Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Agronomía Colombiana
Print version ISSN 0120-9965
Agron. colomb. vol.31 no.3 Bogotá Sept./Dec. 2013
PROPAGATION & TISSUE CULTURE
Anatomy of sugar apple (Annona squamosa L.) seeds (Annonaceae)
Anatomía de semillas de anón (Annona squamosa L.) (Annonaceae)
Fabio Martínez M.1 Diego Miranda L.2 and Stanislav Magnitskiy2
1Tibaitata Reaserch Center, Corporación Colombiana de Investigación Agropecuaria (Corpoica). Mosquera (Colombia). 2Department of Agronomy, Faculty of Agricultural Sciences, Universidad Nacional de Colombia. Bogota (Colombia). svmagnitskiy@unal.edu.coReceived for publication: 6 February, 2013. Accepted for publication: 1 November, 2013.
ABSTRACT
The sugar apple is of great economic importance to the country, however, information on the anatomical and morphological aspects of Annonaceae family seeds is limited to a few species and the anatomical studies refer to very few seeds. Using histological techniques and microscopy, Annona squamosa L. seeds were studied with regard to their anatomical characteristics. In general, they presented the typical anatomical patterns of seeds of the Annonaceae family. The multilayered testa was comprised of exo-, meso- and endotestas, with the presence of longitudinal and oblique fibers. The embryo was small and straight with moderately developed embryonic axis, rudimentary plumule and flat and thin cotyledons, wherein one could observe the procambium. In particular, the perichalaza was observed in the medium longitudinal plane of the endotesta with more than two rows of vascular tissue. The tegmen or inner integument was collapsed. The micropylar plug was tapered, woody and composed of transverse fibers; a fracture line was also observed. The ruminate endosperm interfered with both the inner and external integuments. Oleiferous idioblasts were observed at the outer edges of the ruminations.
Key words: seed coats, ruminate endosperm, micropylar plug, embryo.
RESUMEN
El anón es de gran importancia económica para el país, sin embargo, la información referida a aspectos estructurales de la familia Annonaceae está limitada a algunas pocas especies y los estudios anatómicos referidos a semillas son muy escasos. Empleando técnicas histológicas y microscopia, semillas de las especie Annona squamosa L. fueron estudiadas con respecto a sus características anatómicas. En general, presentan el patrón anatómico típico para semillas de la familia Annonaceae. La testa multiplicativa fue conformada por exo, meso y endotesta, con presencia de fibras longitudinales y oblicuas. Se observó un embrión pequeño, recto, con eje embrionario moderadamente desarrollado, plúmula rudimentaria y cotiledones planos y delgados en el que se puede apreciar el procambium. En particular, se observó la pericálaza en el plano longitudinal mediano de la endotesta, con más de dos filas de tejido vascular. El tegmen o integumento interno se observó colapsado. El plug micropilar fue cónico, leñoso y formado por células fibras transversales, se observó también línea de fractura. El endospermo ruminado se vió interferido tanto por el integumento interno como por el externo. Se observaron idioblastos oleíferos en los bordes externos de las ruminaciones.
Palabras clave: cubierta seminal, endospermo ruminado, plug micropilar, embrión.
Introduction
The Annonaceae family includes about 50 genera. Three genera (Annona, Rollinia, and Asimina) produce edible fruits (George and Nissen, 1987). Two genera are of commercial importance: Annona, comprising about 100 species, and Rollinia, which includes about 50 species (George and Nissen, 1985). The main species are cherimoya (Annona cherimola Mill), the atemoya hybrid (Annona cherimola Mill. × Annona squamosa L.), sugar apple (Annona squamosa L.) and soursop (Annona muricata L.) (Moreno et al., 2013a).
The sugar apple is the species of the Annona genus and is widely distributed in the wild worldwide and in Colombia (Moreno et al., 2013b). It grows in the dry areas of the valleys in the provinces of Valle, Caldas, Tolima, Cundinamarca, and Santander (Guerrero and Fischer, 2007). The sugar apple grows from sea level to 1,000 m a.s.l. (Hoyos, 1989) and requires no cold periods; and so, develops and grows well in relatively stable conditions of temperature (George and Nissen, 1987). The minimum temperature is in a range of 10 to 20°C and the maximum is 22 to 28°C (Guerrero and Fischer, 2007).
The information regarding anatomical and morphological aspects of the Annonaceae family is limited to a few species and anatomical studies related to fruits and seeds are scarce. Likewise for systematic analyses, the anatomical characteristics of seeds are less employed than floral characteristics due to a lower phylogenetic utility and difficulty using these structures (Galastri, 2008; Chatrou et al., 2012; Pirie and Doyle, 2012). However, Garwood (1995) noted that such studies are relevant for the understanding of ecological phenomena, such as frugivory, ecological succession, dispersal and germination of seeds that are of considerable importance for restoration processes.
The anatomical descriptions for the Annonaceae genus are reported by Corner (1949), who made a complete anatomical study of seed integuments in Anaxagorea javanica, A. squamosa, Desmos dasymaschala, D. chinensis and D. dumosa, Xylopia fusca, X. caudata, and other species. This author discussed a number of peculiarities of the seeds of the genus, which Periasamy (1962), Swamy (1961) and Rao (1975, 1979, 1982) and Svoma (1998) subsequently presented in more detail, describing a characteristic anatomical pattern including perichalaza, fibrous mesotesta and ruminations developed by both integuments. Likewise, aryl, thin sarcotesta, that, when fleshy, wraps the entire seed (Judd et al., 1999), and depressions were presented in some taxa, showing different origins and structures.
The seeds of Annonaceae are albuminous, ellipsoid and their lengths range between 5 and 30 mm. They have a ruminate endosperm (Corner, 1949; Garwood, 1995; Judd et al., 1999), on which Van der Heijden and Bouman (1988) described four types of ruminations: ruminations plaques formed by long plates, narrow plates, sticks, or small flattened plates with clasped sticks. The seeds are formed from an anatropous ovary, with bi- or tritegumented perichalaza, crassinucelated, transverse or subbasal, and erect, with micropyle formed by the endostoma, due to fact that the internal integument exceeds the external one (Corner, 1949; 1976).
One aspect found in many kinds of Annonaceae is the occurrence of a lignified tissue in the region that closes the opening micropylar aperture, called the micropylar plug or strophiole (Van der Heijden and Bouman, 1988) or subhilar tissue (Corner, 1949).
Setten and Koek-Noorman (1992) indicated that Annonaceae seeds have a small embryo that is considered underdeveloped and immature. Due to this immaturity, it is likely that more time is needed for embryo growth after dispersal than in other species. Meanwhile, Hayat (1963) reported that A. squamosa seeds have a small embryo with two foliaceous, thin cotyledons that take one to three months to germinate.
In the present study, we described and illustrated the anatomy of A. squamosa seeds, comparing the results with studies done on other species of the family. This research is important not only for ecological and systematical purposes but also because it provides useful information for understanding the phenomena associated with the process of germination and dormancy of these seeds.
Materials and methods
Plant material
The selected material was part of the accessions collected for the development project: Colombian Bank of Anon Germplasm, framed within the program: Planting Material and Breeding of the sugar apple (A. squamosa). The selected accessions were collected in the municipality of Apulo, Cundinamarca from individuals distributed in agroecosystems and not in the field or spontaneous gardens. The A. squamosa seeds were obtained from fruits of collection accessions C2AS224 (C: Cundinamarca; 2: municipality code, AS: A. squamosa species; 224: row number), C2AS226, and C2AS225. From fruits that were ripe, soft to the touch and washed with room temperature water to remove any residues present on the surfaces, the exocarp was separated from the mesocarp (pulp). Subsequently, the seeds were removed manually from the pulp and aryl, after which they were washed with water and placed on absorbent paper towels at room temperature in order to dry to moisture contents of 10%. The seeds were disinfected with 1% sodium hypochlorite for 9 min and washed afterwards with 96% ethanol and three times with distilled water.
Hystological analysis
Given that A. squamosa seminal tissues are highly resistant to cutting due to tough and lignified seed coats, the protocol of Sandoval (2005) for soft plant tissues used in the Laboratory of Microbiology, Faculty of Agronomy, Universidad Nacional de Colombia was adapted and modified for cuts, where the tissue continuity was well-defined and present. The methodology presented below was the result of this adjustment.
Whole, latent A. squamosa seeds with 10% moisture were imbibed in distilled water for 72 h. Subsequently, the seeds were covered with 50% carbolic acid solution for 24 h in order to soften the testa.
The cuts were done in Bouin's fixative (1929) for 48 h at room temperature. Subsequently, the tissues were immersed for 24 h in absolute alcohol and, after that, were was imbibed in tert-butanol for 24 h. The sections were imbibed for 24 h at 60°C in 4 graded concentrations of molten paraffin at 56°C and tert-butanol with increasing concentrations of paraffin and decreasing concentrations of tert-butanol; thus having: a) 25% paraffin – 75% tert-butanol, b) 50% paraffin – 50% tert-butanol, c) 75% paraffin – 25% tertbutanol, and d) 100% paraffin. The blocks were prepared and, prior to the final cutting, were immersed in a solution of 50% hydrochloric acid for 2 h. The cuts were made with a 7 µm microtome. Staining was performed for 24 h with safranin and Fast Green for 10 s. Once the dyeing process was performed, the sheets were cleaned and historesine was applied over the cuts with a foil sealing coverglass. The documentation and observations were made with an Olympus ® CX31 microscope (New York Microscope Company, New York, NY) and the image editing software Image-Pro Plus® (Media Cybernetics, Rockville, MD ).
Results and discussion
Seed coat
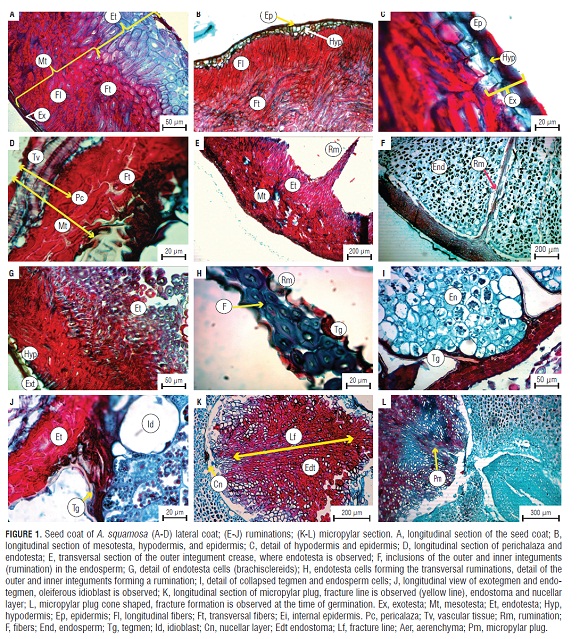
The seed coat was composed of different sections corresponding to the visible outer integument composed of the exotesta, endotesta, and mesotesta, formed by transversal, longitudinal, and diagonal fibers and the inner integument or tegmen (Fig.1A). The outermost epidermis referred to as exotesta was uniseriate, comprised of isoradiometric, tangentially elongated, cuboid cells with thin walls and periclinal thickening in the outer wall and further anticlinally elongated cells as a palisade tissue. The hypodermis was observed to be composed of one or two layers of thin wall isoradiometric cells, compressed and crystalline in appearance (Fig.1B and Fig.1C).
The mesotesta had two layers of lignified fibers interrupted by elongated, longitudinal and transversal, oblique, highly packed cells that might correspond to macrosclereids subsequent to brachysclereids covered with a thin cuticle and one to three layers of hypodermic cells. The endotesta consisted of transverse, isoradiometric fibers with a multilayer of thickened walls, but less packed than the mesotesta (Fig.1A and Fig.1B). In the medium longitudinal plane of the endotesta, the fragment of the perichalaza was observed to be immersed in the outer integument, where it was possible to identify more than two rows of the vascular tissue (Fig.1D) and thin-walled parenchyma cells with small intercellular spaces. The endotesta ruminations comprised both proliferation and extended towards the micropylar area, where the micropylar plug was multiseriate in appearance.
The folds were observed in the inner and outer integuments (tegmen and endotesta), composed mainly of transverse fibers (Fig.1E) penetrating the endosperm to yield the ruminations that were distributed along the length of the seed. Figure 1H shows in detail the cellular structure of the folds that were comprised of the tegmen and endotesta and were in association with the endosperm. Likewise, the regions of the mesotesta were observed to have sections of hypodermis and the least packing of the fibers of the endotesta when included in the endosperm (Fig.1G).
One inner integument composed of a single-layer tegmen can be observed in Figures 1I and J, with a collapsed appearance, arranged after the endotesta and surrounding the ruminations of the endosperm.
The micropylar plug was conical in shape, formed by cells of the transversal, isoradiometrical fibers of the endotesta, containing some crystals. A less dense fiber packing (brachysclereids and macrosclereids) and a thin tissue formation (aerenchyma) were apparent (Fig.1K and Fig.1L). The remains of the internal integument were also observed, mainly tegmen, forming a nucellar layer. In the micropylar plug, it was possible to identify a linear region where the cells were arranged less densely, which might be a fracture line (Fig.1K). Additionally, it was observed that the consistency of the micropylar tissue was soft and viscous.
Corner (1949) reported A. squamosa exotesta characteristics very similar to those found in our study. According to this author, the exotesta was presented by colorless, isoradiometric epidermis cells, with much denser outer walls. Also, in other Annonaceae species, the anatomical characteristics of the exotesta are similar to those of A. squamosa. For example, in Annona dioica, the exotesta is also uniseriate and presents more or less cuboid cells with thin walls, impregnated with phenolic substances. In Duguetia furfuracea and Xylopia emarginata (Annonaceae), the exotesta is composed of phenolic cells of a palisade appearance with thin, pectic walls with amiliferous reserves and without obvious nucellar tissue (Galastri, 2008). Svoma (1997) indicated that, in Bocageopsis canescens (Annonaceae), the exotesta is composed of anticlinal isoradiometric cells, very narrow and thin-walled, generally containing a large vacuole that is surrounded by small taniferous grains and additional elongated, solitary cells. Similar characteristics were also present in Annona sp. (A. stenopetala, A. cherimola, A. montana, A. muricata), Bocagea sp., Bocageopsis canescens, Unonopsis sp., Xylopia aromatica, and X. emarginata. In A. muricata and A. reticulata, the exotesta is consisted of sclereids impregnated with tannins (Corner, 1949; 1976, Van der Heijden and Bouman, 1988; Svoma, 1997; 1998).
The hypodermis traits observed in this study were also consistent with those reported by Corner (1949), Van der Heijden and Bouman (1988) and Svoma (1998) for A. squamosa, who indicated that this tissue was composed of a row of small thin-walled cells, compressed and containing a colorless crystalline material, which is common for the Annonaceae genera. In species such as Annona, Raimondia, Rollinia, D. furfuracea, and X. emarginata, Van der Heijden and Bouman (1988) and Galastri (2008) found anatomical characteristics of the hypodermis similar to those found for A. squamosa. However, in this tissue, Annona dioica differs from the other species because it is formed by thin-walled cells and a bulky, pectic content that are distributed throughout the length of the seed (Van der Heijden and Bouman, 1988).
The mesotesta characteristics of A. squamosa coincided with those reported for Annonaceae by Corner (1949), Van der Heijden and Bouman (1988) and Svoma (1998). These authors reported two distinct regions, one comprising the outer one with longitudinal fibers and the other comprising the inner one with transverse fibers. The fibers exhibited thickened and lignified walls (Svoma, 1997; 1998).
In this study, it was possible to identify two regions in the mesotesta; however, this feature differs from Annona hypoglauca which has mesotesta composed of three regions, while in Annona tenuiflora and Polyalthia purpurea there was no differentiation in the regions (Corner, 1949; Van der Heijden and Bouman, 1988). The presence of longitudinal fibers in mesotesta, uninterrupted by areas of transverse fibers, was reported for other Annonaceae species, such as A. muricata, Annona bicolor, A. senegalensis, A. sericea, Raimondia quinduensi. In these species, mesotesta fibers can pass through the outer section (Van der Heijden and Bouman, 1988).
Unlike that found in Annona bicolor, A. cherimola, Rollinia mucosa, and R. emarginata by Van der Heijden and Bouman (1988), our study did not see parenchymal or nonlignified cells of the mesotesta in A. squamosa but it rather reveal the tissue that had a lignified fibrous appearance, which was also reported in Ancana sp., A. stenopetala, A. cherimola, A. montana, Bocageopsis canescens, Unonopsis sp. by Svoma (1997, 1998), and in Xylopia aromatica and X. emarginata, and by Galastri (2008) in D. furfuracea and X. emarginata.
The endotesta characteristics were consistent with those reported by Svoma (1997) and Van der Heijden and Bouman (1988) for A. squamosa. Meanwhile, Corner, (1949) reported that this tissue is composed of compactly arranged cells, which was similar to that found in Asimina triloba by Rao (1982). We concluded that this fibrous tissue is involved in the formation of ruminations and the micropylar plug and is also observed in the lateral regions of the seed in perichalazal tissue, which agrees with that reported by Corner (1949), who, observing Canangium odoratum, defined the endotesta in terms of an intermediary integument, referring to the integument that is located between the outer and the inner integuments.
Unlike that found by Svoma (1998) in Artabotrys hexapetalus, in the endotesta of A. squamosa, cells pre-swallowed by starch grains and crystalliferous idioblasts could not be observed. These idioblasts occurred as solitary crystals of calcium oxalate and are mostly prismatic (Svoma, 1998). The chalaza in Annonaceae extends around the seed. Corner (1949, 1976) defined perichalaza to describe the condition in which chalaza and vascular package extend around the seed. According to Boesewinkel and Bouman (1984), this occurs up to the median longitudinal plane from the funiculus to the micropyle.
Observations on the anatomy of the perichalaza in this study are consistent with those made by Corner (1949, 1976), Rao (1979, 1982), Van der Heijden and Bouman (1988) and Svoma (1997, 1998) for different Annonaceae species. The authors noted that the perichalaza is consisted of parenchyma cells, including the vascular system. This tissue develops along the seed in the longitudinal median plane and presents a taniferous tissue in the region, where the mesotesta is interrupted by vascular bundles of perichalaza. However, in the present study, it was not possible to identify either the continuity of the perichalaza in the seed perimeter or taniferous cells; however, it was noted that the perichalaza interrupted the median plane of the endotesta.
According to the Annona species, rumination folds developed in the sides of the seed per share of two integuments (Periasamy, 1962). In this study, the ruminations of the A. squamosa seeds were generated from the endotesta and tegmen, similar to that seen by Corner (1949) and Galastri (2008), who noted that the endosperm ruminations are generated by transverse folds and not vascularized by the tegmen, testa, or intermediary integument, which are interrupted in the median plane by the perichalaza (Rao, 1975; Svoma, 1998). However, in some Annonaceae, the ruminations are formed by the testa, as the tegmen is collapsed (Rao, 1975, 1979, 1982; Svoma, 1998). In D. and X. furfuracea emarginata, the ruminations are formed by the internal mesotesta, endotesta, and tegmen. In A. dioica, the median region mesotesta also forms part of the ruminations (Galastri, 2008). Corner (1949) pointed out that the process of formation of ruminations begins in the early stages of seed development, where nucellar tissue is interrupted by parallel and lateral inclusions of the external (endotesta) and internal (tegmen) integuments.
The tegmen characteristics observed in the A. squamosa seeds were similar to those reported by Corner (1949). This author indicated that the tegmen is multiplicative and is generally collapsed, accumulating or not accumulating oil cells. Similar to what was found for A. cherimola (Schroeder, 1951), the seed tegmen of A. squamosa, according to the outcome of the present study, is constituted by a single layer of fibers (Fig.1I), while in Anaxagorea javanica, it consists of four layers of thin-walled cells, some with lipid content (Corner, 1949). In Artabotrys hexapetalus, there are two layers of cells throughout the development of tegmen (Svoma, 1997), while in Xylopia aromatica and Bocageopsis canescens, the tegmen collapses except in the micropylar region (Svoma, 1998). In Annona glabra, a species whose seed dispersion is hidrohoric, the parenchymatous tegmen functions as a fluctuation tissue (Van der Heijden and Bouman, 1988).
An important anatomical finding in this study was the identification of the tissue that forms the micropylar plug, which has similar characteristics to the intermediary integument or endotesta. This tissue is lignified but less packed than the mesotestal tissue, indicating that the micropylar plug is a result of the differentiation and proliferation of the endotesta. This is consistent with reports of Galastri (2008) on D. furfuracea and X. emarginata and of Svoma (1998) on A. squamosa and Xylopia aromatica, where it was revealed that the micropylar plug is formed by the endotesta. On the contrary, Corner (1949, 1976) indicated that the lignified micropylar plug of Annonaceae seeds is derived from the inner integument or, possibly, the intermediate integument when the latter is present. In Ancana sp., the micropylar plug is composed entirely of tegmen.
The characteristics of the fibers, brachy- and macrosclereids were similar to those found in the soursop micropylar plug (A. muricata) by Esau (1972), who reported that the seed micropyle presented a pyramidal plug formed by sclerenchymatous cells of a brachysclereid type. Meanwhile, Van der Heijden and Bouman (1988) indicated that the micropylar plug originates from the exostoma and is consisted of isodiametric sclerenchymatous cells, some of them with crystals.
The cone-shaped micropylar plug found in this anatomical study of A. squamosa seeds was also reported by Svoma (1997, 1998), who also indicated that the plug is not expelled from the seed during germination but breaks down along the fracture line that was formed before germination. The crack region visible in the cuts of the micropylar plug in the present study was also reported in seeds of D. furfuracea and X. emarginata by Galastri (2008).
Consistency of the micropylar plug is another important characteristic in A. squamosa seeds and its appearance and texture were also reported by Svoma (1998), who noted that, in A. cherimola, A. montana, A. muricata, and A. squamosa, a sticky parenchymal mass is present that forms a partial closure of the opening in the testa. In Artabotrys hexapetalus, the closure is formed by multiple overlying endotesta. This closure is formed by a mass of taniferous cells without lignified walls (Svoma, 1997).
Endosperm and embryo
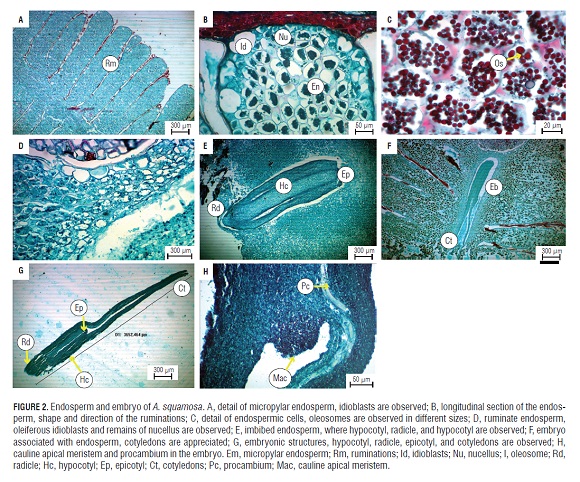
The endosperm was cellular and ruminate, occupied most of the seed and was the main reserve tissue. The ruminations were separated from each other by inclusions or transverse folds of the outer and inner integuments. Spiniforms were more or less flattened and fimbriated (Fig.2A). Structures presumed to be oleiferous idioblasts were observed in the ruminations ends, close to the edge of the seed, where cells were irregularly arranged and of different size and shape than the bulky cells present in most endospermic tissue. These residues might correspond to the nucellus (Fig.2B). The endosperm presented voluminous cells of different sizes, but generally isoradiometric, meristematic, with dense cytoplasm that were divided in an anticlinal sense. Slightly thickened walls presented cells with unidentified reserve structures, presumably of a lipid (oleosomes) and pectin content (Fig.2C) (Corner, 1949, 1976; Svoma, 1997). In the micropylar region, cells of a smaller size and density were located, comprising no more than eight endospermatic cell layers, separating the embryo from the micropylar plug (Fig.2D).
The A. squamosa endosperm showed various characteristics mentioned for other Annonaceae (Corner, 1949, 1976; Schroeder, 1951; Svoma, 1997; Galastri, 2008). The endosperm was ruminate, one typical for Annonaceae according to Boesewinkel and Boumann (1984), and was characterized as having no vascularization and perfectly surrounding the tiny embryo, simulating a system of biological irrigation (Villamil et al., 1999). In soursop, according to Villamil et al. (1999), the seed has an albuminous endosperm, abundant, massive parenchymal cell walls and a thicker consistency of a corneous type.
Although the type of reserve substance in the endospermic cells of the A. squamosa seeds was not studied, some authors (Corner, 1949, 1976; Schroeder, 1951; Svoma, 1997; Galastri, 2008) reported two types of reserve substances in Annonaceae, lipid and amiliferous. However, there is a greater number of reports of lipids as the main reserve substances, with, in addition, proteins and amides. In A. dioica, the endosperm is mainly constituted by proteins, lipids, and amides and D. furfuracea and X. emarginata contain mainly lipids. Likewise, the middle region of the endosperm cells of these species has thinner walls with peptic contents (Galastri, 2008). In some species, such as Polyalthia longifolia (Rao, 1979), Xylopia emarginata (Svoma 1998), Polyalthia purpurea, and Popowia nervosa, the endosperm is rich in starch and oils (Corner 1949). The endosperm of Anonidium mannii has a large reserve of amides and small amounts of lipids (Van der Heijden and Bouman, 1988). Bocageopsis canescens (Svoma, 1998), Xylopia fusca, and Alphonsea elliptica have endosperms with thickened walls and accumulate lipid substances; in particular, walls with crystals were observed (Corner, 1949). Unonopsis sp. seeds also present thickened cell walls but accumulate more lipids and a small quantity of amides and small crystals (Galastri, 2008).
The type of fimbriate rumination present in A. squamosa is found within the types reported by Setten and Koek- Noorman (1992), who stated that there could be lamellated or fimbriated ruminations, or divided into sticks with raphe and antiraphe regions (Ipsis verbis). The hilum is rarely defined (Corner, 1949) or it is basal or hidden and could be exposed by the rudimentary aryl (Garwood, 1995).
Nucellar tissue debris and idioblasts found in the terminal region of the ruminations with a presumed lipid content were also reported by Corner (1949) for A. squamosa. This author indicated that the endosperm has nucellar cells containing three to four drops of oil incorporated in the outermost region of the endosperm, which is a trait generated because, during the seed maturation process, the endosperm replaces all nucellus except the oil cells (idioblasts) (Corner, 1949; Van der Heijden and Bouman, 1988). In some species of Annonaceae, it was also possible to identify the cuticle, nucellar epidermis, and subepidermal oleiferous idioblasts (Van der Heijden and Bouman, 1988).
As for the origin and formation of ruminations, Werker (1997) argued that the ruminations resulted from the development of the integuments during seed development, where the endosperm becomes quiescent. When the seed has reached its final size and the endosperm is activated and becomes internally adjusted to the ruminations after absorbing the nucellus. Meanwhile, Corner (1949) indicated that the process of rumination formation starts at the early stages of development, when the nucellar tissue is interrupted by parallel and lateral inclusions of external (endotesta) and internal (tegmen) integuments.
Svoma (1997) argued that ruminations serve to supply water and nutrients to the embryo and endosperm. However, there are doubts because the supply of nutrients and water during seed development and at early stages of germination is only through the perichalaza. Meanwhile, Bayer and Appel (1996) argued that ruminations, frequently consisted of oils and phenolic substances, make seeds less attractive to seed predators. Also, the increased area of contact between the integuments and the endosperm facilitates the supply of water, oxygen, and nutrients to the endosperm or embryo development. Moreover, ruminations are more permeable to gases than the endosperm, facilitating gas exchange (Svoma, 1997).
The embryo was in the basal central region oriented towards the hilum zone, protected by the micropylar plug, which agrees with the results of Villamil et al. (1999). With a size of about 3.6 mm, it was linear, straight, with two flat, thin, and foliaceous cotyledons and was embedded in the endosperm (Fig.2E). It had tiny, straight with poorly differentiated embryonic axis and rudimentary plumule (Fig.2E-G). The embryo presented isoradiometric and thin-walled meristematic cells in the hypocotyl, cotyledons, and radicle. It was easy to observe the procambium and apical meristem (Fig.2H). The consistency of the embryo was fleshy and it was easily separated from the hydrated endosperm; it had circular cells without a uniform distribution.
The embryo characteristics were consistent with those reported for A. squamosa by Hayat (1963), Hayat and Canright (1965) and for the Annonaceae family by different authors (Corner, 1949; Hayat, 1963; Hayat and Canright, 1965; Garwood, 1995; Svoma, 1997, 1998, Judd et al., 1999) and Galastri (2008) in D. furfuracea, A. dioica, and X. emarginata. Hayat and Canright (1965) also reported the presence of differentiated epidermis and procambium in the embryo for A. squamosa. Hayat (1963) noted that the cotyledons were separated by a thin layer of levulose. According to Villamil et al. (1999), in A. muricata, the cell walls are formed by amyloids and are high in lipids.
Conclusions
The anatomy of the seed coat, endosperm, and embryo corroborates the data reported in the literature for the Annonaceae family.
The outer integument is composed of three layers: exotesta, endotesta, and mesotesta; and contains the inmersed perichalaza which is an association of parenchymal and vascular cells.
The micropylar plug is a proliferation of less dense, endotestal tissue where a fracture line can be seen.
The endotesta is the tissue that is most involved in the formation and constitution of seed organs and confers a ruminate appearance to the seed endosperm. The endosperm is ruminate and has cells of various forms and contents that are bulky and isoradiometric, where idioblasts are present.
The embryo is foliaceous, flat, and underdeveloped.
Literature cited
Bayer, C. and O. Appel. 1996. Ocurrence and taxonomic significance of ruminate endosperm. Bot. Rev. 62(4), 301-310. [ Links ]
Boesewinkel, F.D. and F. Bouman. 1984. The seed structure. pp. 567-610. In: Johri, B.M. (ed.). Embryology of angiosperms. Springer-Verlag, Berlin. [ Links ]
Bouin, P. 1929. Elements of histology . Felix Alcan, Paris. [ Links ]
Chatrou, L.W., M.D. Pirie, R.H.J. Erkens, T.L.P. Couvreur, K.M. Neubig, J.R. Abbott, J.B. Mols, J.W. Maas, R.M.K. Saunders, and M.W. Chase. 2012. A new subfamilial and tribal classification of the pantropical flowering plant family Annonaceae informed by molecular phylogenetics. Bot. J. Linn. Soc. 169, 5-40. [ Links ]
Corner, E. 1949. The annonaceous seed and its four integuments. New Phytol. 4(8), 332-364. [ Links ]
Corner, E. 1976. The seeds of dicotyledons. Cambridge University Press, Cambridge, UK. [ Links ]
Esau, K. 1972. Anatomía vegetal. 2nd ed. Ediciones Omega, Barcelona, Spain. [ Links ]
Galastri, N. 2008. Morfoanatomia e ontogênese de frutos e sementes de Annona dioica (A. St.-Hil.), Duguetia furfuracea (A. St.- Hil.) Saff. e Xylopia emarginata Mart. (Annonaceae). M.Sc. thesis. Institute of Biosciences, Universidade Estadual Paulista, Botucatu, Brazil. [ Links ]
Garwood, N.C. 1995. Studies in Annonaceae. XX. Morphology and ecology of seedlings, fruits and seeds of selected Panamanian species. Bot. Jahrb. Syst. 117, 1-152. [ Links ]
George, A.P. and R.J. Nissen. 1985. The custard apple. Part 1: Species, varieties and rootstock selection. Austral. Hortic. 83, 100-106. [ Links ]
George, A. and R.J. Nissen. 1987. Propagation of Annona species: a review. Sci. Hortic. 33, 75-85. [ Links ]
Guerrero. E. and G. Fischer. 2007. Manejo integrado en el cultivo de anón (Annona squamosa L.). Rev. Colomb. Cienc. Hortic. 1, 154-169. [ Links ]
Hayat, M.A. 1963. Morphology of seed germination and seedling in Annona squamosa. Botan. Gazet. 124, 360-362. [ Links ]
Hayat, M.A. and J.E. Canright. 1965. The developmental anatomy of the Annonaceae embryo and early seedling structure. Amer. J. Bot. 52, 228-237. [ Links ]
Hoyos, J. 1989. Frutales en Venezuela. Sociedad de Ciencias Naturales La Salle, Caracas. [ Links ]
Judd, W.S., C.S. Campbell, E.A. Kellogg, and P.F. Stevens. 1999. Plant systematics: a phylogenetic approach. Sinauer Associates, Sunderland, MA. [ Links ]
Moreno B., N.E., D. Miranda, and F.E. Martínez M. 2013a. La zeatina fomenta la germinación de semillas de anón (Annona squamosa L.). Rev. Colomb. Cienc. Hortic. 7(1), 9-19. [ Links ]
Moreno B., N.E., D. Miranda, and F.E. Martínez M. 2013b. Germinación de semillas de anón (Annona squamosa L.) sometidas a estratificación. Rev. Colomb. Cienc. Hortic. 7(1), 20-30. [ Links ]
Rao, P.R. 1975. Seed anatomy of Artabotrys odoratissimus with a discussion on chalaza, integumentary bundles and ruminate endosperm. Phytomorp. 25, 215- 228. [ Links ]
Rao, P.R. 1979. Seed and fruit anatomy in Polyalthia longifolia (Annonaceae). Bot. Jahrb. Syst. 101, 83-89. [ Links ]
Rao, P.R. 1982. Seed and fruit anatomy in Asimina triloba, with a discussion on the affinities of Annonaceae. Bot. Jahrb. Syst. 103, 47-57. [ Links ]
Periasamy, K. 1962. The ruminate endosperm: development and types of rumination. pp. 62-74. In: Plant Embryology Symposium. Council of Scientific and Industrial Research, Department of Botany, University of Delhi, New Delhi. [ Links ]
Pirie, M.D. and J.A. Doyle. 2012. Dating clades with fossils and molecules: the case of Annonaceae. Bot. J. Linn. Soc. 169, 84-116. [ Links ]
Sandoval, Z.E. 2005. Técnicas aplicadas al estudio de la anatomía vegetal. Institute of Biology, Universidad Nacional Autónoma de México, Mexico DF. [ Links ]
Setten, K. and J. Koek-Noorman.1992. Fruits and seeds of Annonaceae. Morphology and its significance for classification and identification. Bibliot. Botan. 142, 1-101. [ Links ]
Schroeder, C.A. 1951. Fruit morphology and anatomy of the cherimoya. Botan. Gazet. 112(4), 436-446. [ Links ]
Svoma, E. 1997. Seed development and function in Artabotrys hexapetalus (Annonaceae). P1ant. Syst. Evol. 207, 205-223. [ Links ]
Svoma, E. 1998. Studies on the embryology and gynoecium structures in Drimys winteri (Winteraceae) and some Annonaceae. Plant. Syst. Evol. 21(9), 205-229. [ Links ]
Swamy, B. 1961. Studies in the Annonaceae. The development of ovule and seed in Cananga odorata and Miliusa wightiana. Indian Bot. Soc. 41, 206-216. [ Links ]
Van der Heijden, E. and F. Bouman. 1988. Studies in Annonaceae. Seed anatomy of the Annona group. Bot. Jahrb. Syst. 110 (1), 117-135. [ Links ]
Villamil, H., G. Corchuelo, and M. De Valencia. 1999. Morfología, anatomía de la semilla y composición química del endospermo de Annona muricata L. Agron. Colomb. 16(3), 19-23. [ Links ]
Werker, E. 1997. Seed anatomy. Gebrüder Borntraeger, Berlin. [ Links ]





![Gas exchange and mass distribution of the cowpea (Vigna unguiculata [L.] Walp.) under water deficit](/img/en/next.gif)