INTRODUCTION
Some high plants contain biologically active substances with adjuvant-like properties, therefore they can be used in formulation of veterinary vaccines or as supplements in diet 1,2. Furthermore, adjuvants can be used to enhance the immune response to antigens 1,3.
The use of terrestrial plants is the basis of traditional medicine in most ethnic cultures. Of the 300.000 species of flowering plants growing on the planet, studied about 250 000. Of the 350 thousand species of plants in approximately 4% of the studied biological activity 1. A variety of extracts from herbs were discovered that would induce the production of antibodies in various animals 2,4-8. Medicinal herbs play an important role in researching new substances with adjuvant-like properties and are used in veterinary vaccine formulations or as food supplements to reduce the risk of various diseases. Particularly, adjuvants can be used to improve the immune response to antigens for several different purposes, including: 1/ increasing the immunogenicity of weak antigens; 2/ enhancing the speed and duration of the immune response; 3/ modulating antibody avidity, specificity, isotype or subclass distribution; and 4/ promoting the induction of mucosal immunity. IgY antibodies, also called egg yolk immunoglobulins, are the only immunoglobulins in egg yolk and transferred in the female from serum to egg yolk to confer passive immunity to embryos and neonates. According IgY-technology, using birds instead of mammals as the immunization host brings a number of advantages: eggs are cheap and readily available, antibody levels in yolks are high, IgY isolation is fast and simple, IgY as alternative for partually replace of antibiotics 1.
In the recent past, scientific studies on plants used in ethnomedicine have led to the discovery of many valuable drugs. Organic compounds and trace elements are represented in herbs. However, the effect of some extracts from medicinal herbs on immune system has not been studied in a systematic way. The medicinal potential of plants lies in the phytochemical constituents which are responsible for many physiological as well as medicinal activities. Alkaloids are the most diverse type of plant metabolites which possess cytotoxic, analgesic, immunostimulant, antispasmodic and anti bacterial properties. The phenolic compounds like flavonoids, tocopherols and phenolic acids display pharmacological effects such as antiapoptotic, antiaging, anti-inflammatory, cardioprotective, immunomodulatory and anti- angiogenic. However, the effect of some extracts from medicinal herbs on immune system has not been studied in a systematic way 1. Mucosal immunity, in which secretory IgA antibodies play the main important role, is established via the mucosa-associated lymphoid tissues such as those in the respiratory and gastrointestinal tracts. The normal response of the respiratory and gastrointestinal (GI) tracts to any antigens is tolerance rather than immunity and only certain antigens induce a mucosal, peripheral immune response. The normal healthy GI tract is apparently capable to recognize distinguish between safe normal flora and food antigens and dangerous pathogens. The the use of adjuvants-like substances can effectively alter the distinguishing properties of the GI tract 1.
In order to enhance immune response some inorganic substances were applied. In practice different trace elements were used 1, platinum nanoparticles 7,9 or aluminium hydroxide 7,10,11 or iron, selenium and zinc 12 were found to exhibit the stimulating activity to immune system. The extract from Allium sativum and germanium oxide (GeO2) have not been previously examined as potential immunoadjuvants in quails after oral administration. The aim of the present study was to assess the efficacy of quails treated perorally with bovine serum albumin (BSA) in combination with several components: germanium oxide, herb extracts from Allium sativum and Aloe arborescens as mucosal adjuvants The efficiency of the immunity was evaluated by measuring BSA antibodies of IgY in egg yolk and IgA in saliva.
MATERIALS AND METHODS
Animals, feed, and rights statement. Commercial thirty five female Japanese quails (Coturnix coturnix japonica) of seven weeks old were purchased from the local hatchery. Quails each weighing 0.200-0.206 kg were used in the study.
Birds were divided into five groups A (Allium sativum), B (Aloe arborescens), C (germanium oxides), C (IFA) and E (BSA) containing seven birds each. The birds were housed as seven birds per cage of 80 × 60 × 30 cm in a standard animal room on a 16/8 h light/dark cycle (light long 25 lux). As bedding, chips of deciduous trees were used, after sterilization at 120ºC, at a pressure of 1.5 kg/cm², for 20 min. The bedding was changed twice a week. The temperature in the room was 22 ± 1 ºC, with a relative humidity within the range of 55%-60%. The quails were fed with granulated forage (“Kauno grūdai” AB Kaunas, Lithuania), which consisted of digestible energy (880 kJ/100g), crude protein (21.0%), crude fat (3.5%), and crude fiber (15%). The feed was balanced for vitamins and micronutrients, and amino acids. Water was provided ad libitum.
The experiment was approved by authorization and official permissions for growing and commercial use of laboratory animals and products and the permission for the current experimental work (No.: G2-38) were issued by the State Food and Veterinary Service upon the recommendation of the Ethics Commission of Lithuania on the Use of Laboratory.
Antigen. A commercial preparations of BSA (Sigma, USA) was used as a model antigen.
Preparation of herb extract. The spring garlic Allium sativum were collected in 2015 in garden. Leaves of seventh-year-old Aloe arborescens were collected in 2015 in the greenroom of the State Research Institute Centre for Innovative Medicine (Vilnius, Lithuania). And then the 60 g of the garlic cloves of (Allium sativum) and the 250 g of the Aloe arborescens leafs were extracted with 70% ethanol solution during 7 days at room temperature and then remaining aqueous residue were concentrated and dried at 30°C. The yields of the dry extracts Allium sativum and Aloe arborescens were 4.4% and 1.6% respectively.
Germanium oxide. A commercial preparation of germanium oxide (IV), 99.99% (GeO2) was obtained from Alfa Aesar Gmbh and Co KG (Germany) and was used. Incomplete Freund’s adjuvant. The commercial product of incomplete Freund’s adjuvant (IFA) (Bio-Rad, USA) was used.
Experimental design. Quails were randomly divided into five groups with seven quails in each of them. The immunization mixture contained 20 mg of BSA (dose 100 mg/kg) diluted in emulsion of 0.125 mL of phosphate-buffered saline (PBS), pH 7.2 and 0.125 mL of olive oil (Extra Virgin, Carapelli Firenze, Italy). The immunostimulatory components were added additionally.
Group A (Allium sativum). 100.0 mg of dry extract Allium sativum was diluted in 0.25 mL PBS and was mixed with 0.25 mL BSA containing immunization mixture that corresponds to a dose 500 mg/kg.
Group B (Aloe arborescens). 100.0 mg of dry extract Aloe arborescens was diluted in 0.25 mL PBS and was mixed with 0.25 mL BSA containing immunization mixture that corresponds to a dose 500 mg/kg.
Group C (GeO2). 7.0 µg of GeO2 was diluted in 0.25 mL PBS and was mixed with 0.25 mL BSA containing immunization mixture that corresponds to a dose 35.0 µg/kg.
Group D (IFA positive control). The suspension containing 20 mg of BSA in 0.25 ml PBS was emulsified with an equal volume of IFA.
Group E (BSA alone). The mixture contained 20 mg of BSA in 0.5 ml of PBS.
Quails in groups A, B, C and E were administered immunization mixtures orally on days 1, 7 and 14. Quails in groups D were administered intramuscularly into four site of the pectoral muscle of each quial. Inoculation was performed three times (at days 1, 14, and 28). The quails were not anaesthetized during immunizations. Plastic (Luer) was used for oral administration, where hub was supplemented with a feeding needle (length 75 mm, width 15 mm) containing a silicon tip at the end as described previously 6.
Sampling. Eggs and saliva were collected for antibody measurement weekly, beginning 7d after the first injection, and stored at 4°C and -20°C, respectively. The purification of IgY from egg yolk was performed as described earlier 13. Saliva secretion was collected by absorbent filter papers (Whatman No.1, Sigma). Pre-weighed two wicks were placed under the tongue of the hen for approximately 20 s. The wicks were weighed to measure the amount of saliva. The saliva was extracted by adding 400 μL of PBS containing 0.1% Tween 20, pH 7.2, to the Eppendorff tube with the paper wicks and incubating the mixture with slow shaking at 20ºC for 2 h. After this, the extract was used for analysis.
Determination of antibodies. BSA-specific IgA antibodies in saliva and BSA-specific IgY antibodies in egg yolk from all five groups were examined by enzyme-linked immunosorbent assay (ELISA) using Nunc Immuno Plates, (MaxiSorb, F96, Nunc, Denmark) with minor modifications as earlier described 14. Plates were coated with 100 μL/well of BSA (1 mg/mL) diluted in 50 mM sodium carbonate (pH 9.6) and incubated at 4°C overnight. The next day plates were washed with PBS containing 0.05% Tween 20 (PBS-T), pH 7.2 five times. Non-specific protein-binding sites were blocked with PBS containing 7% of skimmed milk (Oxoid, UK), and after 1 h incubation at room temperature, the plates were washed with PBS-T. Extracts of saliva and egg yolk samples were applied to the ELISA plates (100 μL/well), serially two-fold in the blocking solution, and incubated at 4°C overnight. The plates were then washed with PBS-T and then the quail antibodies (IgA and IgY) were detected. To each well of plates 50 μL goat anti-chicken IgA conjugated with horseradish peroxidase (HRP), (Nordic Immunological Lab., The Netherlands) and 50 μL rabbit anti-chicken IgG conjugated with HRP (Nordic Immunological Lab., The Netherlands), were added respectively and incubated at 4°C overnight. After washing, bound antibodies detected with o-phenylendiamine (Sigma, USA) in 0.05 M phosphate-citrate buffer, pH 5.0 containing 0.012% H2O2. Colour developed for 30 min at room temperature. The reaction was stopped with addition of 50 μL per well 1.25 M H2SO4. The antibody titres were determined as the reciprocal of the highest dilution of saliva or yolk (the optical density was measured at 492 nm) (Titertek Multiscan Plus MK II, Labsystems Finland), which generated a 2-fold higher color signal than that of the negative samples. The titres were converted to a base-2 logarithmic scale. The geometric means (GM) were calculated from GM=Σ antilog2/n, where the numerator is the sum of the antilogarithms of all titre values (in log2) and the denominator is number of samples 15.
IgY calculation. The IgY total content (mg/ml) was measured photometrically at λ=280 nm and was calculated according to the Lambert-Beer law with an extinction coefficient of 1.34 for IgY by means of “BioPhotometer” (Eppendorf, Germany).
Statistical analysis. Statistical evaluation of the results was done by one-way analysis of variance ANOVA using PRISM Software (Graph Pad Software, San Diego, CA, USA). The mean of the IgA, IgY antibody titres and total IgY content (were compared using paired, two-tailed t-test. All values were expressed as mean ± standard deviation and were considered to be statistically significant at p<0.05.
RESULTS
Figure 1 shows that antibodies in saliva were detected in groups A, B and C during the observation period. Aside from that in group C the IgA titres in saliva were significantly higher than those in other groups at all days of the observation. In contrast, in groups D and E (negative control) the IgA titres were below log2 2.4 during the observation period. Generally, anti-BSA IgY antibodies in egg yolk were found in groups A, B, C and D throughout the observation period (Figure 2). In part, the IgY titres of group D (positive control) were significantly higher than those in other groups during the observation period. Notably, IgY titres were significantly lower within one to six weeks after the last immunisation only in group E (BSA alone).
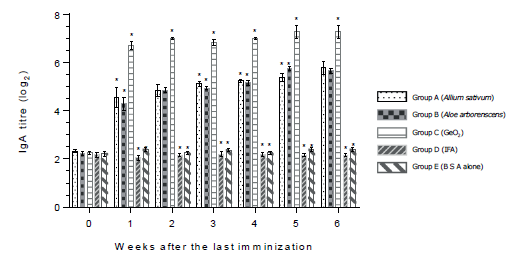
Figure 1 Saliva IgA antibody response (log2) in quails after the oral immunization with bovine serum albumin in combination with herb extract from Allium sativum or Aloe arborescens and GeO2. Asteriks indicate groups the values of which differ significantly from those of other groups on the same week.
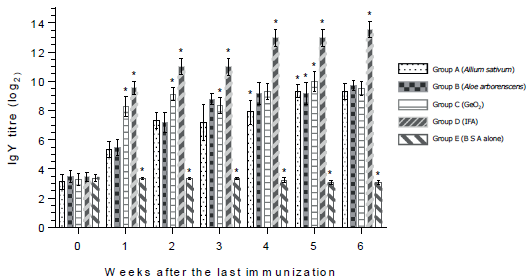
Figure 2 Egg yolk IgY antibody response (log2) in quails after the oral immunization with bovine serum albumin in combination with herb extract from Allium sativum or Aloe arborescens and GeO2. Asteriks indicate groups the values of which differ significantly from those of other groups on the same week.
As seen from Figure 3, the total IgY content in egg yolk among groups A, B, C and D varied from 21 to 40 mg/ml of egg yolk. Beyond that the total IgY concentration in groups C and D reached 41 and 42 mg/mL of egg yolk, respectively. In group E the total IgY content was significantly lower than those in other groups at all time-point of the observation after the last immunization.
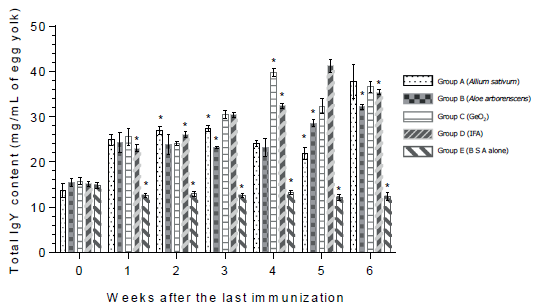
Figure 3 The total IgY content (mg/mL of egg yolk) in quails after the oral immunization with bovine serum albumin in combination with herb extract from Allium sativum, or Aloe arborescens and GeO2. Asteriks indicate groups the values of which differ significantly from those of other groups on the same week.
DISCUSSION
The usual response of the gastrointestinal tract to antigens is rather tolerance than immunity as described formerly 16,17. A combination of soluble antigen, boosting element and the administration protocol have been precisely selected. In the present investigation the quails were used. Our model of study is tightly composed and corresponds to the other studies where BSA was used as the antigen for provoking immune response in hens 5,6,8,18. Firstly, an antigenic capacity of BSA is enough to cause gentle immune response 19, but usually it is combined with the immunostimulator as a booster element. Secondly, the immunization mixture can be enriched additionally by emulsifying with an oil to provoke a prolonged enhancing effect 20.
It occurs due to the position of the soluble antigen in the internal aqueous phase that provides a slow release into the biological fluids. Apart from that an antigen inoculation via oral entry causes a reactivation of secretory as well as humoral components of the immune system. We assessed the immunostimulatory capacity of extracts from Allium sativum, Aloe arborescens, as well as germainium oxides under described conditions. In this study, we have demonstrated that oral administration with BSA in combination with the extracts from Allium sativum, Aloe arborescens, and as well as germanium oxides could also prime the immune system for both secretory and systemic antibody responses. It is shown that the elicited immune response can persist for longer period, even until 1.5 months, following the first administration. These data the supplement previous reports noted that biologically active constituents of plants as Uncaria tomentosa, Vinca major, Vitex agnus-castus, Hedysarum neglectum, Ginkgo biloba and Ocimum sanctum4-7,21 as well as some trace elements (aluminium, platinum) are essential for the development and maintenance of humoral and secretory immunity 6,7.
A large number of sixteen rare earth elements and thirty-one trace elements were found in Shilajit and which caused a strong immune response in chickens 1,7. So it is likely that the high contents of the element are so-called hub-plants can have a high degree of inducing the immune system. In other work was described that feeding with Aloe vera extract stimulated increase of CD4+ lymphocyte in blood and in serum IgM and IgG antibodies over 21 day 22. Using platinum salt-treated animals cellular and humoral responses in guinea pigs 23 and in mice 24 were shown detected. In fact, in Allium sativum, Aloe arborescens was found germanium in concentration of 754 ppm and 80 ppm, correspondingly 25,26. In order to increase the concentration of germanium in extracts from Allium sativum, Aloe arborescens25 higher doses for administration in quails were used.
Trace elements were used for comparative analysis with plant extracts. Also they are important for functioning of various components the immune system. Metals which often concentrated into the medicinal plants perform the functions as cofactors. Such as metals Co, Zn, Fe, Cu, Mn, Cr are participating in the metabolism at various molecular levels. Also divalent metals can complexed with organic compounds of low molecular weight and with proteins 1. It was that between the cadmium or marganese content and IgG concentration inverse correlation. On the contrary a direct correlation between magnesium, calcium and cobalt content and circulating immune complexes had been detected. In addition, was observed that production of cytokines can be activated with manganese. Zinc content in serum elicited IgA, IgG and IgM immunoglobulins 1. Data of the investigations shown that using herb extracts can stimulated immune system after mucosal administration as well as supplements the various plant in feed animals which can magnify for certain physiological functions, such as immunomodulation. May be hypothesized that the degree of influence on the microenviromental in vivo depends from ionization energy. Which is the sum of two parameters the total trace elements content and ionization energy of trace elements. In particularly, our findings show that aluminium, platinum and germanium oxides may be used for immunomodulation in vivo. Furthermore, our data shown that interaction between elements which contain in used plants can influence on the immune response in rabbits and birds. On the other hand, organic components containing in plant such as polysaccharide, alkamides, and triterpenoid saponins would elicited immunity. There is still a need to clarify the phytochemical composition and the mechanisms of action for many herbs, spices and their extracts and furthermore, to assess the appropriate dose that should be safely used in specific circumstances and animal species 1.
Additionally we evaluated the total IgY content in egg yolk. The IgY content follows clear age-dependent kinetics which was confirmed as noted earlier 6. High content of IgY persists in egg yolk longer than one year and later it slightly decreases 27. Furthermore, the fluctuation the immunoglobulin may be reflect the biological activity of quails B lymphocytes, in view of a rhythmic production of different amounts of IgY. Our findings present that all extracts and GeO2 can be advised for immunostimulatory application, as adjuvants in quail models 1. This work is a first report on the influence of germanium oxides on secretory and humoral immune responses in quails.














