Introduction
The umbu (Spondias tuberosa Arruda) belongs to the Anacardiaceae family and is a native, endemic species of the Brazilian semiarid region (Lima et al., 2018). This species has a wide geographical distribution and shows morphologic characteristics that enable its adaptability to various edaphoclimatic conditions. For example, the plant can promote leaf abscission, has roots with structures called lignotubers with the function of storing water and nutrients (Mitchell & Daly, 2015), and efficient control of leaf transpiration (Lima Filho & Aidar, 2016).
The mechanisms of adaptation to water scarcity and irregular rain distribution make it possible to cultivate umbu in several regions of Brazil, promoting food product diversification and increasing food security. Umbu has environmental and socio-economic importance for the producing regions. Its fruits are commercialized as an extract; however, there is great capacity for expansion for commercial growth to meet the demand of the fresh market and the market of processed products for its pulp and peel (Ribeiro et al, 2019).
The fruits are oval or oblong, and their weight can range from 5 to 100 g, according to the genotype (Oliveira et al., 2014). The fruit pulp or edible fraction has bioactive compounds, namely phenolics, carotenoids, and vitamin C that confer its antioxidant potential (Ribeiro et al., 2019). Besides fresh consumption, the fruit can be used in the processing industry to produce juices, beverages, and canned products (Narain et al., 1992; Lima et al, 2018).
Despite the importance of umbu for small and medium farmer economy, its market still faces some difficulties. Some of the problems that this crop must overcome are the lack of efficient methods to monitor fruit quality that can be used to determine the ideal harvest maturity and methodologies to quantify its quality for fresh consumption and for the industry (Marques & Freitas, 2020). Therefore, there is a great need to develop accurate and low-cost methods for assisting the conservation and postharvest quality of fresh fruits, especially those destined to be consumed in natura.
Different postharvest treatments have been studied to control rot and improve fruit shelf life, including the use of modified atmospheric systems (Joshi et al., 2019). The modification of the atmosphere can be easily achieved using polymeric films, such as plastic bags of high or low-density polyethylene (Vieira et al., 2009). Another alternative is the use of edible coatings that have recently gained prominence and are being widely used for fruit preservation (García et al., 2020). These coatings are environmentally friendly and serve as carriers of food additives that facilitate safety and nutrition and enhance the sensory aspects of fruits (Khan et al, 2019).
Edible coatings can be prepared from carbohydrates, proteins, and lipids, individually or in combination (Muley & Singhal, 2020). Some reports suggest that the use of cassava (yuca) starch for fruit coatings can improve their physical and chemical properties and provide greater protection against microbial agents (Aider, 2010; Pimentel et al., 2018). The fruit coating based on cassava starch is of low cost compared to commercial waxes. It also preserves good flavor, is compatible with various processes, and is shiny and transparent (Assis & Britto, 2014; Sousa et al, 2019).
The objective of this research was to evaluate the posthar-vest conservation of umbu (Spondias tuberosa Arruda) fruits coated with a biofilm produced from cassava starch at room temperature. Since cassava starch is a cheap and easily found product, our findings can guide the development of a new methodology to extend fruit postharvest life.
Materials and methods
Experimental design
The experiment was carried out in a completely randomized design in a 2 x 5 factorial arrangement with six replicates. Fruits with and without cassava starch coating (1%) were evaluated during five different storage periods (0, 3, 6, 9, and 12 d of storage).
Plant material
The umbu fruits were harvested on an experimental farm at the State University of Montes Claros - UNIMONTES, Janaúba, Minas Gerais, Brazil, (15°49'44" S and 43°16'09" W at an altitude of 544 m a.s.l.). Sixty fruits were used in the experiment. Twelve fruits (six per treatment, with and without cassava starch coating) were evaluated for each storage period (0, 3, 6, 9, and 12 d). These fruits were harvested at the breaker maturation stage and transported to the UNIMONTES Post-harvest Laboratory where they were graded for uniformity in size, shape, and green color. Fruits free from defects and blemishes were selected. The fruits had an average longitudinal length of 4.5 cm and equatorial diameter of 3.8 cm. The fruits were then washed, immersed in a sodium hypochlorite solution (100 mg L-1 of active chlorine), and dried at room temperature (26.7°C).
Application of cassava starch-based coating
The coating was accomplished by immersing the fruits for 3 min in the biofilm of cassava starch (BT). The solution was made with distilled water + 1% cassava starch. A control treatment (CT) was performed by immersing the umbu fruits in distilled water for 3 min. After immersion, the fruits were placed in a nylon mesh container for draining the biofilm and excess water and drying. The fruits were then stored at room temperature (26.7 ± 3°C) and 75 ± 5% humidity and evaluated at 0, 3, 6, 9, and 12 d after harvest.
Weight loss
Fresh weight loss (WL) was measured using a digital scale with a precision of 0.01 g during the storage periods of 0, 3, 6, 9 and 12 d. The results were expressed as a percentage.
Firmness
Pulp firmness (FIR) was determined using a Brookfield CT3 digital texture analyzer (Brookfield Engineering, Middleboro, MA, USA) equipped with a 7.8 mm diameter stainless steel compression plate. Analyses were carried out in the equatorial region of each fruit. The results were expressed in Newton (N).
pH and total titratable acidity
A solution made from the umbu pulp (5 g) and distilled water (25 ml) was well-mixed. The solution was adjusted to a final volume of 50 ml using distilled water and filtered with Whatman 42 filter paper. The pH of the filtrate was then measured with a digital pH meter (model DM20, Digimed, São Paulo, Brazil).
The titratable acidity (TA, %) was determined by titrating the filtrate (10 ml) against 0.1 N NaOH using 1-2 drops of phenolphthalein as an indicator. The results were expressed as g of citric acid per 100 g of pulp (AOAC, 1997).
Total soluble solids and TSS/AT ratio
Total soluble solids (TSS) were quantified in an N1 bench refractometer (Atago, Tokyo, Japan) with an operating range of 0 to 32% and an accuracy of 0.2%. A few drops of umbu juice were used for the analysis. The results were expressed in °Brix. The TSS/TA ratio was calculated as the relationship between the total soluble solids and titratable acidity.
Color indices
Changes in the control and coated umbu fruit color indexes stored at 26.7°C ± 3°C were determined at 0, 3, 6, 9, and 12 d after harvest. The values of luminosity (L*) to red or green tint (a*) and yellow or blue tint (b*) were measured in the ColorFlex 45/0 colorimeter, stdzMode: 45/0, HunterLab universal software system (Hunterlab, Reston, VA, USA). The chromaticity index or chroma (C*) and hue angle or hue (H*) were determined as previously described by Muley and Singhal (2020).
Data analysis
Data were subjected to an analysis of variance. The means for the qualitative data (with and without the biofilm) were compared using the F-test (P<0.05). Quantitative data (storage days) were subjected to a regression analysis using the Sisvar statistical software (Ferreira, 2014). The figures were made using the SigmaPlot version 11.0 (Systat Software, San Jose, CA, USA).
Results and discussion
The coating of fruits with cassava starch-based biofilm improves WL, pH, and TSS. TA, TSS/TA ratio, and L* were not influenced by the use or absence of the cassava starch biofilm coating. All evaluated parameters were affected by the storage period. There was interaction for WL, FIR, H*, and C* (Tab. 1).
TABLE 1 Summary of analysis of variance of fresh weight loss (WL, %), firmness (FIR, N), pH, total soluble solids (TSS, °Brix), total titratable acidity (TA, g of citric acid per 100 g of pulp), luminosity (L*), hue angle (H*) and chromaticity (c*) of umbu fruits with and without cassava starch biofilm (Biof) during storage.
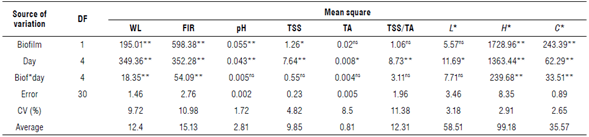
* and ** - significant values at 5% and 1% and ns - not significant values according to the F-test at 5% of probability. CV - coefficient of variation; DF - degrees of freedom.
WL increased in both treatments, reaching, on average, 16% at 12 d of storage (Fig. 1). When using cassava starch biofilm, WL was reduced by 38.8% (Tab. 2). The constant loss of weight showed that even with the starch-based coating, the fruits maintained respiration, but it was slower. Biofilms have the function of contributing to the preservation of texture and nutritional value, reducing surface gas exchange and the excessive loss or gain of water (Assis & Britto, 2014).
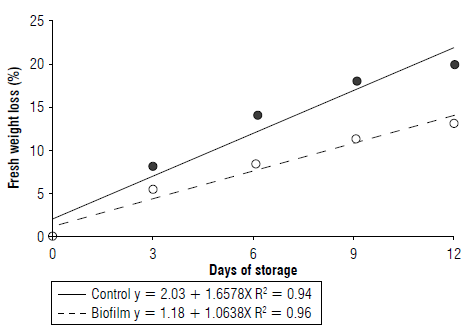
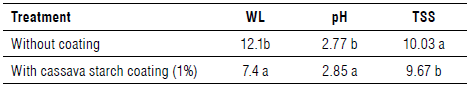
FIGURE 1 Fresh weight loss (%) of umbu fruits over storage time with and without the use of cassava starch biofilm.
TABLE 2 Average fresh weight loss (WL, %), pH, and total soluble solids (TSS, °Brix) of umbu fruits with and without cassava starch coating during storage.
Means followed by different letters in the columns are different by the F-test at 5% of probability.
FIR decreased with storage days and became stable after 9 d (Fig. 2). The FIR of the fruits treated with 1% cassava starch bio film was 67.66% greater than the untreated fruits. Fruits not treated with biofilm obtained FIR values of approximately 5 N on the 9th d of storage that show a high degree of maturation and the beginning of deterioration. In contrast, fruits with biofilm showed FIR of approximately 15 N, with a good appearance and remaining suitable for consumption until the last day of evaluation.
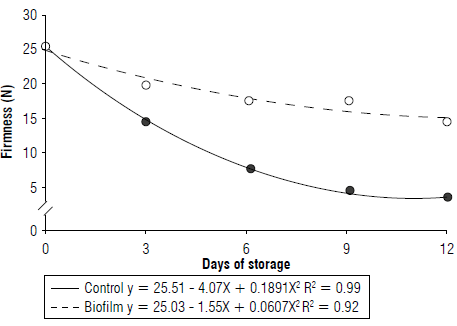
FIGURE 2 Firmness (N) of umbu fruits over storage time with and without the use of cassava starch biofilm.
The greater FIR for the fruits treated with 1% cassava starch biofilm and its reduction over the storage days is attributed to the fruit ripening process over time. The softening of the fruit during maturation is due to the activity of hydrolyses on the fruit cell wall polysaccharides leading to a modification of the cell wall composition (Yao et al., 2014). Thus, the application of a biofilm based on cassava starch favors the transport of fruits, avoiding mechanical damage and, consequently, increasing their shelf life.
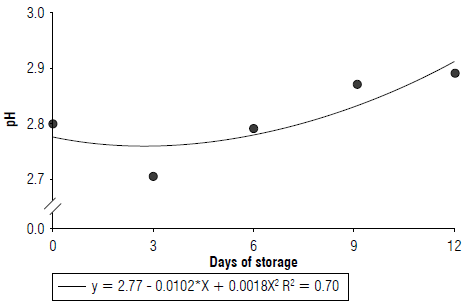
The fruit pH increased 4.33% up to the last day of storage; the increase was more marked after the 6th d (Fig. 3). Fruits treated with cassava starch had a pH 3% lower than those not treated (Tab. 2).
The increase in fruit pH with storage is due to organic acid consumption due to breathing and converting organic acids into sugars (Chitarra & Chitarra, 2005). The higher pH in fruits not treated with 1% starch shows a higher consumption of organic acids due to faster ripening.
The TSS had a significant increase of 25.5%, from 8.4 to 10.57 °Brix during storage, decreasing after the 10th d (Fig. 4A). On the other hand, the TSS showed values 4% lower when the bio film was used. The lower concentration of TSS is due to the delay in fruit ripening provided by the biofilm that reduces the synthesis of sugars.
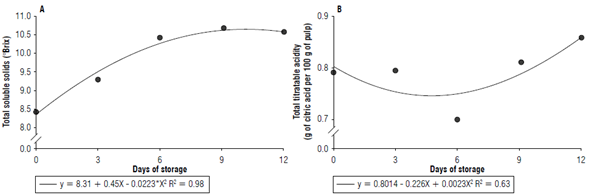
FIGURE 4 A) Total soluble solids (TSS) and B) total titratable acidity (TA) of umbu fruits over storage time.
The TA of fruits treated with starch was similar to that of untreated fruits, differing only according to the storage time. The highest TA occurred at 12 d of storage with increasing values from the 5th d of evaluation (Fig. 4B). Similar results have also been reported in umbu in a modified environment (Silva et al., 2019). The fluctuation in the TA levels with the fruit storage time can be attributed to the biochemical processes of respiratory metabolism when synthesizing and consuming organic acids (Chitarra & Chitarra, 2005).
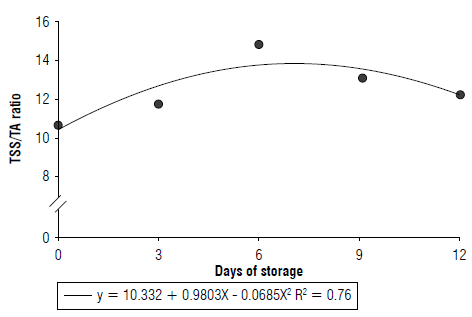
The TSS/TA ratio increased by 36.7% over fruit storage, regardless of the use or absence of biofilm and obtained a maximum value on the 7th day of evaluation (Fig. 5). The TSS/TA ratio is related to the sugar and acid balance of fruits and is a critical flavor indicator (Freitas et al., 2017). The determination of this relationship is essential, as some fruits containing low levels of acids and soluble solids have high TSS/TA ratios that can lead to misinterpretations of organoleptic characteristics.
Fruit peel color is related to attractiveness by the consumer. The use of biofilm and the period of storage interfered in the skin color of the fruits. L* decreased significantly until the 9th d of storage, after which it stabilized (Fig. 6A). The use or absence of starch did not interfere with the fruits L* that maintained an average of 58.5 (Tab. 2).
FIGURE 6 A) Luminosity, B) hue angle, and C) chromaticity of umbu fruits over storage time with and without the use of biofilm.
H* decreased 1.62% and 3.75% for each day of storage with and without the biofilm, respectively (Fig. 6B). For both treatments, fruits remained green until the 3rd d of storage, showing that the fruit's ripening process was delayed. From that period, lower H* values without the biofilm were registered, indicating the change from green to yellow, together with the loss of firmness and the increase of soluble solids that was evidence of fruit ripening. However, the use of starch provided a H* 45.8% greater in relation to untreated fruits and indicated the cassava starch-based biofilm action delayed the degradation of chlorophyll and fruit ripening. In fruits not treated with starch, chlorophyll degradation was more intense. The change in the color of the skin was characterized by a decrease in the chlorophyll content that caused the appearance of a yellow color. This color change is due to the enzymatic action on chlorophyll's structure, enabling the expression of carotenoids (Forato et al., 2015).
C* expresses the color intensity, i.e., the saturation in terms of pigments of this color. The C* in the external periderm of fruits without biofilm increased gradually up to 12 d of storage, reaching a value 28.7% higher than the treatment with biofilm (Fig. 6C). This showed that uncoated fruits had a more intense color; these fruits also showed lower H*, exhibiting a yellow color at the end of storage. Fruits with biofilm showed lower levels of C*, indicating lower color intensity and higher levels of H*. These fruits showed lighter skin color, confirming the biofilm action.
In fruits and vegetables, color is the result of pigments such as chlorophyll, carotenoids in chloroplasts, chro-moplasts, and phenolic pigments (proanthocyanidins, anthocyanins, and flavonols) in the vacuole that are degraded during ripening. The pigment color expression is also influenced by physical factors, such as the presence of cuticular waxes, trichomes, shapes, and orientation of cells in the epidermis and subepidermis (Lancaster et al., 1997). These changes are associated with the ripening of fruits; climacteric fruits generally lose their green color during ripening. Lawson et al. (2019) evaluate the effect of edible coatings based on starch extracted from tropical fruits applied to mangoes and the results of that study mention that the color of the cuticle in mango shows high values for L*, less green (increased values of a*), and higher values of b* as the ripening progresses. These fluctuations coincide with those reported in this research. L* values are associated with the sugar content in the fruits. In contrast, the values of a* and b* are associated with the content of chlorophyll and carotenoids, gradually increasing the total sugar, chlorophyll, and carotenoids as the fruit ripens (Corzo & Alvarez, 2014).