Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
CT&F - Ciencia, Tecnología y Futuro
Print version ISSN 0122-5383On-line version ISSN 2382-4581
C.T.F Cienc. Tecnol. Futuro vol.1 no.5 Bucaramanga Jan./Dec. 1999
PRESSURE CONTROL IN VACUUM TOWERS
2Ecopetrol - Instituto Colombiano del Petróleo, A.A. 4185 Bucaramanga, Santander, Colombia
Abstract
The increase in severity of visbreaking relates to an increase in olefin concentration in the visbreaking pitches. To conditions of the recovery of gas oils in the vacuum distillation tower, these olefins undergo oligomerization reactions, which produce sludge/polymers. The oligomerization reactions are favored by high temperatures, increment in the times of residence, free radicals and metals that can act as catalysts. The polymers are deposited in the Glitsch meshes and they become traps for the asphalt that are carried by the vapor currents. As a consequence of this process, the free area of the meshes is decreased and the linear rates of the vapors are increased. In time, an increment is registered in the pressure drops of the meshes as well as a growing contamination of the gas oils. The experimentation demonstrated high solubility of the components that cause this contamination. As a result of the study, a methodology for online tower clearing was put into practice and the problem indicated was controlled.
Keywords: visbreaking, sludge, resins, vacuum tower.
Resumen
Se implementa una nueva metodología para lavar el empaque de la torre de vacío de una planta viscorreductora evitando su ensuciamiento. El incremento de la severidad en la viscorreducción conlleva a un aumento en la concentración de olefinas en las breas viscorreducidas. A condiciones de la recuperación de gasóleos en la torre de destilación al vacío, estas olefinas se oligomerizan produciendo gomas. La reacción de oligomerización se favorece por las altas temperaturas, incremento en los tiempos de residencia, presencia de radicales libres o de trazas de metales que pueden actuar como catalizadores. Las gomas se depositan en las mallas Glitsch y se convierten en trampas para los asfaltenos arrastrados por las corrientes gaseosas. Como consecuencia de este proceso, se va reduciendo el área libre de las mallas y se incrementan las velocidades lineales de los vapores. En el tiempo se va registrando un incremento en las caídas de presión de las mallas y una creciente contaminación en los gasóleos. La experimentación demostró una alta solubilidad de los componentes causantes de este ensuciamiento. Como resultado del estudio se puso en práctica una metodología de lavado en línea de la torre y se controló el problema enunciado.
Palabras clave: viscorreducción, gomas, resinas, torre de vacío.
Introducción
The visbreaking process is based upon the thermal breaking of the heavy hydrocarbons that make up the bottoms of the barrel Table 1 to obtain more valuable products as naphtha, fuel oils and gas oils for the subsequent catalytic upgrading of these distillates (Yan, 1987 and 1989; Dwijen et al., 1986; Cano et al., 1981; Bravo y Campos, 1984; Kuo, 1984).
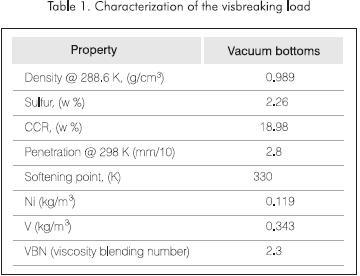
The visbreaking run length depends on the fouling rates of the furnace and distillation towers Figure 1. These rates are favored by the high severity of the operation and content of asphaltenes in the feedstock. During the length of the runs pressure drops (DP) are registered at the inlet and outlet of the furnace and distillation towers. In the visbreaking processes at moderately high severity, as the ones described befw, the length of a run prior to the decoking of the furnace is up to 365 days, while the cleaning of the vacuum distiller is carried out every 120 days. When operational problems, changes in the quality of the feedstock or sudden operational changes arise, the length of the run decreases forcing a cleaning of the towers every 90 days (Del Bianco, 1993).
PROBLEM DESCRIPTION
Unpredictable situations arise in some visbreaking plants hindering normal operation and demanding an individual analysis in order to discover their influence and effects on the plant. Some of these situations are:
Pressure drop increments (ΔP) in the furnace
They are caused by increments in the temperature and/or time of residence (severity) of the operation, and by the accumulation of mineral salts on the internal wall of the tube, which progressively reduce its effective diameter and also increase the roughness that acts as a promoter for coke deposits.
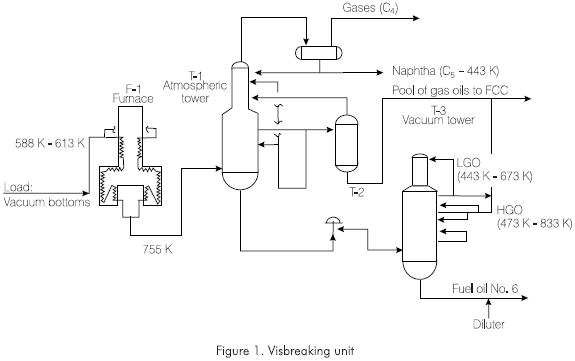
The severity is controlled by means of the Merite test (Kuo, 1984), on a 0 - 10 scale, trying to run the furnace so as to obtain a 6.5 - 7.5 interval (Figure 2). For the Merite test, 2 g of visbreaking pitch are dissolved in 1·10-5 m3 (10 ml) of a solvent mixture composed by oxylene and ioctane. A drop of this solution is deposited on filter paper where it spreads forming one or two concentric circles. The value of this test corresponds to the proportion of oxylene in relation to the mixture that blurs the internal concentric circle (Figure 3). The ideal value of the Merite test is 7 or 70% oxylene. A higher value indicates overcracking of the vacuum bottoms in the furnace tubes. On the other hand, a lower one indicates mild cracking.
Additionally, the severity is controlled by the stability factor (Kuo, 1984) and by the coking index. This index is obtained by dividing the pressure drop (Pa) by the load flow (m3/h). The coke formation is controlled by means of the usage of additives such as the AVB-95 developed by the Instituto Colombiano del Petróleo that can reduce coke formation up to 48% (Carrillo and Pantoja, 1998).

Increments in the pressure drops (ΔP) of the towers
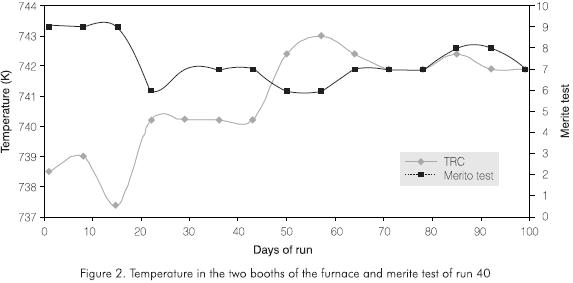
They are caused by the fouling of the first fractionating trays or of the Glitsch mesh beds as the run advances Figures 4 and 5.
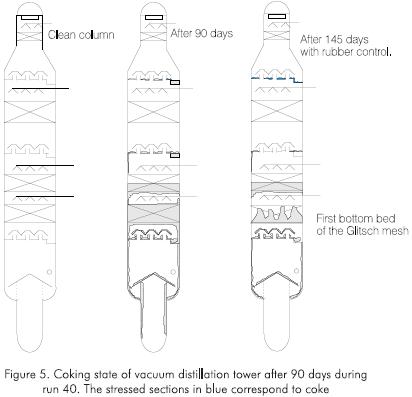
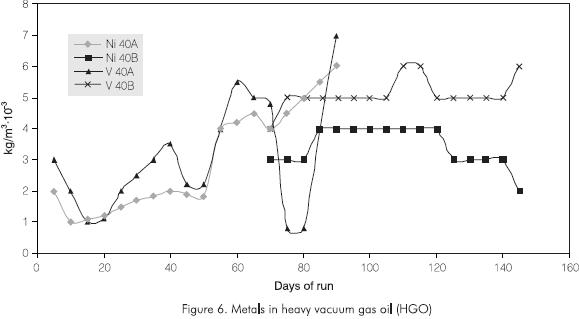
The fouling increases in the trays or beds closer to the bottom of the columns. As the run advances, nickel (Ni), vanadium (V) and Conradson Carbon (CCR) concentration increases in gas oils because the flow is channeled causing an increase in the speed of the vapors that favor the entrainment of volatile materials Figure 6.
This fouling is caused mainly by:
- Entrainment of asphaltenes and resins from the area of instantaneous vaporization (flash area).
- Formation of rubbers as a result of the reactions of oligomerization of olefines present in the products of the reaction (Zanotti and Magri, 1997).
- Entrainment of solids together with the cycling gas oils due to detachments of hcoke from the Glitsch mesh, which arrive to the jdowncomer of gas oils feeding the cycling pump.
In the case of the vacuum tower, the critical fouling point corresponds to the Glitsch meshes closer to the bottom. At this point, the DP is 0.33 kPa (2.5 mmHg) at the beginning of the run increases to a 5.08 kPa (38.1 mmHg) value (Figure 4). When the maximum allowed pressure is reached, the operation of the tower is stopped in order to clean the tower. At this time, the contamination of the gas oil exceeds the values allowed for nickel, vanadium and CCR.
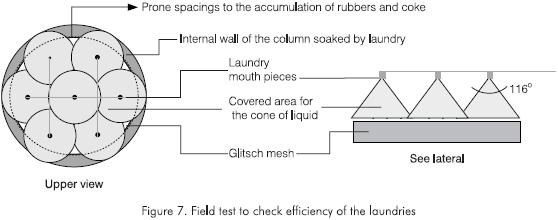
The first tray is provided with a cleaning system whose effectiveness and coverage is controlled through dispersion mouthpieces and through their location in relation to the tray. The liquid used for the cleaning the first bottom tray is a heavy oil. As the run advances, the mouthpieces clog partially and the coverage drops below 70%, a situation that facilitates the deposition of resins and asphaltenes on the grills of the Glitsch trays increasing the ΔP. The flow through the cleaning mouthpieces is controlled according to the recommendations of the makers of the outlets selected for each tray (Koch Engineering and Spraying System) and ranges between 1.4 ·10-4 m3/m2s and 4.1·10-4 m3/m2s. The compliance with this requirement is verified during the inspection of the tower Figure 7.
The cleaning oil, with an olefin concentration of about 50% in weight (Koch Engineering and Spraying Systems) due to its visbreaking product properties, has a sharp polymerization tendency. This tendency increases as the paraffin content of the feedstock increases. The oligomerization not only affects the fouling of the Glitch tray, but it also increases the viscosity of the visbreaking bottom, thus demanding larger quantities of diluent for the fuel oil preparation causing an economic strain.
It is therefore advisable to control the conditions favoring oligomerization. Some of these factors are:
- Catalysts. The oligomerization reactions can be homolytic (by means of free radicals) or heterolytic (by means of catalysts). Temperatures of about 613 K - 630 K (340°C - 357°C) can be found under the operating conditions of the vacuum tower bottoms. At these temperatures, the thermal bond rupture reactions are slow. However, it is accepted that at 623 K (350°C), thermal decomposition of the barrel bottoms begins therefore, this temperature is the maximum permissible one for the distillation. This thermal decomposition of the hydrocarbons is so negligible that is not expected to contribute in the formation of coke, nevertheless, it is a source of free radicals. The oligomerization reactions by heterolytic mechanisms are catalyzed by the iron and the transition metals such as nickel and vanadium present in the reaction (Fred and Patrick, 1988). The iron catalyzes the decomposition reactions of the peroxides and increases the speeds of the Diels- Alders reactions, which generate the initiators of the free radicals.
- Oxygen content. The oxygen present in the feed- stock causes the formation of peroxides, which that initiate fouling even at relatively low temperatures (Yan, 1987).
- Temperature. Temperatures ranging from 273 K (0°C) to some hundreds of degrees centigrade favor the initiation of the oligomerization reactions. The condensation reactions, which are responsible for the coke formation, present high activation energy (63.9 kcal.mol-1). As a consequence, these reactions accelerate as the temperature increases (Fred and Patrick, 1988).
- Residence time. The molecular mass growth in the products of the reaction is favored by the increment in the time of residence, which is directly proportional to the diameter of the heavy oil recirculation line and to the level of the chimney plate.
- Cleaning of the trays. The hydrocarbon vapors can drag some porphyrin asphaltenes that have a certain volatility as well as resins and light rubbers from olefin polimerization. These substances are dragged as tiny drops that stick to the Glitsch tray and as the run advances, the quantity of rubbers increases reducing the free area of the mesh. The asphaltenes dragged by the vapors are caught by the resins forming a mortar similar to cement. In time, this mass becomes the mesophase (incipient coke), which hardens progressively. A faulty cleaning of the trays is the main cause for the presence of oligomerization reactions. This faulty cleaning can be caused by:
- Operation under cycle oil flow rates below the conditions required to guarantee a total and even coverage of the channel or with cycle oil flow rates above the maximum allowed for the mouthpieces that causes excessive cloudiness, dragging of liquids towards the upper meshes and loss of cleaning efficiency.
- Use of mouthpieces below specifications causing conditions of excessive cloudiness before reaching the minimum flow required to obtain an appropriate cleaning of the meshes.
- Mistakes in the design or installation of the cycle oil flow plate.
Additives such as alkylsuccinamides for naphtenic crude oils and pyrocatechol byproducts for all types of crude oils are utilized to control the polymerization reactions. These substances not only serve to block the polymerization reactions, but also reduce the production of asphaltenes allowing larger conversions.
ANALYSIS OF RESULTS
Pilot plant tests.
Two pilot plant tests were carried out to obtain a better understanding of the coking phenomenon of the distillate column beds. The industrial plant conditions were simulated for the operation with three-minute residence times, as well for sudden shutdowns caused by electric failures that cause a 24-hour residence period Table 2.
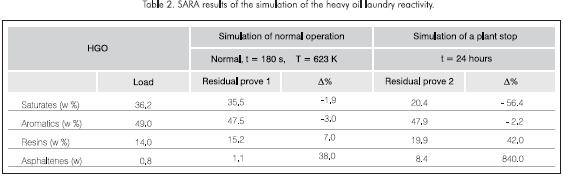
In both simulations a reduction in the contents of saturates and aromatics as well as an increase in the concentration of resins and asphaltenes was detected.
These changes are dramatic for the simulation of a sudden stop of the plant for 24 hours and indicate the presence of an olefin oligomerization reaction.
The results shown in Table 3 indicate that after the normal cleaning of the tower, there is 33% w - 56% w of toluene solubles and between 52% w - 65% w toluene + N-methyl-pyrrolidine (NMP) solubles in the coke.
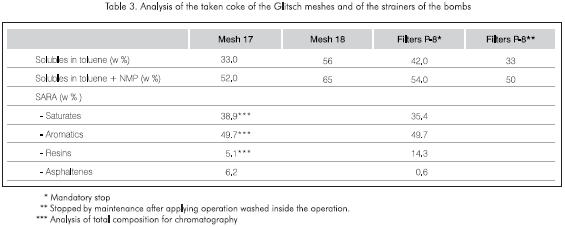
The SARA analysis of the cleaning oil subjected to the simulation of operation conditions in the industrial column has a composition similar to that of the solubles from the coke removed from the Glitsch meshes, thus confirming that the clogging of the Glitsch mesh is mainly caused by olefin oligomerization, which in turn has an effect on the concentration of resins and asphaltenes.
Industrial test
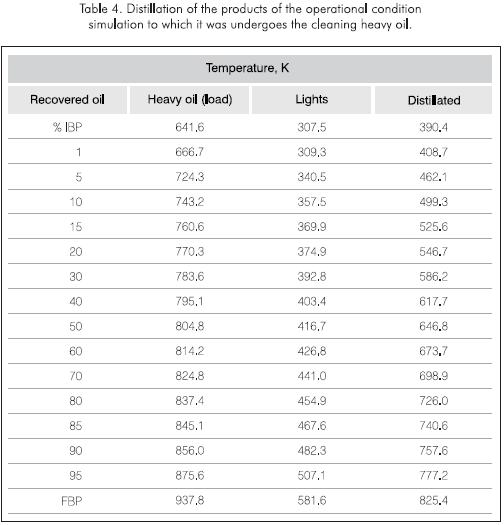
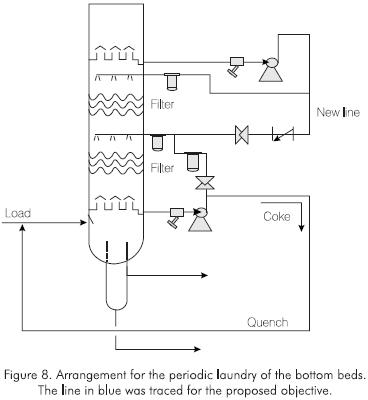
The quantity of soluble material in the coke from the industrial tower was verified after shutdown and cleaning to begin the maintenance procedures (Table 3). The smaller quantities of resins and the larger concentrations of asphaltenes in the meshes in regard to the filters are an indication of their evolution towards asphaltenes during the run (Figure 8). In both simulations, similar quantities of gases (0.5% w), light products (0.6% w - 1.0% w) and medium distillates (1.5% w - 2.0% w) were produced (Table 4).
Based on the information given above, the following changes were considered to control the coke formation mechanisms during the second half of run 40B in the visbreaking plant at Ecopetrol's refinery in Cartagena:
- To set up a cleaning line for the bottom tray with light vacuum gas oil from the same tower. This line is showing in Figure 9. The purpose of this arrangement is to have easy access to a clean and light aromatic solvent to clean the tower every two weeks. With this arrangement, the coke from the Glitsch mesh is sent to the bottoms with the quench of the feedstock.
- To increase the cleaning flow for the bed from the 381 m3 - 636 m3 interval to the 668 - 986 m3 interval. According to the field tests, mouthpieces with a 0.00254 m diameter guarantee even coverage of the mesh hindering oligomer deposition. The flow control restriction plate was recalculated for this purpose. With the implementation of these changes, the operation of the plant was optimized under the following factors:
- Conversion was kept at 38% (typical value 39%).
- Heavy gas oil contaminants remained within the interval allowed: Ni < 6·10-3 kg/m3, V < 5·10-3 kg/m3 and CCR < 0.5% w, with lower values in comparison to run 40A.
- The inspection carried out at the end of the run showed no clogging of the mouthpieces, an unprecedented fact since normally two out of seven get clogged.
- The first Glitsch mesh bed was filled up to 50% with coke in its lower section, which rose to the upper section of the mesh forming small mounds in areas not covered by the mouthpieces. This coke has a porous and fragile appearance. The second Glitsch mesh bed was filled up to 40% with coke in its lower section. When comparing the fouling pattern of the Glitsch meshes against the reference runs, we find that the quantity of coke showed a 60% decrease in the first bed and that a porous, fragile and easily removable coke had formed instead of a bituminous and soft one. It is necessary to consider that run 40B was prolonged for more hours than run 40A.
- The ΔP of the vacuum tower that normally started a sudden increase after 50 days remained constant up to 80 days at 2,4 kPa (18 mmHg). From day 80 to 110, the DP increased and reach to a permanent value of 3,3 kPa (25 mmHg) until the end of the run at day 145.
CONCLUSIONS
- The pressure drop in the Glitsch meshes are caused by the formation of sludge that stick to the meshes and to the deposition of asphalts.
- The oligomerization reactions are favored by long residence periods, high temperatures, and the presence of metals that act as catalysts as well a faulty cleaning of the Glitsch meshes.
- The cleaning of the meshes controls the formation of rubber as it dissolves them thus decreasing the pressure drops and the contamination of the gas oils.
REFERENCES
Bravo, M. M. y Campos, B. P., 1984. "Sistema de cálculo para la estimación de rendimientos y propiedades de productos en una unidad reductora de viscosidad", Revista del IMP, XVI (3): 76 - 83. [ Links ]
Cano, J. L. et al, 1981. Incorporación del proceso de reducción de viscosidad a la tecnología del IMP, Revista del IMP, XIII (1): 53 - 62. [ Links ]
Carrillo J. and Pantoja F., 1998. "Aplicación del aditivo AVB- 95 a las plantas de viscorreducción. 216 TH", American Chemical Society, National meeting and exposition program, Boston Massachussets (August 23 - 27). [ Links ]
Del Bianco, A. et al, 1993. "Thermal cracking of petroleum residues", Fuel, 72: 75 - 80. [ Links ]
Dwijen, K. et al, 1986. "Kinetic studies of coke formation in hydrocarbon fractions of heavy crudes", Fuel, 65 (April): 480 - 484. [ Links ]
Fred, M. and Patrick, L., 1988. "Treating the train", ECN Chemscope (May): 8, Industrial Spray Nozzles, Systems and Accessories. LECHER. Catalog N°140. [ Links ]
Koch Engineering Company Inc. and Spraying Systems Co. "Nozzle Data", Mass transfer, Refining application, bulletin N° WZ - 1. [ Links ]
Kuo, C. J., 1984. "Effect of crude types on visbreaking conversion", Oil & Gas Journal (Sept 24):100 - 102. [ Links ] Yan, T. Y., 1987. "Coke formation in visbreaking process. Symposium on advances in resid upgrading", American Chemical Society, Denver meeting (April 5 - 10): 490 - 495. [ Links ] Yan, T. Y., 1989. "Characterization of visbreaking feeds. Symposium on correlation between resid characterization and proccessability", American Chemical Society, Dallas meeting (April 9 - 14): 428 - 433. [ Links ] Zanotti, A. and Magri, F 1997. "Method and additive to increase yield in thermal conversion operations within petroleum plants", European Patent EP 0818524 A1. [ Links ]














