Introduction
The weed Digitaria insularis, known as sourgrass, is native to tropical and subtropical areas of America. It can be found in pasture lands, coffee plantations, orchards, crop fields, roadsides, and in abandoned fields. It is referred to as rhizomatous and produces seeds throughout the summer, growing in clumps. These traits allow them to aggressively compete against cultivated crops (Moreira and Bragança, 2011; Lorenzi, 2014). There are four known D. insularis herbicide-resistant biotypes, all found in South America, from which three are glyphosate-resistant and one is resistant to haloxyfop and fenoxaprop. Weed resistance is a problem across the globe, and Brazil is ranked fifth with 50 registered resistance cases to EPSPs, ALS, ACCase, PSII, PSI, PPO, and auxins (Heap, 2020).
In addition to resistance cases, as a perennial grass D. insularis is hard to control, especially during flowering development (Zobiole et al., 2016; Canedo et al., 2019). A study by Gazziero et al. (2019) shows that a cohabitation of six D. insularis plants rrf2 among soybean crops reduces its yield by 40%. Thus, appropriate managing practices including different control methods are shown to be important to have an efficient outcome.
The association of different herbicides, rotation of sites of actions, preventive methods, among other practices are essential to manage and prevent new weed biotypes resistant to herbicides (Green, 2018; Heap and Duke, 2018; Neve et al, 2018; Frisvold et al., 2020). However, in the literature, there are reports of the antagonist effect of 2,4-D on the action of ACCase inhibitor graminicides, due to the translocation reduction and increase in the metabolism of herbicides from the ariloxifenoxipropionics group (Trezzi et al., 2007). Thus, there is a need for studies on the different mechanisms of control of D. insularis.
Pre-emergent herbicides used before sowing are emphasized as an important tool to weed management, especially in crop fields (Byker et al, 2013; Belfry et al, 2015). In this sense, ALS inhibitors are highlighted for weed control as pre-emergence herbicides used in soybean fields (Braz et al, 2017; Underwood et al, 2017). Imidazolinone herbicides are part of the group of the acetolactate synthase (ALS) (also called acetohydroxyacid synthase [AHAS]) inhibiting herbicides which hamper the synthesis of the branched-chain amino acids valine, leucine, and isoleucine. After their absorption, the herbicides are translocated to meristems and apexes that are actively growing areas, inhibiting the growth of susceptible plants. Chlorosis happens to sensitive plants followed by death in 7 to 14 d after treatment (Oliveira Júnior, 2011; Shaner and O'Connor, 2017).
These herbicides are applied at pre- and post-emergence, controlling a range of monocotyledons and dicotyledons in cereal and soybean crops and nonagricultural areas (Oliveira Júnior, 2011; Rodrigues and Almeida, 2018). Piasecki and Rizzardi (2016) report that imazapic/imazapyr (premix formulation) is efficient for controlling volunteer corn at soybean pre-emergence. Likewise, studies by Melo et al. (2017) show the efficiency of imazapic/imazapyr on the control of D. insularis.
It is believed that the imazapic/imazapyr is effective in controlling D. insularis when applied at pre-sowing burn-down. Thus, this study aimed to evaluate the efficiency of imazapic/imazapyr (premix formulation) and other herbicides in mixtures to control D. insularis at burndown before soybean sowing.
Materials and methods
Conditions and experimental design
This field research was conducted in Umuarama, State of Parana (PR), Brazil (23°50'25.23" S, 53°13'45.70" W) during the 2018/19 crop season. The soil was classified as sandy (11% clay, 6.5% silt, and 82.5% sand) with the following chemical properties on the 0-20 cm layer: pH (CaCl2) of 4.4, 1.33% organic matter, and 5.78 cmolc dm-3 cation exchange capacity. Under the Koppen classification, the climate is classified as Cfa, and Figure 1 shows the weather conditions during the time of the research.
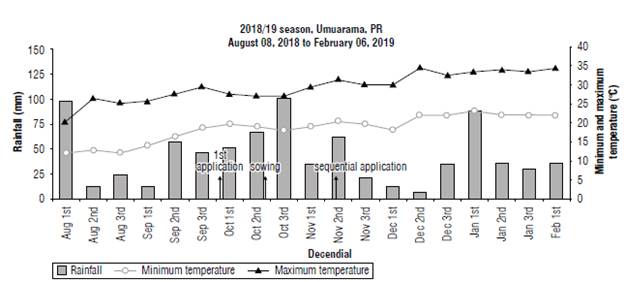
FIGURE 1 Representation of rainfall and minimum and maximum temperature for the study site. 2018/19 season, Umuarama, PR, Brazil.
The trial site was infested by D. insularis, identified as indicative of glyphosate resistance. Before application, the plants were at the flowering stage with 3.5 clumps m-2. The area was previously cultivated with maize. A no-till sowing system was used for soybean, using 0.45 cm row spacing. The cultivar used was Monsoy® 6410 IPRO (Monsanto Co. do Brasil, São Paulo, SP, Brazil). The experiment was conducted in a randomized block experimental design, with four replications and 5x3 m plots. For the useful area, the four central lines were used, discarding the first and last meter of the plot. The treatments are described in Table 1. The commercial products used in the trial are presented in Supplementary material 1.
TABLE 1 Treatments consisting of herbicide mixtures for the control of D. insularis. 2018/19 season, Umuarama, PR, Brazil.

1 Sequential application in all herbicide treatments of haloxyfop (66 g acid equivalent (a.e.) ha-1) + glyphosate (720 g a.e. ha-1), at post-emergence (V3) of soybean.
2Rates at g a.e. ha-1 for glyphosate, haloxyfop, 2,4-D and triclopyr. Rates at g active ingredient (a.i.) ha-1 for other herbicides. Addition of adjuvants: 3Lanzar® 0.5% v/v; Joint® 0.5% v/v; 5Dash® HC 0.5% v/v.
The first application was performed on October 5th, 2018. The soybean was sown on October 18th, 2018, and the sequential application was carried out on November 16th, 2018 when soybean plants were at the V3 growth stage (Fehr et al., 1971). One hundred mm of rainfall was reported in the area between the first application and seeding.
Both applications were performed using a CO2 backpack sprayer at 3.6 km/h with AIXR 110.015 spray nozzles pressured at 2.5 kgf cm-2 with a volume of 150 L ha-1. The weather conditions were a temperature of 23.1oC, 62.3% relative humidity, and wind speed of 6.8 km/h. The sequential application had the following conditions: temperature of 23.1oC, 60.1% relative humidity, and wind speed of 2.2 km/h.
Evaluations
Weed control was visually evaluated at 7, 14, 21, and 35 d after application (DAA) and on the 14th and 28th d after the sequential application. Soybean crop injury was evaluated at 7, 14, 21, 28, and 35 d after sowing (DAS). Percentage grades from 0% (absence of symptoms) to 100% (death of the plant) were given, based on the apparent symptoms related to the plant growth stage (Velini et al., 1995).
By the time of harvest, at the R8 stage (full maturation) of soybean plants (Fehr et al., 1971), the yield was calculated using only the plot's two central lines of 4 m long each. The grain moisture was corrected to 13% and the results converted to kg ha-1.
Data analysis
An analysis of variance and the F-test (P≤0.05) were performed as described by Pimentel-Gomes and Garcia (2002). The means of treatments were grouped together using the Scott-Knott (1974) test (P≤0.05). For this purpose, the software Sisvar 5.6 was used (Ferreira, 2011).
Results
There was little or no control observed in the first application at 7 DAA (all below 35%); however, at 14 DAA all treatments associated with auxinic herbicides and saflufenacil showed inferior control compared to the others. At 21 DAA, the best results were observed for treatments with the application of ACCase inhibitors (haloxyfop and clethodim). Control superior to 80% was reported at 28 and 35 DAA by all herbicide combinations. It is important to highlight that at 35 DAA only the treatments involving glufosinate and 2,4-D showed lower performance (Tab. 2).
TABLE 2 Control (%) of D. insularis under application of herbicides mixtures. 2018/19 season, Umuarama, PR, Brazil.
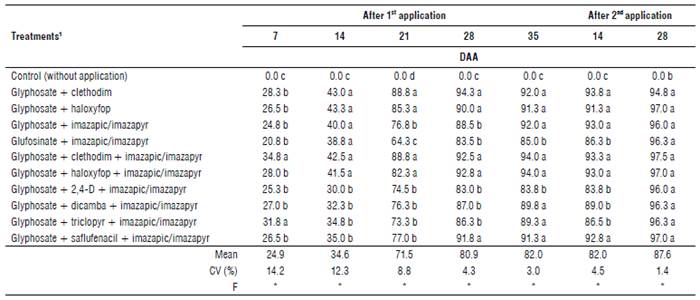
DAA: days after application; CV: coefficient of variation.
1Sequential application in all herbicide treatments of haloxyfop (66 g acid equivalent (a.e.) ha-1) + glyphosate (720 g a.e. ha-1), at post-emergence (V3) of soybean.
*Means followed by the same letter in the column do not differ from each other by the Scott-Knott (1974) test (P<0.05).
After the sequential application of glyphosate + haloxyfop at 14 DAA, weed control of at least 83.3% was observed. Besides that, the treatments involving auxinic herbicides and the ones involving glufosinate were considered inferior as grass regrowth was frequently observed (≥89%). However, at 28 DAA efficient D. insularis control was observed by all the tested combinations of herbicides, computing a rate control of at least 94.8%. The application of glufosinate + imazapic/imazapyr is highlighted for its control levels of 85% at 35 DAA and 96.3% after the sequential application at 28 DAA (Tab. 2), indicating the importance of adding sequential applications to weed management practices.
Lower crop injury by all evaluations was also observed. In general, symptoms were characterized by small chloroses and yellowing of leaves. The application of glyphosate with clethodim or haloxyfop did not cause symptoms (Tab. 3).
TABLE 3 Crop injury (%) and yield (kg ha-1) of soybean plants under application of herbicide mixtures, for the control of D. insularis. 2018/19 season, Umuarama, PR, Brazil.
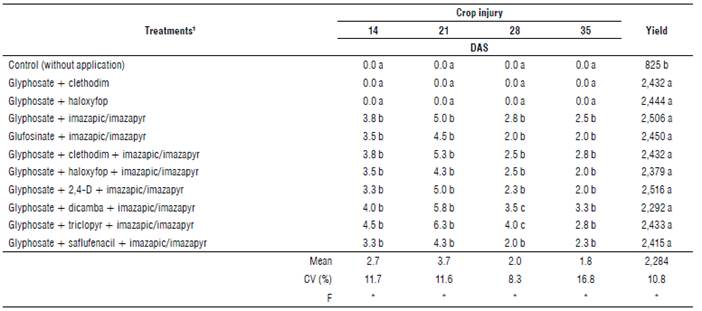
DAS: days after sowing; CV: coefficient of variation.
1 Sequential application in all herbicide treatments of haloxyfop (66 g acid equivalent (a.e.) ha-1) + glyphosate (720 g a.e. ha-1), at post-emergence (V3) of soybean.
*Means followed by the same letter in the column do not differ from each other by the Scott-Knott (1974) test (P≤0.05).
The treatment glyphosate + triclopyr + imazapic/imazapyr showed the greatest values of injury, with 6.3% at 21 DAS, followed by the treatment glyphosate + dicamba + imazapic/imazapyr (5.8%). However, from 21 DAS the symptoms of all treatments started to decrease, and all percentages were considered acceptable and not harmful to the crop. Despite the symptoms, there was no change in yield (Tab. 3).
Discussion
Less than 30 d passed between the application of imazapic/ imazapyr and the sowing of soybean, in accordance with the label recommendations (Rodrigues and Almeida, 2018). Additionally, about 100 mm of precipitation were observed in this period, also a necessary volume according to the label (Rodrigues and Almeida, 2018). This precipitation combined with the sandy texture of the soil may have favored the leaching of the product, which favored the selectivity for soybean.
Every herbicide mixture performed in this study showed efficient performance on D. insularis control with minimum injury while maintaining soybean yield. The use of imazapic/imazapyr and other herbicides in mixtures was potentially selective at burndown before soybean sowing. Melo et al. (2017) observed 100% of D. insularis control at 35 DAA using imazapic/imazapyr (52.5/17.5 g a.i. ha-1) at postemergence. Francischini et al. (2012) also noted imazapic/imazapyr efficiency controlling D. insularis along with other weeds that is similar to that verified in the present study.
Besides successfully controlling D. insularis in different mixtures as shown in this study, imazapic/imazapyr is also efficient in the control of volunteer corn (Piasecki and Rizzardi, 2016) and is selective to soybean. Additionally, post-application of imazapic/imazapyr was also reported as efficient in controlling different grasses and broadleaved weeds, such as eudicotyledons Bidenspilosa and Raphanus raphanistrus (Santos et al., 2012) and monocotyledons Hymenachne amplexicaulis (Silva et al., 2012), Oryza sativa, Echinochloa crus-galli and Cyperus esculentus (Helgueira et al., 2018).
Regarding effectiveness, the glyphosate + imazapic/ima-zapyr mixture is highlighted when combined with auxinic herbicides, especially triclopyr and dicamba. This mixture is equivalent to that of glyphosate + imazapic/imazapyr with ACCase inhibitors (haloxyfop or clethodim). The application of these mixtures shows a broad control spectrum considering the effectiveness of auxins for the control of eudicotyledons combined with the control of D. insularis verified in this study.
High levels of infestation of weeds such as D. insularis and other grasses require ACCase inhibitor herbicides such as haloxyfop and clethodim. However, applying synthetic auxins such as 2,4-D and dicamba is not always an option because of the antagonism that can be created between these herbicides and ACCase inhibitors (Trezzi et al., 2007; Pereira et al., 2018; Gomes et al., 2020). The antagonism between these mixtures is possibly explained by the reduction of translocation of these herbicides to their action sites, compared to the cases when herbicides are pulverized alone (Scherder et al., 2005). Thus, eudicotyledon control is hampered and glyphosate or some other herbicide is used as an option for chemical management. There are few studies in the literature that provide information on antagonism, additive effect, or synergism of imazapic/imazapyr in a mixture with synthetic auxins, glufosinate, or saflufenacil, so the present work is unprecedented.
The mixture of saflufenacil, glyphosate, and imazapic/ima-zapyr was effective on the control of D. insularis (>91.3%) at 28 and 35 DAA. In general, saflufenacil does not show high control of Digitaria spp. (Soltani et al., 2014). However, in this study, it was identified as a tool for D. insularis control when used in mixtures with glyphosate and imazapic/ imazapyr since mixtures like this have a broad spectrum of action. Dalazen et al. (2015) observe synergism for saflufenacil + glyphosate, and Datta et al. (2013) observe it for imazapic + saflufenacil. It is noteworthy that these treatments had slower action, compared to treatments with ACCase inhibitors (haloxyfop and clethodim). Bianchi et al. (2020) observe a synergistic effect of clethodim + glyphosate for the control of D. insularis at 21 DAA. In the present study, treatments without ACCase inhibitors achieved greater levels of control after 28 DAA.
The results of this study showed that glufosinate + imazapic/imazapyr application as a burndown technique prior to sowing, and when applied along with glyphosate (only at soybean post-emergence) associated with haloxyfop they were efficient for controlling D. insularis at a 96.3%. Studies by Everman et al. (2007), Melo et al. (2012), Gemelli et al. (2013), and Silva et al. (2017) also emphasize the use of glufosinate in the control of D. insularis in different chemical weed management programs.
In this sense, it is important to emphasize the four biotype resistance cases of D. insularis in the world. Three of these biotypes are resistant to glyphosate and one to haloxyfop and fenoxaprop (Heap, 2020). Therefore, other mechanisms of action are useful to manage this grass; thus, imazapic/ imazapyr (ALS inhibitors) and glufosinate (GS inhibitor) can be used to control D. insularis offering soybean selectivity as verified in this study.
Conclusion
The application of imazapic/imazapyr in mixtures with other herbicides was effective in controlling glyphosate-resistant D. insularis in burndown before soybean sowing and with sequential application of haloxyfop + glyphosate at the V3 stage of soybean. This chemical management was also selective for soybean.