Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Agronomía Colombiana
Print version ISSN 0120-9965
Agron. colomb. vol.29 no.3 Bogotá Sept./Dec. 2011
SOILS, FERTILIZATION and MANAGEMENT OF WATER
Biological nitrogen fixation by Rhizobium sp. native gliricidia (Gliricidia sepium [Jacq.] Kunth ex Walp.) under greenhouse conditions
Fijación biológica de nitrógeno por Rhizobium sp. nativo de matarratón (Gliricidia sepium [Jacq.] Kunth ex Walp.) bajo condiciones de invernadero
Juan Guillermo Cubillos-Hinojosa1,3, Pablo Ernesto Milian-Mindiola1, and Jorge Luis Hernández-Mulford 2
1Program of Microbiology, Universidad Popular del Cesar. Valledupar (Colombia).2Centre Biotecnology of Caribbean, Regional of Cesar, Servicio de Enseñanza Nacional (SENA). Valledupar (Colombia).
3Corresponding author. jcubillosh@misena.edu.co Received for publication: 31 August, 2010. Accepted for publication: 2 November, 2011.
ABSTRACT
An experiment was conducted under greenhouse conditions in order to evaluate the association and biological nitrogen fixation (BNF), six native strains of Rhizobium sp isolated from nodules gliricidia and a commercial strain (Rhizobiol©). Suspensions of 106 and 108 cells/mL for each of the strains studied and applied in two separate trials: the first with seeds and the second with seedlings 35 days of age gliricidia. In the seed test is calculated the cumulative percentage of germination for 7 days. In both trials after 42 days in seeds and seedlings 52 days in length was measured and stem diameter, leaf number, dry weight of aerial parts, number of nodules/plant and percentage of nitrogen. All treatments stimulated germination, growth and BNF, being better in the native strains at a concentration of 108 cells/mL, compared with the commercial strain and the control. These results indicate the positive effect exerted by Rhizobium sp., in promoting growth and BNF, which could be used for further studies to establish the production of a biofertilizer, allowing potentiate the production of crops by gliricidia for food arich cattle as source of protein in the Caribbean Biotechnology Center.
Key words: biofertilizer, leguminous tree, sustainable agriculture, Caribbean.
RESUMEN
Se realizó un experimento bajo condiciones de invernadero con el fin de evaluar la asociación y fijación biológica de nitrógeno (FBN) en seis cepas nativas de Rhizobium sp. aisladas de nódulos de matarratón y una cepa comercial (Rhizobiol©). Las suspensiones de 106 y 108 células/mL para cada una de las cepas estudiadas y aplicadas en dos ensayos separados: el primero con semillas y la segundo con plántulas de 35 días de edad matarratón. En el ensayo de semilla se calcula el porcentaje acumulado de germinación durante 7 días. En ambos ensayos, después de 42 días en las semillas y 52 días en plántulas se determinó la altura y diámetro del tallo, número de hojas, peso seco de la parte aérea, número de nódulos por planta y el porcentaje de nitrógeno. Todos los tratamientos estimularon la germinación, el crecimiento y FBN, siendo mejor en las cepas nativas a una concentración de 108 células/mL, en comparación con la cepa comercial y el control. Estos resultados indican el efecto positivo ejercido por Rhizobium sp., en la promoción del crecimiento y FBN, que podrían ser utilizados para estudios adicionales para determinar la producción de un biofertilizante, permitiendo potenciar la producción de cultivos de matarratón como alimento de ganado bovino como fuente de proteína en el Centro de Biotecnología del Caribe.
Palabras clave: biofertilizante, árbol leguminoso, agricultura sostenible, Caribe.
Introduction
In the department of Cesar, located northwest of the country on the plains of Colombian Caribbean, its main economic lines are the farm from which it derives 47% of their income. The cattle industry ranks first order, with an estimated population of 1,513,149 heads (Rey et al., 2005; Rojas et al., 2009). For this reason it is estimated that Cesar contributes about 38% of domestic production of meat and 43% of milk production depends mainly on the grazing of native and introduced grasses occupies 1.4 and 1.9 million have respectively (Rey et al., 2005). However, currently being carried out through integrated management system spontaneous cultivation of legumes associated with these grasses, because soil conditions limit the availability and quality of forage during dry periods in the year.
The cattle population is one of the most demanding with respect to food, is currently promoting the use legumes because grasses (conventional plant) are scarce and insufficient in the dry months. As the implementation of this system (or agroforestry legume-grass) is relatively new, the constant waves of summer during the year have caused disruptions in the lives of animals, reflecting disease which has led to low production yield in the single purpose in cattle (dairy), in addition to that you consider that there are currently more than 100 animals formed homogeneously pure breeds such as Guzerat, Gyr-Gir and Brahman, this has led to implementing such a system as an alternative to counter the problem of shortage of fodder (Centro Biotecnológico del Caribe, 2009).
Gliricidia (Gliricidia sepium [Jacq.] Kunth ex Walp.) cultivation, the alternative has proven to be more objective and conducive to overcome the shortage of fodder in dry seasons. The relevance is that the leaves of these legumes, have high concentrations of crude protein, energy and digestibility by cattle (Pedraza et al., 2001; Rey et al., 2005; Mahecha, 2002; Diouf et al., 2008). Furthermore, it is believed that the implementation of this system, legume-grass (agroforestry), provides key components to improve the physicochemical and biological properties of soil, having the ability to fix atmospheric nitrogen (N2), leaving aside the use indiscriminate synthetic fertilizers have generated some extent a negative impact on soil ecology, which is currently expressed in erosion, desertification and depletion of vegetation cover (Mahecha et al., 2002).
The ability of legumes to fix N2, is given through a partnership that is built with own soil symbiotic bacteria belonging to the genus Rhizobium, which lets you pick from nodular simbiosomas all nitrogen necessary for their physiological functions and formation of structures vegetative, besides this, let legume fixed nitrogen made available to other plants nearby (Döbereiner, 1997; Ferrera and Alarcón, 2007; Mahecha, 2002).
Isolation and selection of native strains of Rhizobium sp., intended for creation of bio-fertilizers, is seen as a safe, effective and economical way to secure and restore soil productivity, as well as fix nitrogen produce phytohormones, are involved in the solubilization of phosphorus and backlash (Akhtar and Siddiqui, 2009; Mourad et al., 2009; Puertas et al., 2006; Santillana et al., 2005, Bardin et al., 2004). This complex set of features makes the host plant set up in any type of soil; improve all biological processes manifest in maximum productivity.
The present investigation was to assess the potential for infectivity, promoting plant growth and effectiveness in N2 fixation of native strains of Rhizobium sp. isolated from nodules gliricidia, by application of two bacterial concentrations (106 and 108 cells/mL) in seeds and seedlings under greenhouse conditions, this will continue with a period of consolidating field that supports a byproduct based symbiotic nitrogen-fixing microorganisms that contribute to a sustainable agro-ecological system.
Materials and methods
This research was conducted at the Microbiology Laboratory and greenhouse Caribbean Biotechnology Center (CBC), located in the municipality of Valledupar (Cesar) a 10°27'20'' N and 73°15'30'' W, with an average temperature of 28.4°C and a height of 169 m, annual rainfall of 961 mm and relative humidity of 67%. In the area, wind speeds reach 14.76 km h-1, moderate rainfall spread between April and November with peak in May and October, this is provided by a slightly warmer climate (Ideam, 2005; Alcaldía de Valledupar, 2011)
Six strains were isolated native Rhizobium sp. from nodules of seedlings established crops gliricidia in the CBC for the evaluation and more commercial strain Rhizobiol© (CS). The strains were rectivaron in Petri dishes containing yeast extract mannitol agar (YMA) and incubated at a temperature of 28°C for 3-5 d (Wang and Martínez, 2003). From these cultures were adapted inocula at concentrations of 106 and 108 cells/mL suspended in sterile distilled water.
Seed bioassay
Seed germination in greenhouse
Bags were made with 1 kg of previously sterilized soil, seeds were placed gliricidia three, 1% hypochlorite and 70% alcohol to a depth of 2 cm. Experimental design was applied in a randomized complete block of 7x2, with three replicates per treatment and five experimental units each repetition, using as factor the native strains and the CS, two concentrations 106 and 108 cells/mL. The inoculation was done aseptically using 2 mL of the respective bacterial suspension 106 and 108 cells/mL of each of the strains of Rhizobium sp. and the CS; prepared from pure cultures by counting in Newbauer chamber, applying directly on the seed inoculum. The control treatment (without inoculation) alone was added 2 mL of sterile distilled water.
Observations were made after 7 d, checking the number of seeds germinated seeds considering that emerged to the surface. For this assay were established 15 treatments including the control without inoculation. From the data obtained, we calculated the percentage of germination expressed as a percentage cumulative germination (PG) at 7 d after inoculation. With reference to the total seeds sown and germinated seeds total, as indicated by the following formula:
Infectivity and growth promotion effectiveness in biological nitrogen fixation (BNF) After 2 weeks after it is determined the percentage of germination, seedlings were thinned each pot leaving the most vigorous (Mora, 1995; Matos et al., 2007). The seedlings were maintained under greenhouse conditions with characteristics in 70-80% humidity, temperature of 31-34°C where the air volume, brightness and attack by pests are controlled, spray irrigation was applied twice daily for 2 h. After 42 d it took the data on stem length (SL) with the help of a tape measure and stem diameter (SD) with the help of a caliper, was recorded the number of true leaves (NTL) dry weight of aerial parts (DW) based on the methodology proposed by Reyes et al. (2008). We determined the number of nodules/plant (NNP) and percentage of leaf nitrogen by the Kjeldahl method (Jiménez, 2007).Seedling bioassay
Were taken perforated polythene bags and filled with 1 kg of previously sterilized soil. Each pot was planted with three seeds gliricidia previously disinfected with iodine 70%, 1% sodium hypochlorite and 70% alcohol and subjected to pretreatment in sterile distilled water for 2 h before planting. After two weeks of emerged seedlings were thinned each pot leaving the most vigorous (Mora, 1995; Rincón, 2000). After a period of 35 d inoculation was performed by carefully removing the soil at the level of the stem base and adding 2 mL of the respective bacterial suspension (106 and 108 cells/mL), each of the strains of Rhizobium sp. and the CS, prepared from pure cultures (Matos and Zúñiga, 2002). The control treatment alone was added 2 mL of sterile distilled water. For this assay were established 15 treatments including the control without inoculation. Also in the seed test variables were measured for this test. The results for both tests was performed comparing averages by multiple range test of Duncan (P=0.05) and ANOVA, using the statistical package Statgraphics Plus version 5.1 and tested for the statistical package SPSS version 15.
Results and discussion
Seed bioassay
Seed germination
According to the results of all treatment levels were 100% except G21-106 and G34-106 who obtained values of 93.3% in both cases, with values over the control (80%), this is possibly due to the production of phytohormones such as indole acetic acid (IAA) and gibberellins (Peña and Reyes, 2007). The concentration factor led to differences between treatments obtained a germination rate of 100% in all treatments the concentration treated with 108 cells/mL versus 80% obtained by CS-106 concentration 106 cells/mL, obtaining notice that the more cell the greater the effect on germination.
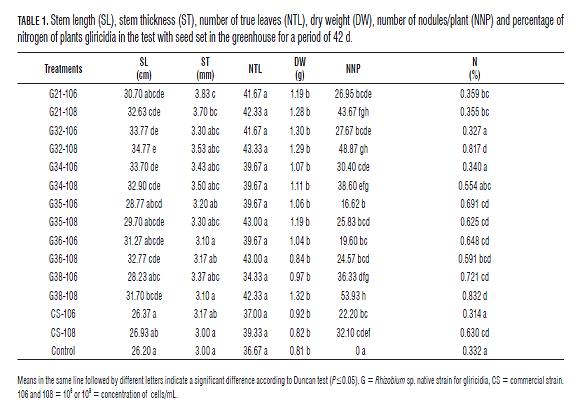
Some investigators have reported favorable results when assessing the effect of strains of Rhizobium sp. in the germination of seeds. Santillana et al. (2005) evaluated strains of Rhizobium sp., isolated from Vicia faba and Pisium sativum var. macrocarpum at concentrations of 108 cells/mL in seeds of Solanum lycopersicum, finding values of 100% in 9/19 strains evaluated with respect to the control of 80%. Infectivity and growth promotion effectiveness in BNF The Tab.1 presents the means for measuring values obtained after 42 of observation. The stem length (SL), shows statistically significant differences (P=0.05) better treatment G32-108 (34.77 cm), compared with the control (26.20 cm) and the commercial strain in both concentrations and CS-106 CS-108 with values of 26.4 cm and 26.9 cm respectively, with these results we can deduce that the strains of Rhizobium sp. native generated a positive impact in terms of their length. The statistically significant differences (P=0.05) for SL, showing that treatments with higher bacterial concentrations (108 cells/mL), differ statistically from the other treatments except for G35-108 (29.70 cm) and CS-108 (26.93 cm), who are not significantly different (P=0.05) compared with the control (26.20 cm). For the ST, was found better results in treatment G21-108 and G21-106, with respective values of 3.7 mm and 3.83 mm (Tab.1), presenting significant differences only for the latter with compared to the control (3.0 mm) in both the commercial strain CS-106 concentrations (3.17 mm) and CS-108 (3.0 mm).
The NTL, no significant differences compared with the control, however the strains G32-108 (43.33), G35-108 (43.0), G36-108 (43.0), G21-108 (42.33), G38-108 (42.33) showed higher values compared with the control (36.67), CS-106 (37.0) and CS-108 (39.33) and can be noted that the inoculum of 108 cells/mL had a positive influence on this variable.
These results contrast with the DW, the best treatments for this analysis were G21-108 (1.28 g), G32-108 (1.287 g), G32-106 (1.30 g) and the highest value was for G38-108 (1.32 g) with respect to the control (0.81g), CS-106 and CS-108, with values respectively 0.92 and 0.82 g (Tab.1), also showed good results for G34-106 (1.07 g), G34-108 (1.107 g), G35-108 (1.190 g), G21-106 (1.190 g), however there were no significant differences according to Duncan test (P=0.05). These results are similar to those found by Mora (1995), who managed to obtain significant differences in dry matter of common bean plants huasteco black variety (Phaseolus vulgaris L.), inoculated with strains of Rhizobium leguminosarum biovar phaseoli native, with respect to control without inoculation.
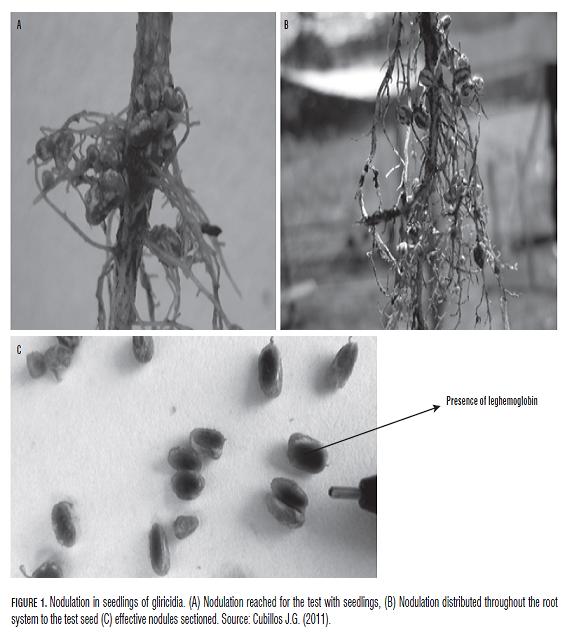
The NNP and %N, makes clear in the first instance, that the strains of Rhizobium sp., generated positive effect on the nodulation process ( Fig.1), relative to uninoculated control (Tab.1), which showed no nodulation. The highest values were driven by G34-108 (38.60 NNP), G21-108 (43.67 NNP), G32-108 (48.87 NNP) and the highest value was obtained for G38-108 (53.93 NNP), showing significant differences according to Duncan test (P=0.05), with respect to CS-106 and CS-108 with values of 22.2 and 32.10 NNP, respectively. This shows the points made by some researchers (Rincón et al., 2000), which indicated that in most soils can be found indigenous populations of rhizobia, which will differ in their ability to infect legumes according to the chemical and soil physical and due to the slow process of natural selection are subject to stress conditions and excretion of substances carried out by the roots as flavonoids and glycoproteins that attract or select the different strains of Rhizobium sp., living in the rhizosphere of these plants. For the %N, was found better results for CS-108 (0.63%), G36-106 (0.648%), G35-106 (0.691%), G38-106 (0.721%), G32 -108 (0.817%) and the highest percentage was for G38-108 (0.832%), showing significant differences compared with the control without inoculation (0.331%) and CS-106 (0.314%). It was found that G38-108 (0.832%), with significant differences in the concentration commercial strain 106 cells/mL CS-106 (0.314%), but not statistically different CS-108 (0.63%). On the other hand the values obtained by CS-106 and control without inoculation (0.331%), not significantly different from each other (Tab.1). It can be inferred that the strains of Rhizobium sp. native generated a very positive effect in terms of biological nitrogen fixation in plants gliricidia, applied at concentrations of 106 and 108 cells/mL directly on the seeds, being even favorable results in the treatment with commercial strain concentration of 108 cells/mL. Nitrogen levels for the best treatments (G32-108 = 0.817% and G38-108 = 0.832%) could be found up to 150% more nitrogen accumulated, compared to the control without nitrogen. Similar results were reported by Matos and Zúñiga (2002), who found that native strains of Rhizobium sp. were highly efficient nitrogen content increased up to 76.8% in Pallar Criollo Iqueño (Phaseolus lunatus) and 76.2% in Pallar ICA 450; with respect to a control added with nitrogen. Moreover Urzúa et al. (2001), noted a correlation between the aerial dry matter production, produced by forage vetch (Vicia benghalensis) and accumulation of nitrogen in greenhouse conditions.
Seedling bioassay
Infectivity and growth promotion effectiveness in BNF In Tab.2 presents the means for measuring values obtained after 17 d after application at 35 d, measurements were made (Mora, 1995).
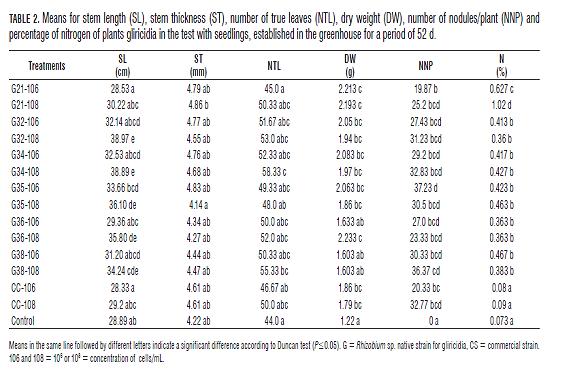
The result obtained for SL in the seedlings at 52 d, shows that all treatments had a positive effect on this variable, except G21-106 (28.53 cm), G21-108 (30.22 cm), G36-106 (29.36 cm), CS-106 (28.33 cm) and CS-108 (29.2 cm) (Tab.2), who did not differ significantly according to Duncan test (P=0.05), compared with the control (28.89 cm), but there were differences with the G36-108 treatments (35.8 cm), G35-108 (36.1 cm), G34-108 (38.89 cm), G32-108 (38.97 cm), showing these last two treatments higher values ( Fig.2). Also is found that the concentration produced a significant effect on the SL showing better results in 108cells/mL except the treatment with the commercial strain CS-108 and G21- 108 (Tab.2). Similar results were reported by Matos and Zúñiga (2002), who found that the length of the stem in two varieties of Pallar Criollo iqueño (P. lunatus) and ICA 450, was significantly higher compared with the control, after being inoculated with native strains of Rhizobium sp., and BradyRhizobium sp.
For the ST was found that the strains did not generate a significant effect on this variable compared with the control, however the highest values in terms of stem diameter (Tab.2) were found in G21-108 (4.86 mm ), G21-106 (4.79 mm), G35-106 (4.83 mm), G32-106 (4.77 mm), G34-108 (4.68 mm) and G34-106 (4.76 mm) compared with the control (4.22 mm), CS-106 and CS-108 who gave values of 4.61 mm in both cases but there were no significant differences. With respect to NTL gave better results in the treatment G34-108, with 58 leaves/ plant, which presents significant differences with respect to G21-106 (45.0), G35-108 (48.0), CS -106 (46.67) and the control (44.0), who had the lowest number of leaves. At this point there is the relationship between the variables SL and NTL for the treatment G34-108 who showed the highest values in both analyzes 38.89 cm and 58.33 leaves/plant, respectively. These results are due to greater leaf area, provide greater availability of photosynthate to the plant for nitrogen fixation, which is reflected in a better development (Matos and Zúñiga, 2002).
As relates to the DW found significant differences only in G36-108 (2.23 g), G21-106 (2.123 g), G21-108 (2.193 g), compared with the control (1.22 g). The treatments CC-106 (1.86 g) and CC-108 (1.79 g), showed no statistical differences with the native strains according to Duncan (P=0.05). With the above we can say that there is strong relationship between the variables ST and DW, as the treatments G21- 106 and G21-108, were filed within the highest values in both cases. These results are similar to those obtained by Urzúa et al. (2001) who found in the dry matter yield better results for native strains of Rhizobium leguminosarum bv. viceae inoculated in Vicia benghalensis, whose values were above the control and the reference strain R. leguminosarum bv. viceae, obtaining significant differences with native strains analyzed.
In analyzing the NNP, we found that the native strains of Rhizobium sp., generated a positive effect in nodulation compared to the control treatment who did not submit nodulation. This is attributed to good soil sterilization process, which eliminated any possible presence of indigenous strains in soil (Mora, 1995). To measure this variable did not take into account data such as the size of nodules and redness to cut them, only the total count of nodes present in the root of each seedling.
The highest levels of nodulation were found in G34-108 (32.83 NNP), CS-108 (32.77 NNP) G38-108 (36,37 NNP) and G35-106 (37 NNP), appearing only in last treatment significant difference compared with the commercial strain CS-106 (20.33 NNP), G21-106 (19.87 NNP) and with the control. But some researchers say the ability of native strains to form nodules, does not necessarily indicate the efficiency of biological nitrogen fixation (Mora, 1995). Legumes is common "promiscuous" presented a nodulation "semi-effective" where there is a good nodulation, but set a small amount of N, which prevents them from reaching their yield potential. Thus, not all strains of Rhizobium sp. are effective in fixing nitrogen, although the infectivity is high (Mora, 1995).
For the %N values were lower in CS-106, CS-108 and the control with respective values of 0.08, 0.09 and 0.073%, with significant differences (P=0, 05) with the other treatments, being highest levels in G21-108 and G21-106, with values of 1.02 and 0.63% respectively. Also is found good results for G34-108 (0.427%), G35-108 (0.463%) and G38-106 (0.467%), who despite being below G21-106 and G21-108, showed significant differences compared to CS-106 (0.08%) CS-108 (0.09%) and the control (0.073%). Is observed that G38-106 (0.467%), showed better results with respect to G38-108 (0.383%) but no significant differences (P=0.05). This may be due to the settling time of the nodules, because the higher the concentration of rhizobia greater the competition for space and nutrients in the root, indicating more time to set when the inoculum was found in higher concentrations. According to Matos and Zúñiga (2002), who argue that early nodulation could induce an earlier onset of nitrogen fixation, therefore a greater amount of nitrogen accumulated in the plant. Infectivity early could increase the effect of the native strains inefficient. (Matos and Zúñiga, 2002; Rincón et al., 2000; Mora, 1995).
Conclusions
The native strains of Rhizobium sp., generate a positive effect on seed germination gliricidia, stimulating germination to levels of 100% in most treatments compared with the control with 80%. Is found that the best results in treatments who receiving inoculation of 108 cells/mL, also had a significant effect on infectivity, effectiveness and growth promotion highlighting the strains G34, G38, G21, G32 which showed better results with respect to accumulated dry matter, percentage of nitrogen in concentrations 108 cells/mL, found results well above the commercial strain and the control.
The nodulation was found in relation to leaf nitrogen accumulation was much better in the test with seed, due to increased settling time of the rhizobia strains as were distributed throughout the radicle. Is found that commercial strain Rhizobiol© in the highest concentration also stimulate the growth and development of seedlings, but these values were below the native strains. The inoculum of 108 cells/mL significantly influenced the growth and development of seedlings, with respect to the concentration106 cells/mL for both strains native to the commercial strain. This indicates the possibility of advancing the study and evaluation of field due to its better adaptation to agro-ecological and climatic conditions of the region and subsequently the development of a bioproduct to mechanize farming gliricidia, roll feed,emphasizing harmony with environmental policies, and improving the development and sustainability of agriculture in the Colombian Caribbean area.
Acknowledgements
The authors of this paper express their appreciation to the Caribbean Biotechnology Center (CBC), SENA - Regional Cesar where this research was developed.
Literature cited
Akhtar, M. and Z. Siddiqui. 2009. Effects of phosphate solubilizing microorganisms and Rhizobium sp. on the growth, nodulation, yield and root-rot disease complex of chickpea under field condition. Afr. J. Biotechnol. 8(15), 3489-3496. [ Links ]
Alcaldía de Valledupar. 2011. Información general municipio de Valledupar. In: http://www.valledupar-cesar.gov.co; consulted: November, 2011. [ Links ]
Bardin, S., H. Huang, J. Pinto, E. Amundsen, and R. Scott. 2004. Biological control of Pythium damping-off of pea and sugar beet by Rhizobium leguminosarum bv. viceae. Can. J. Bot. 82, 291-296. [ Links ]
Centro Biotecnológico del Caribe. 2009. Informe anual Centro Biotecnologico del Caribe 2009. Servicio Nacional de Aprendizaje (SENA), Valledupar (Colombia). [ Links ]
Diouf, A., T. Abdoulaye Diop, I. Ndoye, and M. Gueye. 2008. Response of Gliricidia sepium tree to phosphorus application and inoculations with Glomus aggregatum and rhizobial strains in a sub-Saharian sandy soil. Afr. J. Biotechnol. 7(6), 766-771. [ Links ]
Döbereiner, J. 1997. Biological nitrogen fixation in the tropics: social and economic contributions. Soil Biol. Biochem. 29(5/6), 771-774. [ Links ]
Ferrera, R. and A. Alarcón. 2007. Microbiología agrícola. Editorial Trillas, Mexico DF. [ Links ]
Ideam, 2005. Ubicación de Valledupar en Cesar. In: http://www. pronosticosyalertas.gov.co; consulted: November, 2009. [ Links ]
Jiménez, J. 2007. Caracterización molecular de cepas nativas de Azotobacter ssp mediante análisis de restricción del DNA ribosomal 16S. Undergraduate thesis. Pontificia Universidad Javeriana, Bogota. [ Links ]
Mahecha, L. 2002. El silvopastoreo: una alternativa de producción que disminuye el impacto ambiental de la ganadería bovina. Rev. Colomb. Cienc. Pecu. 15(2), 226-231. [ Links ]
Mahecha, L., L. Gallego, and F. Peláez. 2002. Situación actual de la ganadería de carne en Colombia y alternativas para impulsar su competitividad y sostenibilidad. Rev. Colomb. Cienc. Pecu. 15(2), 213-225. [ Links ]
Matos, G. and D. Zúñiga. 2002. Comportamiento de cepas nativas de Rhizobium aisladas de la costa del Perú en dos cultivares del pallar (Phaseolus lunatus L.). Ecología Aplicada 1(1), 19-24. [ Links ]
Matos, G., E. Ormeño, and D. Zúñiga. 2007. Diversidad de los rizobios que nodulan el cultivo de pallar (Phaseolus lunatus L.) en la costa central del Perú. In: Congreso Peruano de Ecología. Lima. [ Links ]
Mora, F. 1995. Selección de cepas nativas de Rhizobium leguminosarum bv phaseoli eficientes en fijación biológica de nitrógeno en suelos de Costa Rica. Agron. Mesoam. 6, 68-74. [ Links ]
Mourad, K., K. Fadhila, M. Chahinez, R. Meriem, D. Philippe, and B. Abdelkader. 2009. Antimicrobial activities of Rhizobium sp. strains against Pseudomonas savastanoi, the agent responsible for the olive knot disease in Algeria. Grasas y Aceites 60(2), 139-146. [ Links ]
Pedraza, R., F. de Castro, and E. Ørskov. 2001. Valor nutritivo de las fracciones solubles e insolubles del follaje de Gliricidia sepium (Jacq.) Kunth ex Walp. In: http://www.reduc.edu. cu/147/03/2/14703207.pdf; consulted: November, 2011. [ Links ]
Peña, H. and I. Reyes. 2007. Aislamiento y evaluación de bacterias fijadoras de nitrógeno y disolventes de fosfatos en la promoción del crecimiento de la lechuga (Lactuca sativa L.). Interciencia 32(8), 560-565. [ Links ]
Puertas, A., B. de la Noval, B. Martínez, I. Miranda, F. Fernández, and L. Hidalgo. 2006. Interacción de Pochonia chlamydosporia var. catenulata con Rhizobium sp., Trichoderma harzianum y Glomus clarum en el control de Meloidogyne incognita. Rev. Protección Veg. 21(2), 80-89. [ Links ]
Rey, M., D. Chamorro, and M. Ramírez. 2005. Efecto de la doble inoculación de Rizobios y micorrizas sobre la producción y calidad del forraje de Leucaena leucocephala. Rev. Corpoica 6(2), 52-59. [ Links ]
Reyes, I., L. Álvarez, H. El-Ayoubi, and A. Valery. 2008. Selección y evaluación de Rizobacterias promotoras del crecimiento en pimentón y maíz. Bioagro 20(1), 37-48. [ Links ]
Rincón, J., T. Clavero, R. Razz, S. Pietrosemoli, F. Mendez, and N. Noguera. 2000. Efecto de la inoculación con cepas nativas e introducidas de Rhizobium sobre la fijación de nitrógeno en Leucaena (Leucaena leucocephala (Lam) de Wit). Rev. Fac. Nal. Agron. Medellin 17, 342-357. [ Links ]
Rojas, D., M. Garrido, and R. Bonilla. 2009. Estandarización de un medio de cultivo complejo para la multiplicación de la cepa C50 de Rhizobium sp. Undergraduate thesis. Pontificia Universidad Javeriana, Bogota. [ Links ]
Santillana, N., C. Arellano, and D. Zúñiga. 2005. Capacidad del Rhizobium de promover el crecimiento en plantas de tomate (Lycopersicon esculentum Miller). Ecología Aplicada 4(1/2), 47-51. [ Links ]
Urzúa, H., J. Urzúa, and R. Pizarro. 2001. Pre-selección de cepas de Rhizobium leguminosarum bv. Viceae en vicia forrajera, para abonos verdes. Cien. Inv. Agr. 28(1), 3-6. [ Links ]
Wang E. and J. Martínez. 2003. Taxonomía de Rhizobium. In: http://www.microbiologia.org.mx/microbiosenlinea/CAPITULO_ 12/Capitulo12.pdf; consulted: November, 2011. [ Links ]