageal foreign body perforation (6.3%), traumatic perforation (penetrating and closed trauma), and finally perforation due to ingestion of caustics (frequent in children)1,5,6. Typically, when esophageal perforation secondary to barotrauma is mentioned as the causal pathophysiological mechanism, the literature refers to spontaneous esophageal perforation or Boerhaave syndrome (BS), in honor of the Dutch physician Herman Boerhaave, who described the entity for the first time in 1724. It refers to the longitudinal and transmural rupture of the esophagus (previously healthy), secondary to an abrupt increase in intraluminal esophageal pressure caused by an increase in gastric content towards the esophagus due to an increase in intragastric pressure often triggered during vomiting. However, it is also reported in other events that require a Valsalva maneuver and even during sleep7-11.
In this entity, the thickness rupture occurs more frequently in the distal 1/3 of the thoracic esophagus (80%), 3 to 6 cm above the diaphragm, on the left posterior wall (90%), with an average damage length of 2-10 cm7,8. Its clinical manifestations depend on the location of the perforation and the time of evolution8. Patients with perforation of the cervical esophagus exhibit pain in the neck and the upper half of the chest, while in those with more distal perforation, the pain is in the lower half of the chest and the upper left side of the abdomen. Mackler’s triad (vomiting, chest pain, subcutaneous emphysema in the neck-thorax) and Anderson’s triad (subcutaneous emphysema, tachypnea, abdominal muscle tension) are described as clinical aids to the diagnosis of this pathology. Nonetheless, subcutaneous emphysema, the characteristic sign of both triads, is present in only 14%-30% of cases, with an atypical course in 30%-50%7. Among the initial diagnostic supports is radiography (subcutaneous emphysema, pneumomediastinum, pneumothorax, subdiaphragmatic air, pleural effusion), computerized axial tomography (CAT), and esophagogastroduodenoscopy, which, in cases of iatrogenic cause, is capable of detecting the defect promptly during the intervention3,5,6,12.
As we said before, although BS is the representative pathology of barogenic perforation of the esophagus, the medical literature mentions mechanisms of barotrauma different from this entity13-21. In 1990, Gubbins reported the case of a 77-year-old patient with mid-esophageal stenosis, which was bridged over during gastroscopy. He retched during the procedure, with subsequent evidence of a perforation near the esophagogastric junction. The hypothesis of esophageal barotrauma as a causal mechanism is established. In 2002, Van de Louw published the case of a 56-year-old patient with a 3 cm linear esophageal perforation in the distal esophagus after requiring non-invasive mechanical ventilation, where the barotrauma mechanism was triggered by the positive pressure exerted13. Thus, in the case of our patient, considering the type of procedure and the site of the perforation (8 cm above the dilated stenosis), we hypothesize barotrauma as the causal mechanism of the esophageal rupture, triggered by an abrupt increase in esophageal intraluminal pressure during balloon inflation.











 text in
text in 



