Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Agronomía Colombiana
Print version ISSN 0120-9965
Agron. colomb. vol.31 no.1 Bogotá Jan./Apr. 2013
CROP PHYSIOLOGY
Stomatal behavior in fruits and leaves of the purple passion fruit (Passiflora edulis Sims) and fruits and cladodes of the yellow pitaya [Hylocereus megalanthus (K. Schum. ex Vaupel) Ralf Bauer]
Comportamiento estomático en frutos y hojas de gulupa (Passiflora edulis Sims) y en frutos y cladodios de pitahaya amarilla [Hylocereus megalanthus (K. Schum. ex Vaupel) Ralf Bauer]
Camilo Sánchez1, Gerhard Fischer1 and Danny Wilson Sanjuanelo 2
1Department of Agronomy, Faculty of Agronomy, Universidad Nacional de Colombia. Bogota (Colombia). camilosanc@gmail.com2Faculty of Science and Technology, Universidad de Ciencias Aplicadas y Ambientales (UDCA). Bogota (Colombia).
Received for publication: 3 December, 2012. Accepted for publication: 29 March, 2013
ABSTRACT
Plants as C3 and CAM react photosynthetically different but both can grow in the same agroecological zone in the tropics. Therefore we studied the behavior of stomatal opening in fruits and leaves of the purple passion fruit and fruits and cladodes of the yellow pitaya was studied under natural growing conditions in Granada and Fusagasuga, Cundinamarca (Colombia). Imprints were made on the surface of leaves, fruits and cladodes using cosmetic enamel impressions. Three cycles were carried out, each cycle took 72 hours, obtaining three different samples every 3 hours; then the impressions were observed by microscope and the opened and closed stomata were counted in each species. In each sampling, data of solar radiation, temperature and relative humidity (RH) were measured. The purple passion fruit had the typical behavior of a C3 plant in the leaves as well as the fruits, and a positive correlation between the stomatal aperture and radiation and temperature was found, along with a negative correlation between stomatal aperture and RH. The pitaya showed the typical behavior of a CAM plant with a negative correlation between the stomatal opening and radiation and temperature, as well as a positive correlation between stomatal opening and RH. Radiation, temperature and RH affected the stomatal opening in the fruits and cladodes. Stomatal densities differed greatly between the species and plant organs. In the purple passion fruit, 106.53 stomata per mm2 leaf surface were found, but only 12.64 stomata per mm2 fruit surface; whereas in the pitaya, 11.28 and 1.43 stomata per mm2 were found on the cladodes and fruits, respectively.
Key words: tropical fruits, C3 plants, CAM plants, stomata, stomatal movement.
RESUMEN
Las plantas C3 y CAM reaccionan fotosintéticamente diferentes, sin embargo, ambas pueden crecer en la misma zona agroecológica en los trópicos. Por esta razón se evaluó el comportamiento de estomas en frutos y hojas de gulupa y en frutos y cladodios de pitahaya amarilla bajo condiciones naturales en los municipios de Granada y Fusagasugá, departamento de Cundinamarca (Colombia). Se hicieron improntas sobre la superficie de las hojas, frutos y cladodios. Se realizaron tres lecturas, cada una duró 72 horas, haciendo impresiones cada 3 horas sobre tres muestras diferentes para luego observarlas en microscopio, haciendo un conteo de estomas abiertos y cerrados en cada especie. En cada muestreo se obtuvieron datos de radiación solar, temperatura y humedad relativa (HR). En gulupa se encontró un comportamiento típico de una planta C3 tanto para las hojas como para los frutos, además de encontrar una correlación positiva entre la apertura estomática y la radiación y temperatura, así como una correlación negativa entre la apertura estomática y la HR. En pitahaya se encontró un comportamiento típico de una planta CAM, observando una correlación negativa entre la apertura estomática y la radiación y temperatura, así como una correlación positiva entre apertura estomática y HR. La radiación, la temperatura y la HR afectaron la apertura estomático en frutos y cladodios. Las densidades estomáticas contrastaron altamente entre las especies y los órganos de la planta. En gulupa fueron encontrados 106,53 estomas/mm2 en la superficie de las hojas, pero solo 12,64 estomas/mm2 en la del fruto; mientras que en pitahaya fueron encontrados 11,28 y 1,43 estomas/mm2 en cladodios y frutos, respectivamente.
Palabras clave: frutas tropicales, plantas C3, plantas CAM, estomas, movimiento estomático.
Introduction
The purple passion fruit (Passiflora edulis Sims) is a vigorous woody perennial vine which belongs to the Passifloraceae family, which includes 550 species and is represented by more flowering species than probably any other plant family (Menzel and Simpson, 1994). It is an exotic fruit from the region between the Brazilian Amazon, the north of Argentina and Paraguay (Morton, 1987). In Colombia, the production area of the purple passion fruit was estimated by Agronet (2012) for 2011 at 520 ha, principally in the departments of Antioquia, Boyacá, Cundinamarca, Huila, Quindío and Santander. These areas are located at altitudes between 1,800 and 2,400 m, with a medium temperature range of 15-20°C (Jiménez et al., 2012) and 1,500-2,200 mm of rainfall per year (Fischer et al., 2009).
The pitaya or dragonfruit [Hylocereus megalanthus (K. Schum. ex Vaupel) Ralf Bauer], from the tropical Americas, was introduced to Colombia in the mid 1980s due to the interest of Japanese experts and the Federación Nacional de Cafeteros de Colombia. Production in Colombia is concentrated in the departments of Boyacá, Huila, Santander, Valle del Cauca, Cundinamarca, Tolima, Caldas, Bolivar, and Quindío, reaching an area in 2011 of 691 ha (Agronet, 2012). The best cultivating areas are located at altitudes between 1,500 and 1,850 m, with a medium temperature range of 18-21°C and 1,500-2,000 mm of rainfall per year (Corredor, 2012).
Stomata are formed in the epidermis of all plant organs, except the roots (Kramer, 1989), preferentially on the leaves. Stomata are small pores or valves that control the entry of carbon dioxide assimilated in photosynthesis and optimize water use efficiency, essential for plant survive and performance (Bergmann and Sack, 2007). Adjusting the number or movements (opening and closing) of stomata is essential for regulating the water use of crop plants (Pritchard and Amthor, 2005). As specialized structures in the epidermis, they are formed by two elongated guard cells enclosing a pore between them, across which plants absorb CO2 and liberate O2 to the environment (Hetherington and Woodward, 2003). Each stoma is surrounded by two guard (occlusive) cells, which change their shape to open and close the pores (Evert and Eichhorn, 2012). The apertures are so small that at most 3% of the total leaf surface is occupied by the pores (Öpik and Rolfe, 2005).
The stoma opens due to water absorption by the guard cells (Assmann, 1993; Wang et al., 1998), which in turn is caused by an increase in the solute; potassium ions moving towards the occlusive cells as the stoma opens (Salisbury and Ross, 1992). This behavior is the result of interactions between physiological factors and environmental conditions (Assmann and Wang, 2001; Hetherington and Woodward, 2003).
Environmental factors that affect stomatal movement include light, temperature and CO2 concentration (Evert and Eichhorn, 2012). Also, the stomatal response plays a key role in the economy of water in plants, since more than 90% of plant water is lost through leaf stomata (Hess, 2004). When stomata are closed, the main pathway for water loss is through cuticles (Fageria et al., 2006).
Wong et al. (1978) described three mechanisms which cause stomatal response to light. First, the guard cells seem to possess receptive pigments of light that directly detect changes in light intensity and change the stomatal conductance accordingly. Second, the guard cells can indirectly feel a change in the intensity of light through a change in the partial pressure of intercellular CO2 (Ci) that is caused by a change in the photosynthetic rate of the mesophyll tissue. Third, the guard cells can indirectly respond to changes in the rate of photosynthesis by a chemical transmissible signal that is transported from the mesophyll cells to the guard cells. In many species, stomata show a diurnal rhythm, opening by day and closing by night, with light stimulating stomatal opening, with blue wavelengths being most effective (Öpik and Rolfe, 2005).
Within normal ranges (10-25°C), changes in temperature have little effect on stomatal behavior (Evert and Eichhorn, 2012), but temperatures higher than 30 to 35°C can lead to stomatal closure which can be due to an indirect response from the water stress or from an increase in the respiratory rate that induces an increase in the CO2 inside the leaf (Salisbury and Ross, 1992).
Stomata regulate water loss (Pospílová and Dodd, 2005), and stomatal closure responds to the decrease of the moisture in the environment (Kearns and Assmann, 1993). Mott and Parkhurst (1991) suggested that a stomatal closing in response to increasing water stress involves a diminished availability of water for the guard cells and thus the turgor decreases. The apoplastic abscisic acid (ABA) concentration around the guard cells is crucial in determining stomatal responses; when roots are stressed through water deficit, ABA travels by the xylem to the leaves, signaling stomata closure (Pospílová and Dodd, 2005).
In almost all plants, low concentrations of CO2 cause the opening of stomata, even in darkness (Kramer, 1989); if CO2 circulates freely through the leaves, they will open more. On the contrary, a high concentration of CO2 in the leaves can provoke stomatal closing, in the presence of light or darkness (Salisbury and Ross, 1992; Kearns and Assmann, 1993).
The number of stomata varies greatly (Kramer, 1989). Lake et al. (2001) determined that stomatal density (or frequency) is influenced by an environmental signal perceived in mature leaves and transmitted to young leaves. The stomatal frequency is affected by gaseous exchange, transpiration and stomatal conductance (Woodward and Bazzaz, 1988). Usually, the stomatal frequency per mm2 of leaf surface ranges from 10 to 2,000 (Öpik and Rolfe, 2005). In fruits, the number of stomata is determined in anthesis and remains constant during development (Hieke et al., 2002); nevertheless, with the expansion of the fruit, the stomata are diluted on the surface, causing a decrease in stomatal density (Blanke, 1992). After anthesis, fruit stomata appear as regulatory as in the respective leaf, but some stomata may lose their regulatory ability towards maturation (Blanke, 1990).
CAM (crassulacean acid metabolism) plants are different from C3 or C4 plants, in which the stomata typically open at night (when temperatures are lower and humidity higher) and close by day, an adaptive strategy of succulent plants to conserve water in the heat of the day (Hess, 2004; Taiz and Zeiger, 2006). Each of these plant types develops in different climates, and each one represents an adaptation to the respective climate (Salisbury and Ross, 1992).
The purpose of this research was, therefore, to improve knowledge on the behavior of stomatal opening in the fruits and leaves of the purple passion fruit, a C3 plant, and in the fruits and cladodes of the yellow pitaya, a CAM plant, under natural growing conditions in order to better understand their adaptation to their environments and analyze the reaction of stomata to different environmental factors.
Materials and methods
Location: The study was developed in the Cundinamarca department of Colombia, in the municipalities of Granada and Fusagasuga, on the farms: "Chinchiri" and "El Jardin", respectively. The purple passion fruit farm "Chinchiri" is located on the Granada-Silvania road, El Ramal district, at an altitude of 2,150 m with an average temperature of 18°C. The yellow pitaya farm "El Jardin" is located on the Fusagasuga-Silvania road, Jordan Alto district, at 1,200 m from Fusagasuga, at an altitude of 1,700 m, with 20°C medium temperature.
Plant material: The plant species were purple passion fruit (Passiflora edulis Sims) planted in December 2003 at a distance of 8 x 2 m and yellow pitaya [Hylocereus megalanthus (K. Schum. ex Vaupel) Ralf Bauer] planted in December 2005 at 1 x 3.2 m.
Experiment design: The experiment was developed in a spatial arrangement of unique plots for each of the species. The experimental unit constituted all of the plants planted on each farm, 450 plants of the purple passion fruit and 1,980 plants of the yellow pitaya.
Stomatal behavior: The evaluation of the closing and opening of the stomata was carried out by making impressions on the surface of the leaves and cladodes (flattened stems of pitaya) and green fruits. In the purple passion fruit, the chosen leaf was located around the middle-third of the plant and the fruits were completely developed green fruits. In the yellow pitaya, the fruits were completely developed green fruits. The impressions were obtained during cycles of 72 h, gathering samplings every 3 h and repeating the cycle three times every 8 d. In the purple passion fruit, the experiment started at 0:00 AM (midnight) and in the yellow pitaya at 12:00 M (midday).
The technique of enamel imprints (nail polish) (Brewer, 1992) was used, applying enamel on the central part of the abaxial (lower) leaf surface, beside the principal vein, and on the surface of the cladodes and the fruits, allowing it to dry for 10 min. The enamel was applied on the leaf and the cladode nearest to the target fruit.
A completely developed leaf and a cladode were taken from the external part of the plant, and three different plants were sampled following a random systemic sampling. The already dry enamel was removed with an adhesive double sided tape, and the imprints were placed on glass plates.
The identification of the opening and closing states of the stomata was done with the visual scale established by Laurin et al. (2006). Opened stomata were grouped by completely opened (100-90%) and partially opened (89- 40%), and closed ones by partially closed (39-10%) and completely closed (9-0%).
Environmental variables: The environmental variables like solar radiation, temperature and relative humidity (RH) were recorded at the moment of gathering each impression. Solar radiation was measured using a LI -COR LQA 1319 quantic bar connected to a LI -COR L1-189 digital radiometer. Temperature and RH were measured with a COLE-PAR MER higrotermograph.
Stomatal density: The stomatal density (number of stomata per mm2) in the leaves, fruits and cladodes was recorded by observing the imprints under a microscope and counting the stomata in a 1 cm2 area.
Statistical analysis: The SAS ® v. 8.0 analytical program was used and an analysis of variance (ANAVA) following the normal GLM procedure and Duncan's test was realized to show the differences between the variables of the study.
Results and discussion
Stomatal behavior on fruits and leaves of the purple passion fruit
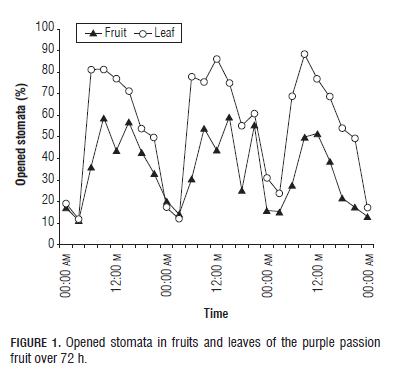
Stomata on the fruits and leaves of the purple passion fruit had a very similar behavior (Fig.1), opening stomata from very early in the morning, reaching maximum opening about noon and initiating closing during the entire afternoon (Salisbury and Ross, 1992; Willmer and Fricker, 1996), the same behavior reported Guaquetá (2007) in the leaves and fruits of banana, starfruit, guava, lime, calamondin and grapefruit.
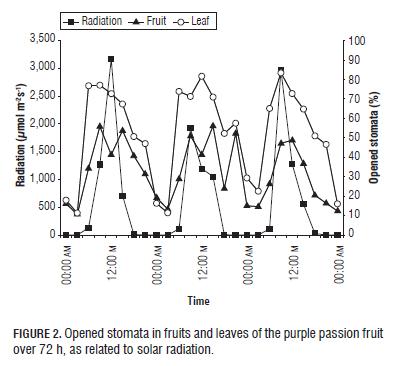
With respect to solar radiation, the opening of the stomata, both in fruits and leaves, responded positively to the change of radiation (Fig.2), so as the radiation increased, the number of opened stomata also increased (Guaqueta et al., 2007). Light has an important effect on stomata, possibly acting on the cells of the mesophyll which send a message to the occlusive cells that lead to opening (Salisbury y Ross, 1992; Taiz y Zeiger, 2006). Also, Sharkey and Ogawa (1987) demonstrated that cloroplast is the major recipient of blue light, which stimulates stomatal opening. Nevertheless, Willmer and Fricker (1996) reported that stomata can open and close in response to an endogenous rhythm, the so-called circadian rhythm (Brinker et al., 2001).
Light not only regulates stomata movements on the purple passion fruit plant, but, in general, passion fruit species are very exigent in light conditions, only flowering with full light, producing the highest yield, whereas leaf area growth and floral bud production are stimulated by =60% of full light; and for net CO2 assimilation, >25% is necessary (Menzel and Simpson, 1994).
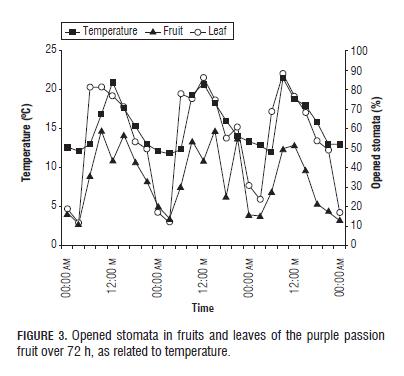
The stomata of leaves and fruits responded to temperature just as with radiation, i.e. they opened with increasing temperatures (Fig.3), a result which is confirmed by Hall (2001), who described that over a certain range of temperatures (which probably varies from crop to crop), temperature increases cause stomata to open, increasing conductance.
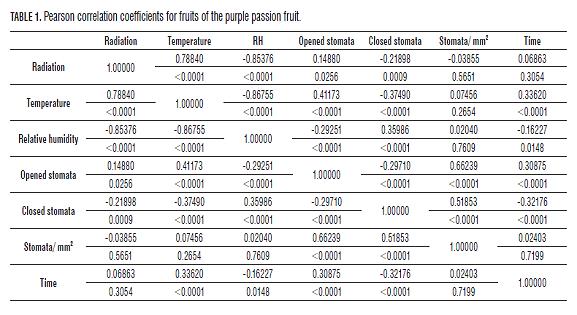
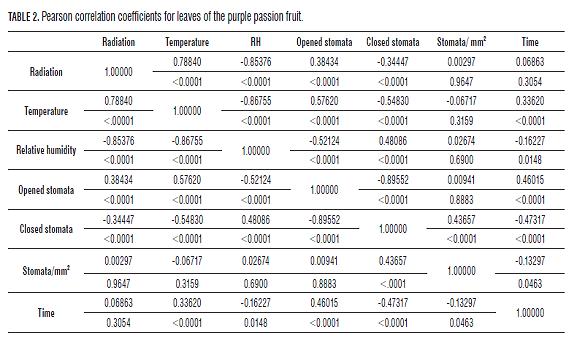
Zeiger et al. (1987) reported that foliar temperatures lower than 30°C lead to stomatal opening, and temperatures higher than 30°C almost always lead to stomatal closing (Brewer, 1992). Also, a positive correlation (R2 0.78840) was observed between radiation and temperature, suggesting that each variable depends on the other (Tab1. and 2).
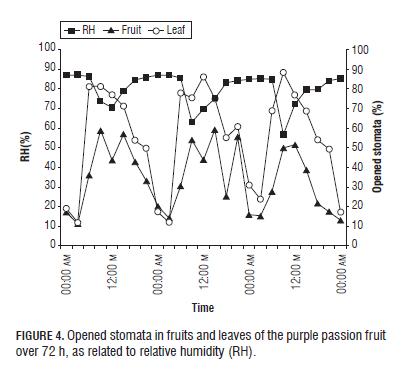
It was observed that when there were more open stomata, the percentage of RH was the lower (Fig.4), as confirmed by Kearns and Assmann (1993). Salisbury and Ross (1992) attributed this to the fact that when the gradient between the content of the air water vapor and that of the intercellular spaces is high, stomatal closing is induced; but when the CO2 content in the leaves is exhausted, in turn causes the opening of the stoma. Closed stomata during dry air conditions at noon reduce photosynthesis of fruit plants (Fischer and Orduz-Rodríguez, 2012). At noon, mild water stress prevails, which causes stomatal closure to reduce water loss, simultaneously decreasing the CO2 influx and limiting photosynthesis (Pospísilová and Dodd, 2005).
In the fruits, a negative small interrelation was found between the RH and stomatal opening (R2 -0.29251 for opened stomata and 0.35986 for closed stomata), which can suggest a low influence of RH on the opening and closing mechanism of stomata in this organ. With the leaves, the correlation (R2) between opened stomata and RH was -0.52124 and 0.48086 for closed stomata (Tabs.1 and 2).
Variance analysis shows for the radiation variable P 0.0001 and P 0.0005 indicating that the stomatal opening of fruits of the purple passion fruit depends on radiation and temperature (Evert and Eichhorn, 2012).shows that for the radiation variable, P>F is less than 0.0001 and 0.0005 for temperature,
Variance analysis shows for the radiation variable, P=0.0001and P=0.0005 for temperature, indicating that the stomatal opening of fruits of the purple passion fruit depends on radiation and temperature (Evert and Eichhorn, 2012). Also in the leaves, with P≤0.0001 for radiation, P≤0.0005 for RH, and P≤0.05 for temperature, showing that the variables radiation and relative humidity have a significant effect on the stomatal opening of this species.
Stomatal behavior on the fruits and cladodes of the yellow pitaya
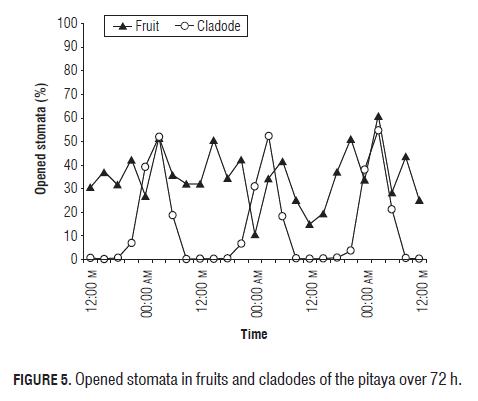
In the case of the cladodes, a typical behavior of a CAM plant was observed (Fig.5), in which stomata were opened at night and completely closed in the day (Wilkins, 1959, 1962; Salisbury and Ross, 1992). In the fruits, the behavior was not so clear but an increase of stomatal opening was observed at night and a small decrease of pore closing during the day, although the closing never appeared to be total.
In leaves of the orchid Arachnis cv. Maggie Oei (CAM plant), Goh et al. (1977) observed that the stomata started opening at about 14 h, with a maximum opening at 18 and 20 h; but the stomata of Aranda cv. Deborah started opening at 20 h, reaching the maximum opening between 21 and 24 h and closing at 4 h; and in Cattleya, the stomata started opening at 21 h, reaching the maximum opening at 3 h. In this case, the behavior was the same as in Cattleya, where the stomata opening started at 21 h, reaching the maximum opening at 3 h and closing at 6 h.
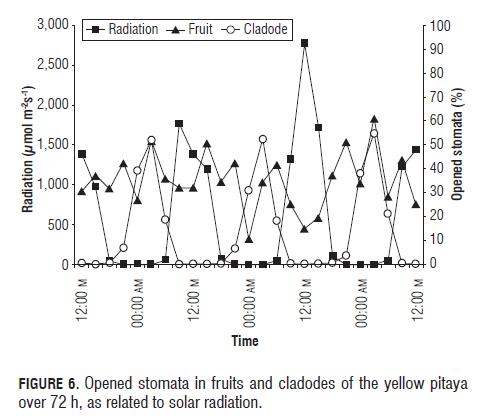
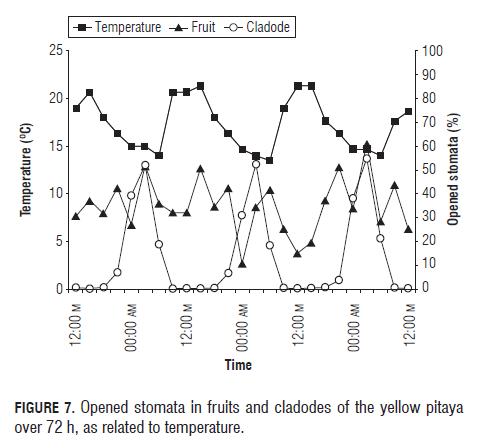
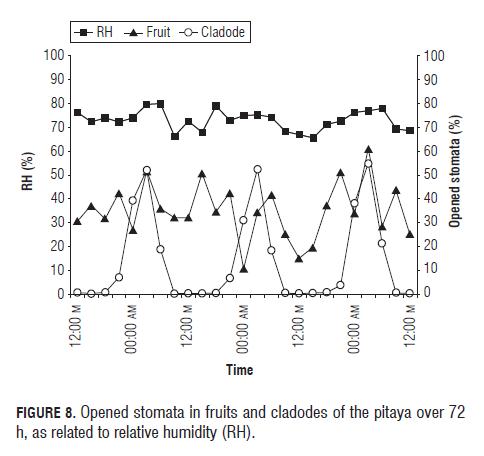
With respect to solar radiation, the opening of the stomata, both in fruits and leaves, responded negatively to the change of the radiation (Fig.6), R2 -0.47694 for cladodes and R2 -0.27155 for fruits (Tabs.3 and 4) where the stomata stayed closed in the presence of light and began opening in the absence of light, as reported Taiz and Zeiger (2006).
Radiation had a very high positive correlation (R2 0.80790) with the temperature, a value that indicates that the one depends on the other, therefore, temperature is going to have a similar effect on the stomata as radiation. Temperature had a negative correlation (R2 -0.67165 for cladodes and R2 -0.17615 for fruits) (Tab.3 and 4) with the aperture, which is shown in Fig.7, where the stomatal aperture began when the temperature decreased.
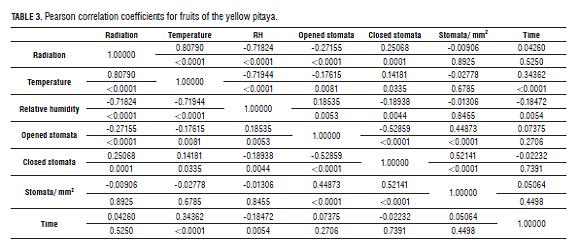
RH had little variation over time during the experiment, but a positive correlation (R2 0.45065 for cladodes and R2 0.18535 for fruits) (Tab.3 and 4) with the stomatal aperture was found, which indicates that stomata respond positively to a RH increase (Fig.8) as confirmed by Salisbury and Ross (1992), a different situation from that found in the purple passion fruit.
According to variance analysis, the P was less than or equal 0.0001 in all the factors, which indicates that significant differences exist between the variables, therefore the three measured environmental factors have an effect on stomatal aperture in yellow pitaya fruits.
The analysis for cladodes showed a P≤0.0001 for the three environmental variables, which indicates that radiation, as well as temperature and RH, had an effect on stomatal aperture in pitaya cladodes.
Comparison of stomatal behavior between the purple passion fruit and the yellow pitaya
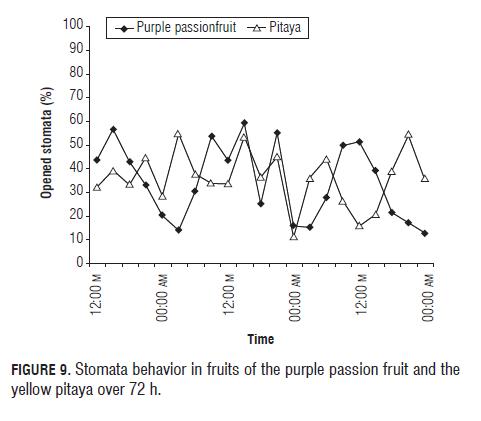
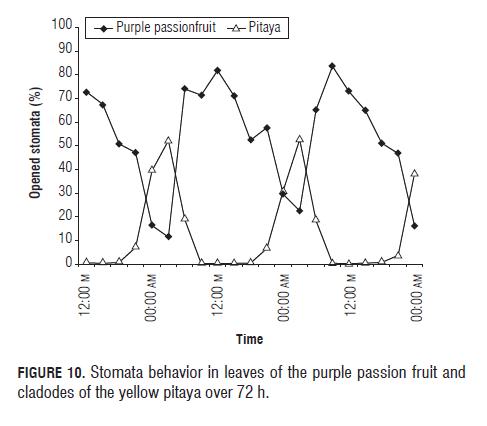
Stomatal behavior, both of yellow pitaya and purple passion fruit fruits, is shown in Fig.9, where we can see the behavior of a C3 and a CAM plant, showing that while the stomata of purple passion fruit fruits are opening, the stomata of pitaya fruits are closing, therefore the purple passion fruit behavior is totally opposite to the pitaya behavior (Salisbury y Ross, 1992). We suppose that the certain percentage of opened stomata in the day of the pitaya fruit (Fig.9) signifies that the fruit (Fig.10) has a higher ability of fixing CO2, in contrast to the cladode, via the Calvin cycle, especially at the end of the day; and this behavior is encouraged by the wet conditions (Taiz and Zeiger, 2006) in which the studied pitaya plants were grown.
The stomatal behavior of purple passion fruit leaves as compared to pitaya cladodes is illustrated in Fig.10, where the behaviors are a lot more contrasting then in fruits, where the behaviors are exclusively, totally inverse, while the stomata of pitaya cladodes are closed, the stomata of purple passion fruit leaves are opened and vice versa (Osmond, 1978; Salisbury y Ross, 1992; Winter y Smith, 1996).
Stomatal densities in studied C3 and CA M species
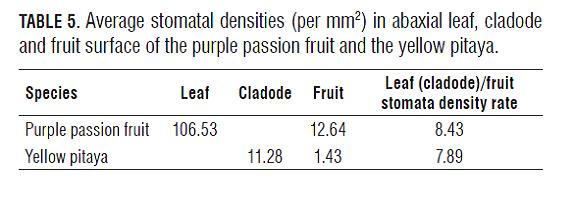
In the two species, stomatal density was much lower in fruits than in leaves, however, these are two different CO2- fixing plant types, a similar fruit/leaf stomata frequency rate was observed (Tab.5). Our results coincide with Blanke (1990), that the stomatal frequency is 5-10 fold lower in subtropical fruits than in the abaxial epidermis of their respective leaves. A similar rate (8.57) was observed by Moreshet and Green (1980) in Shamouti' oranges with 70 stomata/mm2 in fruits and 600 in leaves, whereas Blanke (1990) reported 60 and 730 stomata per mm2 in fruit and leaves of the avocado (rate: 12.17), respectively.
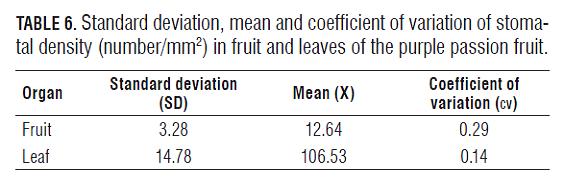
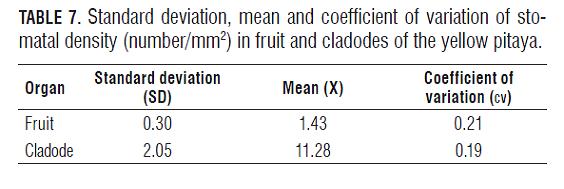
According to the analysis, the cv of the stomatal density of the fruits and leaves in the purple passion fruit were very low (Tab.6), which indicates a low variation between the data found. A similar situation with the fruits and cladodes of the pitaya was found (Tab.7). The standard deviation, both of the purple passion fruit and the pitaya, was very low too, indicating a very large similarity between the data and a high correlation between
The stomatal density in purple passion fruit leaves (106.53) was similar to those commonly found in plant leaves (100), as reported Salisbury and Ross (1992). Also in pitaya cladodes, the stomata frequency agrees with those of other CAM plants. Goh et al. (1977) observed, in orchids Arachnis cv. Maggie Oei, Aranda cv. Deborah, Cattleya howringiana, Arundina graminifolia, Bromheadia finlaysoniana and Spathoglottis plicata, densities of 4, 3, 11, 18, 18 and 14 stomata/ mm2, respectively. These low stomatal densities (and water conserving big vacuoles) allow these plants to achieve higher water-use efficiency than C3 or C4 plants (Evert and Eichhorn, 2012) and to inhabit arid environments with seasonal water availability (Taiz and Zeiger, 2006).
In studies of this kind, it should be taken into account that stomatal densities can be influenced by a lot of factors such as: genetic (variety) and environmental, including CO2 concentration, irradiancy and humidity (Royer, 2001). Generally, few studies about plant photosynthesis have included stomata on fruit surfaces although they are very important in the initial stages of fruit development where gas exchange can compensate for the respiration loss of this organ through photosynthesis (Blanke and Lenz, 1989).
Conclusions
Stomata behave in a similar way between the same species, with a rhythm of stomatal closing and opening almost coordinated between their organs.
A high correlation between radiation and temperature with stomatal aperture was found, suggesting the influence of these on stomata, but was not found with RH.
The pitaya stomata show a typical behavior of a CAM plant, in which they open their stomata at night and close them during the day as a adaptation mechanism to environmental conditions.
The stomatal density in the leaves and cladodes was almost 10 times higher than in fruits in each species, which shows the functional differentiation of these organs in plants.
Changes in the stomatal aperture grade reflect the cumulative effect of many physiological responses by a leaf or a fruit to the environment, meaning that knowledge of the stomatal response increases the understanding of the rates of carbon dioxide assimilation and transpiration, as well as the nature of the ecophysiological adaptations of plants to their environment.
Acknowledgements
The authors wish to thank to Dr. Tom Lee Davenport (University of Florida) and Nelson Guaquetá for their important advice in planning and executing this ecophysiological study.
Literature cited
Agronet. 2012. Producción de Gulupa en Colombia, 2007-2011; producción de pitahaya en Colombia, 1992-2011. In: http://www.agronet.gov.co/www/htm3b/ReportesAjax/parametros/reporte16_2011.aspx?cod=16; consulted: March, 2013. [ Links ]
Assmann, S.M. 1993. Signal transduction in guard cells. Annu. Rev. Cell. Biol. 9, 345-375. [ Links ]
Assmann, S.M. and X.Q. Wang. 2001. From milliseconds to millions of years: guard cells and environmental responses. Curr. Opin. Plant Biol. 4, 421-428. [ Links ]
Bergmann, D.C. and F.D. Sack. 2007. Stomatal development. Annu. Rev. Plant Biol. 58, 163-81. [ Links ]
Blanke, M.M. 1990. Photosynthesis in subtropical fruits. Acta Hort. 275, 435-439. [ Links ]
Blanke, M.M. 1992. Photosynthesis of avocado fruit. pp. 179-189. In: Proc. Second World Avocado Congress. Vol. I. University of California, Davis, CA. [ Links ]
Blanke, M.M. and F. Lenz. 1989. Fruit photosynthesis - a review. Plant Cell Environ. 12, 31-44. [ Links ]
Brewer, C.A. 1992. Responses by stomata on leaves to microenvironmental conditions. pp. 67-75. In: Goldman, C.A. (ed.). Tested studies for laboratory teaching. Vol. 13. Proc. 13th Workshop/ Conference of the Association for Biology Laboratory Education. http://www.ableweb.org/volumes/vol-13/3-brewer.pdf; consulted: February, 2013. [ Links ]
Brinker, M., W. Engelmann, J.W. Kellmann, and B. Piechulla. 2001. Circadian rhythms of leaf and stomatal movements in gymnosperm species. Biol. Rhythm Res. 32(4), 471-478. [ Links ]
Corredor, D. 2012. Pitahaya amarilla (Hylocereus megalanthus [K. Schum. ex Vaupel] Ralf Bauer). pp. 802-824. In: Fischer, G. (ed.). Manual para el cultivo de frutales en el trópico. Produmedios, Bogota. [ Links ]
Evert, R.F. and S.E. Eichhorn. 2012. Raven biology of plants. 8th ed. W.H. Freeman, New York, NY . [ Links ]
Fageria, N.K., V.C. Baligar, and R.B. Clark. 2006. Physiology of crop production. Food Products Press, New York, NY . [ Links ]
Fischer, G., F. Casierra-Posada, and W. Piedrahíta. 2009. Ecofisiología de las especies pasifloráceas cultivadas en Colombia. pp. 45-67. In: Miranda, D., G. Fischer, C. Carranza, S. Magnitskiy, F. Casierra, W. Piedrahíta, and L.E. Flórez (eds.). Cultivo, poscosecha y comercialización de las pasifloráceas en Colombia: maracuyá, granadilla, gulupa y curuba. Sociedad Colombiana de Ciencias Hortícolas, Bogota. [ Links ]
Fischer, G. and J.O. Orduz-Rodríguez. 2012. Ecofisiología en frutales. pp. 54-72. In: Fischer, G. (ed.). Manual para el cultivo de frutales en el trópico. Produmedios, Bogota. [ Links ]
Goh, C.J., P.N. Avadhani, C.S. Loh, C. Hanegraaf, and J. Arditti. 1977. Diurnal stomatal and acidity rhythms in orchid leaves. New Phytol. 78, 365-372. [ Links ]
Guaquetá, N.O. 2007. Comportamiento estomatal en frutas y hojas de banana, carambola, guayaba y cítricos en condiciones naturales y bajo almacenamiento hipobárico. Undergraduate thesis. Agronomy Faculty, Universidad Nacional de Colombia, Bogota. [ Links ]
Hall, A.E. 2001. Crop responses to environment. CRC Press, Boca Raton, FL. [ Links ]
Hetherington, A.M. and F.I. Woodward. 2003. The role of stomata in sensing and driving environmental change. Nature 424, 901-908. [ Links ]
Hess, D. 2004. Allgemeine Botanik. Verlag Eugen Ulmer, Stuttgart, Germany. [ Links ]
Hieke, S., C.M. Menzel, and P. Lüdders. 2002. Effects of leaf, shoot and fruit development on photosynthesis of lychee trees (Litchi chinensis). Tree Physiol. 22, 955-961. [ Links ]
Jiménez, Y., C. Carranza, and M. Rodríguez. 2012. Gulupa (Passiflora edulis Sims). pp. 579-599. In: Fischer, G. (ed.). Manual para el cultivo de frutales en el trópico. Produmedios, Bogota. [ Links ]
Kearns, E.V. and S.M. Assmann. 1993. The guard cell-environment connection. Plant Physiol. 102, 711-715. [ Links ]
Kramer, P.J. 1989. Relaciones hídricas de suelos y plantas. Harla, México DF. [ Links ]
Lake, J.A., W.P. Quick, D.J. Beerling, and F.I. Woodward. 2001. Plant development. Signals from mature to new leaves. Nature 411, 154. [ Links ]
Laurin, É., M.C.N. Nunes, J.P. Emond, and J.K. Brecht. 2006. Residual effect of low-pressure stress during simulated air transport on Beit Alpha-type cucumbers: Stomata behavior. Postharv. Biol. Technol. 41, 121-127. [ Links ]
Menzel, C.M. and D.R. Simpson. 1994. Passionfruit. pp. 225-241. In: Schaffer, B. and P. Andersen (eds.). Handbook of environmental physiology of fruit crops. Vol. II . Sub-tropical and tropical crops. CRC Press, Boca Raton, FL. [ Links ]
Moreshet, S. and G.C. Green. 1980. Photosynthesis and diffusion conductance of Valencia orange fruit under field conditions. J. Exp. Bot. 31, 15-27. [ Links ]
Morton, J.F. 1987. Fruits of warm climates. Creative Resources Systems, Winterville, NC. [ Links ]
Mott, K.A. and D.F. Parkhurst. 1991. Stomatal responses to humidity in air and helox. Plant Cell Environ. 14, 509-515. [ Links ]
Öpik, H. and S. Rolfe. 2005. The physiology of flowering plants. Cambridge University Press, Cambridge, UK. [ Links ]
Osmond, C.B. 1978. Crassulacean acid metabolism: a curiosity in context. Annu. Rev. Plant Physiol. 29, 379-414. [ Links ]
Pritchard, S.G. and J.S. Amthor. 2005. Crops and environmental change. Food Products Press, New York, NY . [ Links ]
Pospísilová and Dodd. 2005. Role of plant growth regulators in stomatal limitation to photosynthesis during water stress. pp. 811-825. In: Pessarakli, M. (ed.). Handbook of photosynthesis. 2nd ed. CRC Press, Boca Raton, FL. [ Links ]
Royer, D.L. 2001. Stomatal density and stomatal index as indicators of paleoatmospheric CO2 concentration. Rev. Palaeobot. Palynol. 114, 1-28. [ Links ]
Salisbury, F. and C.W. Ross. 1992. Plant physiology. 4th ed. Wadsworth Publishing, Belmont, CA. [ Links ]
Sharkey, T.D. and T. Ogawa. 1987. Stomatal response to light. pp. 195-208. In: Zeiger, E., G.D. Farquhar, and I.R. Cowan (eds.). Stomatal function. Stanford University Press, Stanford, CA. [ Links ]
Taiz, L. and E. Zeiger. 2006. Plant physiology. 4th ed. Sinauer Associates, Sunderland, MA. [ Links ]
Wang, X.Q., W.H. Wu, and S.M. Assmann. 1998. Differential responses of abaxial and adaxial guard cells of broad bean to abscisic acid and calcium. Plant Physiol. 118, 1421-1429. [ Links ]
Wilkins, M.B. 1959. An endogenous rhythm in the rate of dark fixation of carbon dioxide in leaves of Bryophyllum: II . The effects of light and darkness on the phase and period of the rhythm. J. Expl. Bot. 10, 377-390. [ Links ]
Wilkins, M.B. 1962. An endogenous rhythm in the rate of dark fixation of carbon dioxide in leaves of Bryophyllum: III . The effects of temperature changes on the phase and period of the rhythm. Proc. Royal Soc. London B 156, 220-241. [ Links ]
Willmer, C. and M. Fricker. 1996. Topics in plant functional biology, 2. Stomata. 2nd ed. Chapman and Hall, London. [ Links ]
Winter, K. and J.A.C. Smith. 1996. Crassulacean acid metabolism: Biochemistry, ecophysiology and evolution. Ecological Studies Vol. 114. Springer-Verlag, Berlin. [ Links ]
Wong, S.C., I.R. Cowan, and G.D. Farquhar. 1978. Leaf conductance in relation to assimilation in Eucalyptus pauciflora Sieb. ex Spenge. Influence of irradiance and partial pressure of carbon dioxide. Plant Physiol. 62, 670-674. [ Links ]
Woodward, F.I. and F.A. Bazzaz. 1988. The responses of stomatal density to CO2 partial-pressure. J. Expl. Bot. 39, 1771-1781. [ Links ]
Zeiger, E., G.D. Farquhar, and I.R. Cowan. 1987. Stomatal function. Stanford University Press, Stanford, CA. [ Links ]