Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em
SciELO
Similares em
SciELO -
 Similares em Google
Similares em Google
Compartilhar
Agronomía Colombiana
versão impressa ISSN 0120-9965
Agron. colomb. v.23 n.2 Bogotá jul./dez. 2005
FISIOLOGÍA Y TECNOLOGÍA POSCOSECHA
Almacenamiento de frutos de mango (Mangifera indica L.) 'Van Dyke' en empaques de atmósfera modificada
Jesús Antonio Galvis1, Harvey Arjona2, Gerhard Fischer2 and Ricardo Martínez3
1 Profesor asociado, Instituto de Ciencia y Tecnología de Alimentos, Universidad Nacional de Colombia, Bogotá. e-mail: jagalvisv@unal.edu.co
2 Profesor asociado, Facultad de Agronomía, Universidad Nacional de Colombia, Bogotá. e-mail: hearjonad@unal.edu.co, gfischer@unal.edu.co
3 Investigador Programa Nacional de Biometría, Corporación Colombiana de Investigación Agropecuaria (Corpoica), C.I. Tibaitatá, Mosquera (Colombia). e-mail: rmartinez@cenipalma.org
Fecha de recepción: 26 de agosto de 2005. Aceptado para publicación: 21 de noviembre de 2005
Abstract
'Van Dyke' mango fruits were harvested at physiological maturity and stored in active modified atmosphere conditions using different plastic packages (modified atmosphere packaging, MAP). Two-fruit-samples were placed in sealed caliber 2 and caliber 3 low density polyethylene packages (LDPE), using two initial atmosphere gas mixtures. Wrapped fruit and control fruit (without bagging) were kept at 12 °C and 90% relative humidity (RH) for 30 days plus 5 days' shelf life at 18 °C and 70% RH. Caliber 3 LDPE film ensured that injected O2 concentration remained more stable and increased CO2 content during storage time than caliber 2 film. MAP treatment increased fruit shelf-life by delaying the ripening process. MAP drastically reduced fruit weight loss and increased ascorbic acid content compared to control. Negative correlation was found between fruit weight loss and fruit ascorbic acid content and O2 concentration within the plastic film and fruit ascorbic acid content. By contrast, there was direct correlation between CO2 content within MAP and the ascorbic acid content of fruit. Mango fruit wrapped in caliber 2 PE film ensured the longest extension of the ripening period, without incurring fruit skin lesions. Fruit in caliber 3-PE-film failed to ripen due to flesh softening being inhibited and organic acid degradation.
Additional key words: polyethylene, ripening, ascorbic acid, citric acid, malic acid, succinic acid.
Resumen
Frutos de mango 'Van Dyke' se cosecharon en madurez fisiológica y se almacenaron en condiciones de atmósfera modificada activa, empleando diferentes empaques de plástico (modified atmosphere packaging, MAP). Muestras de dos frutos fueron empacadas en bolsas de polietileno de baja densidad (LDPE) calibre 2 (0,0350 mm) y calibre 3 (0,0722 mm), utilizando dos mezclas de gases iniciales. Los frutos embolsados y los testigos (sin embolsar) se colocaron a temperatura de 12 °C y 90% de humedad relativa (HR) durante 30 d, más 5 d en anaquel (shelf life) a 18 °C y 70% HR. Las bolsas calibre 3 mantuvieron más estable la concentración del O2 inyectado y aumentaron más el contenido de CO2, que los empaques calibre 2. Los tratamientos en atmósfera modificada incrementaron la vida útil de los frutos retardando el proceso de maduración. El MAP disminuyó drásticamente la pérdida del peso de los frutos e incrementó el ácido ascórbico, respecto al testigo. Hubo correlación inversa entre pérdidas de peso del fruto y contenido de ácido ascórbico y entre concentración de O2 en el empaque y contenido de ácido ascórbico. En contraste, hubo correlación directa entre concentración de CO2 en el MAP y contenido de ácido ascórbico. Los mangos embolsados en PE calibre 2 maduraron en un mayor tiempo, respecto al testigo, sin presentar lesiones sobre la piel. Los frutos empacados en bolsas de PE calibre 3 no maduraron, debido al no ablandamiento de la pulpa y a la no degradación de los ácidos orgánicos.
Palabras claves adicionales: polietileno, maduración, ácido ascórbico, ácido cítrico, ácido málico, ácido succínico.
Introduction
DOMESTIC AND INTERNATIONAL TRADE in fresh mangoes has been limited by the highly perishable nature and susceptibility of this fruit to postharvest diseases, extremes of temperature and physical injury (Gomez-Lim, 1997). Mangoes are commercially grown on 12,707 ha in Colombia (data for 2001, after Food and Agricultural Organization [FAO], 2003), especially for domestic consumption; however, there are good prospects for the export market. Paull (1994) characterized the storage potential of mangoes (postharvest life) as being 14 to 25 d at 10 to 12 °C.
Mango is a climacteric fruit and modified atmosphere storage using plastic bags or wrapping has shown some delay in ripening (Nakasone and Paull, 1998). Protective films modify the fruit's internal atmosphere and have great potential as shelf-lifeextending treatment for many fruit species (Saftner, 1999; Arjona et al., 1994; Nisperos-Carriedo et al., 1990; Kader et al., 1989; Ben-Yehoshua, 1985). Film treatments function as barriers against water vapor, gases, volatile compounds and ethylene transmission (Kester and Fennema, 1986).
Low O2 concentrations generally reduce respiration and ethylene production rates; high CO2 concentrations also hinder ripening, possibly because CO2 acts as a competitive ethylene inhibitor (Yahia, 1997). Many fruits tolerate a minimum 2% O2 concentration; lower O2 levels induce anaerobic respiration, which could result in the development of undesirable off-flavors and odors due to the accumulation of ethanol, acetaldehyde and other volatiles in these conditions (Kader et al., 1989). Tolerance limits to elevated CO2 concentrations vary widely between fruit species; some fruit tolerate CO2 concentrations higher than 5%, whereas others develop disorders at this CO2 level (Yahia, 1997). Tolerance to elevated CO2 decreases with reduced O2 level and, similarly, tolerance limits to low O2 concentration increase with increased CO2 level (Kader et al., 1989).
Specialized postharvest treatment for extending fruit shelf-life in Colombia has not been sufficiently developed as yet (Galvis, 2002). This study was aimed at examining the value of using plastic film packaging by injecting two types of atmosphere and storing the plastic bags in refrigerated conditions in order to improve the shelf-life quality of 'Van Dyke' mangoes.
Materials and methods
'Van Dyke' mango fruits, grown in a commercial orchard in El Espinal, Tolima (Colombia) -located at 4° 12' N 74° 56' W, 431 meters above sea level, having 1,369 mm rainfall, 29 °C mean annual temperature and 70% RH-, were harvested at physiological maturity and stored in the Universidad Nacional de Colombia's Planta Piloto de Vegetales, Instituto Colombiano de Tecnología de Alimentos (ICTA), in Bogotá. Healthy fruit were selected, disinfected with thiabendazole (200 mg· L-1) and twofruit-samples were wrapped in caliber 2 (0.0350 mm) and caliber 3 (0.0722 mm) low density polyethylene (LDPE) packages (30 x 20 cm plastic films).
Bagged mangoes were placed in vacuum packing equipment (Minipack) to evacuate the air within the bag and inject the gas mixtures (table 1). Each package, containing two fruits, constituted an experimental unit and the treatments were replicated four times. Treated (packaged) and non-treated (control) fruit were then stored at 12 °C (± 1 °C) in air with 90% RH (± 2%) for 10, 20 or 30 d. Following storage, plastic films were removed and fruit were kept at ambient temperature (18 °C, 70% RH) for 5 d.
The following variables were measured for fruit quality analysis at 15, 25 and 35 d: fruit weight loss, fruit firmness and ascorbic acid, citric acid, malic acid and succinic acid content. Oxygen and CO2 concentration within packages was measured every 5 d during the storage period using a portable gas analyzer (Fa. Abbis, type Tom 12 N 28). The whole fruit's firmness (without removing the fruit skin) was measured at three points of the equatorial fruit zone (on the fruit's surface area) using a manual penetrometer (Ballarfmfg) with a 7.9 mm tip.
Samples were prepared according to Senesi et al. (1999) for ascorbic acid analysis. 20 µL aliquots of the aqueous solution were filtered through a 0.45-µm filter and injected into a high performance liquid chromatograph (HPLC, Jasco) equipped with a 30 x 0.78 cm Aminex 87-H column and UV detector (VIS UV-9757, Jasco) at 241 nm. The solvent was 4 mM sulphuric acid 0.01 N solution, using a 0.6 mL· min-1 flow rate at 20 °C.
Citric, malic and succinic acid samples (prepared following Senesi et al., 1999) were determined by HPLC (Waters), equipped with a Waters column, packed with 30 x 0.65 cm microparticulated calcic resin and UV detector (Waters). The solvent was bidistilled H2O using 0.8 mL· min-1 flow rate at 20 °C and 2,600 psi pressure.
Data were analyzed by variance analysis. Treatment means were separated at 5% significance level using orthogonal contrast tests. A completely randomized experimental design was employed; SAS PROC GLM software (version 8.2) was used for analyzing band structure variance, subdivided by time (Martínez and Martínez, 1997).
Results and discussion
CO2 and O2 content inside the package
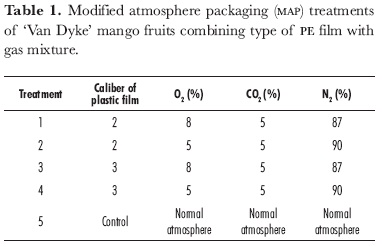
In-package CO2 content increased with storage time in all four modified atmosphere packaging (MAP) treatments, confirming the results of Gonzalez et al. (1990), who wrapped 'Keitt' mangoes in LDPE film. CO2 concentration was significantly higher in caliber 3 film (figure 1) following 10 days' storage. CO2 equilibrium was reached after 5 days' fruit storage inside caliber 2 packaging; elevated CO2 content in these packages did not inhibit mango ripening but did delay it, compared to control fruit.
CO2 concentration markedly increased inside caliber 3 packaging, starting from the 10th day of storage by up to 23%; this was 1.7-fold higher than in caliber 2 films. High CO2 content from day 15 onwards caused abnormal (inhibited) ripening of mangoes in the two atmospheres in caliber 3 packaging. Gautan and Lizada (1984) reported that increased CO2 concentration over prolonged periods inhibits the ripening of mango fruit and causes undesired odors and off-flavors due to the possibility of fermentative decarboxylation at CO2 concentration higher than 15%. Kader et al. (1989) reported that mango fruit tolerate a maximum 5% CO2 concentration.
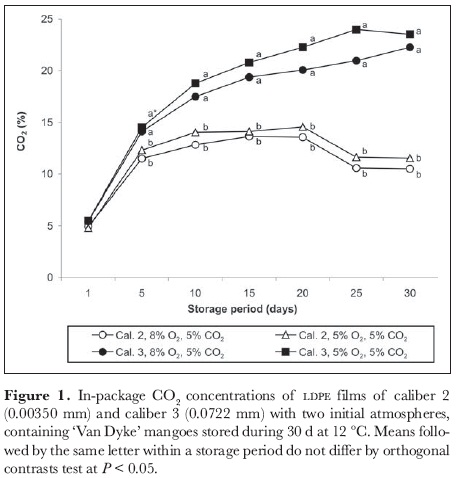
As for CO2 content, O2 concentration was more affected by film thickness than by the type of atmosphere injected (figure 2). Caliber 3 packages maintained lower O2 content than those of caliber 2. Following 15 days' storage, O2 concentration within caliber 2 films reached a maximum of 14.6 and 13.9% for 8 and 5% O2-atmospheres, respectively, and then slowly decreased. By contrast, O2 equilibrium within the thicker film (caliber 3) was reached starting from the 5th day of storage. After 20 days' storage, O2 content dropped significantly in caliber 3 packages; it reached 4.2 and 3.8% by the end of the storage period for 8 and 5% O2-atmospheres, respectively.
The low O2 content in caliber 3 packaging could have caused the poor fruit quality in this experiment (flavor, odor and color) and inhibited ripening. Sy and Mendoza (1984) do not recommend O2 concentrations lower than 5% for 'Carabao' mango storage due to fruit fermentation. Bender et al. (2000) observed that the tolerance of 'Haden' and 'Tommy Atkins' mangoes to low O2 decreases at 2, 3, 4 or 5 kPa O2 plus N atmospheres as fruit ripens.
Fruit weight loss
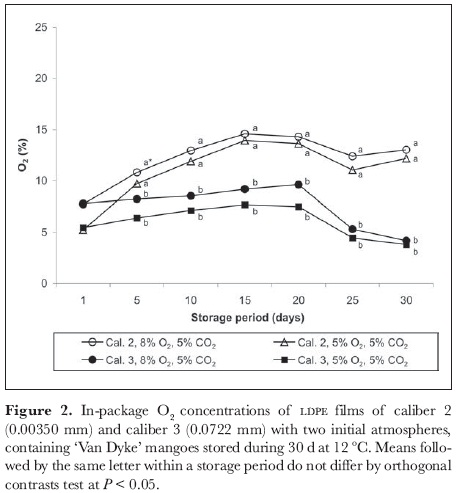
Percentage of fruit fresh weight loss increased in all treatments the longer the storage time (figure 3) as a consequence of transpiration and respiration (Wills et al., 1998). MAP reduced weight loss considerably compared to control fruit. This result is of special interest since mango is a fruit having a high rate of moisture loss during postharvest period (Paull, 1994). Fruits in caliber 3 packaging (initial 8% O2 concentration) lost less weight than those in caliber 2 film packaging with initial 5% O2. The highest losses (14.6%) were found after 35 d in control (unwrapped) fruit, followed by mangoes in caliber 2 packaging (5.6%). There was not any difference in weight loss amongst fruit stored at different gas atmospheres within the packages. Our results coincided with those of Yantarasri et al. (1995), who observed no reduction in mango fruit weight (var. 'Nam Dork Mai') when using PVC films having low water vapor permeability.
Weight loss had a positive correlation with O2 content inside the packaging (r2 = 0.6903), whereas correlation of weight loss with CO2 content was negative (r2 = -0.6839). It may have been that fruit weight loss diminished at low O2 and high CO2 contents within the packaging due to decreasing transpiration and respiration rates. Fruit without MAP lost 8.8% of their initial fresh weight after 15 days' storage, presenting initial signs of skin wrinkling, as described by Paull (1994) for mangoes that lost 6-8% of their weight. Smith et al. (1987) directly associated fruit weight loss to the film's water vapor transmission rates when film barriers are used, these being independent of CO2 and O2 permeability.
Whole fruit firmness
Fruit in all treatments lost firmness the longer the storage time (table 2). Unwrapped fruit exhibited the highest flesh softening, whereas mangoes in caliber 3-film-packaging softened less than those wrapped in caliber 2 film. These results demonstrate the great influence of MAP on delaying mango ripening. Control fruit and those having caliber 2-PE-film MAP had the similar fruit softening after 35 d. By contrast, mangoes wrapped in caliber 3 packaging conserved their high flesh firmness. This fact probably indicates that the elevated CO2 concentration inside the package affected polygalacturonase activity (Kader et al., 1989). Galvis (1995) also observed that 'Tommy Atkins' mangoes, stored for 4 weeks in modified atmosphere packaging at 12 °C, had greater fruit firmness in the treatment which had the highest respiration intensity, corresponding to bags where CO2 content was greater. González et al. (1990) found that low film permeability decreased fruit softening in 'Keitt' mangoes stored in MAP conditions. These authors also observed a direct relationship between loss of weight and firmness.
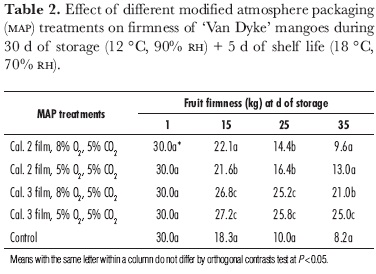
Siriphanich (1998) observed higher fruit firmness during storage (20% CO2 atmosphere at 15 ºC for 2 d) of mature 'Nam Dok Mai' mangoes, but not in immature or fully mature fruit. This author supposed that a high CO2 atmosphere could have a direct influence on fruit firmness by reducing the pH of various cellular compartments including the intercellular space. Change in pH may destroy the ionic balance between compartments and allow calcium ion to move to the cell wall area. Sornsrivichai et al. (1992) described that film packaging for 'Nam Dork Mai' mangoes only retarded fruit softening of the outer portion of the flesh but accelerated softening in the inner portion (where the fruit is more mature).
Ascorbic acid content
Oxidation of ascorbic acid in plant tissue depends on temperature, fruit water content, pH, storage atmosphere, and storage period (Braverman, 1980; Barth et al., 1993).
Ascorbic acid content in fruit varied considerably, depending on the thickness of the plastic film used (tabla 3). Mangoes packed in caliber 3 films maintained higher ascorbic acid concentrations than those in caliber 2 films. Our results correspond to those of Barth et al. (1993), who detected much higher ascorbic acid levels in MAP of broccoli (wrapped in semipermeable polymeric film) as compared to non-packaged bunches. These authors attributed the higher ascorbic acid concentration to lower peroxidase activity (an important factor for ascorbic acid oxidation in plant tissue) in packaged broccoli in response to the low O2 and high CO2 concentration inside the packaging.
There was strong negative linear correlation (r = -0.850) between percentage of fruit weight loss and ascorbic acid content. Control fruit therefore exhibited the lowest vitamin C content during the whole experiment (tabla 3). Our results agree with those of Nunes et al. (1998) who reported negative correlation between fruit weight loss and vitamin C content of strawberries stored in MAP, independent of the storage temperature.
A negative correlation (r = -0.732) was found between O2 content in packages and ascorbic acid concentration in fruit, whereas there was direct correlation between CO2 content and ascorbic acid (r = 0.722). Our results are supported by the data of Kader et al. (1989), who reported that reduced O2 and elevated CO2 content in MAP decreased vitamin C loss in fruit.
Citric acid content
Citric acid is the dominant organic acid in mangoes (Gálvis, 2002; Wickham and Mohammed, 1999), found in all treatments (tabla 3). Percentage citric acid loss during storage was higher than that for other organic acids analyzed here; however, acid loss was less than in control fruit. Citric acid content of fruit wrapped within caliber 2 film was lower than those wrapped in caliber 3, probably due to higher citrate dehydrogenase activity in LDPE film. After 35 d of storage, fruit in MAP treatment maintained their high citric acid concentration; this was 3-fold higher as compared to control fruits. Otherwise, immature green mangoes wrapped in LDPE packages developed and maintained a lower citric acid content as compared to waxed or non-treated fruit (Wickham and Mohammed, 1999).
Yantarasri et al. (1995) found that ripening delay in 'Nam Dork Mai' mangoes wrapped in PVC film induced delay in the decrease of citric acid content, particularly in non-perforated packaging and wrapping having a small degree of perforation. The decline in acidity level was significantly delayed in packages where O2 was below 7%. Yantarasri et al. (1995) therefore supposed hindering of the ripening process caused by anaerobiosis because a fermented flavor was always noticeable during evaluation.
Malic acid content
Malic acid had the lowest content compared to the other acids analyzed in the experiment's fruits (table 3). Control fruit (without MAP) had a much lower organic acid concentration than that wrapped in LDPE film. Control fruit and those wrapped in caliber 2 PE film decreased malic acid during storage time. Mangoes packaged in caliber 3 film having an 8% O2 and 5% CO2 atmosphere maintained acid concentration or increased it (5% O2, 5% CO2). This result might indicate that these atmospheres have affected malate dehydrogenase activity (Kader, 1986). Contrasting with our results, Shipway and Bramlage (1973) found that CO2 content above 6% stimulated malate oxidation and suppressed the oxidation of citrate, α-ketoglutarate, succinate, fumarate and pyruvate in mitochondria isolated from apples. Inhibiting succinate oxidation to fumarate by CO2 has been related to the inhibition of succinic dehydrogenase (Mathooko, 1996), thereby leading to succinate accumulation and a depletion in malate levels.
Succinic acid content
Succinic acid content in fruit decreased drastically during storage without MAP but presented the lowest decline in caliber 3 LDPE films (table 3). Being inside the thick PE film, probably inhibited succinate dehydrogenase activity more than caliber 2 films by increasing the CO2 concentration within such packaging. Kader (1986) reported that a high CO2 concentration (5-20%) inhibited Krebs cycle enzymes accumulating succinic acid (toxic to plant tissues, Matthooko, 1996) due to inhibiting succinic dehydrogenase activity in fruit like apples (Monning, 1983) and pears (Frenkel and Patterson, 1973). There is no evidence that low O2 concentrations can maintain or increase succinic acid content (Kader et al., 1989).
Conclusions
• MAP of 'Van Dyke' mangoes in LDPE films was effective in increasing postharvest life by delaying ripening, as shown by few changes in firmness.
• Wrapping fruit with plasticized film slowed fruit weight loss and maintained ascorbic acid levels.
• The best MAP treatments were those using LDPE caliber 2 films (0.035 mm thickness) with initial 8% O2, 5% CO, 87 N or 5% O, 5% CO, 90% N atmospheres, not only maintaining fruit quality but also its shelf-life.
• Postharvest treatment using caliber 3 LDPE packaging became detrimental for 'Van Dyke' mangoes, independently of the gas mixture inside, since firmness was kept too high and CO2 stress inhibited organic acid degradation in these fruit.
Literature cited
Arjona, H.E., F.B. Matta and O. Garner, Jr. 1994. Wrapping in polyvinyl chloride film slows quality loss of yellow passion fruit. HortScience 29(4), 295-296. [ Links ]
Barth, M.M., E.L. Kerbel, S. Broussard and S.J. Schmidt. 1993. Modified atmosphere packaging protects market quality in broccoli spears under ambient temperature storage. J. Food Sci. 58(5), 1070-1072. [ Links ]
Bender, R.J., J.K. Brecht, S.A. Sargent and D.J. Huber. 2000. Mango tolerance to reduced oxygen levels in controlled atmosphere storage. J. Amer. Soc. Hort. Sci. 125(6), 707-713. [ Links ]
Ben-Yehoshua, S. 1985. Individual seal-packaging of fruits and vegetables in plastic film - a new technique. HortScience 20, 32-37. [ Links ]
Braverman, J.B.S. 1980. Introducción a la bioquímica de los alimentos. Ed. El Manual Moderno, Mexico. pp. 177-178. [ Links ]
Food and Agricultural Organization (FAO). 2003. FAOSTAT Database results mango. Rome. [ Links ]
Frenkel, C. and M.E. Patterson. 1973. Effects of carbon dioxide on activity of succinic dehydrogenase in 'Bartlett' pears during cold storage. HortScience 8, 395-396. [ Links ]
Galvis, J.A. 2002. Análisis de crecimiento del fruto de mango (Mangifera indica L.) var. Van Dyke y estudio de algunos métodos de conservación durante la poscosecha. Ph.D. thesis. Facultad de Agronomía, Universidad Nacional de Colombia, Bogota. [ Links ]
Galvis, J.A. 1995. Conservación del mango (Mangifera indica L.) variedad Tommy Atkins mediante la técnica de atmósfera modificada. In: Proc. Int. Symp. "Manejo postcosecha y perspectivas de comercialización de frutas y verduras". 27-30 June 1995, Bogotá. Lecture N° 19. [ Links ]
Gautan, D.M. and M.C. Lizada. 1984. Internal breakdown in 'Carabao' mango subjected to modified atmosphere: storage duration and severity of symptoms. Postharvest Horticultural Training and Research Center 2, 28-32. [ Links ]
Gomez-Lim, M.A. 1997. Postharvest physiology. pp. 425-445. In: Litz, R.E. (ed.). The mango - production and uses. CAB International, Wallingford, UK. [ Links ]
González, G., E.M. Yahia and I. Higuera. 1990. Modified atmosphere packaging (MAP) of mango and avocado fruit. Acta Hort. 269, 335-344. [ Links ]
Kader, A.A. 1986. Biochemical and physiological basis for effects of controlled and modified atmospheres on fruits and vegetables. Food Technol. 40(5), 99-100; 102-104. [ Links ]
Kader, A.A., D. Zagory and E.L. Kerbel. 1989. Modified atmosphere packaging of fruits and vegetables. Crit. Rev. Food Sci. Nutr. 28(1), 1-30. [ Links ]
Kester, J.J. and O.R. Fennema. 1986. Edible films and coatings; a review. Food Technol. 42, 47-59. [ Links ]
Martínez, R. and N. Martínez. 1997. Diseño de experimentos. Fondo Nacional Universitario, Ed. Guadalupe, Bogotá. pp. 271-289. [ Links ]
Mathooko, F.M. 1996. Regulation of respiratory metabolism in fruits and vegetables by carbon dioxide. Postharv. Biol. Technol. 9, 247-264. [ Links ]
Monning, A. 1983. Studies on the reaction of Krebs cycle enzymes from apple tissue (cv. 'Cox Orange') to increased levels of CO2. Acta Hort. 138, 113-119. [ Links ]
Nakasone, H.Y. and R.E. Paull. 1998. Tropical fruits. CAB International, Wallingford, UK. [ Links ]
Nisperos-Carriedo, M.O., P.E. Shaw and E.A. Baldwin. 1990. Changes in volatile flavor compounds of pineapple orange juice as influenced by the application of lipid and composite films. J. Agr. Food Chem. 38, 1382-1387. [ Links ]
Nunes, M.C., J.K. Brecht, A.M. Morais and S.A. Sargent. 1998. Controlling temperature and water loss to maintain ascorbic acid levels in strawberries during postharvest handling. J. Food Sci. 63(6), 1033-1036. [ Links ]
Paull, R.E. 1994. Tropical fruit physiology and storage potential. In: Champ, B.R., E. Highley and G.I. Johnson (eds.). Proceedings of the International conference of postharvest handling of tropical fruits. 19-23 July 1993, Chiang Mai, Thailand. ACIAR Proc. 50, 198-204. [ Links ]
Saftner, R.A. 1999. The potential of fruit coating and film treatments for improving the storage and shelf-life qualities of 'Gala' and 'Golden Delicious' apple. J. Amer. Soc. Hort. Sci. 124(6), 682-689. [ Links ]
Senesi, E., A. Galvis and G. Fumagalli. 1999. Quality indexes and internal atmosphere of packaged fresh-cut pears ('Abate Fetel' and 'Kaiser' varieties). Italian J. Food Sci. 11(2), 111-120. [ Links ]
Shipway, M.R. and W.J. Bramlage. 1973. Effect of carbon dioxide on activity of apple mitochondria. Plant Physiol. 51, 1095-1098. [ Links ]
Siriphanich, J. 1998. High CO2 atmosphere enhances fruit firmness during storage. J. Jpn. Soc. Hort. Sci. 67(6), 1167-1170. [ Links ]
Smith, S.M., J. Geeson and J. Stow. 1987. Production of modified atmospheres in deciduous fruits by the use of films and coatings. HortScience 22, 772-776. [ Links ]
Sornsrivichai, J., S. Gomolmanee, P. Boon-Long and H. Gemma. 1992. Seal packaging by plastic film as a technique for limiting fungal decay of mangoes. Acta Hort. 296, 23-31. [ Links ]
Sy, D.A. and D.B. Bendoza. 1984. Respiratory responses of 'Carabao' mango to different levels of oxygen. Postharvest Horticultural Training and Research Center 1, 3-8. [ Links ]
Wickham, L.D. and M. Mohammed. 1999. Storage of immature green mango (Mangifera indica, L.) fruit for processing. J. Food Qual. 22, 31-40. [ Links ]
Wills, R., B. McGlasson, D. Graham and D. Joyce. 1998. Postharvest - an introduction to the physiology and handling. CAB International, Wallingford, UK. [ Links ]
Yahia, E.M. 1997. Modified and controlled atmosphere for tropical fruits. Hort. Rev. 22, 123-183. [ Links ]
Yantarasri, T., S. Ben-Yehoshua, V. Rodov, W. Kjumpuan, J. Utahaibutra and J. Sornsrivichai. 1995. Development of perforated modified atmosphere package for mango. Acta Hort. 398, 81-91. [ Links ]














