Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em
SciELO
Similares em
SciELO -
 Similares em Google
Similares em Google
Compartilhar
Vitae
versão impressa ISSN 0121-4004
Vitae v.13 n.1 Medellín jan./jun. 2006
Gloria RUÍZ1* y Evone S. GHALY1
1 School of Pharmacy, Medical Sciences Campus, University of Puerto Rico, G.P.O. Box 365067, San Juan, Puerto Rico
* Autor a quien se debe dirigir la correspondencia: eghaly@rcm.upr.edu
Recibido: Noviembre 09 de 2005 Aceptado: Febrero 28 de 2006
SISTEMAS DE LIBERACION CONTROLADA DE MUCOADHESIVOS UTILIZANDO CARRAGEENAN Y RLPO EUDRAGIT
RESUMEN
El objetivo de esta investigación es desarrollar una tableta de dos capas bioadhesive de liberación controlada. El comportamiento de la liberación de la droga y las propiedades físicas de las formulaciones de las tabletas usando dos polímeros (Carrageenan 934 y Eudragit RLPO) a tres niveles y a diferentes razones, a un nivel total de polímero de 40% p/p fueron comparados. Las formulaciones con dos polímeros a un nivel de 40% controlaron la liberación de la droga mejor que las que contenían un polímero. Las tabletas a razón (1:1) liberaron 46.4% mientras que las formulaciones a razones 3:1 y 1:3 liberaron 58.9% y 72.9%. La formulación con razón (1:1) fue seleccionada la mejor y probada para la disolución en HCl 0.1N y una solución amortiguadora de fosfato a pH 7.4 en adición a agua destilada. La liberación de la droga en la solución amortiguadora fue 96.3%, en HCl y agua destilada fue 59.1% y 46.4 %. Este estudio demuestra la importancia del uso de la combinación de polímeros para obtener una matriz bioadhesiva de liberación controlada y realzar las características de cada polímero.
Palabras clave: matrices de carrageenan, matrices de wudragit RLPO, “Chlorpheniramine Maleate”, bioadhesion, mucoadesivo, liberacion prolongada, liberacion controlada.
ABSTRACT
The objective of this research is a bioadhesive two layers controlled release tablets. The drug release and the physical properties of tablet formulations using two polymers (Carrageenan 934 and Eudragit RL PO) at three levels and at a combination of different ratios of the two polymers were evaluated. Formulation containing 40% total polymer level and a ratio of 1 Carrageenan to 1 Eudragit RLPO was the best formulationa. This formulation was tested in different dissolution medium and at different rotational speed. The drug release was 96.3% in phosphate buffer pH 7.4; 59.1% in 0.1 N HCl and 46.4% in distilled water. This study demonstrates the significance of the combination of two polymers for obtaining controlled release bioadhesive matrix and enhancing the characteristic of each polymer.
Keywords: Carrageenan matrices, eudragit RLPO matrices, chlorpheniramine maleate, bioadhesion, sustained release, controlled release, mucoadhesive.
INTRODUCTION
For systemic delivery, the oral route has been the preferred route of administration for many systemically active drugs. However when administered by oral route many drugs have been reported to be of low systemic bioavailability, short duration therapeutic activity and or formation of inactive toxic metabolites (1).
Sustained or time-release systems are methods of drug delivery in which one preparation will accomplish the desired medicinal effect with more efficiency and longer duration than multiple dosage forms of the same drug (2). Recently extensive efforts have been focused on the developing of a drug delivery system which utilize the principles of bioadhesion for optimum delivery of drug from device (3-8).
Mucoadhesive delivery systems were proven to be suitable for the purpose of reduction of transit time of the dosage form through the gastro-intestinal tract (9-11), and increasing bioavailability of drug (12-13). One of the most important factor in the preparation of sustained release mucoadhesive dosage form is the bioadhesive power of the polymer. Various synthetic as well as natural polymers have been examined in drug delivery applications (14-20).
The overall goal of this research is to design and develop a bioadhesive controlled release two layers chlorpheniramine maleate tablets using direct compression technique. This is the first paper to investigate the effect of different level of binary mixture of Carrageenan and Eudragit RLPO on the physical properties and on drug release from bioadhesive tablets compared to a single bioadhesive polymer.
Finally, to investigate the in vitro adhesion of the bioadhesive dosage form to rats stomach tissues
MATERIALS AND METHODS
Materials
Chlorpheniramine maleate, lot Number 750605 (supplied by Glaxo-SmithKline, Cidra Puerto Rico); Carrageenan NF, Type GP-812 NF, lot Number Zb 502 (Marine Colloids, FMC Inc., Philadelphia, PA, USA); Eudragit RLPO, lot Number 0481236209 (supplied by Rhom, GmbH, Darmstadt, Germany); lactose fast flo, lot Number 64044-51-5 (Foremost Farm, Wisconsin, USA) and magnesium stearate, lot Number H20237J07 (supplied by Ruger Chemical Co., New Jersy, USA). All ingredients used in the manufacture of the emulsion were of compendial grade.
Preparation of Biadhesive Tablets
Direct compression method was used to prepare nine formulations (table 1) where the level of Eudragit RLPO or Carrageenan was varied (10% w/w, 30 and 40% w/w) in order to evaluate the effect of polymer level on drug release. Also the last three formulations were containing a combination of the two polymers at different ratios of Carrageenan:Eudragit (1:1; 1:3 and 3:1) and the total polymer level was 40%m to investigate the effect of combination of two polymers on the mechanical properties and drug release from the bioadhesive tablets. Also, control tablets and tablets containing high polymer level (table 2) were prepared and evaluated.
Two Layers Tablets
Batch size was 1 Kg, the first layer weight is 100 mg containing 3 mg chlorpheniramine maleate as a loading dose for rapid initial release of drug to reach therapeutic blood level immediately and 1 mg magnesium stearate. The drug and lactose were pass ed through screen # 12 in order to break agglomerates, weighed, introduced into V blender ( Blend Master, Lab Blender P-K Ref. # C419379, Patterson Kelly Co., Division of Harsco Corporation, PA, USA) and mixed by geometrical dilution for 20 minutes. Magnesium stearate passed through screen #30 was added and mixed for further 5 minutes.
The second layer, maintenance dose weight is 300 mg containing 9 mg chlorpheniramine maleate, different levels of polymer, 9 mg magnesium stearate and lactose fast flo sufficient quantity to complete up to 300 mg.
The drug, lactose and the polymer were passed through screen #12, weighed and introduced in V blender, mixed with geometrical dilution for 20 minutes. Magnesium stearate passed through screen #30, weighed, added to the mixture in V blender and mixed for further 5 minutes
Weight, Thickness and Hardness
Ten tablets from each lot were measured for weight , thickness and hardness (Schleuniger, serial number 4676, Vector Corporation, Iowa, USA).
The range, mean and standard deviation were calculated.
Friability
Three sets of five tablets were tested for friability. The initial weight was recorded and the tablets were placed in friabilator (Erweka , Heusenstamm, Germany) and were submitted to 100 rotations. Tablets were reweighed to determine friability percent according to the following equation:
Percent Friability = ((Initial Weight – Final Weight)/(Initial Weight))x100
Disintegration
Six tablets from each formulation was placed on the basket tube of the disintegration apparatus (Erweka, Model ZT3-2, serial # 53354, Heusenstamm, Germany)and were tested for disintegration in 900 ml distilled water at 370 C ±20 C.
Drug Content
Five tablets from each batch were grinded to a fine powder using mortar and pestle. Three samples each of 0.4 g were transferred to 1000 ml volumetric flask and completed to volume with distilled water. Samples were stirred for six hours. A portion from each solution was filtered and analyzed for drug content using UV spectrophotometer (DU – 65, serial # 4310231, Bechman, California, USA).
Dissolution Testing
All batches were tested for dissolution using Apparatus 1, basket method (model SR2. serial # 21-78 – 29, Hansen Research , California, USA).at 50 rpm using 500 ml distilled water at 370C ± 0.5ºC. Samples volume of 10 ml were withdrawn at different time intervals over 6 hours period and were replaced with 10 ml distilled water at room temperature. The drug concentration in the different samples was determined by measuring the absorbances at 264 nm and using the slope and intercept obtained from the standard curve of chlorpheniramine maleate in distilled water . The best formulation that controlled the drug release was tested in 0.1 N HCl and phosphate buffer pH 7.4 in addition to the distilled water. Also, was tested using two additional basket rotational speeds (100 rpm and 150 rpm).
Diffusion Study
The glycerin was removed from the dialysis tubing in distilled water for 4 hours and then all sulfur compounds were removed by treating the tubing with 0.3 % w/v solution of sodium sulfite at 80ºC for 1 minute.
The dialysis tubing was then washed with hot distilled water for 2 minutes, followed by acidification with 0.2% w/v solution of sulfuric acid and finally, all sulfuric acid was rinsed using hot distilled water.
The best tablet formulation was weighed and introduced in a dialysis bag containing 500 ml phosphate buffer and magnetic bar. The dialysis bag was introduced in a beaker containing 500 ml phosphate buffer pH 7.4 and was stirred using magnetic bar.
Samples of 5 ml were withdrawn from the beaker at different time intervals up to 12 hours and were replaced with 5 ml buffer at room temperature.
The drug concentration in the different samples was calculated by measuring absorbance at 264 nm using the slope and intercept obtained from standard curve of chlorpheniramine maleate solutions. The diffusion study was performed in two different mediums: 0.1 N HCl and phosphate buffer pH 7.4 and also, was done in triplicates.
Bioadhesion Study
A modified tensioneter, Fisher surface tensiometer (model 21, Fischer Scientific Co.) was used to measure mucoadhesive strength between bioadhesive tablets and G.I.T. tissues. Skin from the abdominal region of six hairless CD rats (Norvergious species, 6-8 weeks old, weight 400 g to 450 g, brought from Charles River Laboratories, Wilmington, Boston, USA) were used. Rats were euthanized in pre-filled carbon dioxide chamber for 5 minutes. The pressure of the chamber was between 20 PSI to 25 PSI. Skin was lift, cut and removed from the abdominal region, soaked in a 0.9% isotonic sodium chloride solution, cut in 2.5 cm X 2.5 cm pieces.
The rat skin tissue (0.96 mm thickness) was attached to the stopper (weight 2.96 g height in 0.092 cm and diameter 0.0885 cm) and secured with aluminum vial cap with a hole of 10 mm diameter in the center. The stopper was hanged to torsion arm of the instrument using a platinum – iridium wire of 3.5 cm length and 0.026 cm diameter.
Another 2.5 cm X 2.5 cm section of the rat skin tissue was secured in the vial by using a rubber. The vial was placed in a beaker (52 mm diameter) containing 100 ml phosphate buffer pH 7.4.
Sixty mg (60 mg) of the gel formulation was uniformly spread over the exposed tissue in the stopper. The stopper was submerged in the beaker and the knob was adjusted to zero. The scale was locked and the vial was moved, so it was centered in the beaker and the beaker was moved to align the stopper and the vfial. The beaker was raised slowly to bring tissue in contact. After 1 minute of contact, the lock was released and a constant force of 10 mg/cm2 was converted into dyne/cm2.
Statistical Analysis
The ANOVA one way analysis was used to determine if there is significant differences between the two variables and the t-test was used to examine if there is significant differences between pairs. (Table 3 and table 4).
RESULTS AND DISCUSSION
The simple factorial design was used to determine number of variables, levels and effect of variables on response.
Pn = 31A + 31B + 31C where: n = number of variable; p = levels of variables; A = Carrageenan at 3 levels (10%, 30 and 40%); B = Eudragit RLPO at 3 levels (10%, 30 and 40%); C = polymer combination (1Carrageenan:1 Eudragit RLPO; 3 Carrageenan : 1 Eudragir RLPO; 1 Carrageenan : 3 Eudragit RLPO).
In phase 1 of the study, six formulations were prepared. Two polymers were used: Carrageenan and Eudragit RLPO each at 3 levels (10%, 20 and 40%) to determine the best level that will give lowest drug release. In the second phase, three tablet formulations containing combination of the two polymers at a total polymer level of 40% and at different ratios of Carrageenan:Eudragit RLPO (1:1; 1:3 and 3:1) were prepared. The nine tablet formulations are shown in Table 1. All tablet formulations were assayed for drug content including control tablets without polymer as shown in Table 2.
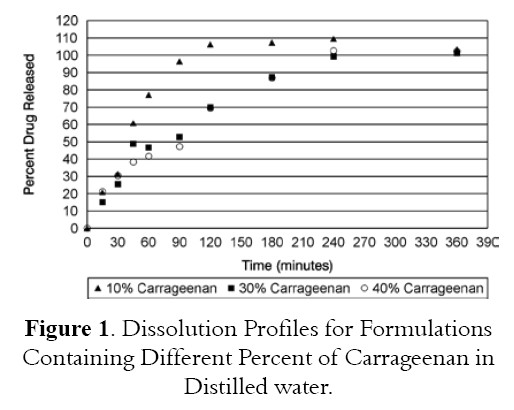
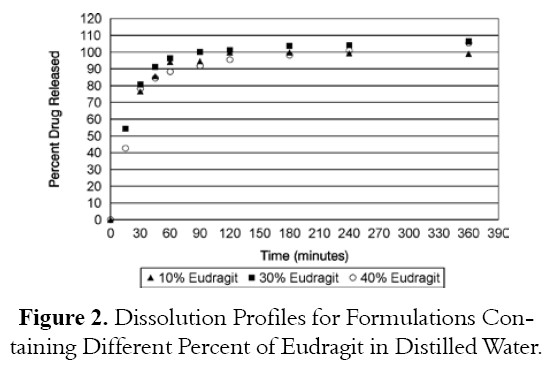
Figures 1 and 2 show the dissolution profiles for formulations containing different levels of Carrageenan and Eudragit RLPO respectively. Figure 1 shows the dissolution profiles for Bioadhesive tablets containing different levels of Carrageenan polymer. Formulation containing 10% Carrageenan release all drug at 2 hours of testing dissolution while formulations containing 30% or 40% Carrageenan released 69.8% and 69.34% drug respectively. Figure 2 depicts the dissolution profiles for formulations containing different levels of Eudragit RLPO. All formulations containing different levels of Eudragit RLPO released all drugs at 2 hours of testing dissolution. Formulation containing 10% Eudragit RLPO released 99.8% while formulation containing 30% and 40% Eudragit RLPO released 101.24% and 95.51% drug at 2 hours of testing dissolution. It appears that the characteristics of the polymers used affect drug release.
ANOVA analysis showed no sigmificant difference between formulations containing 10, 30 and 40% Carrgeenan and also ANOVA analysis showed no significant difference between formulations containing 10%, 30 and 40% and Eudragit as shown in Tables 3 and 4.
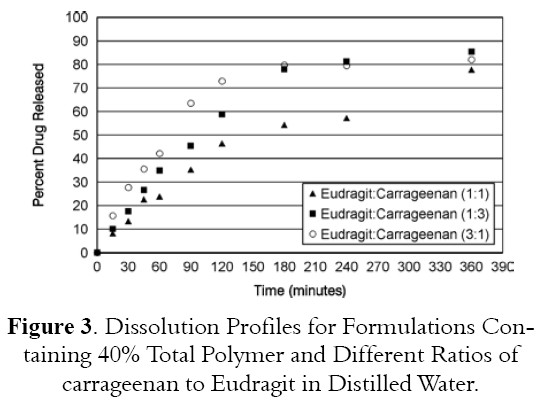
Figure 3 shows the dissolution profiles for formulation containing combination of two polymers at 40% total polymer and different ratios of Carrageenan:Eudragit RLPO(1:1; 3:1 and 1:3). Formulation containing 1 Carrageenan : 1 Eudragit RLPO released 46.4% drug at 2 hours of testing dissolution while formulation containing 1 Carrageenan : 3 Eudragit RLPO and or 3 Carrageenan : 1 Eudragit RLPO released 59.9% and 72.9% drug respectively. These data indicated that best ratio of combination is 1 Carrageenan : 1 Eudragit RLPO and as the ratio of Carrageenan to Eudragit RLPO increases, the drug release increases.
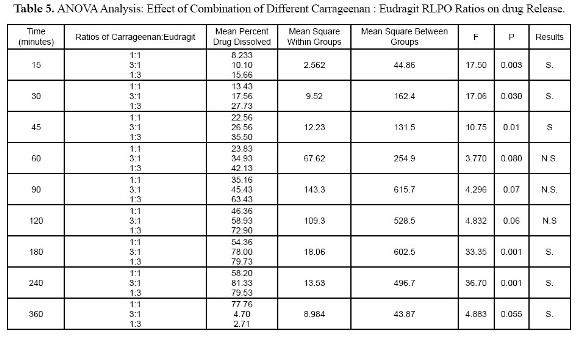
Table 5, one way ANOVA analysis shows significant differences in drug release between formulations containing different ratios of Carrageenan to Eudragit RLPO.
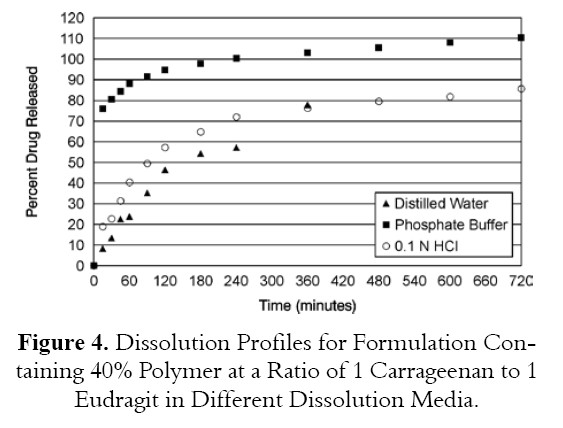
The best formulation containing total polymer level of 40% and combination of Carrageenan and Eudragit RLPO at 1:1 ratio was tested in two additional dissolution medium: 0.1 N HCl and phosphate buffer pH 7.4.
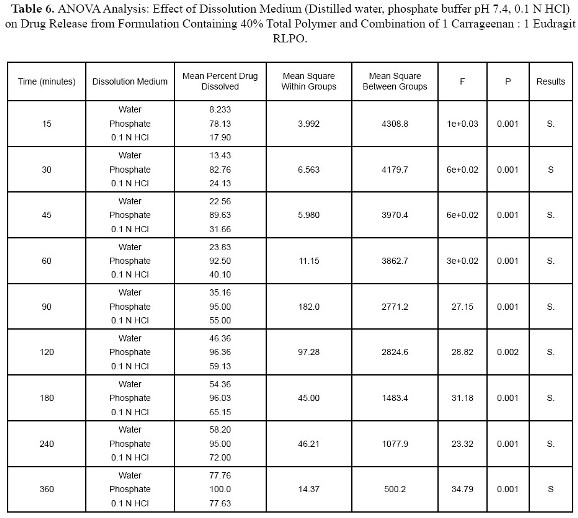
Tablets tested in phosphate buffer pH 7.4 released 96.3% of drug at 2 hours while tablets tested in 0.1 N HCl and distilled water released 59.1% and 46.4% respectively as shown in Figure 4. Eudragit RLPO is water insoluble polymer and is not pH dependent and consequently drug release from Eudragit RLPO is not affected by changing pH of dissolution medium. Carrageenan is a sulfated polymer obtained from Algae and is hydrophilic polymer. Apparantly Carrageen differ from other hydrophilic polymers which their gels strength are disrupted in acidic media but Carrageenan maintain its gel strength in acidic medium.and gave lower drug release in 0.1 N HCL than in phosphate buffer pH 7.3
ANOVA analysis supported the dissolution data and showed significant differences between tablets tested in different dissolution medium as shown in Table 6.
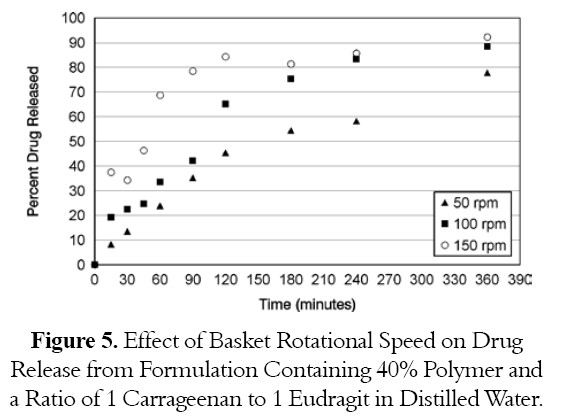
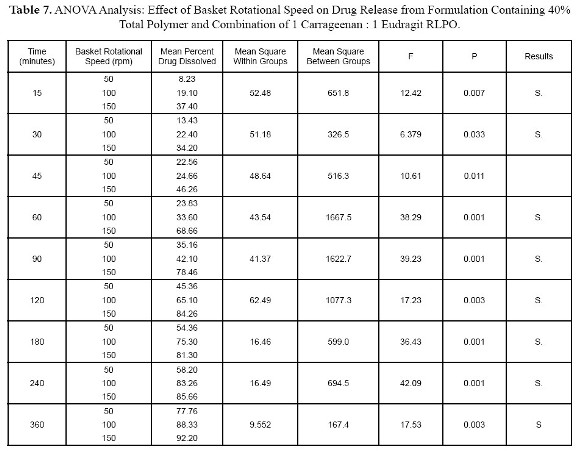
The best formulation was also, tested at different basket rotational speed. At 50 rpm, the percent drug release was 46.4% at 2 hours while percent drug release at 100 rpm and 150 rpm was 65.1% and 84.3% respectively as shown in Figure 5. As the rotational speed opf the basket is increased, the drug release is increased.
ANOVA one way statistical analysis supported the dissolution data and showed significant differences in drug release between tablet tested at different rotational speeds as shown in Table 7.
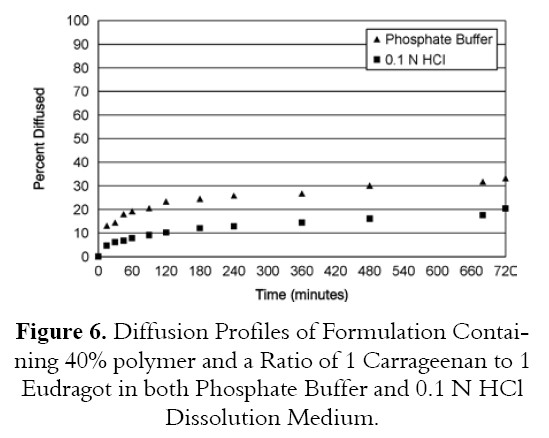
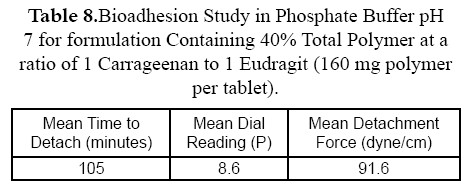
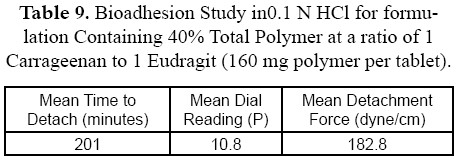
The diffusion study for the best formulation showed that percent drug diffused in phosphate buffer pH 7.4 was 33.2% at 12 hours while percent drug diffused for the same best formulation in 0.1 N HCl was 20.4% (Figure 6). These data are similar to that obtained from dissolution data in different pH dissolution medium. Amount of drug dissolute or diffused in 0.1 N HCl is lower than that dissolute or diffused in phosphate buffer pH 7.4. as shown in Tables 8 and 9.
The bioadhesion study (n = 3) indicated that force needed for detachement of the bioadhesive tablet from the rat mucous membrane in phosphate buffer pH 7.4 was only 91.6 dynes/cm2 compared to 182.8 dynes/cm2 force needed to detach the same bioadhesive tablet from the rat skin in 0.1 N HCl.
SUMMARY AND CONCLUSION
The two layers bioadhesive delivery system has potential to be an effective sustained release system over a long period of time for the antihistaminic chlorpheniramine maleate.
The type and level of polymer used are fundamental factors that can affect the drug release and also the physico-chemical properties of these mucoadhesive devices.
The combination of carrageenan and Eudragit RLPO enhanced the characteristics of each polymer. The combination of 1 Carrageenan to 1 Eudragit RLPO was found to be the best candidate to develop a suitable bioadhesive dosage form.
Finally, the in-vitro study demonstrated the efficiency of the bioadhesion of the developed dosage form.
BIBLIOGRAPHIC REFERENCES
1. Chien W. Yin. Novel Drug Delivery Systems, Marcel Dekker Inc.,, New York, p. 169-238. [ Links ]
2. Perioli, L.; Ambrogi, V.; Angelici, F.; Rioci, M.; Giovagnoli, S.; Capuccella, M.; Rossi, C. (2004) Development of Mucoadhesive Patches for Buccal Administration of Ibuprofen. J. Control Release. 99 (1), 73-82... [ Links ]
3. Ranga, K.N.; Buri, P. (1989) A Novel In-Situ Method to Test Polymers and Coated Particles for Bioadhesion. Int. J. Pharm. 52, 265-270. [ Links ]
4. Yamamoto, H.; Takeuchi, H.; Kuno, Y. (2000) Mucoadhesive DL-Lactide/Glycolide Copolymer nanospheres Coated with Chitosan to Improve Oral Delivery of Elcatonin. Pharm. Dev. Technol. 5, 77-85. [ Links ]
5. Luessen, H. L.; De Leeww, B. J.; De Boer, A. B.; Verhoef, J. C. (1996) Mucoadhesive Polymers in Peroral Peptide Drug Delivery. VI. Carbomer and Chitosan Improve the Intestinal Absorption of the Peptide Drug Buselin In-Vivo. Pharm. Res., 13, 1668-1672. [ Links ]
6. Moratzavi, S.A.; Smart, J.D. (1993) Investigation Into the Role of Water Movement and Mucus Gel Hydrationin Mucoadhesion. J. Controlled Release. 25, 197-203. [ Links ]
7. Alireza, S.; Smart, J. (1994) An Invitro Method for Assessing the Duration of Mucoadhesion. J. Controlled Release. 31, 207-212. [ Links ]
8. Bani-Jaber, A.; Al-Ghazawi, M. (2005) Sustained Release Characteristics of Tablets Prepared with Mixed Matrixz of Sodium Carrageenan and Chitosan L Effect of Polymer Weight Ratio, Dissolution Medium and Drug Type. Drug Dev. Ind. Pharm. 31 (3), 241-247. [ Links ]
9. Varma, M.; Singla, AK.; Dhawan, S. (2004) Release of Diltiazine Hydrochloride from Hydrophilic Matrices of Polyethylene Oixide and carbopol. Drug Dev. Ind. Pharm. 30 (5), 545-553.. [ Links ]
10. Chi-Hyun, L.; Yie, W. C. (1996) Development and Evaluation of Mucoadhesive Drug Delivery System for Dual Controlled Delivery of Nonoxynol-9. J. Controlled Release. 39, 93-103. [ Links ]
11. Leimer, VM.; Marschutz, MK.; Bernkop-Schmurch, A. (2003) Mucoadhesive and Cohesive Properties of Poly (Acrylic Acid)Cystein Conjugate with Regards to their Molecular Mass. Int. J. Pharm. Sci. 18 (1), 89-96. [ Links ]
12. Liabot, JM.; Manzo, RH. Allemandi, DA. (2004) Drug Release from Carbomer:Carbomer Sodium Salt Matrices With Potential Use as Mucoadhesive Drug Delivery System. Int. J. Pharm. 276 (1-2), 59-66. [ Links ]
13. Remunan_Lopez, K.; Portero, A.; Vila-Hato, J. L.; Alonso, M. J. (1998) Design and Evaluation of Chitosan / Ethyl Cellulose Mucoadhesive Bilayer Devicesfor Buccal Drug Delivery. J. Controlled Release 55, 143-152. [ Links ]
14. Singh, B.; Ahuja, N. (2002) Development of Controlled Release Bucoadhesive hydrophilic Matrices of Diltiazem Hydrochloride Optimization of Bioadhesion, dissolution and Diffusion Process. Drug Dev. Ind. Pharm. 28, 431-442. [ Links ]
15. Rily, R.G.; Smart, J. D.; Tsibouklis, J.; Detmar, P.W.; Hampson, F.; Davis, J.A. (2001) An Investigation of Mucus/Polymer rheological Synergism using Synthesised and Charactarised Poly(Acrylic Acid). Int. J. Pharm. 217, 87-100. [ Links ]
16. Betageri, G.V.; Deshmukh, D.V.; Gupta, R.B. (2001) Oral Sustained Release Bioadhesive Tablet Formulation of Didanosine. Drug Dev. Ind. Pharm. 27, 129-136. [ Links ]
17. Picker, K.M. (1999) Matrix Tablets of Carrageenans, I.A. Compaction Study. Drug Dev. Ind. Pharm. 25, 329-337. [ Links ]
18. Mehta, K.A.; Kisla lioglu, M. S.; Phuapradit, W.; Malick, A.W.; Shah, N. H. (2001). Release Performance of Poorly Soluble Drug from a Novel Eudragit-Based Multi-Unit Errosion Matrix. Int. J. Pharm. 213, pp 7-12. [ Links ]
19. Boza, A.; De La cruz, Y.; Jordan, G.; jauregui-Haza, U.; Aleman, A.; Caraballo, I. (2000). Statistical Optimization of a Sustained-Release Matrix Tablets of Lobenzarit Disodium. Drug Dev. Ind. Pharm. 26, 1303-1307. [ Links ]
20. Khan, M. Z.; Stedul, H.P.; Kurjakovic, N. (2000). A pH Dependent Colon Targeted Oral Drug Delivery System Using Methacrylic Acid Copolymer. II. Manipulation of Drug Release Using Eudragit L 100 and Eudragit S 100 Combinations. Drug Dev. Ind. Pharm. 26, 549-554. [ Links ]