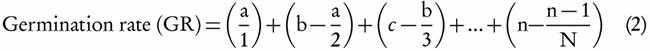Introduction
Engineered nanomaterials (ENMs) have many unique properties such as their small size, large surface area, chemical composition, surface reactivity, charge, shape, and media interactions. These physical, chemical, thermal, magnetic, optical, and biological properties have resulted in numerous applications including use in food and common everyday products [1]. Titanium is the ninth most abundant element and the second most abundant transition metal in the earth’s crust [2]. Anatase, rutile, and brookite are common forms of TiO2 [3]. Titanium dioxide NPs are among the ten most commonly produced ENMs that are used worldwide [4] with global production of this particle now standing at 10 000 tons per year [5]. Commercial production of TiO2 NPs has been estimated to reach approximately 2.5 million tons by 2025 [6]. Titanium dioxide NPs are commonly used in a broad range of products such as paints, papers, inks, coatings, catalysts, solar cells, plastics, soaps, antimicrobial and antifungal agents, alloys, textiles, food products, toothpaste, sunscreen and cosmetics, medicines and pharmaceuticals [7], in certain sectors of agriculture [8], and in environmental cleanup technologies [9]. Recently, TiO2 NPs have been reported to improve seed germination and plant growth [10-12]. For example, simultaneous application of SiO2 and TiO2 nanoparticles accelerated germination and growth of soybean (Glycine max) plants by improving water uptake and increasing the activity of nitrate reductase and antioxidant system [13]. Positive effect of TiO2 nanoparticles on spinach germination is likely related to the small particle size which enables TiO2 NPs to penetrate into the seed and promote germination and growth [10].
Abiotic stresses such as drought, salinity, heavy metals, and temperature affect germination, growth, and some physiological processes in plants [14]. Cadmium (Cd) is considered to be one of the most phytotoxic heavy metals which can be released into the environment via different agricultural or industrial activities. Soil pollution by Cd in many agricultural fields is mainly attributed to the prolonged application of phosphate fertilizers or field irrigation through sewage sludge or waste water [15,16]. Nitric oxide (NO) is known as a gaseous signaling molecule that participated in some critical physiological process within the plants such as seed germination, cell death or expansion, stomata closure and root growth [17-20]. Nitric oxide plays a key role in seed germination of different species under stress condition [21,22]. Nitric oxide is well known to play a dual role in the stress responses of plants. In particular, NO can directly scavenge certain reactive oxygen species (ROS), such as superoxide anions and lipid-derived radicals, and was proven to induce antioxidant enzymes thereby decreasing oxidative damage [17,23]. In recent years, there is abundant evidence to support the protective role of exogenous NO released by a NO donor; such as SNP in alleviating heavy metals-induced phytotoxicity. For example, pretreatment with SNP ameliorated toxic effects of Cd on wheat [24,25] barley [26] and rice [27] by boosting the antioxidant defense system.
Seed germination and root/shoot elongation tests are considered as sensitive, fast and effective methods to assess the phytotoxicity of nanoparticles in plants [28]. Different test media such as culture solution [29], agar medium [30], filter paper on petri dish [31] and soil [32] have used in literature to evaluate the impact of nanoparticles on seed germination and early growth of various plant species. Filter paper on petri dish method which used in current survey appeared to be simple and widely-used method in nanoparticle phytotoxicity experiments [33]. Wheat (Triticum aestivum) was selected as plant material for the current study as recommended for the testing of chemicals by USEPA [34] and OECD [35].
Although both stimulatory and inhibitory impacts of nanoparticles on various crop species have been reported in the literature, however information regarding nanoparticles function under heavy metal stress is still limited. To the best of our knowledge, this study is the first report on the protective role of TiO2 NPs and SNP in ameliorating the adverse effects of Cd on seed germination and seedling growth of wheat. The data obtained in this survey can pave the way for further investigations in this field and shed lights on new applications of nanoparticles, in particular, TiO2 nanoparticles accompanied by stress-ameliorating compounds such as SNP in alleviating Cd stress in plants and improving plant growth under unfavorable condition.
Materials and methods
Plant material
Wheat (var. Pishgam) seeds were supplied by the Seed and Plant Improvement Institute (SPII), Karaj, Iran. Seeds were stored in a dry place at room temperature before use. The average germination rates of the tested seeds were greater than 90%, according to our preliminary study.
Nanoparticle Characterization
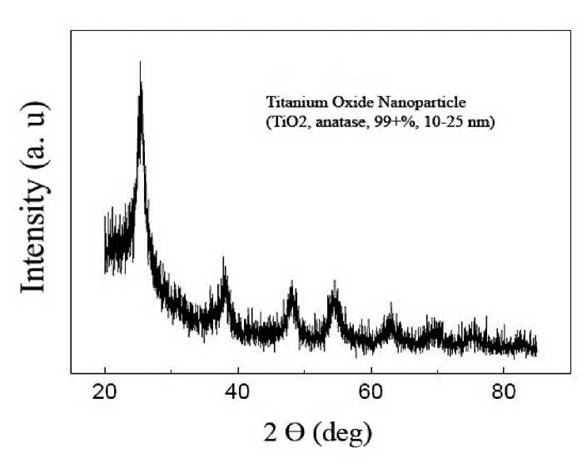
Titanium dioxide NPs were procured from Iranian Nanomaterials Pioneers Company (NANOSANY), Mashhad, Iran. According to the data provided by the manufacturer, TiO2 NPs used in this study had the following features: primary size: 10-25 nm, surface area: 200-240 m2. g-1, pH: 6-6.5, bulk density: 0.24 g. cm-3, true density: 3.9 g. cm-3, and 99% purity. A transmission electron microscopy (TEM) image of the TiO2 particles is shown (Fig. 1). The crystal properties of TiO2 NPs were examined by X-ray diffraction (XRD). The XRD measurement showed that used TiO2 NPs were all present in the anatase form (Fig. 2).
Preparation of nanoparticle suspensions
Stock suspensions of TiO2 NPs (2 000 mg/L) were freshly prepared by dissolving the nanoparticles directly in deionized water and dispersed by ultrasonic vibration (100 W, 40 kHz) for 30 min. Titanium dioxide NP suspensions at the concentration of 0,500, 1000, and 2000 mg/L were prepared by dilution of the stock suspensions with deionized water. The suspensions were stirred for 1 min using small magnetic bars before use to avoid the aggregation of nanoparticles.
Stress treatments
Cadmium stress imposed by 50 and 100 mM CdCl2. Sodium nitroprusside was used as NO donor at concentrations of 0 and 100 μM [36]. Then, seeds were subjected to the following basic treatment protocols as (a) TiO 2 , b) Cd stress, c) TiO2 + Cd stress and d) TiO2 + Cd stress + SNP. The treatment with deionized water served as the control.
Seed germination and seedling growth
Seeds of similar size were randomly selected and immersed in a 5 % sodium hypochlorite solution for 10 min and rinsed several times with distilled water to ensure surface sterility [34]. Sterilized seeds were evenly arrayed on 9 cm Petri dishes containing filter papers moistened with 9 mL of test solution. Petri dishes were sealed with tape to avoid evaporation and placed in a germinator under dark at 25 ± 1 °C for seven days. Germination data were recorded at every 24 h interval following the International Rules for Seed Testing Association, ISTA [37]. Seeds were considered germinated when the radicle reached at least 2 mm in length [38]. At the end of the exposure, length and fresh weight of roots and shoots were measured using a ruler and digital balance respectively.
Germination percentages was calculated by the following equation [39]:
Where, Ni is the total number of germinated seeds at the end of experiment and N is the total number of seeds used in the test.
Germination energy was computed as describe by Amooaghaie et al. [40]: GE = number of germinated seeds after three days/number of total seeds
Germination rate was determined based on Maguire [41]:
Where, a, b, c, and n are numbers of germinated seeds after 1, 2, 3, and N days from the start of imbibition. Mean Germination Time was recorded according to Ellis and Roberts [42]:
Where, Ni is number of germinated seeds till ith day, Di is number of days from start of experiment till ith counting, and T is total germinated seeds. MGT is a measure of the average length of time required for maximum germination of a seed lot.
Seedling vigor was computed based on Vashisth and Nagarajan [43]:
Vigor index (VI) = germination % x seedling length (root + shoot)
Experimental design and statistical analysis
A factorial experiment was arranged based on a completely randomized design (CRD). All germination tests were performed in triplicate and data were reported as the mean of three replicates. To compare treatments, least significant difference (LSD) tests were performed using the Statistical Analysis System (SAS, version 9.3). In all cases, p < 0.05 was considered significant.
Results and Discussion
Titanium dioxide and SNP suspensions either solely or in combination efficiently influenced seed germination under normal condition (Fig. 3). Germination percentage significantly was promoted up to 100 % upon seed exposure to 1 000 mg/L TiO2 + SNP (100 μM) treatment while germination reached 90 % in the unexposed control treatment. A dose-dependent sharp decline was recorded in seeds treated by CdCl2 as a Cd stress agent, so germination percentages dropped to as low as 41 % while the Cd concentration increased in the test medium (Fig. 3). Nevertheless, treatment by TiO2 and SNP solutions in mixture neutralized Cd-induced adverse effects on seed germination. For instance, 1 000 mg/L TiO2 combined with 100 μM SNP promoted seed germination percentage up to 100% and 91% under 50 mM and 100 mM Cd, respectively (Fig. 3). Root and shoot length effectively influenced by TiO2 and SNP suspensions in the absence of Cd toxicity (Fig. 3). Cadmium stress immensely diminished root and shoot length in wheat seedlings, especially under higher Cd concentration. However, TiO2 + SNP treatments significantly ameliorated reductions in root and shoot length triggered by Cd stress. Similar to the response of root and shoot length to TiO2 + SNP treatment, both root and shoot fresh weight responded positively to TiO2 NP and SNP suspensions. As shown in Fig. 3, 500 and 1 000 mg/L TiO2 + SNP (100 μM) treatments notably enhanced root and shoot fresh weight under normal conditions. Cadmium treatment dose-dependently decreased root and shoot fresh weight in wheat seedlings; however, declining trends in root and shoot fresh weight caused by Cd toxicity significantly reversed by protective role of TiO2 and SNP suspensions in alleviating negative effects of Cd on root and shoot fresh weight. Compared to untreated control seeds, mean germination time decreased in seeds exposed to TiO2 + SNP treatments. In contrast, 500 mg/L TiO2 combined with 100 μM SNP significantly accelerated mean germination time in treated seeds under non-stress conditions. Exposure to different Cd concentrations postponed mean germination time in wheat seeds. Nevertheless, mean germination time in Cd-exposed seeds was positively accelerated by adding TiO2 and SNP suspensions in the test medium. Germination rate was also impressed by TiO2 and SNP solutions under both control and cadmium stress in the same way previously stated regarding mean germination time (Fig. 3). Regarding seed vigor index, a statically significant difference was observed followed by seed exposure to TiO2 and SNP suspensions either solely or in combined compared to untreated seeds. Cd stress induced by CaCl2 led to extremely reduced seed vigor which intensified as Cd concentrations increased in the test solution. However, seed vigor dramatically affected by TiO2 plus SNP treatments (Fig. 3). For example, vigor index in seeds treated by combined TiO2 (1000 mg/L) + SNP (100 μM) suspensions experienced up to two-fold increase under 50 mM Cd concentration. Likewise, seed vigor remarkably promoted up to nineteen-fold under 100 mM Cd stress when seeds were exposed to TiO2 (2 000 mg/L) + SNP (100 μM) solutions in combination. Germination energy showed positive, null and even negative responses to different TiO2 + SNP combinations under control condition. Cadmium chloride concentrations induced significant reductions in germination energy of wheat seeds. Unlike trends observed in normal condition, TiO2 + SNP suspensions considerably relieved Cd-stimulated decline in germination energy (Fig. 3).
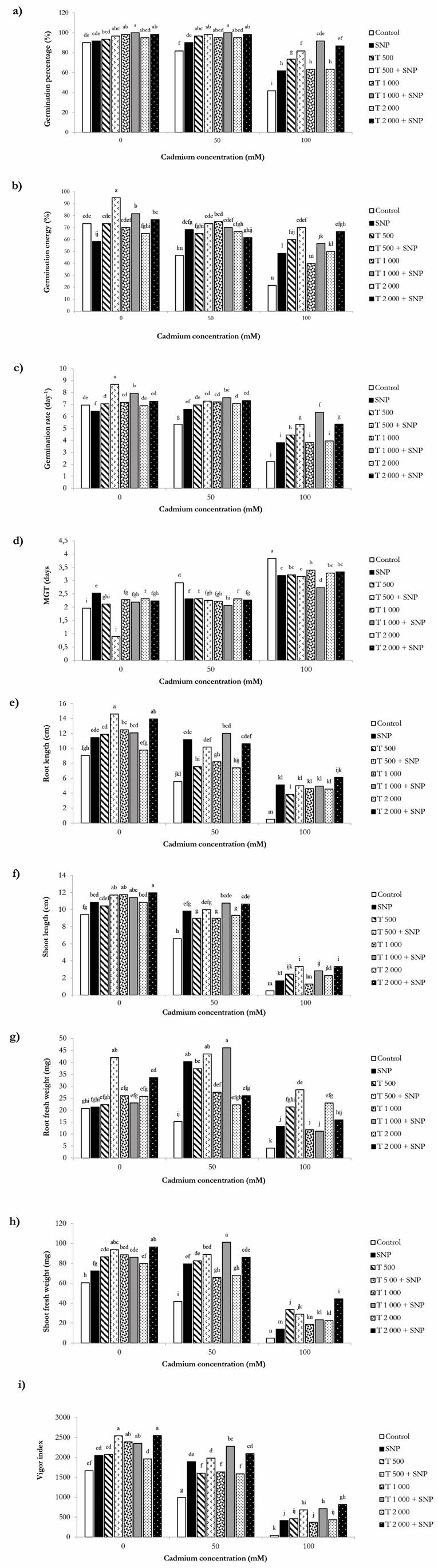
Figure 3 Impacts of TiO2 NPs (T: 500, 1 000 and 2 000 mg/L) and SNP (100 μM) suspensions on a) Germination percentage, b) Germination energy, c) Germination rate, d) Mean germination time (MGT), e) Root length, f) Shoot length, g) Root fresh weight, h) Shoot fresh weight and i) Vigor index of wheat under Cd stress. Data are the mean of three replicates. Different letters represent significant differences between the treatment means at p < 0.05.
Our results indicated that Cd stress considerably inhibited seed germination and early growth in wheat seedlings. Reduced germination indices such as germination percentage, root and shoot length, root and shoot fresh weight, and vigor in wheat seedlings under Cd stress conditions may be attributed to induced water shortage caused by CdCl2 suspensions. Many germination-related processes such as gene transcription and translation, respiration and energy metabolism, early reserve mobilization, and DNA repair could also occur during seed treatment [44] although often limited due to reduced water supply compared to normal germination [45,46]. The root length reduction may be attributed to lower cellular division and elongation during germination [47]. The root/shoot ratio is an important
index for measuring the development of seedling roots, and plants with higher root/shoot ratios are more capable of absorbing water and minerals [48]. Our results revealed that Cd stress stimulated by CdCl2 concentrations adversely affected all germination and growth parameters in germinating seeds. However, it was found that Cd stress, especially in 100 mM concentrations, had more severe inhibitory impacts on wheat germination and growth. Previous experiments indicated that Cd coud inhibit germination and growth of wheat [49-51]. The decreased growth and germination may be associated with several disturbances in germination metabolism which is a highly complex multistage process. Roots are the first organs subjected to pollutants, and therefore toxic symptoms are more apparent in roots rather than shoots [52]. Reduced shoot length, biomass, protein content, and activities of antioxidant enzymes by Cd toxicity have been documented previously [53,54].
Seed germination and root elongation are two standard criteria of phytotoxicity recommended by U.S. Environmental Protection Agency [34]. Improved root and shoot length accompanied by enhanced root and shoot fresh weight in wheat seedlings in the current study may be as a result of TiO2 NPs uptake via roots. Nanoparticles uptake across the cell wall is mostly dependent on the size of the particles, and the pores of the cell wall [55-57]. Due to the small diameter of TiO2 NPs, these nanoparticles might penetrate plant roots via pores in the cell wall. As demonstrated previously, smaller NP with a higher surface reactivity might enlarge root pores or create new ones, leading to higher hydromineral flow in roots [58]. Subsequently, this elevated nutrient uptake is responsible for the increased root length. Also, increased shoot and root length in the present study may be attributed to the early germination caused by TiO2 NPs and SNP treatments. It was shown that rapid embryo growth occurred when the obstacle to germination was removed [59]. Uptake of TiO2 nanoparticles and increased root length has been recorded in wheat plants after exposure to TiO2 NPs [60]. On the contrary, it was reported that TiO2 NPs had no impact [61] or even negative impact [62] on biomass in wheat.
In the presence of different TiO2 suspensions, most of the germination indices such as germination percentage, germination rate, germination energy, and vigor index promoted in wheat seeds under both normal and stress conditions. Improved seed germination indices in wheat upon exposure to TiO2 NPs have been previously reported by Mahmoodzadeh and Aghili [63], Jaberzadeh etal. [64] and Feizi etal. [2]. According to the obtained results, we postulated that TiO2 NPs ability to enhance seed germination and early growth of wheat under control and stress condition might have originated from its unique structural features (i.e., small particle size, large surface area, and photocatalytic properties). These structural features contribute to some key physiological processes in germinating seeds via facilitating water uptake [65], scavenging stress-induced ROS accumulation [66] and accelerating cellular growth and developments [67] in plants.It is well known that TiO2 NPs stimulate oxidation-reduction reactions via the superoxide ion radical during germination, leading to quenching free radicals in the germinating seeds. In turn, oxygen generated in such process could also be involved in respiration, which would further promote germination [68]. Water uptake in seed germination is critical because mature seeds are relatively dry and require a sufficient amount of water to initiate cellular metabolism and growth. Seed coat plays a unique role in protecting the embryo, and only nanoparticles that pass through the seed coat can affect seed germination and seedling growth [69]. Nanoparticles may create large new pores in the seed coat and therefore facilitate the process of water uptake inside the seed embryo, and accelerate germination rate [70]. Elevated nitrate reductase enzyme, improved seed function regarding absorbing and utilizing water and fertilizer and boosted seed antioxidant system was observed in soybean plants upon exposure to TiO2 and SiO2 nanoparticles [13]. Reduced oxidative stress by reducing H2O2, superoxide radicals, and malonyldialdehyde content, and increasing some enzymes such as superoxide dismutase, ascorbate peroxidase, guaiacol peroxidase, and catalase activities [71] by nanoparticles contributed to improved seed germination in some plant species. Ameliorative effects of TiO2 nanoparticles could also be due to antimicrobial and other properties of anatase crystalline structure of TiO2 that improve plant resistance to stress [72]. TiO2 NPs can also incite cell division and cell size in the root tip during germination process under dark conditions and consequently contribute to rapid emergence by increasing root length [59]. It seems that Titanium dioxide NPs may act as an alternative plant growth regulator and play a role similar to plant hormones such as cytokinin and gyberline [73,74] as indicated by the ability to stimulate plant cell division and induce cellular development. Previous studies have also introduced TiO2-anatase nanoparticles as an effective photocatalyst which can enhance cell growth as well as plant fresh and dry weight by improving photosynthetic efficiency and nitrogen metabolism [29,71,75].
As revealed by our experiment, supplemental SNP, as NO donor, might improve germination and subsequent seedling growth in wheat under either normal or stressed media. However, promoting impacts of SNP on germination indices were more evident when seeds were subjected to Cd stress triggered by CdCl2. The enhancement of wheat seed germination and seedling growth under Cd stress may be attributed to the promoting effect of NO in regulating endogenous auxins and gibberellins [76], increasing pectin and hemicelluloses content in the root cell wall [77], scavenging Cd-stimulated ROS [24], and modifications in gene expression [78]. Exogenous NO donor, SNP, dose-dependently alleviated the inhibition of rice seed germination indices and seedling growth caused by Cd stress via counteracting the adverse effects of Cd on germination index, vigor index, root and shoot length and fresh weight [27].
The positive role of SNP on germination percentage, germination rate, germination energy and vigor index either when applied solely or in combination with TiO2 suspensions shows that NO may be involved in energy metabolism in seeds during germination. Abiotic stresses have been demonstrated to restrict the mobilization of starchy endosperm reserves in several species, as a result of inhibition of different enzymatic activities [79,80]. Starch endosperm is the major part of wheat seeds, so its mobilization by amylase is required for wheat seed germination. It is known that NO is responsible for embryo extension and reserve degradation under normal and stress conditions and plays an important role in a-amylase, μ-amylase and protease mobilization in wheat seeds during early germination [81,82]. Zheng et al. [83] suggested that exogenous nitric oxide improved seeds germination and seedling establishment in wheat under abiotic stress via an increase of amylase and starch metabolism. It seems that NO could participate in the regulation of the energy status of the seeds.
Our results showed that seed germination indices markedly improved upon seed exposure to TiO2 and SNP treatments, in particular, under stress conditions stimulated by CdCl2 concentrations. Moreover, it was previously proved that Cd stress adversely limits water uptake by seeds and consequently inhibits germination and seedling growth of wheat [84,85,86]. Cadmium can make plants highly vulnerable to another sort of environmental constraints, such as drought and salinity, due to the reduced water uptake ability of the minor root system [87,88]. Then the plant roots are not able to take up enough water for metabolism, so the plant suffers physiological drought under Cd stress. In addition to other improving functions of TiO2 NPs and SNP during seed germination which was previously described, it seems that both TiO2 NPs and SNP are also involved in regulating mechanisms leading to the facilitating water uptake and improving tolerance to induced water deficit under Cd stress c ondition. Water deficit stimulates transcription of a large set of plant genes that results in the accumulation of new proteins such as water channel proteins (aquaporins) in plant tissues. Aquaporins which are present abundantly in the plasma and vacuolar membranes can
facilitate and the regulate passive transit of water across membranes and trigger a ten- to twenty-fold increase in water permeability [89,90]. On the other hand, aquaporins are proposed as pathways for nanoparticle uptake by plants [91,92]. Recently, up-regulation of aquaporins upon exposure to nanoparticles has been reported [93]. Interestingly, it has been demonstrated that the NO signaling pathway is participated in regulating aquaporin genes expression in germinating seeds [94]. It was shown that induced seed germination upon exposure to SNP treatment is associated with the increased expression of several aquaporin genes, which possibly leads to an enhanced seed capacity for water uptake during imbibition and its subsequent development. Particular aquaporin isoforms can also enable the transit of small molecules such as NO, CO or H2O2 [95].
Conclusions
Seed germination and early seedling growth of wheat positively affected by TiO2 and SNP treatments under normal and stress condition triggered CdCl2 suspensions. Also, it was also identified that the protective role of TiO2 NPs and SNP on wheat seed germination indices markedly enhanced when TiO2 and SNP solutions were exerted in combination. However, promoting the impact of TiO2 and SNP treatments on wheat seed germination improved when seeds were exposed to Cd stress treatments compared to a non-stress condition. Overall, our results suggest that application of TiO2 nanoparticles and SNP may be a promising approach in ameliorating harmful effects of Cd stress on wheat seed germination and early growth.