Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Boletín de Investigaciones Marinas y Costeras - INVEMAR
Print version ISSN 0122-9761
Bol. Invest. Mar. Cost. vol.27 no.1 Santa Marta Jan./Dec. 1998
KARYOLOGY OF MUGIL LIZA AND M. CUREMA FROM VENEZUELA
CARIOLOGÍA DE MUGIL LIZA Y M. CUREMA DE VENEZUELA
Mauro Nirchio T.1 y Hernán Cequea2
1Universidad de Oriente, Núcleo de Nueva Esparta, Apartado postal 147, Isla de Margarita, Venezuela. E-mail: nirchio@enlared.net.ve (M. N. T.).
2Universidad de Oriente, Núcleo de Sucre, Cumaná, Venezuela (C. H.).
ABSTRACT
Karyotypes of Mugil liza and M. curema from Venezuela were studied. M. liza karyotype is 2n=48 acrocentric chromosomes, but M. curema is 2n=24, containing one submetacentric and 11 metacentric pairs. The chromosomic complement described for M. liza constitutes the first report for the species and coincides with the modal karyotype (2n=48) in the Mugilidae family. Venezuelan M. curema karyotype differs in number and shape from M. curema from Louisiana (USA), which has been reported to have a complement 2n=28 (10 metacentric pairs, two subtelocentric pairs and two acrocentric pairs).
KEY WORDS: Karyotype. Chromosomes. Mugil liza. Mugil curema.
RESUMEN
Se estudió el cariotipo de dos especies de mugílidos presentes en Venezuela: M. liza, que posee un cariotipo 2n=48 con cromosomas acrocéntricos, y M. curema con un complemento 2n=24, constituido por once pares metacéntricos y un par submetacéntrico. El complemento descrito para M. liza constituye el primer registro para la especie y coincide con el cariotipo modal (2n=48) en la familia Mugilidae. El cariotipo descrito para M. curema difiere del presentado en un registro previo en el que se describe para la misma especie de las costas de Louisiana, USA, un complemento 2n=28 (10 pares metacéntricos, 2 pares subtelocéntricos y 2 pares acrocéntricos).
PALABRAS CLAVE: Cariotipo. Cromosomas. Mugil liza. Mugil curema.
INTRODUCTION
Descriptions of karyotypes in teleostean fishes have been published and lists of their chromosome morphology and number are readily available (see Gyldenholm and Scheel, 1971; Gold et al., 1980; Sola et al., 1981; Hartley, 1987). Although some intraindividual and intraspecific variation is commonly found among living teleost fishes, a karyotype with 48 uniarmed chromosomes appears to be predominant and has been proposed as ancestral to modern fishes (Gold et al., 1980; Sola et al., 1981; Doucette and Fitzsimons, 1988).
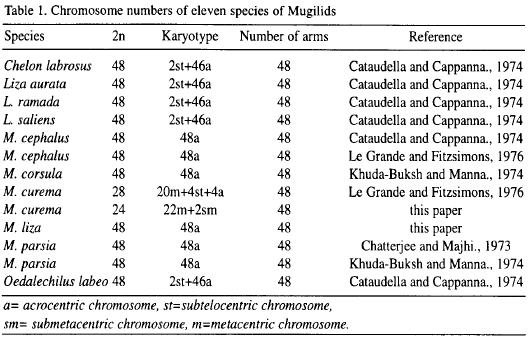
In the case of Mugilidae, reported karyotypes cover the following species: Mugil cephalus, M. corsula, M. parsia, M. curema, Chelon labrosus, Liza ramada, Liza saliens, Liza aurata and Oedalechilus labeo. In Table 1 it can be observed that the modal karyotype is 2n=48 except for M. curema from Louisiana, USA, with a complement 2n=28 (Le Grande and Fitzsimons, 1976). This paper investigates the karyotypes of Venezuelan M. curema and M. liza. It will be shown that the same extensive variation found above is also present in Venezuelan M. curema to even a greater degree and that the M. liza karyotype of Venezuela corresponds to the modal karyotype of the family.
MATERIALS AND METHODS
Sexually immature Mugil curema and M. liza specimens (180-200 mm TL) were collected in coastal waters near La Restinga Lagoon, Margarita Island, Venezuela. Twenty specimens of each species were injected intraperitoneally with 0.1% colchicine and kept in a well aerated aquarium. After 6 h, specimens were sacrificed and the anterior portion of the kidney was removed, placed in 0.4% KC1 solution and cut into small pieces. The chromosome preparations were carried out a technique described by Reddy and George (1987), except that preparations were stained for 20 minutes with FLP orcein (1.5% orcein in 20% formic acid, 80% lactic acid, propionic acid and distilled water; 1:1:1:1). Twenty cells from each specimen were analyzed for chromosome count and chromosome morphology. The size range between the smallest and the biggest chromosomes was measured in the best quality spread. Well spread metaphase plates were photographed and chromosomes arranged according to Levan et al. (1964).
RESULTS AND DISCUSSION
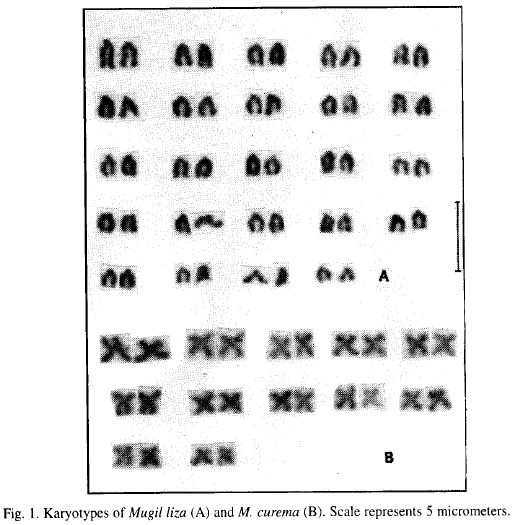
Representative karyotypes from each species are presented in Figure 1. Table 2 shows a summary of karyotypic data for species studied. Mugil liza diploid number ranged from 45-49, while for M. curema ranged from 19-26. The M. liza modal diploid number count was 48 with small acrocentric chromosomes. M. curema had a 2n=24 unimodal complement, with one submetacentric pair and eleven metacentric pairs which were arranged in two groups of different size. The number of arms was the same for both species (NF=48). Counts below the modal numbers are attributed to a loss of chromosomes during slide preparation or to naturally occurring incomplete complements. The few hypermodal counts probably represent additional chromosomes from another spread, a premature separation of chromatids, or additional chromosomes in atypical nuclei.
A common type of chromosomal rearrangement in fishes is a change in chromosome number due to fusion of two one-armed chromosomes into one two-armed chromosome or to fission (the reverse) (Manna and Prasad, 1971; Denton, 1973; Le Grande, 1975; Gold, 1979; Ferguson and Allendorf, 1991), but according to Le Grande and Cavender (1980), centric fission constitutes a more complex event and, as a consequence, it is less probable than centric fusion.
It has been suggested that the deviations from the 2n=48 presumptive fish ancestral karyotype are towards a reduction in the chromosome number (Gold, 1979). Karyotype studies in twenty species of Elopiformes and Clupeiformes, indicated that the association of large metacentric or submetacentric chromosomes with a reduction in chromosome number is consistent with fusion events in the karyotype evolution from a hypothetical ancestral complement (Doucette and Fitzsimons, 1988) .
Since biarmed chromosomes in M. curema are larger than in M. liza (Table 2), it seems reasonable to suggest that the karyotype of the former evolved from an ancestral group like the latter with a chromosome complement of 48 uniarmed elements, by fusion of pairs of acrocentric chromosomes accounted for the formation of biarmed element. This suggestion is consistent with a previous report by Le Grande and Fitzsimons (1976) who observed that the M. curema metacentric chromosomes are about twice the size of M. cephalus uniarmed elements and proposed that the 20 metacentric chromosomes of M. curema from Louisiana evolved from an ancestral group with the M. cephalus chromosome complement probably by centric fusion of 20 uniarmed chromosomes pairs from one with 48 uniarmed chromosomes.
The M. liza karyotype showed in Fig. 1 agrees with the published information, indicating that, with the exception of M. curema which possesses a karyotype strikingly different, mugilids are a fairly homogeneous karyotypic group with 48 chromosomes (Table 1). Nevertheless, our study revealed that the basic chromosome number of M. curema (2n=24) from Venezuela is not in agreement with those reported by Le Grande and Fitzsimons (1976), for M. curema from Louisiana (2n=28).
Although chromosomal polymorphisms among population of fishes are not widespread, there are some documented cases (see Le Grande and Cavender, 1980; Vitturi and Lafargue; 1992; Gyldenholm and Scheel, 1971; Gold et al., 1980; Sola et al., 1981; Ihssen et al., 1981; Hartley, 1987 and references therein). The fact that the modal chromosome number in M. curema from Louisiana (Le Grande and Fitzsimons, 1976), and from Venezuela (present study) were different and are considered to represent the correct diploid chromosome complement for each population, supports the possibility of chromosomal variation within the species which could be used as diagnostic character for fish stock recognition, as it has been described by Ihssen et al (1981).
Althouhg discrepancies between the karyotype reported here and the one reported by Le Grande and Fitzsimons (1976) for M. curema suggest the possibility of chromosomal polymorphisms in the species with fixed karyomorphs in local population, the convenience of a taxonomic revision for the species should not be neglected, since the karyotype differences pointed out could constitute evidence that M. curema denomination could be employed to identify two different species. In this sense, it has been admitted that mugilids belong to one of the most complicated taxonomic group, above all in juvenile stages, and at the present time the classification to specific level is still not completely clear (Cervigón, 1993).
LITERATURE CITED
1 Cataudella, S. and E. Capanna. 1973. Chromosome complements of three species of Mugilidae. Experientia, 29: 489-491. [ Links ]
2 Cervigón, F. 1993. Los peces marinos de Venezuela. 2ª Edición. Volumen II. Fundación Científica Los Roques, Caracas, Venezuela. [ Links ]
3 Chatterjee, K. and A. Majhi. 1973. Chromosomes of Mugil parsia Hamilton (Teleostei, Mugiliformes: Mugilidae). Genen Phaenen, 16(2): 51-54. [ Links ]
4 Denton, T. E. 1973. Fish chromosome methodology. C. C. Thomas Publ., Springfield, Illinois. [ Links ]
5 Doucette, A. J. Jr. and J. M. Fitzsimons. 1988. Karyology of Elopiform and Clupeiform fishes. Copeia, 1: 124-130. [ Links ]
6 Ferguson, M. M. and F. Allendorf. 1991. Evolution of fish genome. In: Hochachka and Mommsen (eds.). Biochemistry and molecular biology of fishes. Vol. 1: 25-42. [ Links ]
7 Gold, J. 1979. Cytogenetics. In: Hoar, W. S. and D. J. Randall (Eds). Fish Physiology, Academic Press, New York. Volume 8: 353-405. [ Links ]
8 Gold, J. R.; W. J. Karel and M. R. Strand. 1980. Chromosome formulae of North American fishes. Prog. Fish. Cult., 42(1): 10-23. [ Links ]
9 Gyldenholm, A. O. and J. J. Scheel. 1971. Chromosome numbers of fishes. I. J. Fish. Biol., 3:479-486. [ Links ]
10 Hartley, S. E. 1987. The Chromosome of salmonid fishes. Biol. Rev., 62:197-214. [ Links ]
11 Ihssen, P. E.; H. E. Booke; J. M. Casselman; J. M. Mcglade; N. R. Payne and F. M. Utter. 1981. Stock identification: materials and methods. Can. J. Fish. Aquat. Sci., 38:1838-1855. [ Links ]
12 King, M. 1987. Chromosomal rearrangements, speciation and the theoretical approach. Heredity, 59:1-6. [ Links ]
13 Khuda-Bukhsh, V. S. and G. K. Manna. 1974. Somatic chromosomes in seven species of teleostean fishes. Chrom. Inf. Serv., 17: 5-6. [ Links ]
14 Le Grande, W. H. 1975. Karyology of six species of Louisiana flatfishes (Pleuronectiformes, Osteichthyes). Copeia, 3: 516-522. [ Links ]
15 Le Grande, W. H. and T. M. Cavender. 1980. The chromosome complement of the stonecat madtom, Noturus flavus (Siluriformes: Ictaluridae), with evidence for the existence of a possible chromosomal race. Copeia, 2: 341-344. [ Links ]
16 Le Grande, W. H. and J. M. Fitzsimons. 1976. Karyology of the mullets Mugil curema and Mugil. cephalus (Perciformes: Mugilidae) from Louisiana. Copeia, 2: 388-391. [ Links ]
17 Levan, A.; A. Fredga and A. Sandburg. 1964. Nomenclature for centromeric position on chromosomes. Hereditas, 52: 201-220. [ Links ]
18 Manna, G. K. and R. Prasad. 1971. A new perspective in the mechanism of the evolution of chromosomes in fishes. Proc. First all India Congr. Cytol and Genet., J. Cytol. and Genet. Congr. Suppl., 237-240. [ Links ]
19 Reddy, P. V. G. K. and J. George. 1987. A method to increase mitotic metaphase spreads in permanent chromosome preparations for karyotype studies of fishes. Proc. World Symp. on Selection, Hybridization and Genetic Engineering in Aquaculture Bordeaux., Vol. II: 199-205. [ Links ]
20 Sola, L.; S. Cataudella and E. Capanna. 1981. New developments in vertebrate cytotaxonomy. III. Karyology of Bony Fishes: a review. Genetica, 54: 285-328. [ Links ]
21 Vitturi, R. and F. Lafargue. 1982. Karyotype analyses reveal inter-individual polymorphism and association of nucleolus-organizer-carrying chromosomes in Capros aper (Pisces: Zeiformes). Marine Biology, 12: 37-41. [ Links ]
DATE RECIEVED: 12/05/1997 DATE ACCEPTED: 24/02/1998