1. Introduction
The word 'textile' refers to a tissue, which includes items such as clothing, ornaments, and upholstery objects, that have been elaborated for utilitarian, ceremonial, funeral, and other purposes.
The pre-Hispanic cultures of the northern coast of Peru, like the Chimu Culture, had among their socio-economic activities the manufacture of tissues. These cultures were careful in the manufacture of textiles, which involved: the selection, spinning, washing, and dyeing of fibers. The Chimu culture developed between 900 - 1470 AD, approximately. Its territory stretched from Tumbes in the north to Carabayllo in the south, its capital was Chan Chan 1.
Most pre-Hispanic textiles from the northern region of Peru were made from cotton fibers 2, decorated with pigments, or dyed with dyes. In ancient Peru, minerals were exploited as pigments 3. Clothing in the Chimu kingdom consisted of loincloths, sleeveless shirts (with or without fringes), small ponchos, and tunics.
Cotton is a plant of the Malvaceae family (genus Gossypium) and is a major source of foreign exchange. Pre-Hispanic textiles are considered as revealing sources of information in the social, symbolic, and economic aspects 4,5. At present, the structure of pre-Hispanic textiles is still being investigated, with the purpose of determining the raw material, quality, manufacture, and obtaining information on social, cultural, and economic aspects.
The present work focused on studying morphology, identifying the atomic composition and the structure of the fibers of a pre-Hispanic textile of the Chimú culture. Instrumental techniques were applied such as Scanning Electron Microscopy (SEM), Energy-dispersive X-ray Spectroscopy (EDS), Laser-induced Breakdown Spectroscopy (LIBS), Attenuated Total Reflection Fourier-Transform Infrared Spectroscopy (ATR-FTIR), and X-ray Diffraction (XRD). Research works involving structural analysis of organic compounds start with the FTIR test because it allows identifying the presence of certain functional groups that characterize an organic substance.
However, it must be accompanied by other spectroscopic techniques to strengthen the scientific evidence of an organic structure. The results of the characterization of the fiber of the Chimú textile were compared with those of the native cotton fiber coming from the northern coast of Peru (Mochumí-Lambayeque). This comparison made it possible to identify the origin and quality of the raw material used to manufacture the studied textile.
2. Materials and methods
Figure 1a shows the Chimú textile (T-shirt) considered for the study. The textile is preserved in the Museum of Natural and Cultural History of the Antenor Orrego Private University (UPAO), Trujillo-Peru. Figure 1b presents the hypothetical reconstruction of the textile.

Figure 1 a) Original Chimú textile preserved in the Museum of Natural and Cultural History of the Antenor Orrego Private University (UPAO), Trujillo-Peru. b) Hypothetical reconstruction of the Chimú textile.
To perform the characterization of the Chimú textile, an area of 1 cm2 was cut out of it (from a deteriorated site, located near the neck). The sample was handled with care to maintain the original texture. For the effect of comparison, native cotton fibers coming from northern Peru (Mochumí-Lambayeque) were analyzed.
The morphology of the fibers was studied by analyzing SEM images. A TESCAN scanning electron microscope, model VEGA 3 LMU, was used, applying acceleration voltages of 5 kV and 10 kV. The samples were placed on a segment of double-sided carbon tape and coated with a thin layer of carbon by sputtering to avoid electron accumulation effects. Additionally, a semi-quantitative elemental analysis of the microfibers was performed by EDS, using an Rx Quantax 200-Bruker detector.
To obtain the emission spectra of the atoms present in the Chimu textile and the native cotton, the LIBS technique was considered. For the laser ablation of the textile and native cotton, a Quantel Q-smart pulsed Nd: YAG laser and a converging lens of 10 cm focal length were used. The sample was located 9.5 cm from the lens. The laser parameters were: wavelength of 1064 nm, energy of 13.2 mJ/pulse, laser pulse duration of 6 ns, and laser pulse repetition rate of 1 Hz. The electromagnetic radiation emitted by the samples was guided through an optical fiber to an HRS-500-Princeton Instruments spectrometer. As a light sensor, an Intensified Charge-Coupled Device (ICCD camera) PI-MAX4 from Princeton Instruments was used. The Princeton Instruments LightField software was used to obtain the emission spectra of the plasmas. For the spectrometer, the grating of 1200 lines/mm was considered, which implied a resolution of 0.02 nm. The delay time to start the light collection (with respect to the arrival of the laser pulse) was 1000 ns. By considering this time, the collection of the braking radiation (Bremsstrahlung) of the ablation process was avoided. The integration time for the light collection was 10000 ns. Event synchronization was performed as follows: Each time the Nd: YAG laser emitted a pulse, an electrical signal was generated and used as a trigger for the ICCD camera. The camera software allowed controlling a delay time with respect to the trigger to start the collection of the light coming from the plasma. Likewise, the software allowed to control the collection time. For the acquisition of the Chimú textile data, the following order was considered: (1) obtaining the emission spectrum of the plasma occasioned by the first incident pulse on the surface of the textile, (2) ablation of an only zone with 30 laser pulses, to obtain a clean zone and acquire the spectrum of the plasma occasioned by a single incident laser pulse on the fiber of the clean textile. For the acquisition of native cotton data, the average spectrum was obtained due to the action of 25 laser pulses.
Infrared spectra were obtained by ATR-FTIR. A Thermo Scientific iS 50 spectrophotometer was used, using the Attenuated Total Reflectance (ATR) mode with diamond crystal, taken in transmittance mode from 500 to 4000 cm-1, 16 scans, and a resolution of 1 cm-1. The ATR-FTIR technique is suitable for the study of archaeological remains because it analyzes the surface composition without destroying the sample.
The degree of crystallinity of the samples was analyzed by XRD. X-ray diffraction patterns were obtained at room temperature, with a D8 ADVANCE ECO - Bruker diffractometer, Cu Kα (λ = 1.5418
Where Ac is the area under the curve of the diffraction peaks, and Aa is the amorphous area.
3. Results and discussions
3.1. Fiber morphology by SEM
Figure 2a shows the image obtained from a thread of the Chimu textile. This figure evidence that each thread of the textile is made up of interlaced microfibers. It is possible that the interlacing of these microfibers has given the Chimú textile a certain mechanical resistance over the years. In Figure 2a, it can be observed that microfiber has a twisted ribbon shape, which is a characteristic of cotton microfibers 6,7.

Figure 2 a) Thread of the Chimu textile, which is composed of several microfibers. b) and c) Microfiber of the Chimu textile. d) Microfiber from native cotton.
Images of a Chimú textile microfiber are presented in Figure 2b and Figure 2c. Figure 2b shows that the average diameter of a Chimu textile microfiber is 15.4 ± 0.2 μm. Figure 2c shows solid particles on the surface of the microfiber of the textile. Unlike the native cotton microfibers, the Chimú textile microfibers showed some solid particles adhered on their surfaces. These solid particles could be dust residues since the textile remained buried for many years. Figure 2d shows a native cotton microfiber, which presented an average diameter of 16.3 ± 0.2 and was homogeneous in appearance.
3.2. Elemental analysis by EDS
Figure 3 shows the results of atomic composition microanalysis of microfibers from Chimú textile and native cotton. To identify atoms present in the microfibers, elemental analysis was performed by scanning. The scanning area was 179 μm x 179 μm.
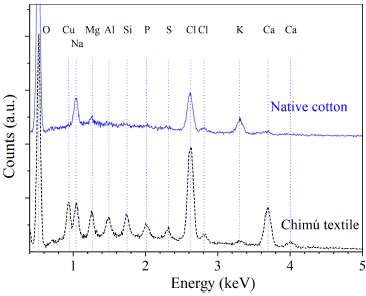
Figure 3 Microanalysis of atomic composition by EDS of microfibers of Chimú textile and native cotton.
Table 1 shows the atomic composition percentages for the microfibers of the Chimú textile and the native cotton. The comparison of both atomic compositions suggests that the raw material of the Chimú textile was cotton.
Table 1 The atomic composition of the microfibers of the Chimú textile and native cotton.
| COMPOSITION | ATOMIC PERCENTAGE (%) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Element | O | Cu | Na | Mg | Al | Si | P | S | Cl | K | Ca |
| Native cotton | 81.4 | 5.1 | 1.0 | 7.4 | 4.1 | 0.9 | |||||
| Chimú textile | 63.2 | 2.9 | 3.3 | 1.7 | 1.2 | 2.1 | 1.1 | 0.9 | 12.3 | 0.6 | 10.3 |
For both microfibers, the scanning analysis revealed the presence of a high oxygen content (characteristic of cellulose) and traces of Na, Mg, Cl, K and Ca.
Only in the microfibers of the Chimu textile, the presence of Cu, Al, Si, P and S was evidenced. These elements would be associated with residues of dust or solid particles adhered to the surface of the microfibers, which were observed in the SEM images (Figure 2b and Figure 2c).
It is likely that some of these elements are associated with metallic mineral pigments such as atacamite Cu2Cl(OH3) or cinnabar HgS 3 and metal salts 8.
3.3. Identification of atoms by LIBS
Figure 4a shows the emission spectrum of the Chimú textile plasma, in the wavelength range from 240 to 340 nm. This spectrum was obtained by considering a single laser pulse, and without any previous preparation or cleaning procedure of the sample. Lines of carbon, silicon, copper, magnesium, sodium, and calcium are observed in the spectrum. The inset in Figure 4a shows the silicon emission lines. It is likely that the silicon lines are due to the presence of microparticles of soil or dust on the surface of the textile.
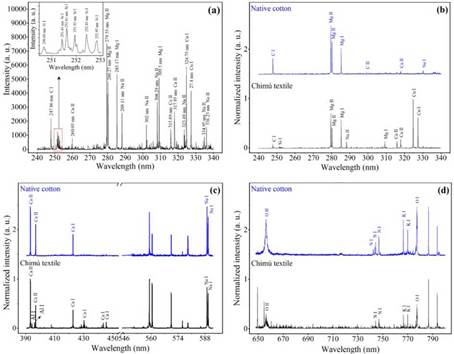
Figure 4 Emission spectra of pulsed laser-induced plasmas of the Chimú textile and native cotton. (a) Spectrum corresponds to the plasma of the Chimú textile when considering a single laser pulse. (b), (c) and (d) Normalized spectra corresponding to Chimú textile and native cotton for different wavelength ranges. The neutral species are specified with the chemical element and to continuation I. The species once ionized, are shown with the chemical element and to continuation II.
Figure 4b, Figure 4c, and Figure 4d show the normalized spectra of the plasmas of native cotton and a clean area (previously irradiated with 30 laser pulses) of the Chimú textile. When comparing Figure 4a with the spectrum corresponding to the Chimú textile in Figure 4b, the same atoms are identified, but the amount of sodium lines decreases significantly. This suggests that sodium is mostly found in the surface layers of the Chimú textile. Figure 4b shows the normalized spectra corresponding to the samples, in the wavelength range from 240 to 340 nm.
By comparing the spectra, several lines of ionic and neutral species are evident in common. The common emission lines are C I 247.86 nm, Mg II 279.55 nm, Mg II 280.27 nm, Mg I 285.17 nm, and Ca II 317.93 nm. Likewise, silicon and copper lines are identified, and these correspond only to the Chimú textile. Figure 4c shows the spectra in the wavelength range from 390 to 454 nm and from 545 to 592 nm. When comparing the spectra, several common emission lines are found. The common emission lines correspond to magnesium, calcium, and sodium. Only the Chimú textile sample presented spectral lines corresponding to aluminum (Al I 394.40 nm and Al I 396.15 nm). Figure 4d shows common elements present in the Chimú textile and native cotton, in the interval ranging from 648 to 800 nm. The spectra are identical and the common spectral lines correspond to oxygen, nitrogen, potassium, and calcium.
The unidentified peaks in Figure 4c, in the wavelength range from 558 nm to 571 nm, correspond to second diffraction orders of magnesium (Mg II 2x279.55 nm, Mg II 2x280.27 nm, and Mg I 2x285.21 nm). The unidentified peaks in Figure 4d correspond to second diffraction orders (Cu I 2x324.75 nm, Cu I 2x327.39 nm, Ca II 2x393.37 nm, and Ca II 2x396.85 nm).
When analyzing the cotton, plasma spectroscopy allowed us to identify the following elements: carbon, nitrogen, oxygen, sodium, magnesium, potassium, and calcium. Likewise, due to the correlation between the atomic lines found for the samples, it can be argued that the raw material of the Chimú textile was native cotton. The presence of copper and aluminum only in the Chimú textile suggests that the cotton was coated with natural dyes. It is worth mentioning that both plasma spectroscopy and EDS evidenced the presence of copper and aluminum only in the Chimú textile.
3.4. Identification of atomic bonds and crystallinity by ATR-FTIR
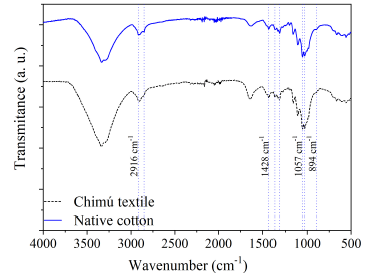
Figure 5 shows the ATR-FTIR spectra of microfibers from the Chimú textile and the native cotton. These spectra are characteristic of cellulose. It can be seen vibration bands of the C-H2 bonds at 2916, 2851, 1428, and 1315 cm-1 (9. The bands correspond to the C-H vibrations at 1370 cm-1 (10 and 894 cm-1 (11. The C-O bands are around 1057 and 1032 cm-1 (12. The broadband between 3600 and 3200 cm-1 corresponds to the O-H vibration modes 9.
The intensities of the bands of both spectra are similar, suggesting little variation in the crystalline structure of the Chimú textile, despite having been underground for hundreds of years. In order to evaluate crystallinity, it was proposed to determine the relationship between absorbances at 1428 cm-1 and 2916 cm-1 (a1428/a2916), associated with the C-H2 bands. The ratios obtained were: 1.31 and 1.67, for the Chimú textile and native cotton fibers, respectively.
Another band that analyzes crystallinity is the one that appears between 1630 and 1650 cm-1, being more intense when it has less crystallinity because it accumulates a greater amount of water 9. This band is more intense for the Chimú textile fiber compared to the native cotton.
The slight loss of crystallinity of the Chimú textile fibers suggests significant structural stability, even though the fabric was underground for hundreds of years.
3.5. Identification of structure and crystallinity by XRD
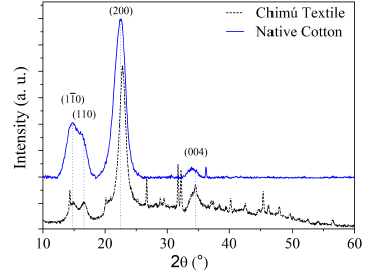
Figure 6 shows the X-ray diffraction pattern of the Chimú textile and native cotton. Both patterns show the presence of the characteristic diffraction planes of the polymorphism of cellulose ) 13,14. These characteristic planes are: (1-10), (110), (200) and (004), located at 2θ = 14.8°, 16.6°, 22.5° and 34.5°, respectively.
Cellulose I ß (C6H10O5)n is a biopolymer composed of ß-glucose molecules present in cotton fibers. The polymorphism of cellulose I ß present a monoclinic structure P21, which can be modeled as a monoclinic unit cell (a = 0.778 nm, b = 0.820 nm and c = 1.038 nm, α = 118.1°, ß = 114.8° and γ = 96.5°) 15.
In Table 2, the lattice parameters, the average crystallite size, and the crystallinity of the Chimú textile and native cotton microfibers are presented. Both samples presented network parameters similar to those of the monoclinic structure of cellulose I ß .
Table 2 Lattice parameters and strúctúral characteristics of the Chimú textile and native cotton microfibers.
| SAMPLE | ESTRUCTURE | LATTICE PARAMETERS ( |
AVERAGE CRYSTALLITE SIZE (nm) | CRYSTALLINITY (%) | ||
|---|---|---|---|---|---|---|
| a | b | c | ||||
| Chimú textile | Monoclinic, γ = 96.50° | 0.781 | 0.819 | 1.021 | 4.67 | 48.00 |
| Native cotton | Monoclinic, γ = 97.38° | 0.785 | 0.818 | 1.017 | 4.30 | 62.30 |
Vegetable fibers, such as cotton, are essentially constitúted of cellulose, which can be crystalline and amorphous 16. A cotton microfiber presents a multilayer structure. In a cross-section, three layers can be identified: external, internal, and lumen. Amorphous cellulose is found in the external layer, while crystalline cellulose is located in the internal layer 6.
The Chimú textile fibers showed larger crystallite size and lower crystallinity compared to native cotton fibers. The difference in crystallinity is slight and suggests good structural stability for the Chimú textile.
4. Conclusions
The SEM images revealed that the microfibers of the Chimú textile look like twisted ribbons, which is characteristic of the microfibers of cotton. EDS and LIBS techniques made it possible to identify characteristic atoms of cellulose (the main component of cotton) in the Chimú textile. The X-ray diffractograms showed characteristic peaks of the polymorphism of cellulose Ig, for the Chimú textile microfibers. The ATR-FTIR and XRD showed a lower crystallinity for the Chimú textile microfibers compared to the cotton microfibers. The raw material of the Chimú textile is native cotton. The colors present in the Chimú textile are due to the use of natural pigments based on metallic salts and mineral. The microfibers of the Chimú textile present significant structural stability.