Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Agronomía Colombiana
Print version ISSN 0120-9965
Agron. colomb. vol.30 no.2 Bogotá May/Aug. 2012
PLANT BREEDING, GENETIC RESOURCES & MOLECULAR BIOLOGY
Genetic diversity in a Colombian bean (Phaseolus vulgaris L.) collection as assessed by phaseolin patterns and isoenzymatic markers
Diversidad genética en una colección colombiana de fríjol (Phaseolus vulgaris L.) usando patrones de faseolinas y marcadores isoenzimáticos
Gustavo Ligarreto1 and César Ocampo2
1Department of Agronomy, Faculty of Agronomy, Universidad Nacional de Colombia. Bogotá. galigarretom@unal.edu.co2Genetics resources Unit, International Center for Tropical Agriculture (CIAT ). Palmira. Colombia. Received for publication: 30 April, 2010. Accepted for publication: 31 August, 2012.
ABSTRACT
The knowledge of genetic diversity patterns increase the efficiency of the conservation and the enrichment of the genetic resourses. This study allowed the discrimination of the existing genetic variability in a Colombian collection of shrub bean by phaseolin patterns and isoenzymatic markers. Bean seed proteins revelated that the phaseolin patterns types T and C are predominant in the Andean pool, type S in the Meso-American pool and type B in Colombian and Central American accessions, with a predominance of 81% of phaseolin T in the Andean pool, and 78% of phaseolin B in the Meso-American pool. The accessions of cultivated and wild beans showed variation in 10 of the studied enzymatic systems: αβ-EST , GOT, αβ-ACP, DIA , PRX, AS D, 6-PGDH, MDH, IDH and ME; and monomorphism in the PGI and PGM systems. The isozyme systems presented 19 bands of activity, of which 74% were polymorphic loci. Both in the Andean and Meso-American genetic pools, the loci Mdh-1, Mdh-2, β-Est-1, Skdh and Me exhibited polymorphisms. Single alleles in the Meso-American pool were found in 6-Pgdh-2103, Mdh-1100, Idh100, α-Est-1100, α-Est-2100, and Dia-195; and in the Andean pool, in 6-Pgdh-1100, and Acp-2100. For degree of domestication, the wild and cultivated accessions presented polymorphisms in 58 and 47% of the lOCi, respectively. The enzymatic relationship cluster analysis of the studied bean collection revealed three distinct groups of accessions; namely the Meso-American pool, including its cultivated and wild accessions; the Andean pool, which is mainly comprised of cultivated accessions, plus the wild DGD-626; and finally, featured by a high degree of enzymatic polymorphism and by the presence of the type I phaseolin, a third group that contains only a wild accession from the northern, Peruvian Andes.
Key words: genetic variability, germplasm, isozymes, seed proteins.
RESUMEN
El conocimiento de los patrones de diversidad genética incrementa la eficiencia de la conservación y el enriquecimiento de los recursos genéticos. Este estudio permitió discriminar la variabilidad genética existente en una colección colombiana de fríjol arbustivo por patrones de faseolina y marcadores isoenzimáticos. Los análisis de las proteínas de la semilla de fríjol revelaron que los patrones de faseolina T y C son predominantes en el acervo andino, el de S en el acervo Mesoamericano y el de B en accesiones de Colombia y de América Central, con representación de la faseolina T en el 81% de los andinos y del 78% de la faseolina B en el acervo mesoamericano. En las accesiones de fríjol cultivado y silvestre, hubo variación en 10 sistemas enzimáticos probados: ab-EST , GOT, αβ-ACP, DIA , PRX, AS D, 6-PGDH, MDH, IDH y ME, y monomorfismo en los sistemas PGI y PGM . Los sistemas isoenzimáticos presentaron 19 bandas de actividad, de las cuales el 74% fueron lOCi polimórficos. En los acervos genéticos andino y mesoamericano los loci Mdh-1, Mdh-2, β-Est-1, Skdh y Me presentaron polimorfismo, fueron alelos únicos en el acervo mesoamericano 6-Pgdh-2103, Mdh-1100, Idh100,α-Est-1100, α-Est-2100, y Dia-195, y únicos en el acervo andino 6-Pgdh-1100, y Acp-2100. Por grado de domesticación, los silvestres presentaron polimorfismo en el 58% de los loci y los cultivados en el 47%. El análisis de agrupamientos de las relaciones isoenzimáticas existentes en la colección de fríjol revelan la diversidad presente en tres grupos de accesiones. Un grupo con características del acervo mesoamericano entre cultivados y silvestres, un segundo grupo lo conforman las accesiones cultivadas del acervo andino y un silvestre (DGD-626), el tercer grupo lo integra el silvestre del norte de los Andes, originario de Perú, con alto grado de polimorfismo isoenzimático y faseolina tipo I.
Palabras clave: variabilidad genética, germoplasma, isoenzimas, proteínas de las semillas.
Introduction
Mesoamerican food agriculture is defined by the milpa cropping system, consisting of maize (Zea mays L.), common bean (Phaseolus vulgaris L.), and squash (Cucurbita sp.) (Sanjur et al., 2002). In recent years, a domestication center for maize has been proposed in the Balsas River basin in west-central Mexico (Kwak et al., 2009). The proposed domestication area for common bean does not overlap with the area proposed for maize domestication, based on genetic data (Matsuoka et al., 2002), nor does it overlap with any of the three major archaeological locations pertaining to the origin of agriculture in the Mesoamerican area: the Oaxaca Valley, the Tehuacán Caves in Puebla, and the ocampo Caves in Tamaulipas. Another important domestication zone in the Americas is found in part of the Andes, from the south of Colombia to Atacama and Jujuy in the northwest of Argentina; and from the Pacific coast to the Amazon valley piedmont (Debouck et al., 1993).
As a low cost protein source, and since the beginning of its domestication process, common beans have been central to the diet of settlers in America. For a long time, the species has been subjected to several selection pressures, thus generating the thousands of known genotypes, existing in the ex situ conservation banks. Beans are cultivated in different environmental conditions and communities, which partially explains the strong differences in growth habits and seed types (Gepts, 1991).
Studies with molecular markers have revealed that many wild bean populations can be classified in two major gene groups: one in the central zone of Meso-America, and the other one in the central and southern Andes (Beebe et al., 1997; Kwak and Gepts, 2009). Domestication was multi-local, that is, it took place many times at different locations, involving populations derived from a single ancestor. In biochemical studies with phaseolins, Koenig and Gepts (1989b) suggested that in the northwest of South America (Colombia, Ecuador and northern Peru), there is a geographical transition area between the Meso-American and Andean populations.
The most important protein stored in bean seeds is phaseolin, which contains 35-50% of the total plant nitrogen (Gepts and Bliss, 1986), and is a useful evolutionary marker due to its high polymorphism and heritability, and nonsensitivity to environmental influences (Triana and Roca, 1989).
More than 40 phaseolin patterns have been identified (Beebe et al., 1997). However, types 'S' (Sanilac), 'Sb' ('S' Brazil), and 'Sd' ('S' Durango) predominate in Meso-American cultivars, while types 'T' (Tendergreen), 'H' (Huevo de Huanchaco) and 'C' (Contender) are more frequent in Andean cultivars. Phaseolin 'B' (Boyaca 22) is found in Colombia and Central America (Koenig et al., 1990). Debouck et al. (1993) showed that wild population isozymes and phaseolins from Ecuador and northern Peru were intermediate between Meso-American and Andean pools. Phaseolin analyses on cultivated populations from Alto Magdalena in Colombia established that the Andean varieties were cultivated at higher altitudes than the Meso- American ones.
Isozymes are useful genotypic markers in studies of plant systematics, evolution, population genetics and conservation (Maquet et al., 1997). In the common bean, they have been mainly used to assess phylogenetic relationships and diversity. Unlike morphological and physiological features, isozymes are little affected by the environment Several bean studies have detected isozyme polymorphisms. Weeden and Liang (1985) found that the enzymes Rubisco (RBCS), shikimato dehydrogenase (SKDH), peroxidase (PRX), malic enzyme (ME) and phospho-gluco isomerase (GPI) are respectively controlled by the single genes Rbcs, Skdh, Prx, Me, and Pgi. Koenig and Gepts (1989b) identified two additional polymorphisms (codified as Lap-3 and Mdh-1) in leucine amino-peptidase (LAP) and malate dehydrogenase (MDH). Except for Diap-1, Diap-2, Rbcs and ME, the studied isozymes presented no linkage, and as a result, they are located in different regions of the genome.
In this context, the objectives of the present study were: (i) to relate bean phaseolin and isozyme types to genetic pools as established by morphological descriptors; (ii) to establish the relative degree of genetic variation of the Colombian common bean collection, as well as its usefulness as a genetic bank for obtaining improved varieties; and (iii) to gather preliminary information about the genetic transition zone featured by allele combinations between the Meso-American and Andean domestication centers.
Materials and methods
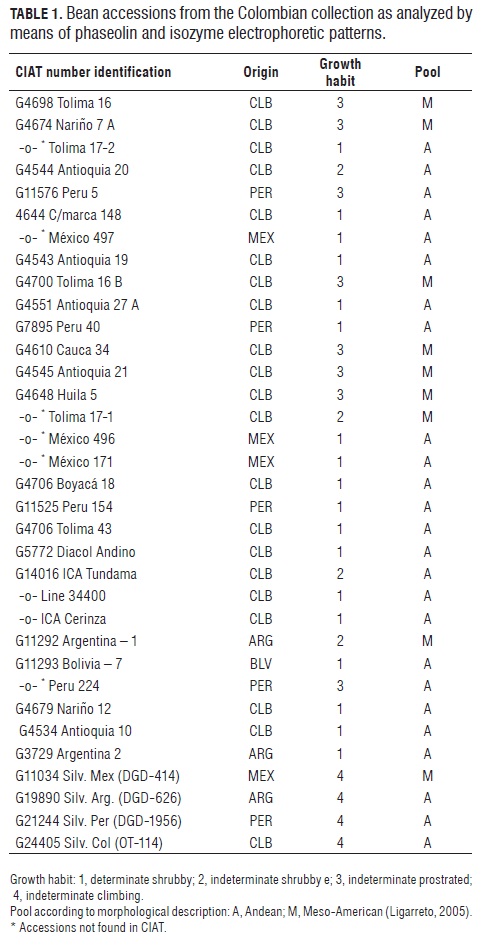
The work was carried out in the Tropical Agriculture International Center (CIAT ), Cali, Colombia, with 30 common bean accessions of shrub (types I and II ) and prostrated (type III ) growth habits, and 4 wild materials described in Tab.1. Out of these 30 accessions, 16 corresponded to regional varieties, 3 to improved commercial varieties (Diacol Andino, ICA Tundama and ICA Cerinza), one was Colombian elite line L-34400, and 10 were introduced varieties from Central and South America.
The studied accessions are part of the germplasm bank of the Corporación Colombiana de Investigación Agropecuaria (Corpoica), located at the Tibaitatá Research Center (Mosquera, Colombia). There, the regional and introduced materials were randomly chosen from the bean collection, which is made up of 165 accessions.
Phaseolin tests
The variability of the collection was analyzed by separating the phaseolins according to their molecular weight, by means of a one-dimension sodium dodecyl sulfate acid polyacrylamide gel electrophoresis system (SDS/ PAG E), as indicated by Hussain et al. (1986).
Fifty mg samples of bean flour ground from ten testas, removed from seeds that were taken from all accessions, were prepared for protein extraction and suspended in 500 mL of a Tris HCl 0.036 M extracting solution (pH 8.5 and a 1:10 w/v ratio), which was then shaken for 5 min. For membrane rupture, the samples were chilled in water and ice and kept in ultra sound chambers for an h. Next they were centrifuged for 20 m at 14,000 rpm. 250 mL of the supernatant were recovered and added to 250 mL of Tris HCl 0.625 M buffer (pH 6.8 and a 1:1 v/v ratio), and shaken for an additional 2 min and kept in bain-marie for 5 min. The samples were kept at -14°C.
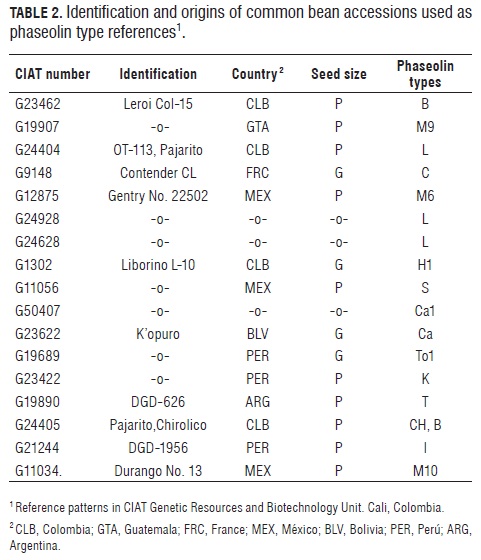
The proteins were separated in a gel in a Miniprotean II chamber by a 180 V current with 60 to 70 mA resistance for 1 h. Gel preparation and staining was done by the method presented by Ma and Bliss (1978) and standardized by CIAT (1996). The analysis of the two phaseolin types found in the collection was made by visual comparison with the center of origin reference patterns that are normally used for these studies in CIAT 's global bean collection (Tab.2)
Isozyme tests
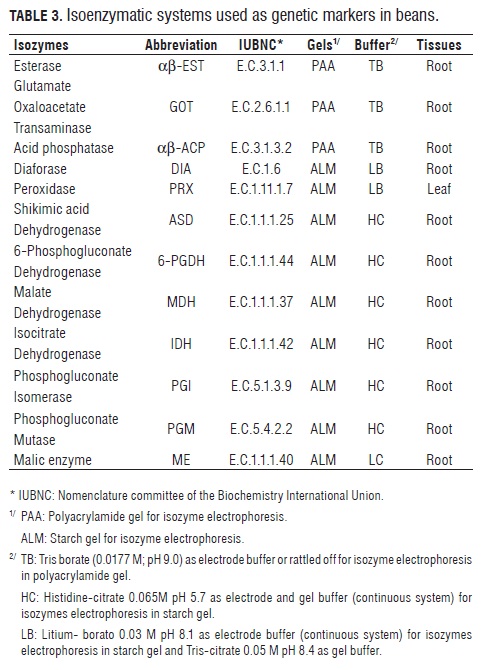
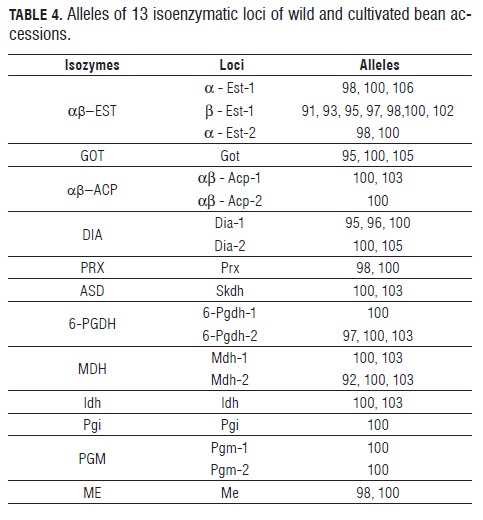
Isozyme analyses of early leaves and root tissue extracts (obtained from 10 seedlings grown in soil for 10 d under greenhouse conditions) were done according to Hussain et al. (1987) as modified by CIAT 's Genetic Resources Unit (CIAT , 1996). Twelve isoenzymatic systems were tested: α-EST , β-EST , GOT, α-ACP, β-ACP, DIA , PRX, AS D, 6-PGDH, MDH, IDH and ME (Tab.3).
The identification of isozyme loci and their respective alleles confirmed those of Koenig and Gepts (1989a). The most common allele was given a 100 value label, from which the others were measured for their difference in millimeters with the wild bean cultivar standards.
Genetic diversity analyses
The Jaccard similarity quotient (sij) was used as a measurement of similarity among isozyme electrophoretic bands. The distance dij = 1-sij (Harch, 1995; Podani, 1993) was calculated for each accession pair. Conglomerate analysis was applied to the distance matrix, using UPGMA between pairs of accessions. The output of that analysis was used to build a dendrogram that provided information about the proximity and dissimilarity between the materials. All these analyses made use of the following statistical packages: SYN -TA X, version 5.0 (Podani, 1993) and NTSYS , version 2.0 (Rohlf, 1998).
Results
Phaseolin genetic diversity
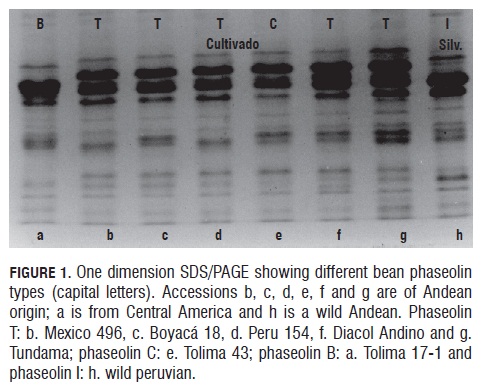
The results of the phaseolin one dimension SDS/PAG E are similar to those obtained in previous studies (Koenig et al., 1990; Beebe, 1997), in which the dominant patterns are T and C for the Andean pool, S for the Meso-American pool, and B in the Colombian and Central American accessions (Fig.1).
Typically, Andean phaseolins were observed in 70% of the studied bean cultivars (21 entries), whereas the remaining 30% (9 entries) corresponded to Meso-American phaseolins. The latter were comprised of phaseolin B (78%) and phaseolin S (22%). The former were dominated by phaseolin T (81%), and some presence of phaseolin C.
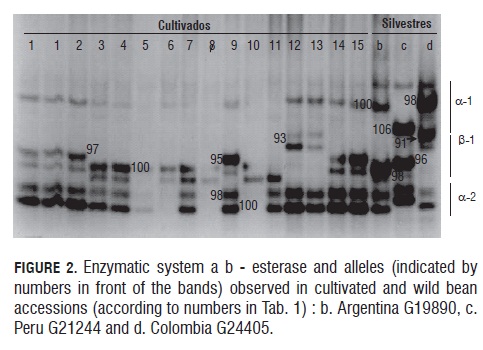
Fig.1 shows the phaseolin zymogram for the studied materials, exhibiting similar band patterns for accessions b, c, d, f and g (which contain phaseolin T) as well as for accession e (with phaseolin C); all of them presenting big red grains. The Wild Peruvian accession (h) (with phaseolin I) lacks a high molecular weight band (52 kD) (Gepts and Bliss, 1986), thus resembling cultivar (a) (Tolima 17-1), which features small red grains and also lacks the mentioned band.
Genetic variability as assessed by isozyme analyses
Both cultivated and wild beans showed considerable variation in the 10 tested enzymatic systems: αβ-EST , GOT, αβ-ACP, DIA , PRX, AS D, 6-PGDH, MDH, IDH and ME; and monomorphism in the PGI and PGM systems. Isozyme αβ-EST exhibited the highest degree of polymorphism, presenting 7 alleles in locus β-Est, 2 in one of the α-Est loCi, and 3 in the other (Tab.4, Fig.2).The 12 isoenzymatic systems presented 19 activity bands, out of which 14 (74%) were polymorphic loci with 43 different band combinations that were used to find genetic distances between the accessions. The activity of these polymorphic systems corresponds with previous reports (Koenig and Gepts, 1989a; Maquet et al., 1997; Avendaño et al., 2004) (Tab.4). The polymorphic loci Mdh-1, Mdh-2, β-Est-1, Skdh and Me were found in both genetic pools.
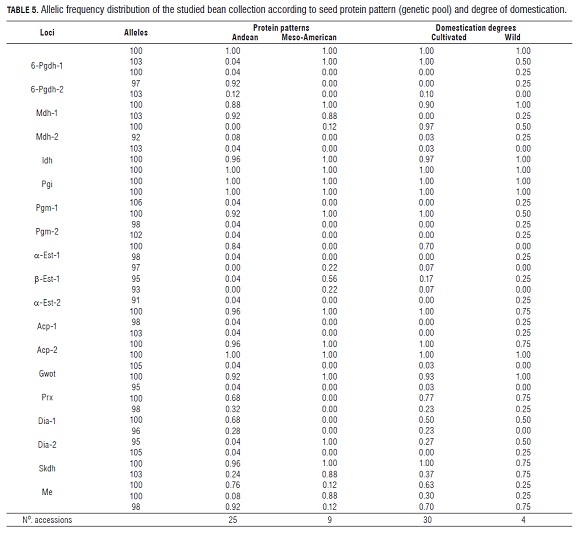
The allelic frequencies of the studied bean collection are presented in Tab.5. Exhibiting polymorphisms in 14 out of 19 loci, the Andean pool accessions registered the highest degree of genetic variability when compared to the Meso- American ones, which presented variation in only 4 loci. The alleles 6-Pgdh-2103, Mdh-1100, Idh100, a-Est-1100, a-Est-2100, and Dia-195 were specific to the Meso-American pool; whereas 6-Pgdh-1100 and Acp-2100 were only found in the Andean pool.
For the degree of domestication, the wild materials showed polymorphism in 58% of the lOCi, while the cultivated ones did so in only 47% of the lOCi. Some isozymes proved to be domestication degree specific. Thus, the alleles 100 and 97 (lOCus 6-Pgdh-2), Mdh-2103, 102, 98 and 91 (lOCus Α-Est-1), a-Est-298, Acp-1103 and Dia-2105 occurred only in wild materials; whereas Mdh-1103, 100, 97 and 93 (lOCus a-Est-1) and Dia-196 were highly frequent in cultivated accessions. Isozyme Skdh was present in both the domesticated and wild materials, with allele 103 exhibiting a high frequency (75%) among the wild accessions, and a low frequency (37%) among the cultivated ones. Conversely, allele 100 shows high frequency (63%) in cultivated materials, but low (25%) in the wild ones (Tab.5).
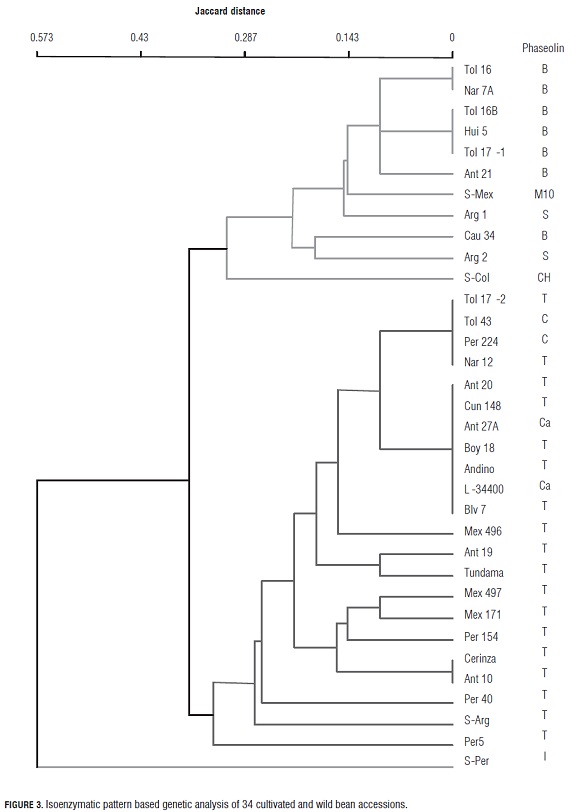
Isozymes Prx and Me had a poor representation in accessions differentiated by domestication degree, because their alleles 100 and 98 presented similar frequencies in both pools. A similar behavior was observed in d-1 with its allele 100, whose frequency was 50% in both domesticated and wild accessions. Also, its allele 96 was found in 23% of the cultivated materials, and allele 95 in 27% of the resting frequency. Among the wild accessions, allele 96 was absent and allele 95 showed a frequency of 50% (Tab.5). The conglomerate analysis of the isoenzymatic relations found in the studied bean collection (Fig.3) showed the existence of three different groups of accessions. The first one is a group including cultivated and wild accessions with typically meso-American isozyme patterns. Its cultivated accessions originated in Colombia and Argentina. The wild ones come mostly from Mexico (DGD-414). The group also includes a Colombian wild cultivar that appears to be distantly related to the rest, and exhibits type CH phaseolin.
Made up of Andean cultivars, the second group includes accessions from Colombia, Bolivia, Mexico and Peru, and a typically Andean wild one from Argentina (DGD- 626). The third group contains a single north Andean wild cultivar native to Peru, with a high degree of intraaccession isoenzymatic polymorphism and an Inca type (I) phaseolin (Fig.3).
Discussion
The presence of type B phaseolins in 78% of the Meso- American bean accessions of the studied Colombian collection confirms assertions by Gepts and Bliss (1986), Koenig et al. (1990), González et al. (2003) and Tofiño et al. (2004). According to Gepts (1988), due to the low frequency of type B phaseolin among Colombian cultivated beans (3%), this geographical zone might be considered as a minor domestication center when compared to Central America and the Andes. This is consistent with the notion that beans weredomesticated several times in the areas of distribution forwild relatives, which are found from Mexico to Argentina.
Type B phaseolin cultivars are also characterized by precocity, type III growth habit (Tab.1), small seeds, and some anthracnose (Colletotrichum lindemuthianum) and bacteriosis (Xanthomonas phaseoli) resistant materials. This does not imply that the phaseolin type is directly involved with the presence of these characteristics (Koenig et al., 1990). The considerable diversity of the studied collection (due to the presence of phaseolins S, T, B, C and Ca in the cultivated materials, and of phaseolin CH in the Colombian wild one), together with the high variability observed between the phaseolin T and phaseolin B accessions (which are typical of Central America and the northern and southern Andes) lead to the conclusion that Colombia can also be a convergence zone between Andean and Meso-American cultivated forms, and that the observed variability results from genetic flows among the cultivated varieties or among the wild and cultivated ones, as suggested by Beebe et al. (1997) in studies with Andean, Peruvian and Colombian materials. In effect, the latter include the wild-weed-cultivated complex, which has enabled the generation of variability by gene introgression from wild to cultivated populations in these geographical zones.
Isozyme analysis complements the genetic diversity and variability information obtained through the study of phaseolins (Gepts and Bliss, 1986). This allowed the separation of the studied materials (both wild and cultivated) into two main groups, as can be seen in the dendrogram in Fig.3. The first group is typically Meso-American, while the second one corresponds to the Andean pool. Additionally, there is a wild accession from the north of Peru (DGD- 1956), which stands apart due to its dissimilarity with the other two groups.
The collection's isoenzymatic pattern diversity is consistent with that of the phaseolins. The Meso-American heritage is characterized by types B and S phaseolins in the cultivated accessions, and types M10 and CH in the wild ones. In turn, the Andean cultivated accessions contain types T, C and Ca phaseolins, while the wild Argentinean one has phaseolin T. These results confirm those of other studies on common bean aloenzyme diversity (Gepts et al., 1986; Koenig et al., 1989b).
According to Koenig et al. (1990), the phaseolin I pattern of some wild materials from northern Peru lacks the (52- kD) high molecular subunit, similar to that of Colombian phaseolin types. As a result, the DGD-1956 accession is from a geographical zone that might be a transitional area between Meso-American and Andean populations. Bitocchi et al. (2012) suggest a clear pattern associated with the Mesoamerican origin of this species from which different migration events extended the distribution of P. vulgaris into South America and that P. vulgaris from northern Peru and Ecuador (type I phaseolin) is a relict population that only represents a fraction of the genetic diversity of the ancestral population and that this population migrated from central Mexico in ancient times.
Genetic variability within genetic pools was relatively high for isozymes, and low for phaseolins. In the case of isozymes, such variability was higher in the Andean pool than in the Meso-American one (Fig.3), due to the low number of Meso-American accessions included in the studied Colombian collection.
In the two studied genetic pools, the lOCi and their alleles were represented by similar frequencies to those obtained by Koenig and Gepts (1989) and Singh et al. (1991). However, allele 100 from lOCus Mdh-1 was the exception. In effect, it was found in 100% of the Meso-American accessions, and in 88% of the Andean ones. This same allele has also been observed to be frequent in the Andean pool, but not in the Meso-American one (Singh et al., 1991). In turn, Koenig and Gepts (1989) report that the genetic variability of lOCus Mdh-1 is low, and appears restricted to the Andean pool (mainly to the south of Peru and to Argentina). Among the wild materials, the highest polymorphism was observed in the Peruvian accession, which combines northern Andean and Meso-American isoenzymatic patterns.
Despite the limitation that is inherent to isozymes as molecular markers in beans (an autogamous species with few lOCi and few alleles per lOCus), an important diversity was observed in the studied collection. This corroborates the notion that Colombia is a domestication center for this species, whose two ancestral branches (Andean and Meso-American) converge in the region; where there has been not only introgression of wild genes into cultivated materials, but also gene flow between the two pools. This might facilitate the transfer of quantitative characteristics between Andean and Meso-American cultivars, where there have been recombination difficulties.
Acknowledgments
The authors thank the Research Division of the Universidad Nacional de Colombia, Bogota and Colciencias for financial support.
Literature cited
Avendaño, C.H., P. Ramírez, F. Castillo, J.L. Chávez, and G. Rincón. 2004. Diversidad isoenzimática en poblaciones nativas de fríjol negro. Rev. Fitotec. Mex. 27(1), 31-40. [ Links ]
Beebe, S., O. Toro, A.V. González, M.I. Chacón, and D.G. Debouck. 1997. Wild-weed-crop complexes of common bean (Phaseolus vulgaris L. Fabaceae) in the Andes of Peru and Colombia, and their implications for conservation and breeding. Gen. Res. Crop Evol. 44, 73-91. [ Links ]
Bitocchi, E., L. Nanni, E. Bellucci, M. Rossi, A. Giardini, P.S. Zeuli, G. Logozzo, J. Stougaard, P. McClean, G. Attene, and R. Papa. 2012. Mesoamerican origin of the common bean (Phaseolus vulgaris L.) is revealed by sequence data. PNAS 109(14), E788-E796. [ Links ]
CIAT , International Center for Tropical Agriculture. 1996. Protocolos de laboratorio de electroforesis. Cali, Colombia. [ Links ]
Debouck, D.G., O. Toro, O.M. Paredes, C.W. Johnson, and P. Gepts. 1993. Genetic diversity and ecological distribution of Phaseolus vulgaris (Fabaceae) in Northwestern South America. Econ. Bot. 47(4), 408-423. [ Links ]
Debouck, D.G. 1996. Observaciones sobre la distribución de los recursos fitogeneticos de Nuevo Mundo en condiciones in situ y ex situ, p. 28. In: Informe de la reunión regional para América Latina y el Caribe sobre recursos fitogenéticos. Corpoica, Bogota. [ Links ]
Gepts, P. and F.A. Bliss. 1986. Phaseolin variability among wild and cultivated common beans (Phaseolus vulgaris) from Colombia. Econ. Bot. 40(4), 469-478. [ Links ]
Gepts, P., T.C. Osborn, K. Kashka, and F.A. Bliss. 1986. Phaseolinprotein variability in wild forms and landraces of commom bean (Phaseolus vulgaris): Evidence for multiple centers of domestication. Econ. Bot. 40(4), 451-468. [ Links ]
Gepts, P. 1988. Genetic resources of Phaseolus beans. Kluwer Academic Publishers, Dordrecht, The Netherlands. pp. 215-241. [ Links ]
Gepts, P. 1991. La biotecnología aclara el proceso de domesticación del fríjol. Diversity, 7(1-2), 52-53. [ Links ]
González, R., E. Gaitán, M. Duque, O. Toro, J. Tohme, and D. Debouck. 2003. Monitoring gene flow between wild relatives and landraces of common bean in Costa Rica. Annual Report Bean Improvement Cooperative 46, 1-2. [ Links ]
Harch, B.D. 1995. Estatistical evaluation of germplasm collections. Ph.D. thesis. Department of Agriculture, University of Queensland, Brisbane, Australia. [ Links ]
Hussain, A., H. Ramírez, W. Bushuk, and W. Roca. 1986. Field bean (Phaseolus vulgaris L.) cultivar identification by electrophoregrams of cotyledon storage proteins. Euphytica 35, 729-732. [ Links ]
Hussain, A., W. Bushuk, H. Ramirez, and W. Roca. 1987. Procedimientos de electroforesis en geles de poliacrilamida para la identificación de cultivares de fríjol, yuca y leguminosas forrajeras. Documento de trabajo No. 22. Cali, Colombia. [ Links ]
Kwak, M., J.A. Kami, and P. Gepts. 2009. The putative mesoamerican domestication center of Phaseolus vulgaris is lOCated in the Lerma–Santiago basin of Mexico. Crop Sci. 49(2), 554-563. [ Links ]
Kwak, M. and P. Gepts. 2009. Structure of genetic diversity in the two major gene pools of common bean (Phaseolus vulgaris L., Fabaceae). Theor. Appl. Genet. 118, 979-992. [ Links ]
Koenig, R., and P. Gepts. 1989a. Segregation and linkage of genes for see proteins, isozymes, and morphological traits in common bean (Phaseolus vulgaris). J. Hered. 80, 455-459. [ Links ]
Koenig, R. and P. Gepts. 1989b. Allozime diversity in Wild Phaseolus vulgaris: further evidence for two major centers of genetic diversity. Theor. Appl. Genet. 78, 809-817. [ Links ]
Koenig, R., S.P. Singh, and P. Gepts. 1990. Novel phaseolin types in wild and cultivated common bean (Phaseolus vulgaris, Fabaceae). Econ. Bot. 44(1), 50-60. [ Links ]
Ligarreto, G.A. 2005. Uso de índices de crecimiento y caracteres relacionados con la fotosíntesis para el análisis de la variabilidad genética de fríjol común (Phaseolus vulgaris L.). Fitot. Colomb. 5(1), 23-35. [ Links ]
Ma, Y. and F.A. Bliss. 1978. Seed proteins of common bean. Crop Sci. 18, 431-437. [ Links ]
Maquet, A., I.Z. Bi, M. Delvaux, B. Wathelet, and J.P. Baudoin. 1997. Genetic structure of a lima bean base collection using allozyme markers. Theor. Appl. Genet. 95, 980-991. [ Links ]
Matsuoka, Y., Y. Vigouroux, M.M. Goodman, G.J. Sanchez, E. Buckler, and J. Doebley. 2002. A single domestication for maize shown by multilOCus microsatellite genotyping. Proc. Natl. Acad. Sci. USA 99, 6080-6084. [ Links ]
Podani, J. 1993. SYN -TA X-PC computer programs for multivariate data analysis in ecology systematic, version 5.0. Scientia Publishing, Budapest. [ Links ]
Rohlf, F.J. 1998. Ntsys. Numerical taxonomy and multivariate analysis system. Version 2.0. Exeter Publ., Setauket, NY . [ Links ]
Sanjur, O., D. Piperno, T.A. and L. Wessel-Beaver. 2002. Phylogenetic relationships among domesticated and wild species of Cucurbita (Cucurbitaceae) inferred from a mitochondrial gene: Implications for crop plant evolution and areas of origin. PNAS 99(1), 535-540. [ Links ]
Singh, S.P., R. Nodari, and P. Gepts. 1991. Genetic diversity in cultivated common bean: I. Allozymes. Crop Sci. 31, 19-23. [ Links ]
Tofiño, A.P., J.A. Gutiérrez, C. ocampo, and V.H. García. 2004. Estudio de la variabilidad genética en habichuela Phaseolus vulgaris L., mediante descriptores morfológicos y bioquímicos. Acta Agron. 53(1), 25-36. [ Links ]
Triana, M. and W. Roca. 1989. Estudio electroforético de germoplasma de fríjol (Phaseolus vulgaris L.) en centros secundarios de diversidad genética: El caso de África sur –oriental. Acta Agron. 39(1-2), 18-23. [ Links ]
Weeden, N.F. and C.Y. Liang. 1985. Detection of a linkage between flower color and Est-2 in common bean. Annu. Rep. Bean Impr. Coop. 27, 87-88. [ Links ]