Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em
SciELO
Similares em
SciELO -
 Similares em Google
Similares em Google
Compartilhar
Revista MVZ Córdoba
versão impressa ISSN 0122-0268versão On-line ISSN 1909-0544
Rev.MVZ Cordoba v.16 n.2 Córdoba maio/ago. 2011
ORIGINAL
Neospora caninum infection in beef cattle reared under grazing conditions in north-central Mexico
Infección por Neospora caninum en ganado de carne mantenido en condiciones de pastoreo en el centro-norte de México
Karina Mondragón-Zavala,1 M.Sc, Carlos Cruz-Vázquez,1* Ph.D, Leticia Medina-Esparza,1 Ph.D, Miguel Ramos-Parra,1 M.Sc, Zeferino García-Vázquez,2 Ph.D.
1Instituto Tecnológico El Llano Aguascalientes. Aguascalientes, México.
2Centro Nacional de Parasitología Veterinaria, Instituto Nacional de Investigaciones Forestales, Agrícolas y Pecuarias (INIFAP), Jiutepec, Morelos, México.
*Corresponding: cruva18@yahoo.com.mx
Recibido: Agosto de 2010; Aceptado: Febrero de 2011.
Abstract
Objetive. To determine the seroprevalence of N. caninum antibodies and prevalence of parasite DNA in blood, and estimate the association between seroprevalence and the potential risk of some factors in beef cattle under grazing conditions in north-central Mexico. Materials and methods. Blood samples from 139 cows and only 10 bulls belonging to 13 farms were collected and evaluated by ELISA test to detect antibodies against N. caninum. Furthermore, to determine the presence of parasite DNA, nested PCR probe was performed on blood samples. Association between potential risk factors and seroprevalence was estimated. Results. Overall seroprevalence was 23% (35/149 samples), while the prevalence of parasite DNA in blood was 28% (42/149 samples). Of the 149 animals examined 28 (19%) were positive to both tests (25 cows and 3 bulls). Concordance between tests was k = 0.63. All herds had seropositive animals with positive parasite DNA detection in blood. The only risk factor identified was the presence of dogs (OR= 2.65). Conclusions. This study showed that bovine neospososis should be considered as an important infectious disease in north-central Mexico herds. Therefore, an epidemiological control should be taken into consideration to avoid the negative effect of this disease on mexican beef industry.
Key words: Bovines, DNA, prevalence, seroprevalence, PCR. (Sources: AIMS,CAB).
Resumen
Objetivo. Determinar la seroprevalencia de anticuerpos contra N. caninum y la prevalencia de ADN del parásito en sangre y estimar la asociación entre la seroprevalencia y algunos potenciales factores de riesgo de ganado de carne mantenido bajo condiciones de pastoreo en el centro-norte de México. Materiales y métodos. Se seleccionaron trece hatos ganaderos, en los cuales se recolectaron muestras de suero sanguíneo de 139 vacas y de solo 10 sementales, que fueron evaluados mediante la prueba de ELISA para detectar anticuerpos contra N. caninum. Por otra parte, se realizó una prueba de PCR anidado en muestras de sangre para determinar la presencia de ADN del parásito. Se estimó la asociación entre la seroprevalencia y algunos potenciales factores de riesgo. Resultados. La seroprevalencia general fue de 23%, mientras que la prevalencia a la presencia de ADN del parásito en sangre fue de 28%. Veintiocho muestras de 149 fueron positivas en ambas pruebas, mientras que 3/10 sementales fueron positivos en las dos pruebas. La concordancia entre las pruebas fue k=0.63. Todos los hatos tuvieron animales seropositivos y con presencia de ADN del parásito en sangre. El único factor de riesgo identificado fue la presencia de perros (OR=2.65). Conclusiones. La infección por N. caninum determinada en este estudio es importante, y deberá de ser más documentada; del mismo modo, algunas medidas de control deberán de considerarse para limitar sus efectos negativos en la industria mexicana de la carne.
Palabras clave: ADN, bovinos, PCR, prevalencia, seroprevalencia (Fuentes: AIMS, CAB).
Introduction
Bovine neosporosis is a parasitic disease caused by Neospora caninum (Apicomplexa, Sarcocystidae), a protozoan that affects a great variety of domestic and wild animals, and is currently considered as one of the main causes of abortion worldwide (1).
In bovine neosporosis epidemiology, transmission mechanisms are diverse, this factor, undoubtedly contributes to the high presence of the parasite in cattle populations. Dogs and coyotes (2) have been described as definitive hosts of N. caninum, and they appear as main factors in the horizontal transmission by excreting oocysts in the feces that contaminate the drinking water and feed. However, there is not doubt that vertical transmission is the main route to maintain the infection in cattle. It has been reported that calves are infected through placenta by chronically infected mothers; however, this is highly efficient allowing the birth of infected but clinically healthy offspring (1,2).
Although there is information about the presence of N. caninum in mexican in dairy cattle (3-5), but data about the presence of this disease in beef cattle raised under grazing conditions is scarce.
The objective of this study was to determine the seroprevalence of N. caninum antibodies and prevalence of parasite DNA in blood, and estimate association between seroprevalence and the potential risk of some factors in beef cattle farms under grazing conditions in north-central Mexico.
Materials and methods
Study site. This study was carried out in the State of Aguascalientes, Mexico, which is located in the north-central part of the country, at 1.885 m above sea level, with an average temperature of 16.9°C, and 475 mm annual rainfall which occurs during summer. This part of the country is semi-arid presenting warm conditions with wide range of temperature variation.
Situation of the farms and animals. A total of 13 beef small cattle farms, with an average herd size of 50 cows and 1 bull per farm from different areas of the State were selected. At the time of this study, farmers raised crossbred (in different proportions) cows (Bos taurus and Bos indicus). Cows were kept, under grazing conditions and together with bulls along the year (mainly together with bulls along the year (mainly Simmental, Charolais and Brown Swiss races), for breeding purposes. No data on gestation status was available and no abortions were notified to be detected in the last 6 months. Blood samples were obtained at random from 139 cows (>2 years old) and from only 10 bulls from each farm selected.
Blood sampling. Blood was obtained from each animal by puncture of the caudal vein and divided into two vacutainer tubes: 1) sample with anticoagulant (EDTA) and 2) sample without EDTA. Samples with anticoagulant were frozen at -20°C and samples without anticoagulant were centrifuged (1000g/15 min) and the sera obtained were stored at -20°C until used.
Serologic test. Serum samples were tested using Enzyme-linked Immunosorbent Assay (ELISA), to determine N. caninum specific IgG which using the Herd Check anti N. caninum commercial kit (IDEXX Laboratories, Westbrook, USA), with 100% sensitivity and 98.9% specificity, according to the manufacturer procedures. To detect positive and negative sera, the test was carried out in paired runs using only one serum dilution (1:100). The cut of value was set at 0.50, considering as positive serum sample that had a mean reading greater than 0.50 (6,7).
PCR probes. Deoxyribonucleic acid (DNA) was extracted from blood samples using the Ultraclean DNA BloodSpin commercial kit (MOBIO Laboratories, Carlsbad, USA) following the manufacturer's instructions, in aseptic conditions. DNA samples were subjected to single tube nested Polymerase Chain Reaction (PCR) probe, with the primers NF1, NS2, NR1 and SR1, using positive and negative controls (5). For PCR assays, DNA concentration in each sample was verified by UV light spectrophotometer and 5 µl of the sample containing 2 µg of DNA, were used in PCR assays. Amplification products were run in 2.5% agarose gels, with a molecular weight marker (Phix 174 DNA, Promega, Madison, USA) in order to estimate product size; gels were stained with ethidium bromide and visualized by UV light lamp. All procedures were carried out in a cabinet under UV light, with new sterile materials and each sample was tested in replicated test. Positive results were those that showed a product with 146 base pairs.
Management practices on farms. A survey was as carried out to each farmer with the purpose to establish the status of each farm, related to health and farm-management conditions. The specific question were related to breeding (use of bulls and/or artificial insemination), presence of other domestic animals in the farm, frequency of abortions, fate of disposable, presence of dogs and/or coyotes in the farm, type of cattle-grazing prairies, use of supplemental feed and sources of drinking water.
Data analyses. Disease seroprevalence, as well as prevalence of parasite DNA in blood were determined and arrange by farm and sex; also animals positive to both tests were identified. Kappa index was calculated to determine concordance between results obtained with both tests (p<0.05). Association between seroprevalence and the factors recorded in the questionnaire was estimated calculating the Odds Ratio (OR) where values above 1 indicated association. The test was corroborated by χ2 test using Yates correction (p<0.05). All tests were done using the EpiInfo software.
Results
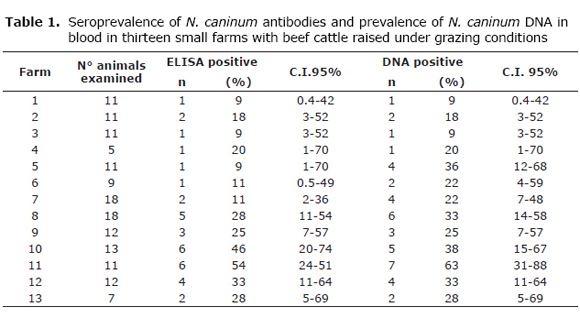
Overall seroprevalence of N. caninum was 23% (35/149); range: 9 to 54%; 95% confidence Interval (C.I.) (17-31). Prevalence of parasite DNA in blood was 28% (42/149; range: 9 to 63%; 95% (C.I. 21-36). (Table 1). All farms studied were found to have positive animals to one or the other test or both.
Seroprevalence in cows was 23% (32/139; 95% C.I. 16-31), and in bulls 30% (3/10; 95% C.I. 0.8-64), while prevalence of parasite DNA in blood was 28% (39/139; 95% C.I. 20-36) in cows and 30% (3/10; 95% C.I. 0.8-64) in bulls.
Twenty eight animals were positive to both tests, corresponding to 25 cows and 3 bulls, representing 19% (28/149) of the animals studied. There were 7 positive animals by ELISA and 14 positive animals by PCR. Concordance between ELISA and PCR was k = 0.63 (95% C.I. 47 - 79).
The presence of dogs was associated with seroprevalence (OR 2.65; C.I. 95% 1.14-6.25; p<0.05). However, it could not be found an association with seroprevalence among farms that presented similar health and farm-management conditions.
Discussion
In different regions of the world the seroprevalence has been found higher in dairy cattle than in beef cattle, mainly attributed to existing differences in management practices (1). In the present study, it was found a higher seroprevalence, more than double of the reported in beef cattle in Mexico (8,9), but lower than observed in dairy cattle (4,10,11).
This results clearly show that animals not only have been exposed to N. caninum infection, as evidenced by the presence of antibodies detected through ELISA assay, but also have a high proportion of active infections supported by parasite DNA detection in blood circulation, therefore real prevalence could be underestimated. This situation also may interfere with the identification of potential infection risk factors. In seropositive animals the parasite DNA detection in blood appears during all gestation, although not of constant form, nevertheless these animals have seropositive offspring and eventually they might abort (12,13). Some studies have reported that seronegative animals, with antibodies level undetectable by the diagnostic test, may be infected and have seropositive offspring (14-16); while another study reported that N. caninum DNA may be identified in heifers' brains without detecting the parasite in blood however their offspring are born infected (12).
Recent study found seronegative animals in which were possible detect N. caninum in the brain tissue by PCR probe (17). Results from this study allow us to conclude that seronegative animals indeed have N. caninum DNA present in their blood and surely are capable of vertical infection transmission or have abortions as the seropositive/positive to DNA detection in blood animals. It is highly likely that some of these animals were probably in the early phases of the infection and therefore diagnostic use of the PCR test may be an aid in the opportune detection of this status with all the epidemiological implications that these results may have.
In this study, 3 bulls came out seropositive and with parasite DNA presence in blood, with a possibility of N. caninum transmission by excretion of viable organisms in semen as has been previously reported (18,19). Currently, there is not much information on what happens in naturally infected bulls, even though the low excretion of tachyzoites in semen as detected in experimental infections hints at a limited possibility for these infections (20), so, further studies in this matter are essential.
The high prevalence found in this study may be only explained by the simultaneous presence of the two transmission routes known for this parasite infection, due to the fact that if vertical transmission were predominant, prevalence would have to be less. Prevalence increases when animals are infected horizontally, as it has already been reported for dairy cattle; dogs and coyotes which have a very important role in horizontal transmission (1). Nevertheless, it has been suggested that this situation is most favored by the coexistence of dogs and cows than by feeding in the same places (21). Most of the farms, raised had dogs, and they were identified as an infection risk factor. Also all the owners indicated that coyotes are frequently seen on the prairies. In this study, dogs and cows were found to share the same areas and sources of drinking water. Also, there was a possible consumption of fetuses and placental waste by dogs. It becomes also important, the needs to be taken into account the presence of coyotes, in order to understand the observed seroprevalence of this parasite.
The seroprevalence in dogs that reside in dairy farms has been reported to be 41% (22). Evidence, that dogs represent an infection risk has not been found in studies carried out in beef cattle in Mexico (8). Similar results have been reported for other regions of the world, where dogs do not live in close contact with cattle. (23-25).
In conclusion, infection by N. caninum in beef cattle belonging to small farms and raised under grazing conditions should be more fully documented. There is not doubt, that an epidemiological control of this disease is necessary to avoid its negative effect on Mexican beef industry.
References
1. Dubey JP, Schares G, Ortega-Mora LM. Epidemiology and control of neosporosis and Neospora caninum. Clin Microbiol Rev 2007; 20: 323-367. [ Links ]
2. Dubey JP, Buxton D, Wouda W. Pathogenesis of bovine neosporosis. J Comp Pathol 2006; 134: 267-289. [ Links ]
3. Morales E, Trigo F, Ibarra F, Puente E, Santacruz M. Neosporosis in Mexican dairy herds: lesions and immunohistochemical detection of Neospora caninum in fetuses. J Comp Pathol 2001; 125: 58-63. [ Links ]
4. García-Vázquez Z, Rosario-Cruz R, Ramos-Aragón A, Cruz-Vázquez C, Mapes G. Neospora caninum seropositivity and association with abortions in dairy cows in Mexico. Vet Parasitol 2005; 134: 61-65. [ Links ]
5. Medina EL, Cruz-Vázquez C, Quezada T, Morales E, García-Vázquez Z. Survey of Neospora caninum infection by nested PCR in aborted fetuses from dairy herds in Aguascalientes, Mexico. Vet Parasitol 2006; 136: 187-191. [ Links ]
6. Wapenaar W, Barkema HW, Vanleeuwen JA, McClure JT, O'Handley RM, Kwok OC, Thulliez P, Dubey JP, Jenkins MC. Comparison of serological methods for the diagnosis of Neospora caninum infection in cattle. Vet Parasitol 2007; 31: 166-173. [ Links ]
7. Wu TJJ, Dreger S, Chow YWE, Bowlby EE. Validation of 2 commercial Neospora caninum antibody enzyme linked immunosorbent assays. Can J Vet Res 2002; 66: 264-271. [ Links ]
8. García-Vázquez, Z., Rosario-Cruz R, Mejía-Estrada F, Rodríguez-Vivas I, Romero-Salas D, Fernández-Ruvalcaba M, Cruz-Vázquez C. Seroprevalence of Neospora caninum antibodies in beef cattle in three southern states of Mexico. Trop Anim Health Prod 2009; 41:749-753. [ Links ]
9. Salinas MJ, Mora J, Zárate J, Riojas V, Hernández G, Dávalos G, Ramírez R, Galán L, Avalos R. Frecuencia de anticuerpos contra Neospora caninum en ganado bovino del noreste de México. Vet Mex 2005; 36: 303-311. [ Links ]
10. García-Vázquez Z, Cruz-Vázquez C, García D, Medina L, Chavarría B. Serological survey of Neospora caninum infection in dairy cattle herds in Aguascalientes, México. Vet Parasitol 2002; 106: 115-120. [ Links ]
11. Gutiérrez GJ, Cruz-Vázquez C, Medina EL, Valdivia A, Islas E, García-Vázquez Z. Factores de manejo asociados con la seroprevalencia a la infección por Neospora caninum, en ganado lechero de Aguascalientes, México. Vet Mex 2007; 38: 261-270. [ Links ]
12. Okeoma CM, Stowell KM, Williamson NB, Pomroy WE. Neospora caninum: quantification of DNA in the blood of naturally infected aborted and pregnant cows using real-time PCR. Vet Parasitol 2005; 110: 48-55. [ Links ]
13. Santana OI, Cruz-Vázquez C, Medina-Esparza L, Ramos PM, Castellanos MC, Quezada GD. Neospora caninum: DNA detection in blood during first gestation of naturally infected heifers. Vet Mex 2010; 41: 131-137. [ Links ]
14. Frössling J, Uggla A, Björkman C. Prevalence and transmission of Neospora caninum within infected Swedish dairy herds. Vet Parasitol 2005; 128: 209-218. [ Links ]
15. López-Gatius F, López-Bejar M, Murugavel K, Pabón M, Ferrer S, Almería S. Neospora-associated abortion episode over a 1-year period in a dairy herd in north-east Spain. J Vet Med B 2004; 51: 348-352. [ Links ]
16. Sager H, Fischer I, Furrer K, Strasser M, Waldvogel A, Boerling P, Audigé L, Gottstein B. A Swiss case-control study to assess Neospora caninum-associated bovine abortions by PCR, histopathology and serology. Vet Parasitol 2001; 102: 1-15. [ Links ]
17. Santos SL, de Souza Costa K, Gondim LQ, da Silva MS, Uzeda RS, Abe-Sandes K, Gondim LF. Investigation of Neospora caninum, Hammondia sp., and Toxoplasma gondii in tissues from slaughtered beef cattle in Bahia, Brazil. Parasitol Res 2010; 106: 457-461. [ Links ]
18. Caetano-da-Silva A, Ferre I, Collantes-Fernández E, Navarro V, Anduriz G, Ugarte-Garagalza C, Ortega-Mora LM. Occasional detection of Neospora caninum DNA in frozen extended semen from naturally infected bulls. Theriogenology 2004; 62: 1329-1336. [ Links ]
19. Ferre I, Anduriz G, Del-Pozo I, Regidor-Cerillo J, Atxaerandio R, Collantes-Fernández E, Hurtado A, Ugarte-Garagalza C, Ortega-Mora LM. Detection of Neospora caninum in the semen and blood of naturally infected bulls. Theriogenology 2005; 63: 1504-1518. [ Links ]
20. Osoro K, Ortega-Mora LM, Martínez A, Serrano-Martínez E, Ferre I. Natural breeding with bulls experimentally infected with Neospora caninum failed to induce seroconversion in dams. Theriogenology 2009; 71: 639-642. [ Links ]
21. Dijkstra T, Barkema HW, Björkman C, Wouda W. A high rate of seroconversion for Neospora caninum in a dairy herd without an obvious increased incidence of abortions. Vet Parasitol 2002; 109: 203-211. [ Links ]
22. Cruz-Vázquez C, Medina-Esparza L, Marentes A, Morales-Salinas E, García-Vázquez Z. Seroepidemiological study of Neospora caninum infection in dogs found in dairy farms and urban areas of Aguascalientes, Mexico. Vet Parasitol 2008: 57: 139-143. [ Links ]
23. Barling KS, McNeill JW, Paschal JC, McCollum III FT, Craig TM, Adams LG, Thompson JA. Ranch-management factors associated with antibody seropositivity for Neospora caninum in consignments of beef calves in Texas, USA. Prev Vet Med 2001; 52: 53-61. [ Links ]
24. Otranto D, Llazari A, Testini G, Traversa D, Di Regalbono AF, Badan M, Capelli G. Seroprevalence and associated risk factors of neosporosis in beef and dairy cattle in Italy. Vet Parasitol 2003; 118: 7-18. [ Links ]
25. Sanderson MW, Gay JM, Baszler BM. Neospora caninum seroprevalence and associated risk factors in beef cattle in the northwestern United States. Vet Parasitol 2000; 90: 15-24. [ Links ]