Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Agronomía Colombiana
Print version ISSN 0120-9965
Agron. colomb. vol.30 no.3 Bogotá Sept./Dec. 2012
CROP PHISIOLOGY
Aspects to consider for optimizing a substrate culture system with drainage recycling
Aspectos a tener en cuenta para optimizar un sistema de cultivo en sustrato con reciclaje de drenajes
William J. Cuervo B.1, Víctor J. Flórez R.1 and Carlos A. González M.2
1Department of Agronomy, Falculty of Agronomy, Universidad Nacional de Colombia. Bogota (Colombia). vjflorezr@unal.edu.co2Department of Civil Engineering and Agricultural, Faculty of Engineering, Universidad Nacional de Colombia, Bogota (Colombia).
Received for publication: 30 Aprol, 2010. Accepted for publication: 30 October, 2012.
ABSTRACT
In Colombia, the soilless system has been implemented in the cut flowers industry, among others, due to soil limitations. The use of substrates as growing media implies to drain around 30% of the applied fertigation solution to avoid the rhizosphere salinization. The drainage solution is spilled out to the soil producing environmental hazards and it might reach the water table; although the drainage solution could be recycled or reuse, depending upon their chemical characteristics. The nutrient uptake by the plants depends upon their phenological stage and the nutrient concentration in the solution; which could lead to ion depletion or accumulation. In general, monovalent ions are withdrawn faster than divalent ones. An efficient drainage treatment involves the automation of sensing and evaluation of ion concentration and recycling the drainage solution. The system should take into account the chemical aspects in the recycled and the new solutions in order to predict the life time of the drainage solution from their EC and pH. The system must be integrated with disinfection methods to avoid the spreading of plant pathogens. This review point out the physiological and technical bases that should be taking into account in a drainage recycling system in established crops under substrates, as a tool to take decisions more efficiently.
Key words: leaching, pollution, automation and control, soilless cultivation, ion imbalance.
RESUMEN
En Colombia, el sistema de cultivo sin suelo es una práctica común en los cultivos de flores de corte, debido entre otras causas, a las limitaciones de algunos suelos para la producción. Utilizar sustratos como medio de crecimiento implica generar como drenaje aproximadamente el 30% del total del fertirriego aplicado, para evitar la salinización de la rizosfera. Comúnmente estos drenajes son vertidos al suelo con los consecuentes problemas de contaminación, que podría alcanzar el nivel freático; aunque sus características químicas son apropiadas para ser recirculados o reutilizados. La absorción de nutrientes por la planta depende del estadio fenológico y de la concentración de iones en la solución; lo que puede llevar al agotamiento o acumulación de iones. De forma general, los iones monovalentes son removidos más rápido de la solución que los divalentes. Un reciclaje eficiente requiere captar, evaluar y reciclar la solución automáticamente; para lo cual es necesario considerar aspectos químicos de las soluciones nueva y reciclada, para estimar la vida útil de la solución drenada a partir de CE y pH. Así mismo, integrar métodos de desinfección para evitar la dispersión de patógenos. Esta revisión presenta las bases técnicas, avances tecnológicos y fundamentos fisiológicos que enmarcan y definen las características de un sistema de reciclaje de drenajes en cultivos establecidos en sustratos, como herramienta en la toma de decisiones de manera eficiente
Palabras clave: lixiviados, contaminación, automatización y control, cultivo sin suelo, desbalance iónico.
Introduction
The rising costs of production due to increased use of protected agriculture have foster the farmers to increase productivity using different technological advances, including the use of substrates (Raviv and Lieth, 2008). One reason that justifies the change from soil culture to substrate cultivation is the proliferation of diseases, as in the case of vascular wilt in cut carnations in Colombia (Pizano de Marquez, 2001). In order to control the causative agent (Fusarium oxysporum f.sp. dianthi), expensive chemical applications were used (Patiño, 2000). Another factor is the control of fertilizer solution variables, such as pH, electrical conductivity (EC) and ion concentration, improving productivity (Olympios, 1999).
The trend to increase productivity for agriculture is encompassed by the growing use of fertilizers, particularly inorganic nitrogen. Use of this component peaked at 11 million t annually in the mid-1980s and recently dropped to about 9 to 10 million t (European Commission, 2000). Since 2000 year, in the Netherlands, the law establishes the use of closed systems or recycling of the nutrient solution in 100% of cultivated areas to mitigate pollution, particularly of surface waters (Van Os, 1999). In Mediterranean countries, the law states that leaching from soilless culture systems (SCS ) should be treated as industrial discharge. For crops growing in nitrate vulnerable zones, it should be applied irrigation methods allowing maximum water use efficiency (Marfà et al., 2000).
In carnation production with open substrate culture systems, it is estimated that 4,200,000 m3 year-1 of water and 2,000 t year-1 of salt fertilizers are applied, based upon 30 to 40% leaching solution (Van Kooten et al., 2004). Fate of contaminant in water solution legislation in Colombia (Vélez, 2010) points out during the diagnostic phase, it should be identified and characterized the problems caused by imbalances in the natural Environment as well as the natural and man-made degradation in quality or quantity of renewable natural resources and environmental impacts, among others. The regulation seeks to establish the parameters and maximum permissible values for discharge to surface and groundwater. It refers to pollution produced by activities such as growing cut flowers and sets up maximum allowable limits for many of the common effluent substances in agroindustrial processes (Ministerio de Ambiente, Vivienda y Desarrollo Territorial, 2011).
The aim of this review is to show the technical basis, technological advances and physiological fundamentals framing the characteristics of a recycling drainage system in substrate cultivation, as a tool for more efficient use of resources in horticultural production.
Soilless culture
From 2,000 m3 ha-1 of waste water (with 20% sewage) and fertilizer losses up to 5 t ha-1 in the Netherlands and Belgium (Marins et al., 1998), they have implemented rules forcing producers to recycle drained solutions. In the Netherlands, the discharge of nutrient solution is allowed only if the sodium in the drainage solution is between 3 and 8 mM, depending upon the crop species, with drainage of sodium concentrations greater than 4 mM for roses and 8 mM for tomatoes (Baas and Berg, 1999).
Implementing a SCS leads to a more rational use of water, fertilizers and pesticides, hence reducing pollution and production costs (Gent and Short, 2012). An automation of recycling for tomato crop, achieved percentages of water savings in the fertigation (31.5), phosphorus (31.4), nitrogen (29.9), potassium (29.8) and calcium (28.2) (Dhakal et al., 2005).
According to Martinez and Morard (1999), the SCS with appropriate recycling of the nutrient solution, could be achieved savings up to 30 and 50% for water and fertilizers, respectively. Richard et al. (2001), in a rose cultivation system using low-tech drainage recycling based on EC control, obtained savings of 42% in water and 55% in nutrients, without affecting the quality of the flowers. Closed SCS in Mediterranean conditions can produce excellent quality carnation flowers using water with average chemical characteristics, with low concentrations of sodium and chlorine, maximizing water and nutrients use efficiency. The recycling system and disinfection of the solution did not require great technical deal, but weekly analysis of the solution and leaching was needed for a proper management (Marfà, 1999).
The yield of plants in SCS is higher than the traditional soil system, which makes this cultivation technique of interest for increasing the food supply (Raziq, 2007). Substrate culture systems use organic or inorganic substrates to replace the soil as mean of anchorage and provider of nutrients to the plants. The physical, chemical and microbiological properties of the substrates are essential, because, due to the reaction with the nutrient solution in the root environment, their characteristics vary over time and may affect the availability of nutrients and water retention (Martínez and Roca, 2011). A growth media that has a proper balance between water availability and aeration of the roots will optimize the yield and quality of horticultural crops (Casadesus et al., 2007).
For proper management of fertigation, it is important to understand the factors affecting the availability of air and water in a substrate. The water retention curve (WRC ) establishes the relationship between the matric potential and volumetric water content and is used to estimate the availability of water and air in substrates (Wallach, 2008). In addition, from the WRC is obtained the container capacity, the pore space occupied by air and water, as well as, the readily and not readily water available to the plant (Murray et al., 2004). The importance of these soil hydraulic functions and their difference with values obtained for mineral soils are described by González et al. (2006a) and the effect of the temporal variation of these features on the substrate, by González et al. (2006b).
In Colombia, since 1992, open systems cultivation in substrates has been used for the production of cut flowers. Previously, SCS was used to produce hydroponic vegetables and forage. The most widely used substrate for growing cut flowers has been burnt rice husk (BR H), at the beginning was used rice husk not burnt; afterwards, it was subjected to fermentation and aging aiming to improve the water retention capacity (Calderón and Cevallos, 2001). The mixture of rice husk with other materials such as coal slag, river sand and husk ash has also been used to improve moisture retention.
Due to higher quantity of fine particles of BR H, it shows higher water holding capacity than coconut fiber (CF), which has coarser particles size, henceforth lower holding retention capacity. In the substrate mixtures studied by Quintero et al. (2009), there was a reduction in the rate of decomposition and washing of BR H; and the variations in volume of air and the amount of readily available water aided the definition of the appropriate conditions in water management for production.
Recycling drainage
One way to counter the environmental pollution generated by the SCS is the use of drainage recycling techniques, which take advantage of their fertilizer potential. This is achieved by implementing a suitable system for drainage isolation and subsequent treatment for recycling the saline solution (Gent and Short, 2012).
Success of closed SCS depends on knowledge and management of the nutrient solution. Most of the water used for irrigation has high concentrations of salts, especially sodium and chloride, elements that are poorly absorbed by most plants, which can affect the reduced volume of roots, characteristic of SCS (Sonneveld and Van der Burg, 1991). This accumulation can be prevented by washing with additional water applications, which also leads to considerable losses of nutrients (Sonneveld, 2000). The non-absorbed salts by the crop are drained and tend to accumulate in the recirculating nutrient solution; therefore it must be frequently leached (removed from the system), particularly under high temperature environments where salt accumulation can reach 2 dS m-1 (Incrocci et al., 2006).
Maas and Hoffman (1977) modeled salt tolerance with the following characteristics: (1) salinity threshold value (ST V), which is the maximum value of salinity which does not present a significant reduction in growth or yield and (2) the salinity yield decrease (SYD), a value that indicates the percentage of decrease in yield per unit increase in EC over ST V.
Sonneveld et al. (1999) concluded that the sodium and chloride absorption by plants increase with increasing concentrations in the root environment, henceforth counteract their accumulation in the rhizosphere, would be an advantage, however, it depends on the species. For carnation the calculated values were: ST V of 4.3 dS m-1 and SYD of 3.9% per dS m-1; and for rose: ST V of 2.1 dS m-1 and SYD of 5.3% per dS m-1. Low EC values can produce inadequate input elements and nutritional deficiencies (Graves, 1983). In contrast, EC values over the ST V decrease performance due mainly to osmotic effects which are influenced by the composition of the nutrient solution (Savvas, 2003). Salinity affects the quality of cut flowers, decreasing the length and diameter of the stem, firmness and vase life (De Kreij and Van Den Berg, 1990). In the case of gerberas and roses, these species respond by reducing the number of flowers more than the average weight of the flower, while in carnations and bouvardias the opposite occurs (Sonneveld et al., 1999).
In some growers of cut flowers in the Bogota plateau, leaching effluent is conducted to the reservoir water supply. This water is used to prepare a new fertigation solution. This is not considered properly recycling since the chemical composition of the reservoir water is altered with ions poorly absorbed by plants. According to this, is necessary to develop techniques of recycling that isolate drainage solutions for recomposition.
In this context, a crop system based on the reuse of the leaching solution or recycling of the drainage solution would contribute to the optimization of the SCS technique in protected agriculture. A cleaner production technique benefits the marketing of products since this production system involves the use of good agricultural practices, which is certifiable by green labels. By adopting this technique, the discharge of water and fertilizer salts will be reduced, especially of nitrate, phosphorus and potassium, mitigating environmental impact, reducing costs and preserving water sources (Sengupta and Banerjee, 2012).
Basic concepts
Some of the most important variables to consider in SCS are: the rhizosphere temperature, pH solution, concentrations and ratios of nitrate and ammonium, container size or root volume, growth media, EC and root aeration (Kafkafi, 2001). In addition to EC and pH to prepare the initial solution, the concentration ratios of nutrients and water quality must be taken into account (Savvas and Adamidis, 1999). Some important concepts for managing closed SCS are defined (Stanghellini and Kempkes, 2002). They are variables that depend on the type of substrate, the weather and the phenological state of the plant; and they are important for determining the water relations and balances over time.
Volume of water used (VU): volume that is used to restore the system volume, equals the volume of the new nutrient solution; irrigation volume (VI): volume of solution applied to the crop in a time interval; pulse volume (VP): volume of solution applied in an irrigation event; volume absorbed (VA): The amount of water used by the crop at a particular time and is equal to the transpiration plus growth in biomass increase, which is usually less than the 10% of the transpiration; drained volume (VD): amount of water flowing out of the root zone; leachate volume (VL): amount of water leaving the system; leaching fraction (LF = VL / VU): ratio between the volume exiting the system and the volume of water used in a given period; leaching requirement (LR = VL / VA): ratio between the volume exiting the system and the volume of water absorbed by the plant, in a given period, Operational EC (ECO): average of the EC desired for the system, leachate EC (ECL): EC at which the system solution should be discarded.
Variations in pH
The pH in the root zone for most hydroponics is between 5.5 to 6.0. Values between 5.0 to 5.5 and from 6.5 to 7.0 would not cause problems in most crops (Graves, 1983), while values greater than 7.0 may cause problems in the absorption of P, Fe and Mn; and sometimes deficiency symptoms for Cu and Zn (de Rijck and Schrevens, 1997). The pH values in a substrate system have large variations during the growing season, partly due to small volume, especially when the substrate used has a low buffering capacity. However, pH variations are greater in inert substrates, and more stable in organic substrates. The buffering capacity of the nutrient solution used is very low and almost only determined by the phosphorus concentrations (Sonneveld, 2002).
Vélez (2012) evaluated recirculation rates and substrates based on BR H and CF in culture system for carnation, and found that the pH in the leachates tended to decrease and in the substrates, was constant during the study period but increased with a rise in percentage of BRH.
In periods of high growth rate with sufficient light intensity, anion absorption normally exceeds cation absorption, which is due to high absorption of nitrate and its use in plant metabolism. This difference in rate of absorption of ions is compensated by the release of HCO3- and OH- to rhizosphere (Ben-Zioni et al., 1971), increasing the pH of the root zone. However, under conditions of low light intensity, this situation is reversed, thus reducing the use of nitrate and increasing the cation:anion absorption ratio; rapid uptake of cations is compensated by the release of H+ by the roots (Graves, 1983).
The pH in the solution can be controlled by: (i) modification of the NH4:NO3 ratio in the recycled solution and (ii) application of the required nitrogen in nitrate form and reducing the pH with acid. By not adding acid, the first method reduces the salt load in the system, however, ammonium may inhibit absorption of Ca, Mg and K and impair the development of the roots at high substrate and solution temperatures (Bar-Yosef, 2008).
Physiological considerations – Concentration of elements in the solution
The composition of the nutrient solution is defined by the known total salt concentration, pH, micronutrients concentrations, and the ratios between the macronutrients and the irrigation water composition (Savvas and Adamidis, 1999).
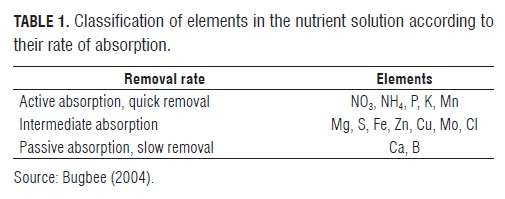
It has been established that the absorption of nutrients by the plant is specific for each solute and follows the Michaelis-Menten dynamic (Claassen and Barber, 1974). In this regard, based on the rate of absorption of the solution, Bugbee (2003) placed essential nutrients into three categories (Tab.1), wherein the monovalent ions are more efficient (Schippers, 1980). However, the ion uptake mechanisms are different for each ion and each species of plant and therefore the passive and active absorption terms should not be taken literally as plant chemistry plays an important role (Marschner, 1995) through the stoichiometry of some elements (Ågren, 2008). One drawback in single ion monitoring is the concentrations of the elements N, P, K, and Mn, which should be kept low to prevent toxic accumulation in plant tissues. The total amount of the nutrient solution can be determined easily and with some precision from the EC of the solution. However, because of the differential removal rate through the absorption of nutrients, EC mainly measures the concentration of calcium, magnesium and sulfate remaining in the solution. Micronutrients contribute less than 0.1% to the EC (Bugbee, 2004).
When irrigation water is poor in quality there is a rapid increase in EC due to the accumulation of sodium, chloride and sulfate. In the case of hard water is due to the accumulation of calcium and magnesium. The bicarbonates commonly present in groundwater are neutralized with acid. On the other hand, micronutrients such as boron or heavy metals may accumulate to toxic levels, even though their concentrations are in the order of micromoles per liter; unlike other ions that are determined indirectly by measuring the EC , these have to be monitored by expensive laboratory analysis (Olympios, 1999; Carmassi et al., 2003). One reason for the rapid buildup of chloride is owed to its weak retention in soil and subsequent leaching due to high mobility (Marschner, 1995). It is an essential element for all plants and its addition may be a strategy to reduce nitrate levels in accumulator crops, due to the antagonistic effects between these two ions (Chapagain et al., 2003).
According to Savvas (2003), in the case of actively absorbed macronutrients such as N, P and K, maintaining low concentrations could be obtained good yields; with the best results when the concentrations of elements in solution correspond roughly to the absorption ratio nutrient: water (Sonneveld, 1981; Graves, 1983). Under these conditions, plants do not consume energy to actively take or exclude ions (Steiner, 1980), however, the absorption ratio nutrient:water fluctuates in response to weather conditions, which makes difficult the preparation of a solution consistent with that ratio (Savvas, 2003).
The excess of nitrate in the restitution solution increases dry mass allocation to the leaves at expense of shoots and inflorescences. pH values between 3 and 4 increase the absorption of P, and its concentration in leaves, reducing the biosynthesis of sucrose and adversely affecting the yield of flowers. Bar-Yosef et al. (2009), using appropriate mixtures of NO3, NH4 and urea reduced the harmful effects caused by changes in pH, which produce ionic imbalances and competition for absorption of Ca, P, Mn, and other elements.
In practice, in systems based on organic substrates is difficult to track the variation of microelements concentration (Cu, Fe, Mn, Zn and B), likely due to changes in physical, chemical and microbiological properties of the substrates. In carnation established system under BR H and CF substrates, with three recycling percentages of the drained solution, Mesa et al. (2011) reported increases in EC and Cu concentration along with a trend of pH reduction during the transition between vegetative and reproductive stages. In the same experiment, Vélez et al. (2012) showed the nitrate concentration in the leaching solution was influenced by levels of recirculation and for the substrates factor this concentration decreased as the BR H content increased. For phosphate, the concentration in the leaching solution was influenced by the BR H percentage. While the potassium concentration in the leachated tended to increase with age of the plant after the "transition" stage; only influenced by the substrates, with significantly higher concentrations in treatments with higher BR H contents.
In a hydroponic system, the lowest nutrient absorption happened when rose flower stems reached the maximum elongation rates; with the reduction in the growth rate nutrient uptake increased, reaching the maximum when the stems were ready to harvest. This contrasts with the common belief that the absorption rate is linked to the phenological development of rose flower stems (Cabrera and Solís, 2011).
The temperature and oxygen concentration of the solution also require control. For instance, the production of Brassica chinensis L. in the nutrient film technique system (NFT), 10°C increase in environment temperature (25 to 35°C) decreased the percentage of dissolved oxygen from 80 to 30% (Kao, 2002). In a nutrient solution containing ammonium and nitrate, the absorption of ammonium is favored when the temperature in the root zone is low, between 3 and 11°C (Clarkson et al., 1986). During the winter season, Rouphael et al. (2008) find out that concentration of the fertigation solution may be reduced about 50% without adversely affecting the plant. They considered the association of the growth index as a function of water use efficiency and the parameters solar radiation, average air temperature and vapor pressure deficit as a tool to reassess fertilizer formulas.
Quantitative information is needed from the absorption of water and ions during crop development. In addition of essential ions, is necessary to consider the absorption of other ions such as sodium and chloride to avoid accumulation in the root environment. Ratios and the amounts of nutrients absorbed vary with the stage and conditions of the plant development. Unexpected changes in the composition of the nutrient solution frequently arise in commercial systems, that is why is necessary to analyze the root environment (Sonneveld, 2000). Similarly, the composition and volume of the drained nutrient solution will vary over time and it would affect the number of cycles of reuse. The correction procedure of the nutrient solution can be performed in two ways: by conventional control based on the adjustment of EC or by programing addition of nutrients. Kläring (2001) provided three ways to control these supplies: adding water and nutrient amounts that are expected to be taken by the plant, control of water content and concentration of nutrients in the root zone as well as in the tissues. One of the requirements for the proper replenishment of drained solution is the addition of nutrients in an appropriate ratio (Savvas and Gizas, 2002).
According to Van Noordwijk (1990), it was critical to synchronize demand and supply of nutrients to increase their use efficiency. One tool is the mathematical modeling in controlled conditions. Models of nutrient uptake have been proposed, such as, Barber - Cushman model (Cushman, 1979; Barber, 1995), the mobile limit (Reginato et al., 2000) and one that considers the increase in root density and competition between them (Hoffland et al., 1990). Carmassi et al. (2005) developed a model for the tomato crop to predict changes in ion concentration and the EC of the recycled solution. It was designed based on a balanced nutrient absorption equation and simulates the accumulation of salts in the solution when it is prepared with low quality water, in order to schedule the solution disposal.
Silberbush et al. (2005) proposed a model of water and nutrient flow, and their uptake by the plant. It takes into account the loss of water through transpiration and the accumulation of salts in the solution, including the interaction of this effect with the absorption of water and nutrients. By developing a similar model and integrating it with a computer-aided control, it is feasible the automation of a closed SCS .
Gieling et al. (1997) proposed controlling closed SCS by maintaining the flow rate and concentration of the drained solution at a fixed point. This concept has the ability to compensate for changes in the absorption of water. If absorption by the plant decreases, the drained volume will be increased, thereby compensating with a low volume.
Pathogen control
It is perceived that one of the common problems in drainage recycling systems is the spread of pathogens in the liquid medium, since the reapplication of a contaminated solution facilitates their dispersal throughout the crop (Poncet et al., 2001). Passive disinfection techniques such as slow sand filtration leave part of the resident microflora alive, while active methods such as UV, pasteurization and chemical products control harmful and beneficial organisms. The use of microbiological methods is environmentally friendly, microorganisms are active throughout the system, but more knowledge is needed about the whole process in the growth media and nutrient solution (van Os and Alsanius, 2004). Ozonation, thermal and UV disinfection methods require a large investment and therefore are used when production justifies the cost; furthermore, they must be used in conjunction with sand filters to increase effectiveness (Ehret et al., 2001; Barth, 1999).
The effectiveness of slow flow sand filter (SS F) has been verified at different latitudes (Hoitink and Krause, 1999) as an appropriate method for the control of microorganisms present in the solution; it is a low cost system whose action lies in natural control exercised physically and biologically (Calvo-Bado et al., 2003). Koohakan et al. (2004) found that cropping systems such as NFT and DFT (Deep Flow Technique) contained high amounts of Pythium sp. compared with substrate systems. The NFT contained the largest population of Fusarium sp. when compared to other SCS . One of the risks of using chemicals for disinfecting nutrient solutions is chemical residue in the leaching solution. The use of compounds with chlorine is not justified due to the environmental and health risks. UV disinfection and SS F appear to allow for a chemical-free management of the recycled solution. Garibaldi et al. (2004) concluded that among the best controls for the incidence of Phytophthora cryptogea in gerbera cv. Goldie cultivated in a recycling system were SS F filter and UV radiation system. It has been found that the proliferation of strains of Pseudomonas fluorescens colonizing roots has controlled pathogens such as Sclerotinia sclerotiorum (Li et al., 2011) and Fusarium oxysporum f.sp. Radicis-lycopersici (M'pinga et al., 1997).
Furthermore, the substances exuded by the roots of the plants could become a major problem in closed hydroponic systems. Temperature and photoperiod are related to the quality and quantity of root exudates, and these compounds can inhibit the growth of roots and produce autotoxicity (Pramanik et al., 2000).
Conclusions
Local research results show the farmers need to know the hydrophysical characteristics of the substrate as well as the interaction of nutrients with the drained solution and the substrate itself in order to improve fertigation application processes.
It is necessary to establish guidelines for the design, monitoring and control of SCS according to their minimum components: substrate, plant, nutrient solution and environment. Understanding the variations in the physical, chemical and biological characteristics, according to the environmental and sustainability standards, will make possible the implementation of controlled systems.
Studies must address the system components in order to: define the physical, chemical and biological characteristics for setting up an appropriate substrate; understand the dynamics of recycled nutrient solutions and thereby control the useful life of the drainage without affecting yields; and determine the influence of the components on the productivity of cultivated plants.
Literature cited
Ågren, G.I. 2008. Stoichiometry and nutrition of plant growth in natural communities. Ann. Rev. Ecol. Evol. Syst. 39, 153-170. [ Links ]
Baas, R. and D. Berg. 1999. Sodium accumulation and nutrient discharge in recirculation systems: A case study with roses. Acta Hort. 507, 157-164. [ Links ]
Barber, S.A. 1995. Soil nutrient bioavailability: A mechanistic approach. 2nd ed. John Wiley. & Sons, New York. [ Links ]
Bar-Yosef, B. 2008. Fertigation management and crops response to solution recycling in semi-closed greenhouses. pp. 383-388. In: Raviv, M. and J.H. Lieth (eds.). Soilless culture: theory and practice. Elsevier, London. [ Links ]
Bar-Yosef, B., N.S. Mattson , and H.J. Lieth. 2009. Effects of NH4:NO3:urea ratio on cut roses yield, leaf nutrients content and proton efflux by roots in closed hydroponic system. Sci. Hortic. 122(4), 610-619. [ Links ]
Barth, G. 1999. Slow flow sand filtration (SSF) for water treatment in nurseries and greenhouses: the nursery papers. In: Nursery and Garden Industry Australia, http://www.ngia.com.au/files/nurserypapers/NP_1999_03.pdf; consulted: October, 2012. [ Links ]
Ben-Zioni, A.Y., Y. Vaadia, and S.H. Lips. 1971. Nitrate uptake by roots as regulated by nitrate reduction products of the shoots. Plant Physiol. 24(2), 288-290. [ Links ]
Bugbee, B. 2004. Nutrient management in recirculating hydroponic culture. Acta Hort. 648, 99-112. [ Links ]
Cabrera, R.I. and A.R. Solís P. 2011. Particularidades de fisiología vegetal y nutrición mineral, con enfásis en estreses, en rosas de invernadero. pp. 91-105. In: Flórez R., V.J. (ed.). Avances sobre fisiología de la producción de flores de corte en Colombia. Editorial, Universidad Nacional de Colombia, Bogota. [ Links ]
Calderón, F. and F. Cevallos. 2001. Los sustratos. p. 21. In: Memorias Primer Curso de Hidroponía para la Floricultura. Bogota. [ Links ]
Calvo-Bado, L.A., T.R. Pettitt, N. Parsons, G.M. Petch, J.A.W. Morgan, and J.M. Whipps. 2003. Spatial and temporal analysis of the microbial community in slow sand filters used for treating horticultural irrigation water. Appl. Environ. Microbiol. 69(4), 2116-2125. [ Links ]
Carmassi, G., L. Incrocci, R. Maggini, F. Malorgio, F. Tognoni, and A. Pardossi. 2005. Modeling salinity build-up in recirculating nutrient solution culture. J. Plant Nutr. 28, 431-445. [ Links ]
Carmassi, G., L. Incrocci, M. Malorgio, F. Tognoni, and A. Pardossi. 2003. A simple model for salt accumulation in closed-loop hydroponics. Acta Hort. 614, 149-154. [ Links ]
Casadesus, J., R. Caceres, and O. Marfà. 2007. Dynamics of CO2 efflux from the substrate root system of container-grown plants associated with irrigation cycles. Plant Soil 300(1-2), 71-82. [ Links ]
Chapagain, B.P., Z. Wiesman, M. Zaccai, P. Imas, and H. Magen. 2003. Potassium chloride enhances fruit appearance and improves quality of fertigated greenhouse tomato as compared to potassium nitrate. J. Plant. Nutr. 26(3), 243-658. [ Links ]
Clarkson, D.T., M.J. Hopper, and L.H.P. Jones. 1986. The effect of root temperature on the uptake of nitrogen and the relative size of the root system in Lolium perenne. I. Solutions containing both NH4+ and NO3 -. Plant Cell Environ. 9(7), 535-545. [ Links ]
Claassen, N. and S.A. Barber. 1974. A method for characterizing the relation between nutrient concentration and flux into roots of intact plants. Plant Physiol. 54(4), 564-568. [ Links ]
Cushman, J.H. 1979. An analytical solution to solute transport near root surfaces for low initial concentration: I. Equations development. Soil. Sci. Soc. Amer. J. 43 (6), 1087-1090. [ Links ]
De Kreij, C. and T.H.J.M. Van Den Berg. 1989. Nutrient uptake, production and quality of rosa hybrida in rockwool as affected by electrical conductivity of the nutrient solution. p. 519. In: Proceedings of the Eleventh International Plant Nutrition Colloquium. Kluwer Academic, Wageningen, The Netherlands. [ Links ]
De Rijck, G. and E. Schrevens. 1998. Elemental bioavailability in nutrient solutions in relation to precipitation reactions. J. Plant Nutr. 21(10), 2103-2113. [ Links ]
Dhakal, U., V. Salokhe, H. Tantau, and J. Max. 2005. Development of a greenhouse nutrient recycling system for tomato production in humid tropics (on line). CIGR Ejournal 7, http://ecommons.library.cornell.edu/bitstream/1813/10429/1/BC%2005%20008%20Salokhe%20final%206Dec2005.pdf; consulted: October, 2012. [ Links ]
Ehret, D.L., B. Alsanius, W. Wohanka, J.G. Menzies, and R. Utkhede. 2001. Disinfestation of recirculating nutrient solutions in greenhouse horticulture. Agronomie 21(4), 323-339. [ Links ]
European Commission. 2000. Aplicación de la Directiva 91/676/ CEE del Consejo relativa a la protección de las aguas contra la contaminación producida por nitratos utilizados en la agricultura. In: European Commission, http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=CELEX:31991L0676:es:HTML; consulted: October, 2012. [ Links ]
Garibaldi, A., A. Minuto, and D. Salvi. 2004. Disinfection of nutrient solution in closed soilless systems in Italy. Acta Hort. 644, 557-562. [ Links ]
Gent, M.P.N and M.R. Short. 2012. Effect on yield and quality of a simple system to recycle nutrient solution to greenhouse tomato. HortScience 47(11), 1641-1645. [ Links ]
Gieling, Th.H., J. Bontsema, T.W.B.M. Bouwmansandand, and R.H. Steeghs. 1997. Modelling and simulation for control nutrient application in closed growing systems. Neth. J. Agr. Sci. 45(1), 127-142. [ Links ]
González M., C.A., H. Tafur H., V.J. Flórez R., and R. Burbano. 2006a. Modelación del movimiento del agua en el suelo. pp. 359-376. In: Flórez R., V.J., A. de la C. Fernández M., D. Miranda L., B. Chaves C., and J.M. Guzmán P. (eds.). Avances sobre fertirriego en la floricultura colombiana. Unibiblos, Universidad Nacional de Colombia, Bogota. [ Links ]
González M., C.A., M.F. Quintero, and V. Meneses. 2006b. Determinación de algunas propiedades hidrofísicas de los sustratos utilizados en cultivos de flores en la Sabana de Bogotá. pp. 427- 450. In: Flórez R., V.J., A. de la C. Fernández M., D. Miranda L., B. Chaves C., and J.M. Guzmán P. (eds.). Avances sobre fertirriego en la floricultura colombiana. Unibiblos, Universidad nacional de Colombia, Bogota. [ Links ]
Graves, C.J. 1983. The nutrient film technique. pp. 1-44. In: Janick, J. (ed). Horticultural reviews. Vol. V. John Wiley & Sons, Hoboken, NJ. [ Links ]
Hoffland, E., H.S. Bloemhof, P.A. Leffelaar, G.R. Findeneggy, and J.A. Nelemans. 1990. Simulation of nutrient uptake by a growing root system considering increasing root density and inter-root competition. Plant Soil 124(2), 149-155. [ Links ]
Hoitink, H. and M. Krause. 1999. New approaches to control of plant pathogens in irrigation water. Special Circular No. 173-00. Landscape, and Turf Team, Ohio State University Extension Nursery, Wooster, OH. [ Links ]
Incrocci, L., F. Malorgio, A. Della Bartola, and A. Paradossi. 2006. The influence of drip irrigation or subirrigation on tomato grown in closet-loop substrate culture with saline water. Sci. Hortic. 107(4), 365-372. [ Links ]
Kafkafi, U. 2001. Root zone parameters controlling plant growth in soilless culture. Acta Hort. 554, 27-38. [ Links ]
Kao, T.C. 2002. The dynamic root floating hydroponic technique: year-round production of vegetables in roc on Taiwan. In: Food & Fertilizer Technology Center, http://www.agnet.org/htmlarea_file/library/20110801154442/eb330.pdf; consulted:October, 2012. [ Links ]
Kläring, H.P. 2001.Strategies to control water and nutrient supplies to greenhouse crops. A review. Agronomie 21(4), 311-321. [ Links ]
Koohakan, P., H. Ikeda, T. Jeanaksorn, M. Tojo, S. Kusakari, K. Okada, and S. Sato. 2004. Evaluation of the indigenous microorganism in soilless culture: occurrence and quantitative characteristics in the different growing systems. Sci. Hort. 101(1-2), 179-188. [ Links ]
Li, H., H. Li, Y. Bai, J. Wang, M. Nie, B. Li, and M. Xiao. 2011. The use of Pseudomonas fluorescens P13 to control sclerotinia stem rot (Sclerotinia sclerotiorum) of oilseed rape. J. Microbiol. 49(6), 884-889. [ Links ]
Maas, E.V. and G.J. Hoffman. 1977. Crop salt tolerance - current assessment. J. Irr. Drain. Div. 103(2), 115-134. [ Links ]
Marfà, O. 1999. Soilless culture and fertilizer efficiency. p. 105. In: IFA Agricultural Conference on Managing Plant Nutrition. Barcelona, Spain. [ Links ]
Marfà, O. 2000. La recirculación en los cultivos sin suelo. Elementos básicos. pp. 21-27.In: Marfà, O. (ed.). Recirculación en cultivos sin suelo. Compendios de horticultura. 2nd ed. Ediciones de Horticultura. Reus, Spain. [ Links ]
Marins-Peil, R., J. López Gálvez, and A. Martins. 1998. Cultivo de pepino con técnica de solución nutritiva recirculante. p. 160. In: I congreso Ibérico sobre Gestión y Planificación de Aguas. Universidad de Zaragoza, Zaragoza, Spain. [ Links ]
Martinez, S. and P. Morard. 1999 Réduction de la consommation en eau d´une culture hors sol de tomates par recyclage de la solution nutritive. pp. 531-536. In : 3ème Congrès International de Recherche: L'eau et sa Réutilisations. INSA, Toulouse, France. [ Links ]
Martínez, P.F. and D. Roca. 2011. Sustratos para el cultivo sin suelo. Materiales, propiedades y manejo. pp. 37-78. In: Flórez R., V.J. (ed.). Sustratos, manejo del clima, automatización y control en sistemas de cultivo sin suelo. Editorial, Universidad Nacional de Colombia, Bogota. [ Links ]
Ministerio de Ambiente, Vivienda y Desarrollo Territorial. 2011. Decreto 3939 del 2010: Plan de gestión del riesgo para el manejo de vertimientos. In: http://www.minambiente.gov.co/documentos/DocumentosBiodiversidad/110211_ter_ref_manejo_vertimientos.pdf; consulted: October, 2012. [ Links ]
Marschner, H. 1995. Mineral nutrition of higher plants. 2nd ed. Academic Press, London. [ Links ]
Mesa, J.M., M.C. Henao, and V.J. Flórez. 2011. Comportamiento de los microelementos en un sistema de cultivo de clavel en sustrato con recirculación de lixiviados. pp. 72-90. In: Flórez R., V.J. (ed.). Avances sobre fisiología de la producción de flores de corte en Colombia. Editorial, Universidad Nacional de Colombia, Bogota. [ Links ]
Murray, J.D., J.D. Lea-Cox, and D.S. Ross. 2004. Time domain reflectometry accurately monitors and controls irrigation water applications in soilless substrates. Acta Hort. 633, 75-82. [ Links ]
M'pinga, P., R.R. Belanger, T.C. Paulitz, and N. Benhamou. 1997. Increased resistance to Fusarium oxysporum f.sp. radicislycopersici in tomato plants treated with the endophytic bacterium Pseudomonas fluorescens strain 63-28. Physiol. Mol. Plant P. 50(5), 301-320. [ Links ]
Olympios, C.M. 1999. Overview of soilless culture: advantages, constraints, and perspectives. pp. 307-324. In: Choukr-Allah, R. (ed.). Protected cultivation in the Mediterranean region. Vol. 31. Colloque sur les Cultures Protégéesdans la Région Méditerranéenne. CIHEAM Agadir, Morocco. [ Links ]
Patiño, M. 2000. Cultivo de clavel sobre sustrato de cascarilla de arroz. pp. 41-43. In: Pizano de Márquez, M. (ed.). El clavel. Ediciones Hortitecnia, Bogota. [ Links ]
Pizano de Márquez, M. 2001. Floriculture and the environment - growing flowers without methyl bromide. United Nations Environment Programme Division of Technology, Industry and Economics. Nairobi. [ Links ]
Pramanik, M.H.R., N. Masayuki, A. Toshiki, and M. Yoshihisa. 2000. Effects of temperature and photoperiod on phytotoxic root exudates in hydroponic culture. J. Chem. Ecol. 26(8), 1953-1967. [ Links ]
Poncet, C., M. Offroy, S. Antonini, A. Bettachini, G. Bonnet, J.M. Drapier, D. Hericher, and P. Julien. 2001. Disinfection systems of recycled effluents in flower crops. Acta Hort. 554, 349-354. [ Links ]
Quintero, M.F., C.A. González-Murillo,V.J. Flórez, and J.M. Guzmán. 2009. Physical evaluation of four substrates for cutrose crops. Acta Hort. 843, 349-358. [ Links ]
Raviv, M. and H. Lieth. 2008. Significance of soilless culture in agriculture. pp. 1-10. In: Raviv, M. and J.H. Lieth (eds.). Soilless culture: Theory and practice. Elsevier, London. [ Links ]
Raziq, A. 2007. Can we solve the problem of hunger and malnutrition? In: Food and Agriculture Organization of the United Nations, http://km.fao.org/fileadmin/user_upload/fsn/docs/CanWe-SolvetheProblemofHungerandMalnutrition.doc; consulted: October, 2012. [ Links ]
Reginato, J.C., M.C. Palumboc, I.S. Moreno, I.Ch. Bernardo, and D.A. Tarzia. 2000. Modeling nutrient uptake using a moving boundary approach comparison with the barber-cushman model. Soil Sci. Soc. Amer. J. 64(4), 1363-1367. [ Links ]
Richard, B., S. André, and C. Corinne. 2001. Recycling of nutrient solutions for rose (Rosa hybrida) in soilless culture. Acta Hort. 554, 183-191. [ Links ]
Rouphael, Y., M. Cardarelli, E. Rea, and G. Colla. 2008. The influence of irrigation system and nutrient solution concentration on potted geranium production under various conditions of radiation and temperature. Sci Hortic. 118(4), 328-337. [ Links ]
Savvas, D. and K. Adamidis. 1999. Automated management of nutrient solutions based on target electrical conductivity, pH, and nutrient concentration ratios. J. Plant Nutr. 22(9), 1415-1432. [ Links ]
Savvas, D. and G. Gizas. 2002. Response of hydroponically grown gerbera to nutrient solution recycling and different nutrient cation ratios. Sci Hortic. 96(1-4), 1-14. [ Links ]
Savvas, D. 2003. Nutritional management of vegetables and ornamental plants in hydroponics. pp. 37-87. In: Dris, R. and R. Niskanen (eds.). Crop management and postharvest handling of horticultural products fruits and vegetables. Vol. 1 - Quality management. Science Publishers. Enfield, NH. [ Links ]
Sengupta, A. and H. Banerjee. 2012. Soil-less culture in modern agriculture. World J. Sci. Technol. 2(7), 103-108. [ Links ]
Schippers, P.A. 1980. Composition changes in the nutrient solution during the growth of plants in recirculating nutrient culture. Acta Hort. 98, 103-117. [ Links ]
Silberbush, M., J. Ben-Asher, and J.E. Ephrath. 2005. A model for nutrient and water flow and their uptake by plants grown in a soilless culture. Plant Soil 271(1-2), 309-319. [ Links ]
Sonneveld, C. 1981. Items for application of macro-elements in soilless culture. Acta Hort. 126, 187-195. [ Links ]
Sonneveld, C. and A.M.M. Van der Burg. 1991. Sodium chloride salinity in fruit vegetable crops in soilless culture. Neth. J. Agr. Sci. 39, 115-122. [ Links ]
Sonneveld, C., R. Bass, H.M.C. Nijssenand, and J. De Hoog. 1999. Salt tolerance of flower crops grown in soilless culture. J. Plant Nutr. 22(6), 1033-1048. [ Links ]
Sonneveld, C. 2000. Effects of salinity on substrate grown vegetables and ornamentals in greenhouse horticulture. Ph.D. thesis. University of Wageningen, The Netherlands. [ Links ]
Sonneveld, C. 2002. Composition of nutrient solutions. pp. 179-210. In: Savvas, D. and H. Passam (eds.). Hydroponic production of vegetables and ornamentals. Embryo Publications, Athens. [ Links ]
Steiner, A.A. 1980. The selective capacity of plants for ions and its importance for the composition and treatment of the nutrient solution. Acta Hort. 98, 87-97. [ Links ]
Stanghellini, C. and F.L.K. Kempkes. 2004. A blueprint for optimal management of multiple-quality water-resources. Deliverable 8. In: Hortimed, http://www.aua.gr/ns/project/hortimed/Deliverable_8.pdf; consulted: October, 2012. [ Links ]
Van Kooten, O., E. Heuvelink, and C. Stanghellini. 2004. Nutrient supply in soilless culture: on demand strategies. Acta Hort. 659, 533-540. [ Links ]
Van Noordwijk, M. 1990. Synchronization of supply and demand is necessary to increase efficiency of nutrient use in soilless horticulture. pp. 525-531. In: Proceedings of the Eleventh International Plant Nutrition Colloquium. Plant nutrition -Physiology and applications. Wageningen, The Netherlands. [ Links ]
Van Os, E.A. 1999. Closed soilless growing systems: a sustainable solution for dutch greenhouse horticulture. Water Sci. Technol. 39(5), 105-112. [ Links ]
Van Os, E. and B. Alsanius. 2004. Disinfestation of recirculated nutrient solution - towards new approaches? Acta Hort. 644, 605-607. [ Links ]
Vélez C., N.A. 2012. Comportamiento de macronutrientes en un sistema de cultivo sin suelo para clavel estándar cv. Delphi con recirculación de drenajes en la sabana de Bogotá. M.Sc. thesis. Faculty of Agronomy, Universidad Nacional de Colombia, Bogota. [ Links ]
Vélez C., N.A., V.J. Flórez R., and S.E. Melo M. 2012. Comportamiento de NPK en un sistema de cultivo sin suelo para clavel estándar cv. Delphi con recirculación de drenajes en la Sabana de Bogotá. In: Encontro Brasileiro de Hidroponia. Florianópolis, Brasil. [ Links ]
Vélez O., M.V. 2010. Guía metodológica para la formulación de planes de manejo ambiental de acuíferos. In: Ministerio de Ambiente, Vivienda y Desarrollo Territorial, http://www.minambiente.gov.co/documentos/DocumentosBiodiversidad/recurso_hidrico/280111_prop_guia_met_pma_acui_280211.pdf; consulted: October, 2012. [ Links ]
Wallach, R. 2008. Physical characteristics of soilless media. pp. 41- 116. In: Raviv, M. and J.H. Lieth (eds.). Soilless culture: Theory and practice. Elsevier, London. [ Links ]