Introduction
The efficient control of spider mites has been reported for invertebrate pathogenic fungi (van der Geest et al., 2000). The entomopathogenic fungi Beauveria bassiana (Bals.-Criv.) Vuill., Hirsutella thompsonii Fischer and Metarhizium anisopliae (Metschn.) Sorokin were observed to be promising controls for T. urticae (Jeyarani et al., 2011). Invertebrate pathogenic fungi are efficient control agents against mites and other arthropods and are increasingly involved in pesticide resistance management in combination with sublethal doses of acaricides (Amjad et al., 2012). Moreover, they can be applied in combined strategies, for example, along with the predatory mites Phytoseiulus longipes (Amjad et al., 2012) or Neoseiulus californicus (Oliveira et al., 2013).
The breakdown of the T. urticae colony in our greenhouse as a result of an entomopathogenic fungal outbreak, apparently from the genus Cladosporium, sparked interest in its identification and evaluation of its potential as a biological control agent for this mite. This fungus has been widely studied against T. urticae (Eken and Hayat, 2009). These authors tested 13 strains of C. cladosporioides in a lab bioassay, and the total mortality percentage varied from 50.95 to 74.76% for T. urticae, with a LT50 range between 2.34-3.9 d. These results led the authors to the conclusion that this fungus is a promising candidate for the biological control of T. urticae. Since no studies on Cladosporium spp. versus mites are known to have been conducted in Colombia so far, the aim of this study was to test the pathogenicity of these Colombian strains against T. urticae.
Materials and methods
Studies were conducted in the Laboratory of Entomology, Faculty of Agricultural Sciences, at the Universidad Nacional de Colombia, Bogota. Deceased mites (T. urticae) were collected from bean plants (Phaseolus vulgaris, variety Cerinza) established in a greenhouse (mean temperature 22°C ± 10°C measured using a TFA Indoor Digital Thermo Hygrometer during the entire assay, 80% ± 3% relative humidity and 12:12 h light:darkness). P. vulgaris in the vegetative stage (three weeks after germination) served as host plants for the mite colony; the plants were irrigated every three days. In order to avoid the presence of any infested mites, healthy mites were obtained from the Center of Biosystems of the Universidad Tadeo Lozano in Chia (Cundinamarca, Colombia) and from Flores de Tenjo Ltda. (Tenjo, Cundinamarca, Colombia).
Strain isolation and identification
The fungal strain was isolated from dead mites in the colony in the entomology greenhouse of the Faculty of Agricultural Sciences, where an apparently epizootic event occurred in 2010. The fungus was cultured in Potato Dextrose Agar (PDA) medium and kept it in an incubator at 25°C. This procedure was repeated until monoconidial cultures were obtained.
The fungus was identified by considering its morphological and genetic characteristics. The morphological part was described using a culture grown on a solid PDA medium following the key developed by Bensch et al. (2012). For the molecular identification of the fungus, genomic DNA was isolated as follows: (i) to a 2 ml Eppendorf tube containing 1000 μ! of lysis buffer (50 mM Tris-HCl [pH 8.0], 100 mM NaCl, 1% Sodium Dodecyl Sulfate, SDS) and 425-600 mm sized acid-washed glass beads (Sigma-Aldrich, St. Louis, USA), a small lump of fresh mycelia was added by using a sterile toothpick. After a quick vortex mix, the Eppendorf tube was left at room temperature for 15 min and centrifuged at 10,000 g for 10 min at 4oC. (ii) The supernatant was transferred to a 1.5 ml Eppendorf tube, and an equal volume of phenol-chloroform-isoamyl alcohol (25:24:1) was added; the sample was vortexed and centrifuged at 10,000 g for 10 min at 4oC; this step was repeated using an equal volume of chloroform-isoamyl alcohol (24:1). (iii) After transferring the supernatant to a new 1.5 ml Eppendorf tube, an equal volume of isopropyl alcohol was added and briefly mixed by inversion and stored at -20oC for at least for 1 h. The sample was centrifuged at 10,000 g for 10 min and the supernatant was discarded. (iv) The resultant DNA pellet was washed in 300 μl of 70% ethanol two times, and the supernatant was discarded each time after the pellet was centrifuged at 10,000 g for 5 min at 4oC. The DNA pellet was air dried and dissolved in 50 μl of Tris-EDTA and incubated with RNAase (10 μg/ml) at 37oC for 30 min. Different dilutions (1:1 and 1:5) of the purified DNA were used in 50 μl of the PCR mixture. The internal transcribed spacer region (ITS) from the rRNA was amplified using the ITS 1-4 primers, following the conditions described by Avellaneda-Torres et al. (2014). The obtained sequences were analyzed using the software Genius, version PRO 5.1.5., and compared with sequences in the NCBI GenBank using Basic Local Alignment Serach Tool (BLAST) (Altschul et al., 1990; Benson et al., 2005).
Preparation of inoculum
The inoculum was produced using pure fungus cultures grown on PDA in sealed Petri dishes and incubated at 25°C. The spore solutions were prepared by adding 10 ml of distilled and sterile water and 0.1% Tween 20 on the pure culture and scraping the medium with a sterilized needle. This solution was passed through a cloth, and the number of conidia per ml was counted in a Neubauer chamber under a light microscope.
Five different concentrations were prepared from a 2x108 conidia ml-1 stock suspension, which were diluted in water at 2x104, 2x105, 2x106, and 2x107 conidia ml-1 for immediate application in the bioassay.
Bioassays under controlled conditions
Humid cotton was placed in a Petri dish and was covered with an upside-down rose leaf (variety Charlotte grafted on the Natal Briar rootstock). Then, 20 mites were placed on each leaf (experimental unit). Every experimental unit was repeated five times. The fungus was applied using a mobile sprayer located in the weed greenhouse of the Faculty of Agricultural Sciences. First, the sprayer was calibrated as follows: 1 kg/cm2 pressure, 0.48 m s-1 speed and 0.57 l min-1 as volume. After spraying the Petri dishes, they were placed in an incubator (25°C, 80 % RH, 12:12 h light:darkness) for 10 d.
The seven treatments were conducted as follows: T1: negative control (water and Tween 20 0.05 %), T2: positive control applying the commercial product Bassianil®, with Beauveria bassiana as the active ingredient (Perkins Ltd., Palmira, Colombia) at a concentration of 1x106 conidia ml-1. Based on previous experiments and the study by Saranya et al. (2010), six conidia concentrations were selected; the treatments (fungus concentrations = conidia ml-1) were T3: 2x104, T4: 2x105, T5: 2x106, T6: 2x107, T7: 2x108. The entire experiment was repeated three times over time. The mites were counted for 10 d, starting on the first day after application, and the accumulated percentage of dead individuals was calculated.
Statistical analysis
Abbott's equation (1925) was used in order to correct the mortality data. The effects of treatments on mite mortality were compared using the general linear model procedure (PROC GLM, SAS Institute 2013); significant differences between the treatments were identified with Tukey's test HSD (P<0.05). The treatments were arranged using a completely randomized design.
Results and discussion
Strain identification
The isolated strain taken from the PDA cultured fungus presented a brownish dark color surface, a black color reverse; the plain surface was haired and occasionally turned to dust. At the microscopic level, we identified hyphae septate, branched and dark (data not shown). These results are corroborated by the observations of Bensch et al. (2012), who described the surface ornamentation of conidia in the C. cladosporioides complex as quite variable, ranging from smooth, or almost so, to irregularly verruculose-rugose, verrucose or rough-walled in some species.
The extracted DNA was visualized with agarose gel electro-phoresis (Fig. 1A), and the size of the successfully amplified sequence, using ITS1-4 primers, was about 513 base pairs (Fig. 1B). After the sequencing and DNA data comparison using BLAST search, highly homologous sequences were obtained with GeneBank samples in NCBI. This indicated that the microorganism corresponds to the fungus of the division Ascomycota, subphylum Pezizomycotina, order Capnodiales, family Cladosporiaceae, genus Cladosporium, and species Cladosporioides. Its coverage was 100%, the Evalue was 0.0, and the identity was 100% (accession number LN808903.1).
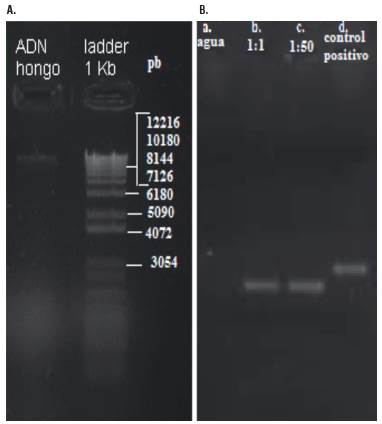
FIGURE 1 Agarose gel analysis of DNA extracted from a fungus strain (A) and Gel electrophoresis of amplicons products with the primers ITS1-4 of the rRNA gene on purified genomic DNA (B). Amplicons (513 bp) from 1:1 (b) and 1:50 (c) ADN dilutions; NC: negative control; no DNA (a). PC: positive control (d). MM: 1 Kb DNA molecular weight marker; DNA lambda (Thermo).
Cladosporium spp. These species are ubiquitous, saprobic, dematiaceous fungi and have been associated with human and animal opportunistic infections (Sandoval-Denis et al., 2015). Their entomopathogenic potential has been described since the early eighties, when their pathogenicity to insects was recorded (Samways and Grech, 1986). Nowadays, Cladosporium spp. are considered effective biological control agents against insects and mites at the laboratory level (Sosa-Gomez et al., 2010; Bahar et al., 2011). Nevertheless, among the arthropod-associated ascomycete fungi, uncertainties remain about the extent to which species in ubiquitous genera, such as Cladosporium, Aspergillus, Penicillium, and Fusarium, are pathogenic to arthropods or might be opportunistic secondary pathogens or saprotrophics that colonize available cadavers (Samways and Grech, 1986). Thus, histopathologic studies are necessary in order to prove whether this organism is the causal agent of an arthropod's death.
Entomopathogenic activity of Cladosporium cladosporioides
It was observed that the dead mites presented a brown or dark green color resulting from the attack of the fungus. When the fungus entered an advanced stage of development, the color turned black. Table 1 shows significant differences between the different conidia concentrations for all days (Tukey's test, P<0.05). The concentration 2x104 conidia ml-1 achieved moderate control of the mites, 62%. The highest mortality (81.7%) was obtained with 2x108 conidia ml-1, suggesting that this fungus might be a promising candidate for the control of mites and should be evaluated under field conditions.
These results surpassed the efficacy of the commercial B. bassiana strain (72.3 %). The negative control (only water + Tween) showed a mortality of 20.7%, which was similar to the results obtained by Chandler et al. (2005). They hypothesized that a high mortality in the control might be associated with an interaction between fungus and non-fungus mortality factors in a treatment bioassay; for instance, lesions caused by transferring the mites to the experiment unit, a short life cycle and a non-uniform age of the mites.
Table 1 gives evidence for the progressive activity of the fungus: the concentration 2x108 ml-1 caused a 50% mortality after 5 d, 2x107 ml-1 after six, 2x106 ml-1 after seven and the treatments with the lowest concentration (2x105 ml-1 and 2x104 ml-1) caused 50% mortality after 8 d. A similar mortality rate was reported by Gatarayiha (2009), who observed that B. bassiana caused a lethal time, LT50 between 5.5 and 8.9 d. Unfortunately, it is impossible to compare our results with other reports because of different experimental designs of bioassays with different fungi and arthropods (Chandler et al., 2005; Bahar et al., 2011). However, other criteria for fungi selection should be considered, such as a high conidia production and high final mortality rates (Chandler et al., 2005; Bahar et al., 2011). Lower conidia concentrations (2x104 and 2x105 conidia ml-1) are able to reduce mite populations. However, higher concentrations (2x106, 2x107 and 2x108 conidia ml-1) definitely shorten the lethal period, which was corroborated by our experiments.
A mean lethal concentration LC50 of 1.95x106 conidia ml-1 was found. This concentration was low compared with the LC50 of other fungi, such as B. bassiana (8.65x107 conidia ml-1), Hirsutella thompsoni (1.06x108 conidia ml1), and M. anisopliae (2.43x107 conidia ml1), on T. urticae (Chandler et al., 2005). Our results indicate that the fungus showed a good control potential towards mites. It is possible that the fungus also acted through toxic bioactive metabolites, increasing the effect with a typical mycoparasitic infection (Bensaci et al., 2015). Samways and Grech (1986) stated that Cladosporium oxysporum is capable of inducing major decreases in the aphids Toxptera cictriculus and T. erytreae. Moreover, Shaker et al. (2019) provided support for this hypothesis through the isolation of two major compounds from C. cladosporioides, which were identified as 3-phenyl propanoic acid and 3-(4p־hydroxy-6-pyranonyl)-5-isopropylpyrrolidin-2-one. These compounds caused 100% mortality for the aphid Aphis gossypii.
The toxic properties and degree of their toxicity vary depending on the administration, chemical structure and concentration (Piecková and Jesenská, 1999). Since Clado-sporium spp. form tiny conidia in high amounts (Piecková and Jesenská, 1999), it can be concluded that this fungus is an efficient candidate for the biological control of arthropods. Singh et al. (2016) described the potential of toxins produced by Cladosporium velox for exhibiting activities of Alpha glucosidase inhibitors, affecting the digestion and moulting of Spodoptera litura.
TABLE 1 Percentage of corrected mortality (Abbott, 1925) of Tetranychus urticae after applications of Cladosporium cladosporioides over 10 days.
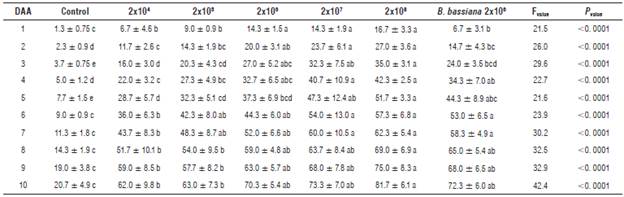
DAA: Days after application. *Means followed by the same letter are not significantly different (Tukey HSD, P≤0.05).
It was observed that the LT50 of the mites oscillated between seven and nine days when treated with Cladosporium and B. bassiana, which was significantly different from the control. The lowest LC50 (1.11x106 conidia ml-1) occurred on d 10. The rate of mortality decreased during the entire observation period, whereas the standard deviation increased. Despite the fact that the differences between the commercial strains and our isolate were not statistically significant because of the high mortality in the control. The results suggest that our tested strain may be a promising candidate for biological control. Another experiment under controlled conditions and with higher conidia concentrations verified under semi-controlled and field conditions may give further evidence of the efficacy of this strain as a control agent of mites. Moreover, further studies on secondary metabolites of this strain and their toxicity to tetranychid mites might create options for biological control strategies against this pest. For example, Shaker et al. (2019) successfully extracted ethyl acetate from C. cladosporioides, leading to 100% control of aphids.















