Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Agronomía Colombiana
Print version ISSN 0120-9965
Agron. colomb. vol.30 no.2 Bogotá May/Aug. 2012
CROP PROTECTION
Monitoring the infective process of the downy mildew causal agent within micropropagated rose plants
Seguimiento del proceso infectivo del agente causal del mildeo velloso en plantas micropropagadas de rosa
Sonia Yamile Gómez1 and Juan José Filgueira-Duarte2
1Faculty of Agricultural Sciences, Universidad Nacional de Colombia. Medellin (Colombia). juan.filgueira@unimilitar.edu.co2Faculty of Science, Universidad Militar Nueva Granada. Bogota (Colombia). Received for publication: 23 July, 2011. Accepted for publication: 29 June, 2012.
ABSTRACT
Downy mildew in the rose caused by a species of the Peronospora genus is a very restrictive disease for the Colombian greenhouse rose production. The damage observed in the susceptible varieties of commercial rose include symptoms affect young steams and tiny leaves causing reddish and brown spots and defoliation; leading to 10% production losses. The infective behavior of this pathogen was studied with the aim of increasing the knowledge about the biology of the rose downy mildew. The study of the infective process was performed on the Charlotte variety using micropropagated roses inoculated with suspensions of sporangia. A germinal tube was observed during the germination process, it came from a lateral papilla and reached up to 300 microns in length. During this study, the ability of the pathogen to use vascular sieves as communication systems within the plant was determined. Oogonia and antheridia were also observed inside the epidermal cells, and oospores inside the parenchymal tissue close to xylem vessels. To the best of our knowledge, these sexual structures have not been reported on in Colombia before. This study verifies the ability of the downy mildew causal agent to move through the xylem vessels and produce sexual structures, such as oogonia, antheridia and oospores within those tissues.
Key words: Peronospora sparsa, infection, sexual structures, in vitro propagation.
RESUMEN
El mildeo velloso de la rosa ocasionado por una especie del género Peronospora, es una enfermedad muy restrictiva para la producción de rosas de corte en Colombia. Los daños que se observan en las variedades susceptibles de rosa comercial, incluyen síntomas que afectan tallos jóvenes y hojas tiernas causándoles manchas rojizas y cafés al igual que defoliación; lo que conlleva a pérdidas de hasta el 10% de la producción. El comportamiento infectivo de este patógeno fue estudiado con el fin de aportar al conocimiento sobre la biología del mildeo velloso de la rosa. El estudio del proceso infectivo fue realizado sobre plantas micropropagadas de la variedad susceptible Charlotte inoculadas con suspensión de esporangios del patógeno. Un tubo germinal fue observado durante el seguimiento al proceso de germinación, provenía de una papila lateral y alcanzó 300 micras de longitud. Durante este estudio pudo ser determinada la habilidad del patógeno para usar los haces vasculares como sistemas de comunicación dentro de la planta. Oogonios y anteridios fueron observados dentro de células epidermales infectadas, al igual que oosporas dentro del tejido parenquimático cerca de los vasos conductores. Hasta donde se sabe en Colombia no se habían reportado estructuras sexuales antes para este patógeno. Este trabajo verifica la capacidad del agente causal del mildeo velloso de la rosa para moverse a través de los vasos del xilema y de producir estructuras sexuales tales como oogonios, anteridios y oosporas dentro de dichos tejidos.
Palabras clave: Perosnopora sparsa, infección, estructuras sexuales, propagación in vitro.
Introduction
Rose downy mildew is caused by Peronospora sparsa Berkeley (Aegerter et al., 2003), an oomycete biotrophic parasite. This organism affects susceptible varieties of roses causing losses by deformity, blights on leaves, stems and twigs and defoliation (Horst, 1998), (Fig.1). To control this disease, intensive applications of foliar fungicides (preventive and curative agrochemical products) and intensive labor is needed to eradicate affected material, causing burdensome costs to the Bogota Plateau rose production (Arbeláez, 1999). According to Suárez (1999), the cost of downy mildew due to losses in the 1,950 ha of roses on the Bogota Plateau was US $5.675.670.
The lack of public information about the biology of this pathogen in particular has forced people in Colombia to use information from other related crops or crops affected by another type of downy mildew, for example onion downy mildew caused by Peronospora destructor in Canada (Hildebrand and Sutton, 1985), or in lettuce caused by Bremia lactucae in Europe (Sargent, 1981). Other major producers of roses, such as the Netherlands, do not consider Peronospora sparsa as a significantly important pathogen and so do not invest economic sources for research into this particular downy mildew. This is because the conditions in the climatic controlled glasshouses where these roses are produced are not favorable for the downy mildew which is especially sensitive to temperature and humidity to complete its reproductive cycle (De Vis, Raf, 1999).
Studies performed in Colombia have tried to clarify different aspects of the biology of this biotrophic pathogen such as the effect of some climatic variables on the disease incidence, the effect of the grafting variety on the sensitivity of the floral variety and the phenological state of the plant in the pathogen life cycle, among others (Giraldo, 1999). Although knowledge about rose downy mildew has advanced, today, there are still too many unanswered questions about its biology. For example, in Colombia, no sexual structures have been observed before and there is no conclusive evidence about its systemic behavior. Although observations of wounds on affected greenhouse plants could show the pathogen was able to spread from this white marks using vascular sieves to distant parts of the plant. However, for other Peronosporales sexual structures have been already reported but systemic behavior has not been proved yet (Arbeláez, 1999; De Vis, Raf, 1999; Giraldo, 1999).
The objective of this study is to present the monitoring of the infective process of the rose downy mildew causal agent within the Charlotte variety in in-vitro microplants under laboratory conditions.
Materials and methods
Rose downy mildew inoculum was collected from sporulating leaves at MG Consultores greenhouses in Chia Cundinamarca, Colombia. In the laboratory, this inoculum was concentrated sweeping the lesson using a little brush over 20 mL of sterile distillated water (SDW), then the concentration was measured using an haemocytometer. The viability of the inoculum suspension was then evaluated on water agar media under room conditions. Every hour, the germination process was observed to obtain the percentage of sporangia germinated after 24 h. Once sporangia viability was confirmed, healthy clean detached leaves of the Charlotte variety were inoculated with the suspension using the fail drop technique over the abaxial side, and they were incubated under room conditions until sporulation was produced 4 or 5 d later. Using this technique to collect and transfer sporangia from detached leaves, the inoculum was also cleaned from spores of any other microorganisms. Rose microplants were produced in the laboratory introducing buds into the supplemented Murashige y Skoog (1962) agar media (4.4 g L-1 MS , 2.5 g L-1 Phytagel, 30 g L-1 sugar, 10 mg L-1 BAP, 60 mg mL-1 AIA and 224 mg L-1AG3, pH 5.6). The buds were incubated with the following climatic controlled conditions: 26°C, 12 d/night h, and 6,000 lux of white light. After 6 weeks, the buds were separated from the woody part and then reintroduced into a new MS supplemented media. Then, the rose microplants were infected with the sporangia suspension adjusted to 1x104 sporangia/mL in 20 mL SDW using the 15 s immersion technique; the youngest leaf from each microplant was inoculated. For drying, the microplants were inverted for 4 h at the center of the flux chamber and then were incubated under room conditions. Every 2 h, raised hand transversal cuttings from a sample were taken and immersed in 3 mL of a fix solution (0.2 M buffer phosphate, 150 mM NaCl and 2% Glutaraldehide). The thinnest cuttings were then dyed with cotton blue dye on a microscope slide. With a optic microscope, the cuttings were observed at x40, to determine absence/presence of the pathogen and then at x100 to establish the sort of plant tissue colonized and the type of pathogen structures. The infection process was monitored continuously for 92 h post inoculation (hpi).
Results and discussion
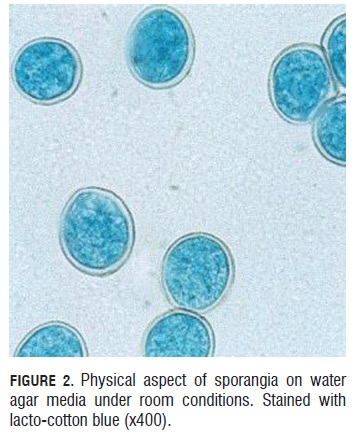
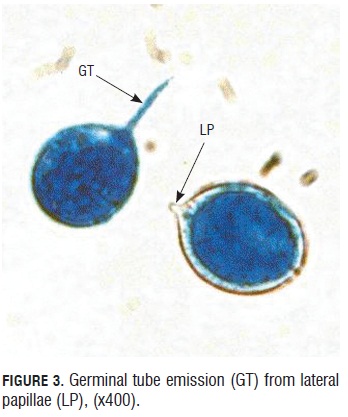
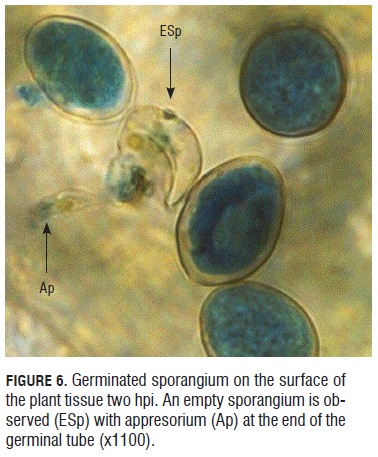
Sporangia viability test. During the germinative capacity of the sporangia evaluation, it was observed that the sporangia that germinated were bigger than 20 microns long by 15 microns wide and also with a double well formed and turgid cell wall that presented a light blue color of the cytoplasm due to the lacto cotton blue stain (Fig.2). Sporangium begins to germinate one hour after under room conditions. They emitted a germinal tube from lateral papillae. (Fig.3).
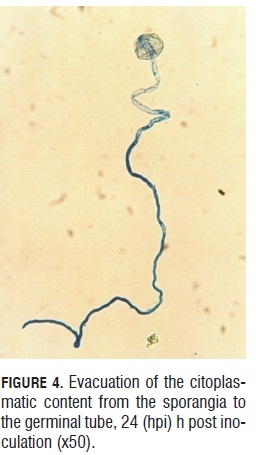
During germination, a cellular content evacuation event from the sporangia to the germinal tube was observed. This can be affirmed because the germinated sporangia lose the blue intensity given by the lacto-cotton blue dye (Fig.4a and 4b). Germinated sporangia can produce germinal tubes up to 300 microns in length in 24 h, allowing translocation for the landing site, searching for more suitable places to initiate the infective process (Fig.4 b).
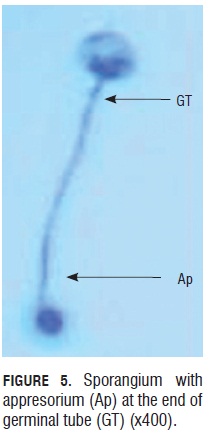
Germination percentages of the five trials had values ranging from 70.5 to 80% in the first six to eight h of monitoring. These are relatively high values if we consider they come from inoculum constantly under the pressure of fungicide application to control this disease in the field. Although Quiroga and Arbeláez (2004) reported that foliar applications of agrochemicals like dimethomorph, aluminum phosetyl and metalaxyl with mancozeb fungicides affected the sporangia shape and turgid, during this study, it was observed that sporangia recollected from the field in MG Consultores were not affected in their shape and still able to germinate, which means the products used for controling the disease during this test either were not effective enough or agrochemicals made a selection of those with better fitness to keep the disease in the field. Furthermore, it was clear that there was a relationship between the size and turgor of the sporangia and the germination capability, thus those small ones and non-turgid ones lacked the ability. In this evaluation, appresorium formation from the germinal tube was observed. These appresoria were eight to 10 microns in diameter and were easy to see at the terminal end of the germinal tube. They were observed in the absence of plants between six to eight hours after the germination process started, under room conditions.
Evaluation of the sporangia germination percentage in the water media agar after 24 h of incubation showed the capacity of these structures to produce long germinal tubes, over 100 microns. This was reported by Shiltz (1981) for other Peronosporales such as Peronospora hyoscyami . The length reached by the germinal tubes in 24 h is a very important advantage for the pathogen, because a long germinal tube has the chance to access places far from the point where it originally fell. Sporangia with more than one germinal tube were also observed during this work (not shown in this document). This characteristic is also reported for other downy mildews such as Bremia lactucae in lettuce according to Sargent (1981) and Reuveni et al. (1986) for Peronospora tabacina.
Monitoring the infective process of downy mildew in rose microplants. Two h after sporangia contact with the youngest leaf of the rose microplants, germination initiated as shown in Fig.6. The pointed sporangium is not turgid as those next to it, on the contrary, it is assumed that it has germinated because it has evacuated its cellular content and an appresorium ready to penetrate the plant tissue is observed at the end of the germinal tube.
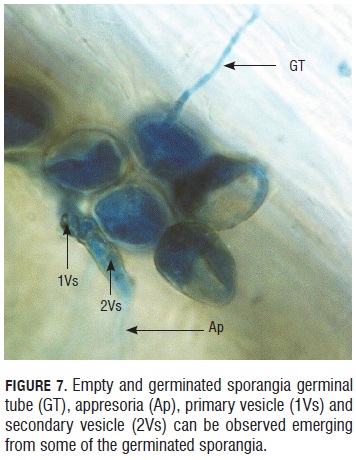
In Fig.7, 4 hour post inoculation (hpi), it was possible to see evacuation from sporangia. Some of them had different structures formed during the germination, for example: appresorium, primary vesicle and secondary vesicle.
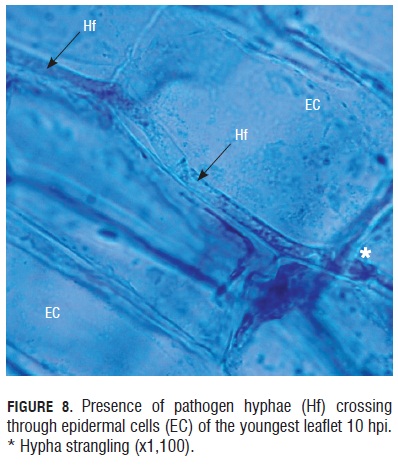
The infective process of the pathogen continued with the presence of hyphae in the epidermal cells after 10 hpi. Hyphae crossed inside, one by one, of the epidermal cells and had the capacity to modify the cells thickness to gain entrance to new plant tissue, thus, strangling the hypha in some portions, as shown in Fig.8.
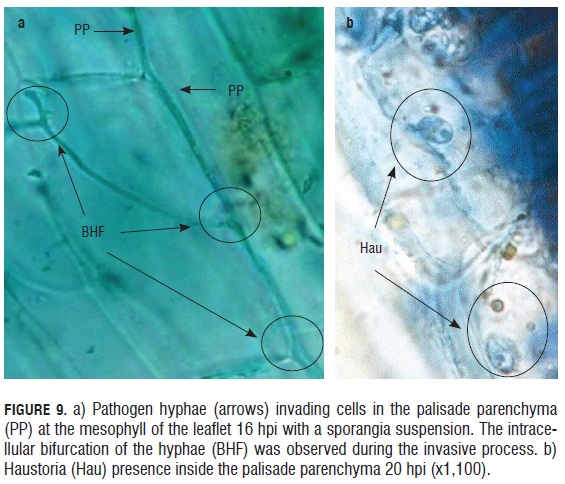
In the monitoring of the infective process of the downy mildew causal agent in in vitro rose microplants, a rapid invasion of the pathogen into the leaflet epidermal tissue was observed in less than 12 h. The advance of the pathogen was registered both inter and intracellular. Although it did not affect the cytoplasmic membrane of the cells, and perhaps for this reason, cellular lysis was not present in the areas infected by the pathogen. Thus, the biotrophic behavior of the pathogen was evident, colonizing the plant tissue without damaging the cells of the host 16 hpi, pathogen hyphae were observed bifurcating within the palisade parenchyma cells (Fig.9). Similarly, 20 hpi, hyphae were evident inside the parenchyma cells along with haustorium within subepidermal cells.
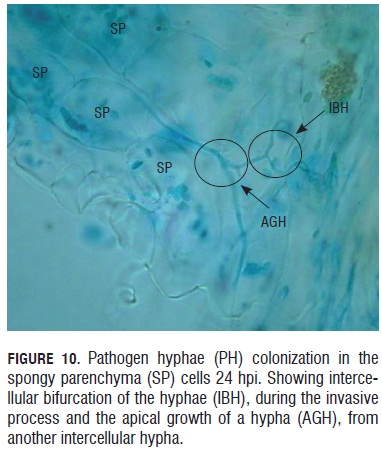
Twenty-four hours after the inoculation event, pathogen hyphae were seen growing and branching in the spongy parenchyma (Fig.10). In this Fig.10, it is possible to observe how a new hypha has an apical growth and is produced from another hypha that is already formed. In this form, the hypha can colonize the neighboring tissue in a fast and effective way.
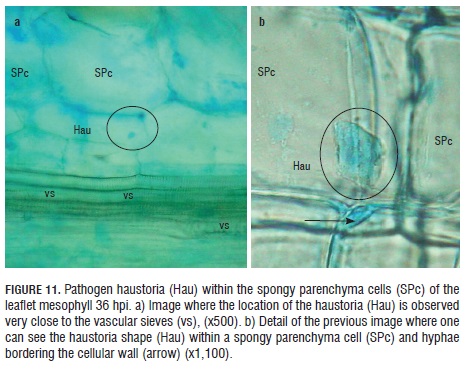
Thirty-six hpi, haustoria were observed inside the spongy parenchyma cells next to the vascular sieves (Fig.11 a). With higher resolution, it was possible to see the appendix shape of the haustoria close to the parenchymal cell wall, where this structure is lodged, as well as the pathogen hyphae, by color contrast, bordering the spongy parenchyma cells, where it is located (Fig.11 b).
The mesophyll images showed the presence of the pathogen hyphae in a short period of time, between 16 and 24 hpi. These hyphae invaded the parenchyma cells in a very efficient way. According to Crute (1981), in the lettuce downy mildew, the advance of B. lactucae is slower, since it occurs in 96 hpi. Besides, in B. lactucae, bifurcations have not been reported yet, while in rose downy mildew, this type of division is evident and could explain the efficiency of the pathogen to colonize the plant tissue.
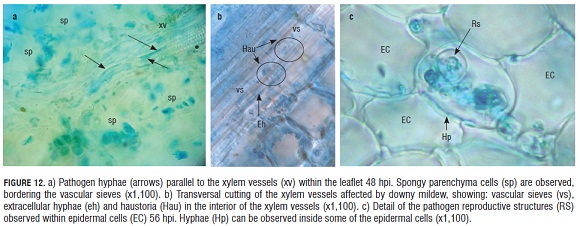
After 48 hpi, hyphae of the pathogen were observed growing in parallel, inside and outside of the xylem vessels within the vascular tissue of the infected leaflet (Fig.12).
Parasite presence in the xylem tissue of the leaflet was evident at 48 hpi, when the pathogen hyphae were observed as aligned to xylem vessels and simultaneously inside this tissue.
There were no reports or photographic evidence about the invasion of Peronosporales into conductive tissues. Although for the lettuce downy mildew, Crute and Dixon (1981) talk about systemicity for the B. lactucae capacity to move from cotyledons to roots, but just for young plants.
Colombian downy mildew reports refer to this physiological trait of systemic growth as a characteristic of the pathogen. Due to the behavior of the pathogen under greenhouse conditions and the presence of symptoms such as white spots on the stems with reinfection quality, it is evident that the pathogen has the capacity to move inside the vascular system and remain in this tissue during difficult weather conditions to later reinfect young buds when environmental conditions get better. In this study, the presence of the rose downy mildew causal agent was reported inside the vascular tissue of the microplants. This shows the capacity of this pathogen to move through vascular sieves as a conductive mechanism to invade other tissues in the plant and perhaps it also uses this tissue as a shelter to remain viable inside the plant during unfavorable weather conditions.
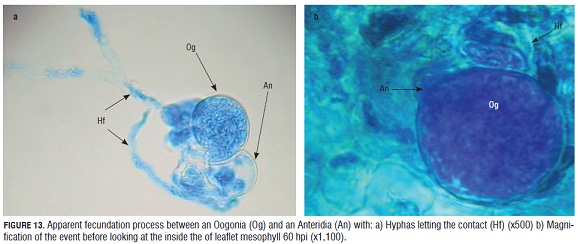
In a more detailed observation of the reproductive structures registered inside the epidermal cells, an antheridia and an oogonia were clearly observed in very close contact (Fig.13).
Between 56 and 68 hpi, sexual structures began to be observed within the epidermal cells of the microplants. There are no previous reports about these kind of structures for this pathogen in roses, in addition, it was believed that it was not necessary for the pathogen to produce these types of structures in the climatic conditions of the Bogota Plateau. Nevertheless, once we compared our data with the reports of Svircev et al. (1989) on the histology of P. hyoscyami and Ingram (1981) for P. parasitica, it is clear to see the similarity between the oogonia and antheridia of these species with those found in the epidermis of the rose microplants which were inoculated with the downy mildew sporangia suspension. The only difference was the speed of oogonia development during P. sparsa infection which occurs four dpi while P. hyoscyami and P. parasitica develop oogonia eight and 12 dpi, respectively. It is possible to conclude that the in vitro rose microplants conditions are preferable for the formation of these sexual reproductive structures.
Fig.13 a shows the contact among the oogonium and the antheridia of the pathogen and some hyphae that connect these two structures. These hyphae come from different origins, thus, it looks like an encounter between two homothallic strains according to Michelmore et al. (1988) for P. parasitica, who observed homothallic and heterothallic strains responsible for the presence of either oogonia, antheridia and oospores or the asexual condition marked by the sporangiophore and sporangia formation respectively. It is not clear yet if the downy mildew causal agent of roses in Colombia has homothallic or heterothallic populations. More studies on this aspect would be important in clarifying the dynamic of this pathogen in the field.
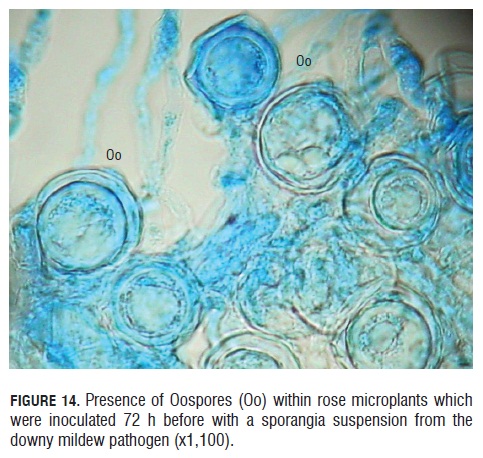
Finally, in the observation from to the 72 hpi, it was seen the product of the sexual reproduction from the antheridium and oogonium, the oospores (Fig.14). These oospores had a very clear round shape and a double wall covering the whole perimeter of the oospore. Many oospores were observed 60 hpi in the in vitro rose microplants, and the vicinity of these oospores to the vascular sieves was also registered (See Fig.14). According to Michelmore et al. (1988), three elements are required to make the establishment of the B. lactucae oospores possible on the foliar tissue and apparently they were present in this study: presence of the pathogen mating types, some infection level and the pathogen growing pattern inside the host ensure an encounter of the two compatible mating types.
Finally, in the observation from to the 72 hpi, it was seen the product of the sexual reproduction from the antheridium and oogonium, the oospores (Fig.14). These oospores had a very clear round shape and a double wall covering the whole perimeter of the oospore. Many oospores were observed 60 hpi in the in vitro rose microplants, and the vicinity of these oospores to the vascular sieves was also registered (See Fig.14). According to Michelmore et al. (1988), three elements are required to make the establishment of the B. lactucae oospores possible on the foliar tissue and apparently they were present in this study: presence of the pathogen mating types, some infection level and the pathogen growing pattern inside the host ensure an encounter of the two compatible mating types.
The importance at the genetic level of a structure produced after sexual reproduction is evident in the case of diseases caused by Oomycetes, as in the case of Phytophthora infestans. In the case of Peronosporales, the role of the oospores in the downy mildew epidemiology is not yet clear and is proposed to be a very susceptible structure to hyperparasites attack (Svircev et al., 1989).
Conclusions
Sporangia from rose downy mildew collected from a commercial rose field were able to germinate in 24 h in spite of the use of agrochemicals to control the disease. Only those sporangia with a big size and turgid shape were able to germinate in the water agar under room conditions. The management of the disease in the field using fungicides has contributed to the selection pressure to favor those with higher fitness. Under laboratory conditions and in invitro microproagated rose plants, the pathogen was able to colonize the host tissue in inter and intracellular manners, reaching the conductive vessels 48 h after inoculation. The biotrophic behavior of the pathogen could be proven with the observation of intercellular hyphas without necrosis in the tissue, as well as haustorium structures inside unaffected cells. In comparison with other Peronosporales such as P. parasitica and P. hyoscyami, the downy mildew causal agent in roses is faster at invading the host tissue when in-vitro micropropagated rose plants are used as the experimental field.
The presence of the pathogen in the xylem vessel 48 h after inoculation could explain its systemic capacity to grow and move inside the plant, reaching very distant places in short periods of time. Under the in-vitro micropropagated conditions and between 56 and 68 hpi, the downy mildew causal agent of the rose produced sexual structures not reported before in Colombia. The presence of sexual structures inside an enclosed controlled space could indicate that the strain of the pathogen used in this study was homothallic. The systemically behavior of the pathogen observed in this study could explain the presence of downy mildew symptoms like white spots in the stems as strategies of the pathogen to remain in the plant when environmental conditions are not favorable.
This new knowledge will facilitate the development, in a short time, of molecular techniques that permit the monitoring of systemic growth throughout the plant, which will change the usual methods of chemical treatment. Just in our laboratory, we are introducing reporter genes into the sporangia using the spheroplast method and Agrobacterium transformation.
Acknowledgements
We would like to express gratitude to the Faculty of Science at the Universidad Militar "Nueva Granada" (UMNG), also, this work was supported by the CIAS of UMNG.
Literature cited
Aegerter, B.J., J.J. Núñez, and R.M. Davis. 2003. Environmental factors affecting rose downy mildew and development of a forecasting model for a nursery production system. Plant Dis. 87, 732-738. [ Links ]
Arbeláez, G. 1999. El mildeo velloso del rosal ocasionado por Peronospora sparsa Berkeley. Acopaflor 6, 37-39. [ Links ]
Crute, I.R. 1981. The host specificity of Peronosporaceous fungi and the genetics of the relationship between host and parasite. pp. 237-250. In: Spencer, D.M. (ed.). The downy mildews. Academic Press, New York, NY. [ Links ]
De Vis, R. 1999. Manejo de mildeo velloso en rosa con control climático. Acopaflor 3, 31-33. [ Links ]
Giraldo, K.C.A. 1999. Control cultural del mildeo velloso y la ecocultura de la capacitación. Acopaflor 6, 4-12. [ Links ]
Gómez, S.M. 2002. Influencia de la temperatura en la germinación de esporangios de Peronospora sparsa, en rosa. Agron. Colomb. 20, 35-38. [ Links ]
Gómez, S. 2004. Determinación de componentes de la biología de Peronospora sparsa Berkeley, y caracterización de la respuesta de tres variedades de rosa a la infección del patógeno bajo condiciones de laboratorio e invernadero. Bogotá. M.Sc. thesis. Faculty of Agronomy, Universidad Nacional de Colombia, Bogota. [ Links ]
Hildebrand, P.D. and J.C. Sutton. 1985. Environmental water in relation to Peronospora destructor and related pathogens. Can. J. Plant Pathol. 7, 323-330. [ Links ]
Horst, R.K. 1998. Compendio de enfermedades de la rosa. APS Press. St. Paul, MN . [ Links ]
Ingram, D.S. 1981. Phisiology and biochemistry of host-parasite interaction. pp. 143-163. In: Spencer, D.M. (ed.). The downy mildews. Academic Press, New York, NY . [ Links ]
Michelmore, R.W., S.H. Hulbert, T. Ilott, and B. Farrara. 1988. The downy mildews. Adv. Plant Pathol. 6, 55-76. [ Links ]
Murashige, T. and E. Skoog. 1962. A revised medium for rapid grown and bioassays with tabacco tissue culture. Physiol. Plant 15, 473-497. [ Links ]
Quiroga B., N.J. and G. Arbeláez T. 2004. Evaluación de la eficacia de fungicidas aplicados al suelo y al follaje para el control de mildeo velloso, ocasionado por Peronospora sparsa en un cultivo comercial de rosa. Agron. Colomb. 22, 110-118. [ Links ]
Reuveni, M., W.C. Nesmith, and M.R. Siegel. 1986. Symptoms development and disease severity in Nicotiana tabacum and Nicotiana remanda caused by Peronospora tabacina. Plant Disease. 70, 727-729. [ Links ]
Sargent, J.A. 1981. The fine structure of the downy mildews. pp. 183- 236. In: Spencer, D.M. (ed.). The downy mildews. Academic Press, New York, NY . [ Links ]
Schiltz, P. 1981. Downy mildew of tobacco. pp. 577-594. In: Spencer, D.M. (ed.). The downy mildews. Academic Press, New York, NY. [ Links ]
Suárez, G.J. 1999. Problemática actual del mildeo velloso en rosa. In: VIII Congreso Acopaflor - Asocolflores. Bogota. [ Links ]
Svircev, A.M., W.E. McKeen, and R.J. Smith. 1989. Host-parasite relations: morphology and ultrastructure. pp. 43-104. In: McKeen, W.E. (ed.). Blue mold of tobacco. APS Press, St. Paul, MN . [ Links ]