Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Agronomía Colombiana
Print version ISSN 0120-9965
Agron. colomb. vol.31 no.1 Bogotá Jan./Apr. 2013
SOILS, FERTILIZATION & MANAGEMENT OF WATER
Assessment of the impact of anthropic activities on carbon storage in soils of high montane ecosystems in Colombia
Determinación del impacto de actividades antrópicas en el almacenamiento de carbono en suelos de ecosistemas de alta montaña en Colombia
Orlando Zúñiga-Escobar1 , Andrés Uribe V. 2, Alba Marina Torres-González2, Ramiro Cuero-Guependo1 and Julián Andrés Peña-Óspina1
1Grupo de Investigación en Ciencias Ambientales y de la Tierra (ILA MA), Facultad de Ciencias Naturales y Exactas, Universidad del Valle. Santiago de Cali (Colombia). orlando.zuniga@correounivalle.edu.co2Grupo de Investigación en Biología Vegetal Aplicada (BVA), Facultad de Ciencias Naturales y Exactas, Universidad del Valle. Santiago de Cali (Colombia).
Received for publication: 3 March, 2011. Accepted for publication: 29 March, 2013.
ABSTRACT
The organic carbon in the soil was quantified to assess the impact of anthropic activities on montane ecosystems in Colombia in Chingaza Parque Nacional Natural (PNN ) and Los Nevados Parque Nacional Natural (PNN ). For the development of the soil samples, a detailed in situ description of the edaphological profile of four ecosystems of paramo and high Andean forest areas, of both disturbed and undisturbed zones, was taken as the base. The calculation of the amount of total carbon stored by the soil profile shows that, in Colombia, undisturbed high montane ecosystems (520.9 t ha-1 in paramos and 323.6 t ha-1 in high Andean forests of Chingaza PNN , and 373.0 t ha-1 in paramos and 254.6 t ha-1 in high Andean forests of Los Nevados PNN ) currently have more carbon than disturbed ecosystems (135.1 t ha-1 in paramos and 141.5 t ha-1 in high Andean forests of Chingaza PNN , and 356.3 t ha-1 in paramos and 217.1 t ha-1 in high Andean forests of Los Nevados PNN ). It is clear that the disturbance of high montane ecosystems decreases the amount of carbon in the soil, a situation that is more concerning in Chingaza PNN where the difference between the disturbed and undisturbed ecosystems is much more marked than in Los Nevados PNN .
Key words: soil microorganisms, Andean zone, paramos, natural parks
RESUMEN
Se cuantificó el carbono orgánico en el suelo para evaluar el impacto de las actividades antrópicas en los ecosistemas de alta montaña en Colombia en el Parque Nacional Natural (PNN ) Chingaza y en el Parque Nacional Natural (PNN ) Los Nevados. Para el desarrollo del muestreo de suelos se tuvo como base la descripción detallada in situ del perfil edafológico en cuatro ecosistemas correspondientes a zonas de páramo y bosque alto andino en sus variaciones intervenido y no intervenido. El cálculo de cantidad de carbono total almacenado por perfil de suelo muestra que en Colombia ecosistemas no intervenidos de alta montaña (520,9 t ha-1 en páramo y 323,6 t ha-1 en bosque alto andino del PNN Chingaza; y 373,0 t ha-1 en páramo y 254,6 t ha-1 en bosque alto andino del PNN Los Nevados) presentan en la actualidad mayor cantidad de carbono que ecosistemas intervenidos (135,1 t ha-1 en páramo y 141,5 t ha-1en bosque alto andino del PNN Chingaza; y 356,3 t ha-1 en páramo y 217,1 t ha-1 en bosque alto andino del PNN , Los Nevados). Es evidente que la intervención en los ecosistemas de alta montaña disminuye la cantidad de carbono presente en el suelo; situación que se hace más preocupante en el PNN Chingaza donde la diferencia entre el ecosistema no intervenido y su correspondiente intervenido es mucho más marcada que en el PNN , Los Nevados.
Palabras clave: microorganismos del suelo, zona andina, páramos, parques naturales.
Introduction
The increasing concentrations of carbon dioxide (CO2) and other gases have produced an altered balance between incoming and outgoing radiation beyond the sum of the remainder of known disturbances and could thus be the dominant cause of the warming observed during the last hundred years (BBVA, 2000).
With respect to the content of carbon dioxide (CO2), the increase has been caused mainly by two human activities: change in land use and combustion of fossil fuels. It is estimated that the conversion of land for agricultural use emits 5.9 Gt CO2-eq/year (1 Gt = 1·109 t) (Seeberg 2010) while the burning of fossil fuels emits 27.7 Gt CO2-eq/ year and deforestation, degradation 8.5 Gt CO2-eq/year (IPCC, 2007).
There are several problems in terms of the various factors that control the level of carbon in soils, such as the type and use of the land, climate and agricultural management practices. The amount of organic carbon in soil worldwide is directly dependent on the changes that are experienced by atmospheric carbon (Kimble et al., 1998).
There are estimates on how much a doubling of atmospheric carbon content could affect high montane ecosystems, but it is also important to know what changes have been generated in recent decades by global warming. Recent estimates conclude that the average global air temperature has increased by 0.6±0.2°C over the last hundred years (Houghton et al., 2001). These trends may be affecting montane ecosystems (IDEAM, 2002). The Primera Comunicación Nacional (CN1) de Colombia report to the United Nations Framework Convention on Climate Change found that the Colombian ecosystem more vulnerable to the effects of climate change is high montane. With a projected increase by 2050 in the average air temperature of the country of between 1 and 2°C and a variation in precipitation of ±15%, it is expected that 78% of the snowcaps and 56% of the paramos will disappear (MAVDT et al., 2001).
The important role of high montane ecosystems at various stages of growth as a sink for carbon dioxide adds to the need for conservation, justified by the fact that they store significant amounts of carbon, whose potential emissions should be avoided by controlling logging and unsustainable management practices (IDEAM, 2002).
While exact scenarios are still uncertain, serious negative effects are expected (although some positive effects are also expected), so it is essential that a number of measures be taken to reduce greenhouse gas emissions and increase their capture in soils and biomass. Therefore, new strategies and policies for the management of agriculture and forests must be developed. Since the Kyoto Protocol, this is known as Land Use, Land-Use Change and Forestation (LULUCF) and concerns Articles 1.3 and 1.4 of the Protocol (IPCC, 2000).
Monitoring the carbon content in high montane ecosystems has gained global importance today given the potential role they can play as sinks or sources of carbon and, consequently, reducing the emission of greenhouse gases (CO2) (Watson et al., 2000; Houghton et al., 2001). This research evaluated the impact of climate change on the carbon and water cycles in high montane ecosystems, conducted between 2007 and 2008, and aimed to determine the influence of human intervention on carbon in the soil. The impact of human activities on carbon storage was evaluated in soils of high montane ecosystems in Colombia in Chingaza (PNN ) and Los Nevados (PNN ) in different paramo and high Andean forest areas, in both disturbed and undisturbed zones.
Materials and methods
A proposed abductive methodology arising from the protocol for the characterization of carbon and water cycles in high, worked montane ecosystems was used.
Area of study
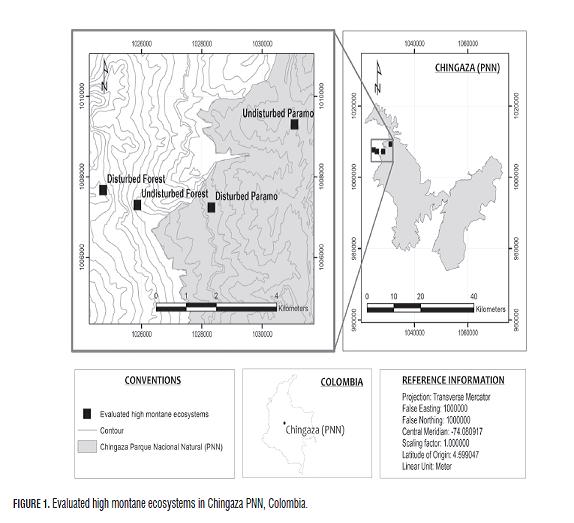
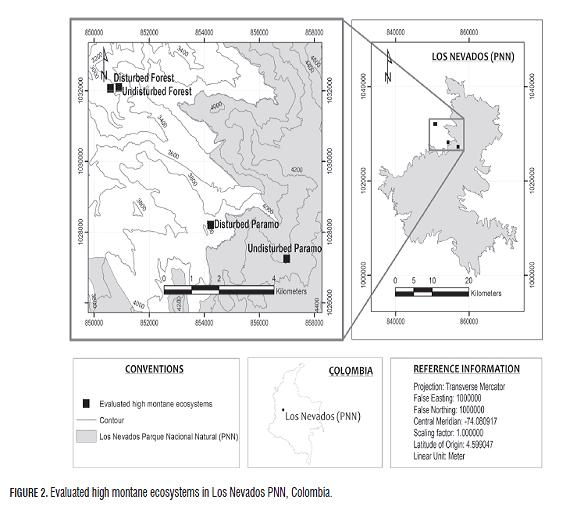
The study was conducted in paramos and high Andean forests in Colombia to evaluate the impact of anthropic activities on carbon storage in soils of Chingaza PNN (Fig.1) and Los Nevados PNN (Fig .2). The ecosystems and monitored study areas were selected using the wind rose methodology, considering technical factors, socioeconomics, logistics and security in the area.
Procedure for sampling in the paramos and high Andean forests
In the quantification and analysis of soil carbon in high montane ecosystems, both disturbed and undisturbed paramos and high Andean forests were evaluated. In each of the ecosystems, two to four pits were assessed according to the agroclimatic variability and in search of uniformity of such conditions. Generally, the terrain is hilly; the topography of the sampling sites is broken up with slopes between 30 and 40%, with vegetation unique to each ecosystem and traces of potato plants and pastures, and extensive development of ranching in disturbed ecosystems.
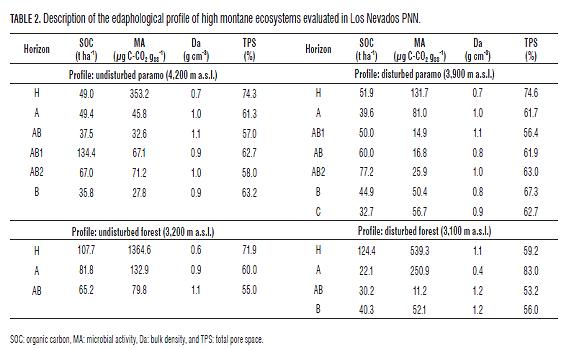
The evaluations were conducted in wetlands with soil of the Inceptisol order, young soils, Cryaquents and Dystrocryepts, for paramos and wetlands of the Andisol order, of recent volcanic activity, Humitropepts and Andaquepts. Soil sampling was performed more accurately considering its anisotropy to differentiate the variability of the horizons in each profile. In each test site defined by the ecosystems representative of high montane, a 1 x 1 x 1 m pit was excavated where a detailed in situ description of the soil of the profile was carried out, discriminated by horizons; and in each defined horizon, specific samples were taken to determine the organic carbon, microbial activity, bulk density and total pore space (UNIVALLE and IDEAM, 2008).
Soil organic carbon (SOC)
The SOC (contents of soil organic carbon in t ha -1) content was calculated from the percentage values of carbon (determined by the Walker - Black method), bulk density (determined by the ETSIA method, Universidad Politécnica de Madrid) and volume of the sample, with equation 1 (Rosenzweig and Hillel, 2000).
Soil microbial activity
To estimate the soil microbial activity through respiration (C-CO2), the Centro de Agrobiología del Brasil methodology was used. The sample was incubated for 5 d in a closed system, NaOH 1N was added and precipitated with BaCl2; and two drops of 1% phenolphthalein were added. A purple color indicates a basic pH by the formation of BaCO3 and NaCl. An d, the sample was titrated with HCl 0.5 N to quantify the amount of hydroxide that was unreacted with CO2. A control sample was always included. The mineralized carbon in these experiments has been reported as an excellent indicator of change in the SOC, which represents the active fraction of soil organic matter (Carter and Rennie, 1982).
Results and discussion
The characteristics of the soil profile in Los Nevados PNN and Chingaza PNN are very different in relation to carbon sequestration.
Soils in Chingaza PNN are deep with organic horizons characteristic of carbon uptake, while soils in Los Nevados PNN have broad horizons that are highly differentiated by the volcanic ash characteristic of the volcanic phenomena of the Cordillera Central Colombiana, alternating with small horizons corresponding to organic soil with carbon deposits.
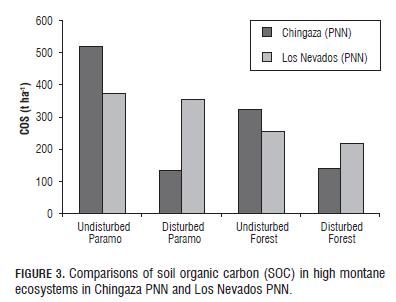
The statistical results of the amounts of SOC, weighted thickness of each horizon to a meter deep in the paramos in Chingaza PNN and Los Nevados PNN (Fig.3) are close to those found in paramos of Ecuador in the provinces of Carchi, Canar, Azuay, Chimborazo, Pichincha and Loja with values of 463, 466, 364, 479, 356 and 239 t ha-1 , respectively (Hofstede et al., 2003).
Amezquita et al. (2006) found SOC in high Andean forests of Colombia at 155-231 t ha-1, close to that found in the same ecosystems in Los Nevados PNN and Chingaza PNN (Fig.3). Kanninen (2003) with data from Brown and Lugo (1992) and Brown et al. (1969), also found SOC stored in cloud forests at 190-230 t ha-1 , close to those found in this study in the two national parks.
This highlights the superiority of the high montane forests of Chingaza PNN and Los Nevados PNN as storers of SOC as compared to the findings of other studies in neotropic, afrotropic, palearctic and nearctic ecozones. In several neotropic studies: Hofstede (1999) found quantities of SOC in rainforests of 50 t ha-1 and Batjes (1999) reported storage at 80-100 t ha-1 , values lower than those found in the forests of this study; in Colombia, SOC storage has been seen in rainforests at 52 t ha-1 (Ibrahim et al., 2007), in jungles at 75 t ha-1 (Escobar and Toriatti Demattê, 1991) and 102 t ha-1 (Carvajal et al., 2009), and in tropical rainforests at 107 t ha-1 and 181 t ha-1 (Amezquita et al., 2006); in Guatemalan rainforests, SOC storage has been seen at 130.4 t ha-1 (Arreaga, 2002); Ibrahim et al. (2007) found quantities of SOC at 139 t ha-1 in tropical rainforests of Nicaragua; SOC storage was determined in Costa Rican tropical jungles at 82.2 t ha-1 (Powers and Schlesinger, 2002), and in tropical rainforests, at 116 t ha-1 (Ibrahim et al., 2007) and 134 t ha-1 (Amezquita et al. 2006); storage of 88.5 t ha-1 was found in tropical forests of Ecuador by López-Ulloa et al. (2005); in tropical forests of Venezuela, SOC values were determined at 125 t ha-1 (Delaney et al., 1997); in Brazilian rainforests, SOC quantities of 162 t ha-1 were found (IP CC, 2000); in Hawaiian forests, Schuur et al. (2001) reported SOC storage at 62.5 t ha-1 .
Tropical soils as carbon sinks are questioned on the grounds that the rates of decomposition of organic matter are very high in these latitudes, due to increased moisture and temperature conditions as compared with temperate regions (Moreno and Lara, 2003). In the afrotropics, Glenday (2006) determined SOC at 100 t ha-1 in forests of Kenya. In the palearctic forests of France, Dupouey et al. (1999) reported SOC stores at 71 t ha-1; this figure is close to the nearctic forests found in the United States of America (IP CC, 2000).
The soils of paramo ecosystems clearly have a higher carbon sequestration capacity than the respective high montane forests in the two national parks (Fig.3), because they are of volcanic origin with a high organic matter content, and maintain the microbial activity at low temperatures in the soil, decomposing the organic carbon at a rate lower than in the said forests (Tabs.1 and 2). This same relationship of microbial activity, temperature and organic carbon is present in studies by Moretto et al. (2005) and Montagnini and Jordan (2002). In addition, volcanic ash aluminum and organic matter combine to form vesicles very resistant to decomposition by microorganisms (Hofstede and Mena, 2003).
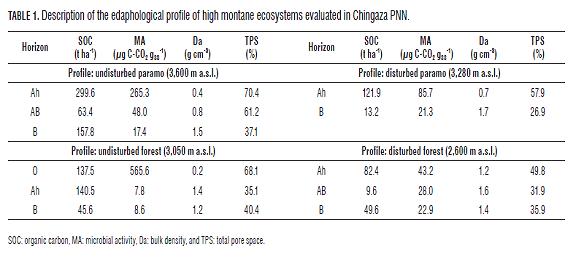
The SOC contents depended on the depth, with the deeper the soil profile, the less accumulated SOC found (Tabs.1 and 2). Similar results were found by Carvajal et al. (2009).
The soils of the undisturbed high montane ecosystems in Chingaza PNN currently have more carbon than those in Los Nevados PNN (Fig.3), this is due to the wetter climatic conditions in Chingaza PNN , with a annual rainfall of 2,700 mm for the study area (Vargas and Pedraza, 2004), compared to the drier environment in Los Nevados PNN with an average annual rainfall of 1,500 mm for the area sampled (Lotero et al., 2006); therefore, Chingaza PNN presents a lower microbial activity than Los Nevados PNN (Tabs.1 and 2); this situation is due to the oxygen concentration in the soil decreasing when the pores are filled with water, preventing the diffusion of atmospheric oxygen, a situation that is further accentuated by the low total pore space in Chingaza PNN as compared to Los Nevados PNN (Tabs.1 and 2); since decomposition is much slower in anaerobic soils, they accumulate more organic matter in a partially decomposed condition (Brady and Weil, 2002; Lal, 2004; Palacios-Bianchi, 2002; Kimmins, 1997). Batjes (1999), IP CC (2000) and Kanninen (2003) showed the same trend in relation to temperature and rainfall; according to Batjes (1999), SOC stored up to one meter in depth range from 40 t ha-1 in arid zones to 210-240 t ha-1 in polar or boreal regions, with intermediate values of 80 to 100 t ha -1 in the tropics. On the other hand, the soils of the mountain range in Chingaza PNN are enriched by deposits of a submarine origin with abundant Cretaceous fossils and glacial valley planes (Garcés and De la Zerda, 1994), in contrast to the sharp relief of the mountain range of Los Nevados PNN .
In wetlands of Los Nevados PNN and Chingaza PNN near the undisturbed paramos in this study, Peña et al. (2009) found that dissolved and total organic carbon were significantly lower in the wetlands of Los Nevados PNN with average values of 1.2 mg L-1 , followed by the wetlands of Chingaza PNN with average values of 2.8 mg L -1 , which coincides with the greater accumulation of SOC seen in Chingaza PNN as compared to Los Nevados PNN , thus demonstrating that Chingaza PNN has the greater potential as a carbon sink.
In Los Nevados PNN and Chingaza PNN , one can observe the superiority of SOC storage in undisturbed ecosystems over disturbed ones (Fig.3). Other studies have found the same trend; Carúa et al. (2008) and Hernández and Triana (2009) determined the organic matter in the soil of paramos, which is a parameter directly proportional to SOC (Lal, 2004), finding a higher content in undisturbed ecosystems verses disturbed ones; undisturbed high Andean forest ecosystems had a higher SOC content than disturbed ecosystems, according to Díaz (2008), this behavior is due to the high percentage of aggregates in the soil of a size <0.125 mm in high Andean forests, within which there is high protection for organic carbon and microbial processes become recalcitrant (Lal, 2004), so that carbon may remain in the soil for long periods of time.
In the two national parks, deterioration in the soil structure was seen in the disturbed ecosystems with the observation of higher values of bulk density and lower total pore space as compared to the undisturbed ones (Tabs.1 and 2). Díaz (2008), in high Andean forests, and Carúa et al. (2008) and Hernández and Triana (2009), in paramos, found the same trend in comparing undisturbed and disturbed ecosystems. Only the soil of disturbed paramo ecosystems of Los Nevados PNN did not present structural damage when compared with the corresponding undisturbed ecosystem.
Land degradation in Chingaza PNN was demonstrated to be great when the levels of organic carbon, bulk density and total pore space were compared for the disturbed and undisturbed ecosystems (Tabs.1 and Fig.3), this is because of the practice of burning, which occurs in these paramos to generate younger, tender grasses that provide food for livestock (Laegaard, 1992, Vargas 2000, Vargas et al. 2002), on the other hand, the use of soils in this area has been, perhaps for more than 300 years, primarily oriented toward agriculture and livestock activities such as potato cultivation and extensive ranching, practices that in some cases involve the introduction of improved pastures (Vargas and Pedraza, 2004). Thus, the disturbed high montane ecosystems of Los Nevados PNN have more SOC and less impact on the structure as compared to those in Chingaza PNN (Fig.3 and Tabs.1 and 2).
Conclusions
High montane ecosystems that undergo land use change to perform agriculture decrease their coverage and there is a danger that much of the carbon will decompose and escape to the atmosphere as carbon dioxide: the main cause of global warming, possibly the most serious global environmental problem.
It is clear that the disturbance of soils in high montane ecosystems decreases the amount of organic carbon and degrades the structure; a situation that is more concerning in Chingaza PNN where the difference in SOC content between the undisturbed ecosystems and the disturbed ones is much more marked than in Los Nevados PNN .
The soils of the undisturbed high montane ecosystems in Chingaza PNN currently have more organic carbon than those in Los Nevados PNN .
The soils of the studied paramo ecosystems obviously have a greater carbon retention capacity than the high Andean forests in both study areas.
Acknowledgements
This research was made possible by the donation from Japan to the Proyecto Piloto Nacional Integrado de Adaptación al Cambio Climático: ecosistemas Colombianos de alta montaña, áreas insulares del Caribe y salud humana and the work of the Grupo Técnico y Científico del Instituto de Hidrología, Meteorología y Estudios Ambientales (IDEAM) and the Grupo de Ecosistemas de Alta Montaña (Ecoalmo).
Literature cited
Amezquita, M.C., B. Van Putten, M. Ibrahim, B.L. Ramírez, H. Giraldo, and M.E. Gómez. 2006. Recovery of degraded pasture areas and c-sequestration in ecosystems of tropical America. WSEAS Trans. Environ. Develop. 2(8), 1085-1091. [ Links ]
Arreaga, W.E. 2002. Almacenamiento de carbono en bosques con manejo forestal sostenible en la Reserva de Biosfera Maya, Petén, Guatemala. M.Sc. thesis. CATIE, Turrialba, Costa Rica. [ Links ]
Batjes, N.H. 1999. Management options for reducing CO2- concentrations in the atmosphere by increasing carbon sequestration in the soil. International Soil Reference and Information Centre (ISRI C), Wageningen, The Netherlands. [ Links ]
Brady, N. and R. Weil. 2002. The nature and properties of soils. 13th ed. Prentice-Hall, Upper Saddle River, NJ . [ Links ]
BBVA. 2000. El Cambio Climático. El campo de las ciencias y las artes. Servicio de Estudio del Banco Bilbao Vizcaya Argentaria, Madrid. [ Links ]
Brown, S. and A. Lugo. 1992 Aboveground biomass estimates for tropical moist forest of the Brazilian Amazon. Interciencia 17(1), 8-18. [ Links ]
Brown, S., A.J. Gilliespie, and A.E. Lugo. 1989. Biomass estimation methods for tropical forests with applications to forest inventory data. Forest Sci. 35(4), 881-902. [ Links ]
Carter, M.R. and D.A. Rennie. 1982. Changes in soil quality under zero tillage farming systems: distribution of microbial biomass and mineralizable C and N potentials. Can. J. Soil Sci. 62, 587-597. [ Links ]
Carúa, J., M. Proaño, D. Suarez, and P. Podwojevski. 2008. Determinación de retención de agua en los suelos de los páramos: estudio de caso en la subcuenca del Río San Pedro, Cantón Mejía, Pichincha, Ecuador. pp. 27-39. In: La Forestación en el Páramo. Serie Páramo 26. GTP; Abya Yala. Quito. [ Links ]
Carvajal, A., A. Feijoo, H. Quintero, and M. Rondón. 2009. Carbono orgánico del suelo en diferentes usos del terreno de paisajes andinos Colombianos. Rev. Cienc. Suelo Nutr. 9(3), 222-235. [ Links ]
Delaney, M., S. Brown, A.E. Lugo, A. Torres-Lezama, N. Bello- Quintero. 1997. The distribution of organic carbon in major components of forest located in five zones of Venezuela. J. Trop. Ecol. 13, 697-708. [ Links ]
Díaz, E. 2008. Distribución del contenido de carbono orgánico en agregados de diferentes tamaños, procedentes de varios sistemas de uso y altitudes en suelos de la cuenca del río Cauca, Colombia. M.Sc. thesis. Universidad Nacional de Colombia, Palmira, Colombia. [ Links ]
Dupouey, J.L., G. Siguand, V. Bateau, A. Thimonier, J.F. Dhole, G. Nepveu, L. Bergé, L. Augusto, S. Belkacem, and C. Nys. 1999. Stocks et flux de carbone dans les forêts françaises. C.R. Acad. Agric. Fr. 85(6), 293-310. [ Links ]
Escobar, C.J. and J.L. Toriatti Dematté. 1991. Distribución de la materia orgánica y del carbono-13 en un Ultisol del piedemonte amazónico. Pasturas Tropicales 13(2), 27-30. [ Links ]
Garcés, D. and S. De La Zerda. 1994. Gran libro de los parques nacionales de Colombia. Intermedio Editores, Bogota. [ Links ]
Glenday, J. 2006. Carbon storage and emissions offset potential in an East African tropical rainforest. Forest Ecol. Manag. 235, 72-83. [ Links ]
Hernández, F. and A. Triana. 2009. Efecto de actividades agropecuarias en la capacidad de infiltración de los suelos del Páramo del Sumapaz. Rev. EIDENAR (8), 29-38. [ Links ]
Hofstede, R. 1999. El páramo como espacio para la fijación de carbono atmosférico. In: Medina, G. and P. Mena (eds). El páramo como espacio para la fijación de carbono atmosférico. Serie Páramo 1. GTP; Abya Yala, Quito. [ Links ]
Hofstede, R. and P. Mena. 2003. Los beneficios escondidos del páramo: servicios ecológicos e impacto humano. Ecociencia Foundation; Mountain Institute; University of Amsterdam, Quito. [ Links ]
Hofstede, R., P. Segarra, and P.V. Mena. 2003. Los páramos del mundo. Proyecto Atlas Mundial de los Páramos. Global Peatland Initiative; NC-IU CN; EcoCiencia, Quito. [ Links ]
Houghton, J.T., Y. Ding, D.J. Griggs, M. Noguer, P.J. Van der Linden, X. Dai, K. Maskell, and C.A. Jonhnson. 2001. The scientific basis. Contribution of Working Group 1. Intergovernmental Panel on Climate Change (IP CC), Cambridge University Press, Cambridge, UK. [ Links ]
Ibrahim, M., M. Chacón, C. Cuartas, J. Naranjo, G. Ponce, P. Vega, F. Casasola, and J. Rojas. 2007. Almacenamiento de carbono en el suelo y la biomasa arbórea en sistemas de usos de la tierra en paisajes ganaderos de Colombia, Costa Rica y Nicaragua. Agrof. Amer. 45, 27-36. [ Links ]
IDEAM. 2002. Páramos y ecosistemas alto andinos de Colombia en condición HotSpot & Global Climatic Tensor. Instituto de Meteorología, Hidrología y Estudios Ambientales (IDEAM), Bogota. [ Links ]
IP CC, Intergovernmental Panel on Climate Change. 2007. Cambio climático 2007. Informe de síntesis. Grupo Intergubernamental de Expertos sobre el Cambio Climático, Cambridge, UK. [ Links ]
IP CC, Intergovernmental Panel on Climate Change. 2000. Land use, land-use change, and forestry special report. Cambridge, UK. [ Links ]
Kanninen, M. 2003. Secuestro de carbono en bosques, su papel en el ciclo global. In: Sánchez, M.D. and M. Rosales (eds.). Agroforestería para la producción animal en América Latina and II Memorias de la Segunda Conferencia Electrónica. FAO, Roma. [ Links ]
Kimble, J.M., R. Lal, and R.B. Grossman. 1998. Alteration of soil properties caused by climate change. pp. 175-184. In: Blume, H.-P., H. Eger, E. Fleischhauer, A. Hebel, C. Reij, and K.G. Steiner (eds.). Towards sustainable land use. Vol. I. Advances in Geoecology 31. Catena Verlag, Reiskirchen, Germany. [ Links ]
Kimmins, J.P. 1997. Forest ecology. A Fundation for Sustainable Management, The University of Bristish; Prentice Hall, Columbia, NJ . [ Links ]
Laegaard, S. 1992. Influence of fire in the grass páramo vegetation of Ecuador. pp. 151-170. In: Balslev, H. and J.L. Luteyn (eds.). Páramo: An Andean ecosystem under human influence. Academic Press, San Diego, CA. [ Links ]
Lal, R. 2004. Soil carbon sequestration to mitigate climate change. Geoderma 123, 1-22. [ Links ]
Lal, R. and J. Kimble 1998. Pedospheric processes and the carbon cycle. pp. 1-8. In: Lal, R., K.M. Kimble, R.F. Follett, and B.A. Stewart (eds.). Soil processes and the carbon cycle. CRC Press, Boca Raton, FL. [ Links ]
López-Ulloa, M., E. Veldkamp, and G.H.J. De Koning. 2005. Soil carbon stabilization in converted tropical pastures and forests depends on soil type. Soil Sci. Soc. Amer. J. 69 (4), 1110-1117. [ Links ]
Lotero J., M. Dossman, C. Castillo, and M. Giraldo. 2006. Plan básico de manejo 2006-2010 Parque Nacional Natural Los Nevados. Parques Nacionales Naturales de Colombia Dirección Territorial Noroccidente, Medellín, Colombia. [ Links ]
MAVDT, IDEAM; PNU D. 2001. Primera Comunicación Nacional ante la Convención Marco de las Naciones Unidas sobre el Cambio Climático. Bogota. [ Links ]
Moreno, F. and W. Lara. 2003. Variación del carbono del suelo en bosques primarios intervenidos y secundarios. In: Orrego, S.A., J.I. Del Valle, and F.H. Moreno (eds.). Medición de la captura de carbono en ecosistemas forestales de Colombia: Contribuciones para la mitigación del cambio climático. Universidad Nacional de Colombia; Departamento de Ciencias Forestales; Centro Andino para la Economía en el Medio Ambiente, Bogota. [ Links ]
Moretto, A., A. Lázzari, and O. Fernández. 2005. Calidad y cantidad de nutrientes de la hojarasca y su posterior mineralización en bosques primarios y bajo manejo con distintos sistemas de regeneración. Módulo Lenga. PIAR FON BAP , Buenos Aires. [ Links ]
Montagnini, F. and C.F. Jordán. 2002. Reciclaje de nutrientes. pp. 167-191. In: Guariguata, M. and Y. Kattan (eds.). Ecología y conservación de bosques neotropicales. Libro Universitario Regional (LUR ), San Jose. [ Links ]
Palacios-Bianchi, P. 2002. Producción y descomposición de hojarasca en un bosque Maulino fragmentado. Biología Ambiental, Universidad de Chile, Santiago. [ Links ]
Powers, J.S. and W.H. Schlesinger. 2002. Relationships among soil carbon distributions and biophysical factors at nested spatial scales in rain forests of northeastern Costa Rica. Geoderma 109, 165-190. [ Links ]
Peña, J.E., H. Sandoval, O. Zúñiga, and A. Torres. 2009. Estimates of carbon reservoirs in high-altitude wetlands in the Colombia Andes. J. Agric. Rural Dev. Trop. 110, 115-126. [ Links ]
Seeberg, C. 2010. Las posibilidades de financiación del carbono para la agricultura, la actividad forestal y otros proyectos de uso de la tierra en el contexto del pequeño agricultor. Oficina de Intercambio de Conocimientos, Investigación y Extensión, FAO, Roma. [ Links ]
Rosenzweig, C. and D. Hillel. 2000. Soils and global climate change: Challenges and opportunities. Soil Science 165, 47-56. [ Links ]
Schuur, E., O. Chadwick, and P. Matson. 2001. Carbon cycling and soil carbon storage in mesic to wet Hawaiian montane forests. Ecology 82(11), 3182-3196. [ Links ]
UNIVALLE; IDEAM. 2008. Validación del protocolo para la caracterización de los ciclos de agua y carbono en ecosistemas de alta montaña y diseño, instalación y puesta en operación de la red de monitoreo para determinar los impactos del cambio climático en dichos ciclos. Cali, Colombia. [ Links ]
Vargas, O. 2000. Sucesión regeneración del páramo después de quemas. M.Sc. thesis. Department of Biology, Universidad Nacional de Colombia. Bogota. [ Links ]
Vargas, O., J. Premauer, and M. Zalamea. 2002. Impacto de fuego y ganadería sobre la vegetación de páramo. pp. 819-841. In: Memorias del Congreso Mundial de Páramos. Tomo II . Ministerio del Medio Ambiente; CAR ; IDEAM; Conservación Internacional, Paipa, Colombia. [ Links ]
Vargas, O. and P. Pedraza, 2004. Parque Nacional Natural Chingaza. Gente Nueva Editorial. Bogota. [ Links ]
Watson, R.T., I.R. Noble, B. Bolin, N.H. Ravindranath, D.J. Verardo, and D.J. Dokken. 2000. IP CC Special Report, Intergovernmental Panel on Climate Change, Cambridge University Press, Cambridge, UK. [ Links ]