Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Agronomía Colombiana
Print version ISSN 0120-9965
Agron. colomb. vol.33 no.1 Bogotá Jan./Apr. 2015
Doi: 10.15446/agron.colomb.v33n1.47199
1 Oil Palm Biology and Breeding Research Program, Colombian Oil Palm Research Center (Cenipalma). Bogota (Colombia). hromero@cenipalma.org
2 Department of Biology, Faculty of Science, Universidad Nacional de Colombia. Bogota (Colombia)
Received for publication: 13 November, 2014. Accepted for publication: 30 March, 2015.
ABSTRACT
The phenological stages of oil palm can be coded using the BBCH scale, which has three digits due to the inclusion of intermediate stages between the principal and secondary stages in order to provide greater detail on each developmental stage. For the phenological description of the reproductive development of Elaeis oleifera, the principal stages used were emergence of inflorescence, flowering, fruit growth and development, and fruit ripening. The observations were made in Colombia over a 12 month-period on E. oleifera palms planted in 1991; the observations were made on the daily course or depending on the development stage. The duration of each phenological stage was measured in days. Thus, the appearance of new leaves took 20.1±2.8 days, reaching preanthesis I (601) took 145.09±19.61 days, from this stage to preanthesis II (602) took 7.50±1.50 days, then to preanthesis III (603) took 7.39±1.56 days and finally to anthesis (607) took 5.74±1.32 days. At the population level, it was found that the phenology cycle of inflorescence is annual and that the production of flowers and the opening of inflorescences with pistils is asynchronous.
Key words: American oil palm, Noli, BBCH scale, Arecaceae, Colombia.
RESUMEN
Los estadios fenológicos de la palma de aceite pueden ser codificados de acuerdo a la escala BBCH, que consta de tres dígitos, debido a la inclusión de mesoestadios entre los estadios principales y secundarios con el fin de presentar más detalles de cada etapa de desarrollo. Para la descripción fenológica del desarrollo reproductivo de Elaeis oleifera se tomaron como base cuatro de estos estadios principales: emergencia de inflorescencias, floración, crecimiento y desarrollo de fruto y maduración del fruto. Las observaciones se realizaron en Colombia sobre palmas de E. oleifera sembradas en 1991, durante un período de 12 meses; las observaciones se realizaron diariamente o dependiendo de la etapa de desarrollo. La duración de cada etapa fenológica se midió en términos de días. Así, la aparición de nuevas hojas tomó 20,1±2,8 días, llegar a preantesis I (601) 145,09±19,61, de allí a preantesis II (602) 7,50±1,50 días, a preantesis III (603)7,39±1,56 y a antesis (607) 5,74±1,32 días. A nivel poblacional se encontró que el ciclo fenológico de inflorescencias es anual y la producción junto con la apertura floral de las inflorescencias pistiladas fue asincrónica.
Palabras clave: palma de aceite americana, Nolí, escala BBCH, Arecaceae, Colombia.
Introduction
According to the current classification of the Elaeis genus, it belongs to the Liliopsida class (monocotyledonous) of the Arecaceae family (Palmaceae). This family includes over 2,500 species distributed throughout all of the tropical and sub-tropical regions of the world. They also offer broad morphological diversity and are confined primarily to tropical rainforests in Asia and the Americas. Though the genus Elaeis has three species, only two are of agronomic and economic importance: Elaeis guineensis, known as the African oil palm (Combres et al., 2013; Dransfield et al., 2005), and Elaeis oleifera, native to tropical America, which is the second most important species in this genus and grows naturally from northern South America (Brazil, Colombia, Ecuador, Venezuela, Guyana, and Suriname) to Central America (Costa Rica, Nicaragua, and Honduras) (Rey et al., 2003; Rey et al., 2004; Rivera et al., 2013). The species Elaeis odora is not cultivated and little is known about it.
The American palm E. oleifera has single stalks that grow 1-3 m high and 25-30 cm wide, with erect or semi-erect growth habits, covered by strong petiole bases and crowned by a rosette of pinnate leaves arranged in a spiral shape; the leaflets are regularly distributed along the rachis and inserted on a single plane (de Andrade Miranda, 2001; Rocha et al., 2006). The flowers, numerous and inconspicuous, are grouped in a sword-shaped inflorescence (spadix) and are protected by two bracts, generally woody. The flowers have three sepals and three petals, are generally single-sex, and may be found on the same tree. The fruit is a drupe, with a meaty and fibrous pericarp (de Andrade Miranda, 2001).
Most commercial plantations are planted with African genetic stocks (Tenera hybrid); however, some commercial plantations are currently planted with inter-specific hybrids (OxG), obtained by crossing a E. oleifera mother and E. guineensis father, which apparently offer tolerance to the most damaging diseases. However, the hybrids obtained to date, in addition to having lower productivity than the African palms, have problems related to abnormal inflorescences, low pollen viability, and some productivity limitations related to the development of bunches (Sánchez, 2008).
Oil palm research has focused on materials produced through crosses with various parents, as in the case of the American palm E. oleifera, whose characteristics offer great potential for genetic improvement, especially because it can be easily crossed with African palms (Meunier and Boutin, 1975; Meunier, 1991). Studies of this species have been undertaken not only because of its genetic potential, but also because its biology is generally not well-known. For this reason, it is necessary to carry out detailed research on this species, particularly on the phenology of the plant development, in order to be able to predict its behavior in different environments.
The BBCH scale is used to identify the development of mono- and dicotyledon plants. Phenological stages are used in several scientific disciplines (crop physiology, entomology, etc.) and in the agricultural industry (application of agro-chemicals, fertilization, agricultural insurance policies, etc.). This scale establishes a coding system that divides growth into principal and secondary stages and is based on the system of Zadoks et al. (1974) for cereals. The BBCH scale offers numeric characteristics for each stage, is universal, and can be standardized for other species. The scale has 10 principal stages (seed germination, leaf development, tillers, stem elongation, development of the plant's harvestable parts, emergence of inflorescence, flowering, fruit development, fruit ripening, seed formation, and senescence), of which 8 apply to oil palms according to Forero et al. (2012).
This paper establishes, in detail, the stages of female floral development and fruit and bunch growth and development for the American Oil Palm as the first step to understanding how the plant phenology changes.
Materials and methods
The study area was in the municipality of Paratebueno (Cundinamarca, Colombia), at the Cuernavaca Estate of the Unipalma Plains Plantation, plot CPT 18, with an altitude of 240 m a.s.l.; the E. oleifera (Brazil) palms were planted in 1991. Climate conditions in the study area are: average temperature of 27°C and average rainfall of 3,800 mm year-1, in a mono-modal distribution; the rainy season occurs from March to November and the dry season occurs from December to March, featuring a very marked drop in precipitation (Ideam, 2012). Five different codes were selected based on the different origins of the parent plants (codes 3531, 3530, 3527, 3551, 3557, and 3563) and a sample of six palms was taken for each one, for a total of 30 palms.
The duration of the phenological stages of leaf opening and development of inflorescence was measured in terms of days, as well as the phenological stages of flower development, flowering and producing fruit. They were described using the BBCH general phenological scale of Bleiholder et al. (1991), Hack et al. (1992), and Hess et al. (1997).
Phenological monitoring was performed on a weekly basis or as required by the speed of growth of the organs over a period of 12 consecutive months (December of 2010 to December of 2011) for 30 individuals (palms) randomly selected in the study area. Data were collected for leaf growth, flowering and fruit development. The BBC phenological scale has been used for the phenological description of the oil palm E. guineensis Jacq. (Forero et al., 2012) and the inter-specific hybrid OxG (E. oleifera × E. guineensis) (Hormaza et al., 2012). For the estimation of leaf growth, data were taken for 5 months; the initial lanceolate leaf was labeled (H0) and the number of days it took to reach the position of leaf 1 was established (leaf with 70% of the leaflets open); subsequent leafs were sequentially labeled (H2, H3, H4... etc.)
For the description of the development of the floral structures (inflorescences), the phenological scale proposed by Hormaza and Romero (2012) was used, making appropriate adjustments to the morphological differences of the species E. oleifera. Additionally, morphological recognition was carried out for the floral structures. For each sample unit (palm), three consecutive inflorescences were studied, beginning with stage 501 (undifferentiated, 10% growth) and starting on December 2, 2010. Observations of the flowering stage were taken on a weekly basis and occasionally more frequently, depending on the rate of changes in the floral structures. A group of 50 inflorescences was selected. Once these inflorescences entered the anthesis stage, the prophyll and peduncle bract were removed to expose the flowers and assisted pollination was performed. The pollen applications were repeated due to the asynchrony in the opening of the stigmata.
Results and discussion
Description of the BBCH scale for E. oleifera
The description of the reproductive phenology of E. oleifera showed four principal stages (emergence of inflorescence, flowering, fruit growth and development and fruit ripening) according to the BBCH scale (Tab. 1).

Principal growth stage 5: emergence of inflorescence
A characteristic of the American oil palm is that it continuously produces inflorescence cycles. Throughout the year of monitoring, the fruits developed at a different rate and at different proportions. This was a long and continuous reproductive pattern that also occurs in other palm species (Barfod et al., 2011). Inflorescence is attached between the petiole base and the stem (stage 500); two fibrous bracts protect the floral structure: the prophyll and the peduncle bract, which are joined in a thick and lignified consistency. Later, the protective bracts that protect the floral structure begin to lose their lignified consistency, and the base of the structure begins to thicken (stage 501). At stage 503, the inflorescence is at nearly 30% of its growth. The floral structure thickens and is also visible in the leaf axil (Fig. 1).

The base and middle sections of the floral structure continue to thicken; the floral structure protrudes along the leaf axil in the opposite direction of the phyllotaxis position of the palm; the floral structure begins to separate from the leaf petiole. The prophyll begins to shred and acquires a fibrous consistency, and both the prophyll and the peduncle bract continue to cover the floral structure, which reaches 50% of its total size (stage 505). Each leaf has a potential inflorescence, which implies that, until the leaf completes its growth, the rapid growth phase of inflorescence may not be started, which is the stage when the axil becomes visible in the leaf.
When the floral structure has reached close to 90% of its final size, two protection bracts are clearly differentiated, which provide adequate conditions for the development of the flower buds; the first bract or prophyll tears and appears shredded; the end section of the second bract, called the peduncle bract, is visible; it is smooth and has no clefts on the surface, which implies that, at this time, the sex of the structure is not yet visible although it has already been differentiated (stage 509).
Principal growth stage 6: flowering
Once growth of the inflorescence axis is finished, the flower buds begin to grow, starting with tearing of the bracts that cover the inflorescence axis; between 10 and 30% of the peduncular bract is torn and the rachillae are visible. The sex of the inflorescence can now be observed, male and/or female, the prophyll continues to cover the floral structure, the rachillae are not visibly separated from one another, and the flower buds are not yet clearly visible (stage 601).
At stage 602, the peduncle bract opens further and the tear appears between 30 and 60%; the prophyll continues to cover the inflorescence. Inside, the separation between the rachillae and the floral bract is visible, as are the flower buds in the axil of the floral bract (Fig. 2). Afterwards, the structure of the peduncle bract becomes fibrous and very strong and continues to cover the inflorescence; cracks appear on the bract, but the rachillae are not visible because the peduncle bract is very thick. The flower buds are observed to protrude and leave the brown tepals uncovered. The floral bract breaks due to the growth of the flower bud. The rachillae are very far apart (stage 603).

At stage 607, the prophyll practically disappears and the peduncle bract is broken but remains firmly attached to the inflorescence, fully covering it. The bract must be manually removed in order to clearly observe the opening of the flower bud, exposing the stigma lobes, which are coated with a mucillagenous substance that facilitates adhesion to pollen (Fig. 2). The flower buds open into an acropetal and, in some cases, in an asynchronous manner, which causes lack of uniformity in the opening of the flower buds. After pollination, the flower secretes a reddish brown substance, possibly indicating the loss of receptivity in the stigma. This receptivity loss is due to the degradation of the pectineal layers of the cells in the stigma groove and the formation of compounds that prevent pollen germination (Hormaza and Romero, 2012). After this, the flower takes on a black color, giving way to senescence (stage 609).
Principal growth stage 7: growth and development of oil palm bunches and fruits in E. oleifera
During the growth and development of the bunches and fruits of the American oil palm, five significant phenological stages were defined with morphological changes between the different phenological stages, which occurred in a continuous and gradual manner. Stage 700 begins between 0 and 2 weeks after pollination. During this stage, the stigmata begin to wilt, taking on a brown to dark brown color and the petals and sepals protect the ovary. This is the early fruit development phase, phase I of rapid fruit growth (Fig. 3).

Between 2 and 4 weeks after pollination, the fruits have reached close to 10% of their final size and are still protected by the sepals and petals; they are a shiny olive green color in the apical and central sections and a parchment color at the base (stage 701). The fruits continue to grow and, between approximately weeks 4 and 8, they reach 30% of their final size (stage 703). Between 9 and 15 weeks after anthesis, the fruits grow to 50% of their size (stage 705). Between 16 and 18 weeks after anthesis, the fruits reach the end of growth phase I and are 90% of their final size. The epidermis color changes slightly, losing the olive green shade and turning yellow (stage 709).
Principal growth stage 8: fruit ripening
Once the fruits have reached their maximum size, the ripening process begins, defined as the physiological, biochemical, anatomic and, in some cases, morphological transformations that lead to the ripe stage, when the fruit is harvested or falls from the tree and decomposes (Gil-Albert, 1995). At stage 800, the fruits are at the end of growth phase II and at the beginning of ripening (approximately 18 WFP), when the color of the fruit epidermis begins to change to the typical ripe color, from olive green to lemon yellow (Fig. 4). Afterwards, a greater range of colors is seen with various shades of green, yellow and pale orange on the fruit epicarp (stage 803).

At stage 805, the entire bunch is pale orange and the endosperm is completely developed; at stage 807, the fruits are very close to falling off or there are between 1 and 10 fruits per bunch already detached. The fruits have grown to their maximum size in weight, thickness and length; the epidermal color is bright orange and the surface of the fruit is smooth and shiny. Afterwards, the over-ripening stage begins, when cracks are displayed in the apical section of the fruits, abscission from the rachis begins, and, consequently, all of the fruits begin to fall off from the bunch; saprophyte fungi begin to grow on the fruits, giving them a dark gray color (stage 809).
Duration of the phenological stages in days
Duration of leaf opening
The average number of days in which the growth of new leaves was observed was 20.1±2.8 for E. oleifera (Tab. 2) and the average rate of new leaf shoots was 1.4 leaves/month. In contrast, for E. guineensis and the inter-specific hybrid, the duration of stage 159 was 12.4-14.3 and 14.4 d, respectively, with approximately 2.0-2.5 new leaf shoots emerging per month.

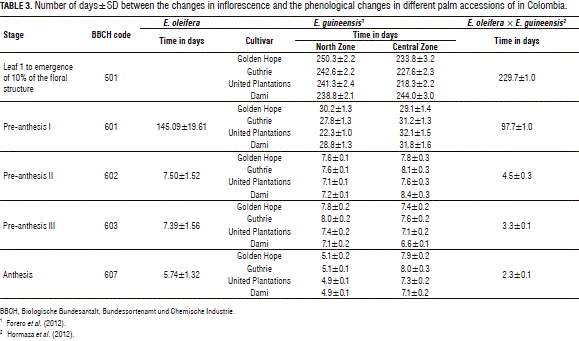
Duration of inflorescence stages
Inflorescence involves a series of changes during development. Table 3 shows the duration in days of the phenological stages of emergence and flowering for oil palm. It was observed that, in E. oleifera, it takes 166 d from when the undifferentiated structure is visible to the start of anthesis, where the period of rapid growth of inflorescence lasts 145 d and flowering lasts 21 d. According to studies by Forero et al. (2012), the hybrid has intermediate characteristics at stage 601 of inflorescence development, as compared to their parents, though there are similarities at stages 602 and 603 between E. oleifera and E. guineensis.
Conclusions
The BBCH scale provides a detailed description of the growth and development events of the various oil palm organs; this scale can be used to facilitate understanding of the various developmental stages and, thereby, widely disseminate the findings. The phenological analysis of the reproductive stages of the American oil palm and their coding using the BBCH phenological scale make it possible to standardize the language of the different disciplines involved in growing oil palm.
The rate of leaf emission for the E. oleifera oil palm is 1.43 leaves per month and a new leaf emerges approximately every 20.1 d, which implies that 18 leaves are produced per year.
The period from when the undifferentiated structure is visible (stage 501) to the start of anthesis lasts 166 d, where the inflorescence rapid growth period lasts 145 d and flowering lasts 21 d.
Acknowledgements
The authors wish to thank the Unipalma S.A. plantation for allowing us to use its facilities to carry out this study. This work was funded by the Oil Palm Development Fund (FFP) managed by Fedepalma.
Literature cited
Barfod, A.S., M. Hagen, and F. Borchsenius. 2011. Twenty-five years of progress in understanding pollination mechanisms in palms (Arecaceae). Ann. Bot. 108, 1503-1516. Doi: 10.1093/aob/mcr192 [ Links ]
Bleiholder, H., H. Kirfel, P. Langelüddeke, and R. Stauss. 1991. Codificação unificada dos estádios fenológicos de culturas e ervas daninhas. Pesq. Agropec. Bras. 26, 1423-1429. [ Links ]
Combres, J.-C., B. Pallas, L. Rouan, I. Mialet-Serra, J.P. Caliman, S. Braconnier, J.C. Soulié, and M. Dingkuhn. 2013. Simulation of inflorescence dynamics in oil palm and estimation of environment-sensitive phenological phases: a model based analysis. Funct. Plant Biol. 40, 263-279. Doi: 10.1071/FP12133 [ Links ]
De Andrade Miranda, I.P. 2001. Frutos de palmeiras da Amazônia. Presidência da República, Ministério da Ciência e Tecnologia, Instituto Nacional de Pesquisas da Amazônia (INPA), Manaus, Brazil. pp. 65-68. [ Links ]
Dransfield, J., N.W. Uhl, C.B. Asmussen, W.J. Baker, M.M. Harley, and C.E. Lewis. 2005. An outline of a new phylogenetic classification of the palm family, Arecaceae. Kew Bull. 60, 559-569. [ Links ]
Forero, D.C., P. Hormaza, and H.M. Romero. 2012. Phenological growth stages of African oil palm (Elaeis guineensis). Ann. Appl. Biol. 160, 56-65. Doi: 10.1111/j.1744-7348.2011.00520.x [ Links ]
Gil-Albert L., F. 1995. Tratado de arboricultura frutal. Vol. I: Morfología y fisiología del árbol frutal. 3th ed. Ministerio de Agricultura, Pesca y Alimentación; Ediciones Mundi-Prensa, Madrid. [ Links ]
Hack, H., H. Bleiholder, L. Buhr, U. Meier, U. Schnock-Fricke, E. Weber, and A. Witzenberger. 1992. Einheitliche codierung der phänologischen entwicklungsstadien mono-und dikotyler pflanzen-erweiterte BBCH-Skala, allgemein. Nachrichtenbl. Deut. Pflanzenschutzd. 44, 265-270. [ Links ]
Hess, M., G. Barralis, H. Bleiholde, L. Buhr, TH. Eggers, H. Hack, and R. Stauss. 1997. Use of the extended BBCH scale-general for the descriptions of the growth stages of mono-and dicotyledonous weed species. Weed Res. 37, 433-441. Doi: 10.1046/j.1365-3180.1997.d01-70.x [ Links ]
Hormaza, P., E. Mesa-Fuquen, and H.M. Romero. 2012. Phenology of the oil palm interspecific hybrid Elaeis oleifera x Elaeis guineensis. Sci. Agric. 69, 275-280. [ Links ]
Hormaza, P. and H.M. Romero. 2012. Escala BBCH para la descripción del desarrollo reproductivo del híbrido OxG. pp. 105-120. In: Romero, H.M. (ed.). Generalidades sobre la morfología y fenología de la palma de aceite. Cenipalma, Bogota. [ Links ]
Ideam, Instituto de Hidrología, Meteorología y Estudios Ambientales de Colombia. 2012. Balance hídrico climático de las estaciones aeropuerto Vanguardia, Barranca de Upía, Carimagua, Lejanías y Villavicencio. Bogota. [ Links ]
Meunier, J. 1991. Una posible solución genética para el control de la pudrición de cogollo en la Palma aceitera. Híbrido interespecífico Elaeis oleifera × Elaeis guineensis. Palmas 12, 39-42. [ Links ]
Meunier, J. and D. Boutin. 1975. L'Elaeis melanococca et l'hybride Elaeis melanococca × E. guineensis. Oléagineux 30, 5-8. [ Links ]
Rey B., L., I. Ayala, W. Delgado A., and P.J. Rocha S. 2003. Colecta de material genético de la palma americana Nolí Elaeis oleifera (H.B.K.) Cortez en el Trapecio Amazónico. Ceniavances 101, 1-4. [ Links ]
Rey B., L., P.L. Gómez C., I.M. Ayala D., W. Delgado A., and P.J. Rocha S. 2004. Colecciones genéticas de palma de aceite Elaeis guineensis (Jacq.) y Elaeis oleifera (H.B.K.) de Cenipalma: características de importancia para el sector palmicultor. Palmas 25, 39-48. [ Links ]
Rivera M., Y.D., G. Cayón S., and J.E. López M. 2013. Physiological and morphological characterization of american oil palms (Elaeis oleifera HBK Cortes) and their hybrids (Elaeis oleifera × Elaeis guineensis) on the Indupalma plantation. Agron. Colomb. 31, 314-323. [ Links ]
Rocha S., P.J., F. Prada C., L. Rey B., and I.M. Ayala D. 2006. Caracterización bioquímica parcial de la colección de Elaeis oleifera de Cenipalma proveniente de la Amazonía colombiana. Palmas 27, 35-44. [ Links ]
Sánchez Ch., G. 2008. Repetibilidade da produção de cachos, anomalias florais e germinação de pólen de híbridos interespecíficos entre o caiaué e o dendezeiro. MSc thesis. Instituto Nacional de Pesquisas da Amazônas, Universidade Federal do Amazonas, Manaus, Brazil. [ Links ]
Zadoks, J.C., T.T. Chang, and C.F. Konzak. 1974. A decimal code for growth stages of cereals. Weed Res. 14, 412-421. Doi: 10.1111/j.1365-3180.1974.tb01084.x [ Links ]














