Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Agronomía Colombiana
Print version ISSN 0120-9965
Agron. colomb. vol.31 no.2 Bogotá May/Aug. 2013
CROP PHYSIOLOGY
Response of the banana passion fruit (Passiflora tripartita var. mollissima) to different levels of nitrogen, potassium and magnesium
Respuesta de la curuba (Passiflora tripartita var. mollissima) a diferentes niveles de nitrógeno, potasio y magnesio
Miguel Ángel Lizarazo1 , Camilo Andrés Hernández1 , Gerhard Fischer1 and Manuel Iván Gómez 1
1Department of Agronomy, Faculty of Agronomy, Universidad Nacional de Colombia. Bogota (Colombia). malizarazot@unal.edu.coReceived for publication: 7 March, 2013. Accepted for publication: 5 June, 2013.
ABSTRACT
In order to observe the effects of N, K, and Mg on dry mass (DM) allocation, canopy growth and nutritional deficiency symptom expression of the banana passion fruit (Passiflora tripartita var. mollissima), three levels of fertilization were evaluated: none (0), low (1) and high (2) of each element, and compared to complete fertilization (control), arranged in a randomized complete block design. Banana passion fruit plants were transplanted to plastic pots, containing a substrate of quartz sand, with irrigation twice a week and fertigation once a week. The evaluated variables were total dry weight (TDW), percentage of dry mass allocation, leaf area (LA), number of leaves (NL), specific leaf area (SLA) and leaf area ratio (LAR). In addition, a photographic record was taken to recognize the symptomatology in the leaves. The absence of N, K and Mg affected the accumulation of DM, whereas the high nutrient level allowed for the greatest gain in DM only with N, while with K and Mg, it was reduced, indicating that these doses may be too excessive. The distribution pattern of DM with the N treatments corresponded to the model proposed by Thornley, accumulating more DM in the root. With the K and Mg levels, the distribution pattern was similar with the absence of these nutrients causing higher DM accumulation in the above-ground plant parts. The leaf variables NL and LA were reduced with all three elements at the zero nutrient level, whereas the high indices of SLA and LAR revealed a low gain of DM. Deprivation of each nutrient showed characteristic symptoms on the leaves, mainly chlorosis with different patterns, necrosis and reduced LA.
Key words: plant nutrition, nutrient deficiency, dosage effects, tropical fruits, dry mass.
RESUMEN
Para conocer el efecto de N, K y Mg sobre la distribución de masa seca (MS ), crecimiento foliar y expresión de síntomas de deficiencia nutricional en curuba (Passiflora tripartita var. mollissima), se evaluaron tres niveles de fertilización: carencia (0), bajo (1) y elevado (2) para cada elemento, comparados con una fertilización completa (testigo), dispuestos en un diseño de bloques completamente al azar. Las plantas de curuba fueron trasplantadas en materas plásticas, con un sustrato de arena cuarcítica, realizando un riego y dos fertirriegos cada semana. Las variables evaluadas fueron peso seco total (PST ), porcentaje de distribución de materia seca, área foliar (AF), número de hojas (NH), área foliar específica (AFE) y relación de área foliar (RAF), además se realizó un registro fotográfico para reconocer la sintomatología de las deficiencias en hojas. La carencia de N, K y Mg afectó la acumulación de MS , mientras que el nivel elevado el nutriente permitió la mayor ganancia de MS sólo en N, pues en K y Mg no fue alta. El esquema de distribución de MS , en los niveles de N, correspondió al planteado en el modelo de Thornley al acumular mayor MS en raíz, en los niveles de K y Mg la distribución fue similar entre ellos, resaltando que bajo carencia se almacenó mayor MS en parte aérea. En todos los elementos, el nivel de carencia redujo el NH y AF, mientras los índices AFE y la RAF fueron altos en este nivel, revelando una baja ganancia de MS en hojas y planta respectivamente. Se observaron síntomas característicos en hojas, bajo el nivel de carencia, principalmente clorosis en distintos patrones, necrosis y AF reducida.
Palabras clave: nutrición de plantas, deficiencia de nutrientes, efectos de dosificación, frutas tropicales, masa seca.
Introduction
Within the genus Passiflora, there are more than 20 species with edible fruits, of which half are grown commercially due to the characteristics of these fruits, such as the banana passion fruit (Passiflora tripartita var. mollisima Holm- Nielsen & Møller Jørgensen) (Primot et al., 2005). It is native to the Andean valleys of high mountainous areas and, in Colombia, is grown throughout the three "Cordilleras", especially in the eastern and central ranges of the departments of Boyaca, Tolima, Cundinamarca, Santander, Valle, Caldas and Antioquia, among others (Campos, 2001), with Boyaca being the principal producer with a harvested area of 663 ha and a production of 6,868 t, contributing 48% of the national production of the banana passion fruit (Agronet, 2012). Agro-ecological conditions in which the passion fruit grows naturally contain an altitude between 2,000 and 3,200 m a.s.l., an optimum temperature between 13 and 16°C and rainfall of 1,000-1,500 mm year-1 (Campos, 2001).
Dry mass (DM) yield is the result of the photosynthetic process; from the quantitative point of view, its accumulation, and hence plant growth, depends on the excess of synthesized carbohydrates in relation to those that are used as a respiration substrate. However, in the particular case of plant structures, this concept is not necessarily strict, because the photosynthate redistribution mechanism may increase or decrease the accumulated biomass in each of the structures (Norero and Pilatti, 2002).
Approximately 90% of accumulated DM during plant growth is the product of photosynthetic activity while the remaining percentage comes from mineral absorption (Benincasa, 2003). For the banana passion fruit, Montenegro (1991) determined that DM accumulation peaks when flowering starts.
The leaves are the main organs that intercept photosynthetically active radiation (PAR) to produce carbohydrates that are then used to build new tissues and both vegetative and reproductive structures. The efficiency in the interception of the PAR and the leaf area are factors that influence the photosynthetic activity and thus, the formation of biomass (Coombs and Hall, 1982).
Specific leaf area (SLA) is defined as the ratio of leaf area and dry weight of leaves, is a foliar thickness and density index or, in other words, is a measure of the plant leafiness based on dry weight (Hunt, 2003). The leaf area ratio (LAR) is the ratio of leaf area and total plant dry weight, a morphological index that indicates the size of the photosynthetic apparatus, and measures the balance between gains of the photosynthetic process and expenditures through respiration in different parts of the plant (Hunt, 2003).
As for the driving variables of plant growth, Lambers et al. (1998) designated leaf area and net assimilation rate, as well as the plant's nutrient concentration and nutrient productivity. In Colombia, in the zones where banana passion fruits are commercially grown, deficiencies of nitrogen, potassium and magnesium have been observed (M.I. Gómez, personal communication, 2010). When mineral nutrients are limiting, plants reduce growth and alter aspects of morphology, as well as acquisition and use of minerals in order to maximize acquisition of these scarce resources (Schachtman and Shin, 2007). Thus, the visualization of plant symptomatology is a valuable tool for morphological characterization (Lizarazo et al., 2013), basic to identifying nutrients as limiting the factors in plant growth and production, and is also useful to differentiate physiological disorders caused by abiotic factors such as climate or toxicities induced by fertilization, among other factors (Gómez, 2012).
This study aimed to establish the effect of different levels of nitrogen, potassium and magnesium fertilization on biomass, leaf growth and symptomatology of the banana passion fruit in order to better understand plant response to nutrition scale and to recognize deficiency and excess symptoms on leaves.
Materials and methods
The experiment was conducted under greenhouse conditions at the Faculty of Agronomy of the Universidad Nacional de Colombia, Bogota, with an average greenhouse temperature of 20°C and relative humidity of 80%. Banana passion fruit plants (Curuba de Castilla' type) were obtained from a commercial nursery 35 d after seed emergence. The plants were pinched apically to encourage lateral budding and then a main stem of uniform height was selected. They were then transplanted into 5 kg plastic pots filled with an inert substrate of quartz sand (≤ 1 mm) and pure quartz (≤ 4 mm), 50% vol. each, and put on the greenhouse floor at 0.5 m between plants and 1.0 m between rows.
We used a tutoring system in vertical trellis with two wire lines. The apical meristem was pruned when the plants reached the 2 m wire line to stimulate bud break of secondary stems, of which two were selected for each line wire (at 1 and 2 m). Periodically, new side shoots were removed from the main stem to support the growth of the four lateral stems.
The treatments were composed of three fertilizer levels for each of the elements N, K and Mg, (0): total absence of the element; (1): each element at less (N, K 30% and Mg 50%) than the commercially recommended dose; (2): each element at a greater proportion (N, K 30% and Mg 50%) than the recommended dose and (control): commercially recommended dose; for a total of 10 (3 x 3 + 1) treatments. For the dosage of each nutrient, a tool guide for fertilization programs of commercial crops in the vegetative growth stage was used (Tab.1). (M.I. Gomez, personal communication, 2010).
The fertilizer sources used in the stock solutions were: urea (CH4N2O), monoammonium phosphate (NH4H2PO4), monopotassium phosphate (KH2PO4), potassium chloride (KCl), potassium nitrate (KNO3), potassium sulfate (K2SO4), phosphoric acid (H3PO4), magnesium sulfate Heptah (MgSO4 7H2O), magnesium nitrate (Mg(NO3)2 6H2O), sodium octaborate (Na2B4O7 10H2O), zinc sulfate (ZnSO4), ferrous sulfate (FeSO4), technical copper sulfate (CuSO4 5H2O), calcium nitrate (Ca(NO3)2, calcium chloride (CaCl2), manganese sulfate (MgSO4 7H2O), sodium molybdate (Na2MoO4), cobalt sulfate (CoSO4), acid anhydrous citric (acidifier) (C6H8O7) and EDTA sodium salt (C10H14N2Na2O8 2H2O).
The evaluation involved a destructive sampling in the ninth month after transplant (at the end of the vegetative cycle of the banana passion fruits), i.e. when the main stems with its side shoots were formed and the emission of floral buds began. The application of nutrition treatments began 68 d after transplant. Weekly irrigation was performed, including two ferti-irrigations, with an initial volume of 300 mL for each application per plant, which increased to 600 mL at the end of the cycle.
The quantification of the number of leaves (NL), leaf area (LA) and dry mass (DM) content was performed in the laboratory of Plant Physiology, Faculty of Agronomy, Universidad Nacional de Colombia, Bogota. Through a visual count of mature, fully-expanded leaves, the NL was determined. For the DM content, the different plant organs (leaves, stems and roots) were taken separately and were placed in a drying oven at 70°C for 48 h for subsequent weighing on an analytical balance. The LA was determined with a LI-3100 planimeter (Licor, Lincoln, NE). With the information obtained from these measurements, the growth indices SLA and LAR were calculated, using the following formulas (Hunt, 2003):
Specific leaf area (SLA): relationship between total leaf area (LA) and total dry mass of leaves (DML):
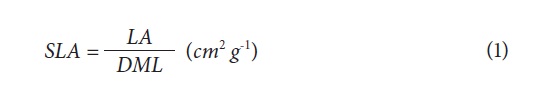
Leaf area ratio (LAR): ratio of leaf area (LA) and total dry mass of plant (DMP):
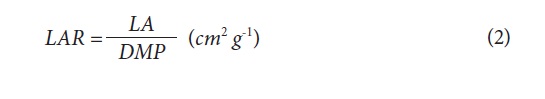
A visual characterization and photographic record of the symptoms presented in leaves of the different deficient nutrients were carried out. The trial was conducted as a randomized complete block design with four replications and one plant per experimental unit. For statistical analysis of the data, the SAS ® software, version 9.1 was used, from which analysis of variance (Anova) and the multiple range test of Duncan were performed to establish the statistical difference between the means.
Results and discussion
Dry mass accumulation
Nitrogen
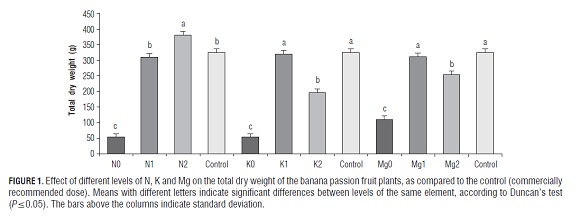
The absence of nitrogen fertilization (N0) presented a plant DM of 53.28 g, significantly lower (P≤0.05) than the plants of the other treatments and the control (Fig.1), a result similar to that reported by Castaño et al. (2008) in the blackberry (Rubus glaucus var. San Antonio) under nitrogen-starved nutrition. This can be explained by the fact that N is one of the elements required in larger amounts in plants, especially in the vegetative stage. Its absence limits the growth of all plant organs, affecting a wide range of organic molecules where N forms a part, such as in proteins, nucleic acids, purines, pyrimidines, coenzymes (vitamins), secondary metabolites and hormones, which are essential in the whole plant metabolism (Barker and Pilbeam, 2007). Additionally, the nitrogen concentration in leaves is closely related to the contents of Rubisco and chlorophyll, the rate of electron transport and the quantum yield of photosynthesis, which directly influence photosynthetic efficiency and in turn growth (Pérez et al., 2007; Sanclemente and Peña, 2008). In fruit orchards, nitrogen deficiency can be found in soils due to low and incomplete decomposition of organic matter, water logging, a high degree of leaching and denitrification by soil microorganisms (Rajendran et al., 2009). For the N1 level and complete fertilization, no significant differences (P≤0.05) in the total DM were registered, while the N2 plants recorded the highest DM yield with 380.63 g, confirming the statements of Lambers et al. (1998) that plants with high nutrient concentrations can grow faster. Also, Vázquez et al. (2008) found a higher DM yield with an increasing nitrogen dose in strawberry plants (Fragaria x ananassa). This high biomass accumulation may be due to enhanced photosynthetic efficiency which was found to be considerably higher in the leaves of plants with high N availability (Pérez et al., 2007).
Potassium
The lowest plant DM was obtained in the K0 treatment (53.35 g; Fig.1), probably because potassium fulfills the main functions in plants, such as enzyme activation, protein synthesis, osmoregulation, absorption, transport and ion balance, and is also involved in photosynthesis, respiration, transportation and long distance distribution of assimilates (Barker and Pilbeam, 2007; Hawkesford et al., 2012). The amount of K absorbed by the plant is closely related to its growth since the accumulation of DM is influenced by its availability and the balance with other nutrients (Baque et al., 2006). In orchards, potassium deficiency occurs in highly leached sandy soils, in red lateritic soils, with high (Mg+Ca)/K ratios in the soil and in cold/ wet or dry soils (Rajendran et al., 2009).
The plants of the K2 treatment accumulated lower DM contents (197.10 g) than the K1 and control plants did. Although excess K is not common in plants since there are mechanisms in which its uptake is strictly regulated, excess K fertilizing, although it does not produce visible symptoms, can depress plant growth (Barker and Pilbeam, 2007). Xu et al. (2011) found, in Houttuynia cordata, that as the K level increased so did the plant DM, to a maximum value, after which, the higher the concentration, the more reduced the accumulation of DM was. These authors mentioned a low water status due to a failure in water absorption and accumulation of H2O2, suggesting oxidative stress, which increases the activity of catalase and peroxidase in response to this condition.
Magnesium
Plants of the Mg0 treatment accumulated only 109.68 g DM, with a significantly lower value (P≤0.05) than the other treatments (Fig.1). The absence of this element affects many physiological and molecular processes in which it participates: as a structural component of the chlorophyll molecule, cofactor of various enzymatic processes associated with phosphorylation, dephosphorylation and hydrolysis of compounds, in the synthesis of protein, in addition to being a structural stabilizer of several nucleotides (Barker and Pilbeam, 2007). Mg-deficiency also affects the photosynthetic capacity and in turn the accumulation of DM, it increases light sensitivity, which occurs due to reactive oxygen species (ROS) that exceed the ability of detoxifying enzymes, leading to a photo-oxidation of chloroplast pigments, producing chlorosis and necrosis of the leaves (Hawkesford et al., 2012).
Fruit plants suffer from Mg deficiency in highly leached acid sandy soils, low soil pH, calcareous soils and through an imbalance of K/Mg and Ca/Mg ratios in the soil (Rajendran et al., 2009).
The DM content of the Mg1-plants did not show significant differences (P≤0.05) compared to the control plants, while the Mg2-plants clearly accumulated less DM than the Mg1 and control treatments. Although no alterations in growth or in the normal leaf characteristics were observed in the Mg2-treatment, there may be other factors that likely affect this variable, for example, a high concentration of Mg2+ can induce deficiencies of other cations such as K+, NH4 +, Ca2+ and Na+ due to competition with Mg2+ for binding sites on soil colloids, a phenomenon known as ion antagonism (Barker and Pilbeam, 2007).
Dry mass distribution
Nitrogen
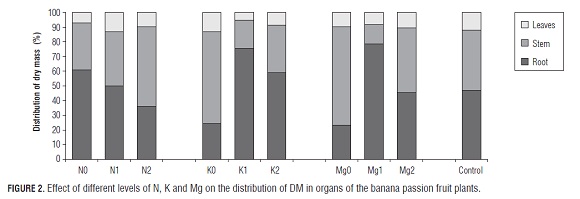
The distribution (or allocation) of DM into the vegetative organs of the N0-plants was mainly towards the roots (60.79%) (Fig.2) because, under a deficiency of N, the majority of this nutrient is used for the growth of the root system (Wilson, 1988). Since one of the initial physiological effects of such deficiency is a change in the routing of the products of primary metabolism and of accumulated sugars in leaves, N deficiency can alter the partitioning of carbon, increasing its transport to the root, which increases the allometric root/shoot ratio and, in conjunction with changes in hormone levels, modifies the root morphology, allowing the plant to respond appropriately to nutritional deficiency with a more efficient exploration of the rhizosphere in search of mineral elements (Hermans et al., 2006).
The pattern of DM allocation in the N1 plants was similar to the control, unlike the N2 plants whose DM was used in greater proportion in the above-ground part (64%) (Tab.2). This pattern is consistent with the mechanistic model of Thornley, evaluated by Wilson (1988), based on the supply, transport and utilization of carbon and nitrogen within the root/shoot ratio system. According to this, the conditions leading to an increased carbon concentration should therefore lead to an increase in the distribution of biomass towards the roots, while an increase in the concentration of nitrogen should contribute to biomass partition towards above-ground organs.
Potassium
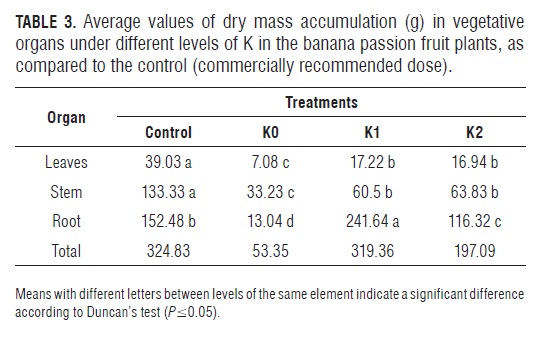
The distribution of DM in the non-K-fertilized plants showed that 62.29% of the DM was allocated towards the above-ground part, compared with only 24.44% towards to the roots (Fig.2). Potassium deficiency alters the partition of assimilates and, in turn, the concentration of metabolites in the vegetative organs (Hawkesford et al., 2012). In this sense, the distribution pattern can be explained by the sucrose accumulation in the leaves, affecting photosynthesis and disrupting the transport chain of sugar towards the root for its growth, due to the fact that K ions act in the loading and transport of sucrose from the mesophyll to the phloem and for this reason the plant cannot expand the exploring root volume (Hermans et al., 2006). Cabezas and Sánchez (2008) found that, under K deficiency, the DM distribution pattern is unaffected in banana passion fruit plants, showing a proportional allometric root/shoot ratio. On the other hand, the K1 and K2 levels tended to accumulate more DM in the roots, as compared to the control plants.Tab.3
Magnesium
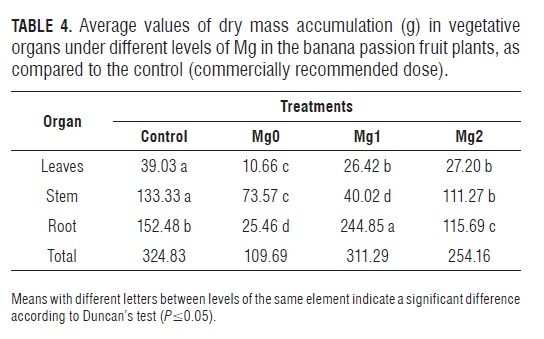
Dry mass distribution in plants lacking Mg, as in the case of K, tended to be higher in above-ground parts (Tabs. 2, 3 and 4), confirming that the effect on root growth from these two elements is similar (Hawkesford et al., 2012). In the physiological process, there is a particularly high accumulation of glucose, fructose and sucrose in the source leaves, which creates problems in phloem loading, apparently related to low concentrations of the Mg-AT P complex in sites where sucrose is loaded into the phloem, consequently altering photosynthesis and the remaining photosynthates for root growth (Hermans and Verbruggen, 2005). Because of this fact, the export of other solutes such as K and particularly amino acids are also negatively affected (Hermans et al., 2006).Tab.4
With a limited supply of Mg, Cabezas and Sánchez (2008) found that 50% of the plants' DM was distributed towards the roots in 1-year-old banana passion fruit plants, in contrast to the results of our study and to Ericsson and Kähr (1995), who reported that the partition of DM towards the root can decrease a plant's DM by up to 8% in Mg-deficient Betula pendula seedlings.
The distribution pattern of DM in the Mg1 plants was similar to that observed in the K1 treatment, storing a high percentage of DM in the roots (Fig.2). The plants with the highest Mg fertilization did not differ in the DM distribution pattern from the control plants.
Leaf parameters of growth
Nitrogen
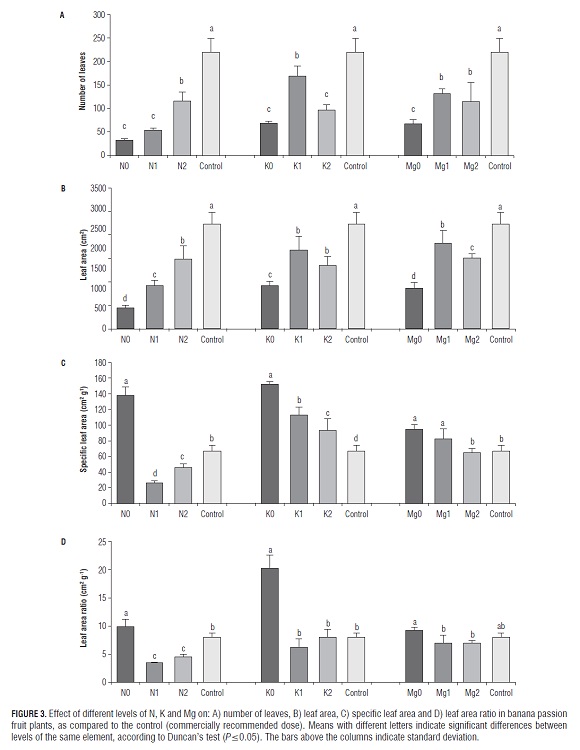
The three levels of nitrogen fertilization showed statistically lower values (P≤0.05) for leaf number (LN) and leaf area (LA) when compared to the control plants (Fig.3A-B).
The same result was found for the SLA (Specific leaf area) and LAR (Leaf area ratio) except for the non N-fertilized plants, which showed a higher magnitude (Fig.3C-D). The results suggest that with the reduction of the element concentration, number of leaves (NL) and LA also decrease, similar to that found by Sanclemente and Peña (2008) in Ludwigia decurrens, who stated that, although photochemical activity does not depend solely on the availability of nitrogen, its absence directly affects chlorophyll content and hence growth.
Compared to the control, NL and LA were lower in the N1 and N2 treatments (Fig.3A-B), but DM accumulation was the same or higher than in the control plants (Fig.1), likely because these treatments used their photosynthetic apparatus more efficiently. Castellanos et al. (2010) reported that a reduction in the number of source leaves, due to different causes, can lead to an increase in photosynthate production by the remaining leaves as a compensatory effect.
As for the SLA and the LAR, N0 was notable because it showed significantly higher (P≤0.05) values than the N1, N2 and control plants (Fig.3C-D). These high values are the result of a limited accumulation of DM in the leaves and its increased fragility due to a smaller amount of structural tissue (consisting mainly of cellulose and lignin) per unit area (Niinemets and Kull, 1998; Castro et al., 2000). It has to be taken into account that these values can vary between species and between leaves of the same plant, depending on their longevity or light exposure (Björkman and Holmgren, 1963).
Potassium
The banana passion fruits without K fertilization, as in the case of N, had the highest SLA and LAR, 151.79 and 20.21 cm2 g-1, respectively, as compared with the K1, K2 and the control plants (Fig.3C-D). This indicates that DM production is low compared to its related leaf area because the absence of K limits stomatal conductance and the level of chlorophyll and photosynthesis, as found by Barragán et al. (2009) in cotton (Gossypium hirsutum) plants. Also, Wright and Cannon (2001) registered lower rates of photosynthesis with a high SLA and, therefore, a lower accumulation of DM. Hawkesford et al. (2012) argued that when there is an adequate supply of K, leaves tend to be rather thick, which explains our finding of decreasing SLA values with the increasing K supply (Fig.3C).
Magnesium
The NL of the Mg0 treatment was 67, equivalent to 1007.06 cm2 of LA, values significantly lower (P≤0.05) than the higher Mg levels (Fig.3A-B). This implies a greater dependency of the plants on these source organs and an effect on photosynthetic activity and subsequent accumulation of DM as mentioned above. No differences between the SLA of Mg0 and Mg1 and of Mg2 and the control plants were observed (Fig.3C), indicating that the leaves with low Mg levels tend to be thinner than those with adequate levels, which is related (Castro et al., 2000) to certain functional aspects of woody plants, such as gas exchange, photosynthetic rate and even the growth rate.
The LAR of the Mg0- plants was similar to that of the control (Fig.3D), although the latter showed values 2-3 times higher for the NL and LA variables (Fig.3A-B), suggesting that the net production efficiency of the photosynthetic apparatus per unit leaf area in plants of the two treatments was similar. No differences (P>0.05) in LAR between the control plants and Mg treatments were registered, similar to the findings of Troyanos et al. (1997) in two cultivars of the genus Prunus, where different levels of Mg did not alter the LAR. However, in our trial, the plants without Mgfertilization had a LAR significantly higher than the Mgfertilized ones (Fig.3D), confirming that the leaf surface of the Mg0 plants produced significantly lower (P≤0.05) plant DM than the other treatments (Fig.1).
Symptoms of nutrient deficiencies
Nitrogen
The symptoms of nitrogen deficiency corresponded to less vigorous plants as compared with the control. Initially, the leaves developed a generalized chlorosis in mature leaves (Fig.4A)) which then became evident in young leaves (Fig.4B), without any type of necrosis (Fig.4C). New shoots at the end of the vegetative cycle presented small leaves with interveinal chlorosis (Fig.4D) and early abscission. These symptoms coincided with the observations of Cabezas and Sánchez (2008) for the banana passion fruit, with widespread chlorosis of the older leaves and reduced leaf area with early foliar abscission, aspects which all affect the LA (Fig.3B). Gómez (2012) found similar symptoms in lulo (Solanum quitoense) plants and Martínez et al. (2009) in the cape gooseberry (Physalis peruviana), while Barker and Pilbeam (2007) generalized that nitrogen deficiency produces a pale green or yellow leaf color due to a loss of chlorophyll that occurs simultaneously with the deterioration of membranes and proteins.
Potassium
The plants with K deficiency demonstrated small speckled or chlorotic zones in the leaves that then formed necrosis as described by Rajendran et al. (2009) (Fig.5A). Typically, a progressive tissue death appeared at the leaf tip (Fig.5B), advancing along the margins, followed by leaf wilting (Fig.5C). These symptoms were observed initially in mature leaves and later in young ones, which in turn showed a clearer tone, light crinkling inward from leaf margins and fragile appearance. The advancement pattern of this deficiency can be explained by the high mobility of K in the phloem where it can be retranslocated due to a limited offering of the nutrient (Barker and Pilbeam, 2007). Similarly, Cabezas and Sánchez (2008) observed smaller leaves and leaf blades of leathery consistence with necrotic margins in K-deficient banana passion fruits.
Magnesium
Magnesium deficiency initially resulted in a generalized chlorosis of adult leaves (Fig.6A) which then accentuated in the form of interveinal chlorosis (Fig.6B), forming necrotic areas bounded by the nerves of the leaves (Fig.6C). This pattern was then expressed in young leaves (Fig.6D) as reported by Martínez et al. (2009) in the cape gooseberry. Finally, the tendrils dried and premature leaf senescence started (Fig.6E). It has been shown that the symptoms of Mg deficiency are highly dependent on light, because high light intensity increases interveinal chlorosis and tissue necrosis of leaves due to the production of reactive oxygen species (ROS), which eventually produce peroxidative damage (Cakmak and Kirkby, 2008).
Conclusions
The deficiency of N, K and Mg in the banana passion fruits reduced the accumulation of DM, demonstrating the importance of these elements in the physiological processes involved in growth.
The high nutrient level produced the highest gain of DM only with N because, in the high K and Mg fertilization, the accumulated DM was not high, indicating that these doses may be excessive and cause metabolic problems.
The allocation pattern of DM, in the case of N, corresponded to that proposed in Thornley's model where a higher concentration of nitrogen favors biomass partitioning towards above-ground plant parts. In the K and Mg treatments, the DM distribution pattern was similar, indicating that most of DM is stored in the above-ground part under deficiency, basically due to problems of excessive accumulation and therefore failure of the mobilization of photosynthates towards different plant organs.
The absence of the three elements in fertilization reduced NL and LA but increased SLA and LAR to the highest levels, revealing a low DM gain in the leaves and plants, respectively.
Deprivation of the three nutrients expressed characteristic symptoms in the leaves, mainly chlorosis in different patterns, fragile appearance, crinkling, necrosis, growth restriction and reduced LA.
Acknowledgements
The authors thank Microfertisa and ING EPLANT Ingeniería en nutrición de cultivos, Bogota for providing nutrient solutions; the Department of Research of Bogota (DIB) of the Universidad Nacional de Colombia for co-financing this study; and Gilberto Sánchez of the Research and Rural Extension Centre (CIER), Faculty of Agronomy, as well as Sergio Hernández, for taking photographs.
Literature cited
Agronet. 2012. Comparativo en la producción de curuba por departamento 1992-2011. In: http://www.agronet.gov.co/agronetweb1/Estadísticas.aspx; consulted: August, 2012. [ Links ]
Baque, A., A. Karim, A. Hamid, and H. Tedsushi. 2006. Effects of fertilizer potassium on growth, yield and nutrient uptake of weath (Triticum aestivum) under wáter stress conditions. S. Pac. Stud. 27(1), 25-35. [ Links ]
Barker, A. and D. Pilbeam. 2007. Handbook of plant nutrition. CRC Press, Taylor and Francis Group, Boca Raton, FL. [ Links ]
Barragán, E., C.A. Rosolem, and L.M. Melgarejo. 2009. Efecto de la nutrición con potasio en el crecimiento, partición de biomasa, tasa de fotosíntesis, y calidad de fibra en algodón (Gossypium hirsutum L.). p. 176. In: Book of Abstracts, Congreso Colombiano de Botánica, Pasto, Colombia. [ Links ]
Benincasa, M.M.P. 2003. Análise de crescimento de plantas (noções básicas). 2a ed. Funep, Jaboticabal, Brazil. [ Links ]
Björkman, O. and P. Holmgren. 1963. Adaptability of the photosynthetic apparatus to light intensity in ecotypes from exposed and shaded habitats. Physiol. Plant. 16, 889-914. [ Links ]
Cabezas, M. and C. Sánchez. 2008. Efecto de las deficiencias nutricionales en la distribución de la materia seca en plantas de vivero de curuba (Passiflora mollissima Bailey). Agron. Colomb. 26(2), 197-204. [ Links ]
Cakmak, I. and E. Kirkby. 2008. Role of magnesium in carbon partitioning and alleviating photooxidative damage. Physiol. Plant. 133(4), 692-704. [ Links ]
Campos, T. 2001. La curuba: su cultivo. IICA, Bogota. [ Links ]
Castaño, C.A., C. S. Morales, and F.H. Obando. 2008. Evaluación de las deficiencias nutricionales en el cultivo de la mora (Rubus glaucus) en condiciones controladas para bosque montano bajo. Agronomía 16(1), 75-88. [ Links ]
Castellanos, M., M. Segura, and C. Ñústez. 2010. Análisis de crecimiento y relación fuente-demanda de cuatro variedades de papa (Solanum tuberosum L.) en el Municipio de Zipaquirá (Cundinamarca, Colombia). Rev. Fac. Nal. Agr. Medellin 63(1), 5253-5266. [ Links ]
Castro, P., J. Puyravaud, and J. Cornelissen. 2000. Leaf structure and anatomy as related to leaf mass per area variation in seedlings of a wide range of woody plant species and types. Oecologia 124, 476-486. [ Links ]
Coombs, J. and D.O. Hall. 1982. Whole plant photosynthesis and productivity. in: techniques in bioproductivity and photosynthesis. Pergamon Press, Oxford, UK. [ Links ]
Ericsson, T. and M. Kähr. 1995. Growth and nutrition of birch seedlings at varied relative addition rates of magnesium. Tree Physiol. 15, 85-93. [ Links ]
Gómez, M.I. 2012. Fertilización de frutales. pp. 141-168. In: Fischer, G. (ed.). Manual para el cultivo de frutales en el trópico. Produmedios, Bogota. [ Links ]
Hawkesford, M., W. Horst, T. Kichey, H. Lambers, J. Schjoerring, I. Skrumsager Møller, and P. White. 2012. Functions of macronutrients. pp. 135-189. In: Marschner, P. (ed.). Marschner's mineral nutrition of higher plants. 3rd ed. Academic Press, London. [ Links ]
Hermans, C., J. Hammond, P. White, and N. Verbruggen. 2006. How do plants respond to nutrient shortage by biomass allocation? Trends Plant Sci. 11(12), 610-617. [ Links ]
Hermans, C. and N. Verbruggen. 2005. Physiological characterization of Mg deficiency in Arabidopsis thaliana. J. Exp. Bot. 56(418), 2153-2161. [ Links ]
Hunt, R. 2003. Plant growth analysis: individual plants. In: Thomas, B., D.J. Murphy, and D. Murray (eds.). Encyclopedia of applied plant sciences. Academic Press, London. [ Links ]
Lambers, H., F.S. Chapin III, and T.L. Pons. 1998. Plant physiological ecology. Springer, New York, NY . [ Links ]
Lizarazo, M.A., C.A. Hernández, G. Fischer, and M.I. Gómez. 2013. Biomasa, crecimiento foliar y symptomatología en curuba (Passiflora tripartita var. mollissima) afectada por diferentes niveles de manganeso, zinc y boro. Rev. Colomb. Cienc. Hortic. 7(1), 31-45. [ Links ]
Martínez, F.E., J. Sarmiento, G. Fischer, and F. Jiménez. 2009. Síntomas de deficiencia de macronutrientes y boro en plantas de uchuva (Physalis peruviana L.). Agron. Colomb. 27(2), 169-178. [ Links ]
Montenegro, J.W. 1991. Estudio de crecimiento de dos tipos de curuba (Passiflora mollissima B.) en la zona de Usme. Undergraduate thesis. Faculty of Agronomy, Universidad Nacional de Colombia, Bogota. [ Links ]
Niinemets, Ü. and O. Kull. 1998. Stoichiometry of foliar carbon constituents varies along light gradients in temperate woody canopies: implications for foliage morphological plasticity. Tree Physiol. 18, 467-479. [ Links ]
Norero, A.L. and M.A. Pilatti. 2002. Enfoque de sistemas y modelos agronómicos. Necesidades, métodos y objetos de estudio. Universidad Nacional del Litoral, Santa Fe, Argentina. [ Links ]
Pérez, J.M., E. Peña, and C. Torres. 2007. Efecto del nitrógeno y la irradianza en la eficiencia fotosintética del Anamú Petiveria alliacea (Phytolaccaceae). Rev. Acad. Colomb. Cienc. 31(118), 49-55. [ Links ]
Primot, S., V. Rioux, G.C. d`Eeckenbrugge, F. Garcin, and J.A Ocampo. 2005. Variación genética de tres especies de curubas (Passiflora tripartita var. mollissima, Passiflora tarminiana y Passiflora mixta) y sus híbridos en el Valle del Cauca. CIRADFLHOR, IPGRI, CIAT , Cali, Colombia. [ Links ]
Rajendran, C., K. Ramamoorthy, and S.J. Hepziba. 2009. Nutritional and physiological disorders in crop plants. Scientific Publishers, Jodhpur, India. [ Links ]
Sanclemente, M.A. and E. Peña. 2008. Crecimiento y eficiencia fotosintética de Ludwigia decurrens Walter (Onagraceae) bajo diferentes concentraciones de nitrógeno. Acta Biol. Colomb. 13(1), 175-186. [ Links ]
Schachtman, D.P. and R. Shin. 2007. Nutrient sensing and signaling: NPKS. Annu. Rev. Plant Biol. 58, 47-69. [ Links ]
Troyanos, Y.E, N.A. Hipps, J. Moorby, and M.S. Ridout. 1997. The effects of external magnesium concentration on the growth and magnesium inflow rates of micropropagated cherry rootstocks "F.12/1" (Prunus avium L.) and "Colt" (Prunus avium L. x Prunus pseudocerasus L.). Plant Soil 197, 25-33. [ Links ]
Vázquez, G., R. Cárdenas, and P. Lobit. 2008. Efecto del nitrógeno sobre el crecimiento y rendimiento de fresa regada por goteo y gravedad. Agr. Tec. Mex. 34(2), 235-241. [ Links ]
Wilson, J.B. 1988. A review of evidence on the control of shoot: root ratio, in relation to models. Ann. Bot. 61, 433-49. [ Links ]
Wright, I.J. and K. Cannon. 2001. Relationships between leaf lifespan and structural defenses in a low-nutrient, sclerophyll flora. Functional Ecol. 15, 351-359. [ Links ]
Xu, Y., Y. Zou, A. Husaini, J. Zeng, L. Guan, Q. Liu, and W. Wu. 2011. Optimization of potassium for proper growth and physiological response of Houttuynia cordata Thunb. Environ. Exp. Bot. 71, 292-297. [ Links ]