Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Boletín de Investigaciones Marinas y Costeras - INVEMAR
Print version ISSN 0122-9761
Bol. Invest. Mar. Cost. vol.35 no.1 Santa Marta Jan./Dec. 2006
CAPRELLIDS FROM THE CARIBBEAN COAST OF COLOMBIA, WITH DESCRIPTION OF THREE NEW SPECIES AND A KEY FOR SPECIES IDENTIFICATION
CAPRÉLIDOS DE LA COSTA CARIBE DE COLOMBIA, CON LA DESCRIPCIÓN DE TRES ESPECIES NUEVAS Y UNA CLAVE PARA LA IDENTIFICACIÓN DE LAS ESPECIES
José M. Guerra-García1, Traudl Krapp-Schickel2 and Hans G. Müller3
1Laboratorio de Biología Marina, Departamento de Fisiología y Zoología, Facultad de Biología, Universidad de Sevilla, Avda Reina Mercedes 6, 41012, Sevilla, Spain. E-mail: jmguerra@us.es
2Museum A. Koenig, Adenauerallee 160, D-53113 Bonn, Germany
3Institut für Allgemeine und Spezielle Zoologie der Justus-Liebig-Universität, Heinrich-Buff-Ring 29, D-6300 Giessen, Germany
ABSTRACT
The Caprellidae (Crustacea: Amphipoda) of shallow waters of the Caribbean coast of Colombia, South America, are investigated. This is the first study dealing with the caprellidean fauna of this area. A total of 45 stations from Punta Brava to Punta Cañaveral, at Santa Marta region, Colombia, was selected, and samples of algae, coral rubble, sponges, hydroids, bryozoans and sediments were collected from the intertidal zone to 30 m depth. Ten species in seven genera were found (three of which are new to science): Aciconula sp., Caprella cf. penantis Leach, 1814, Caprella danilevskii Czerniavskii, 1868, Deutella caribensis n.sp., D. incerta (Mayer, 1903), D. mayeri Stebbing, 1895, Hemiaegina minuta Mayer, 1890, Paracaprella pusilla Mayer, 1890, Pseudaeginella colombiensis n.sp., and Tritella chibcha n.sp. All the species are fully illustrated, and a key to species level for all species is also provided.
KEY WORDS: Amphipoda, Caprellidae, Taxonomy, New species, Caribbean, Colombia.
RESUMEN
Se estudiaron los Caprellidae (Crustacea: Amphipoda) de las aguas someras de la costa Caribe de Colombia, Sudamérica. Este es el primer estudio sobre la fauna de caprélidos de la zona. Se seleccionaron 45 estaciones distribuidas desde Punta Brava hasta Punta Cañaveral y se recolectaron muestras de algas, corales, esponjas, hidrozoos, brioozoos y sedimentos, desde el intermareal a los 30 m de profundidad. Se encontraron diez especies pertenecientes a siete géneros distintos (tres de las especies son nuevas para la ciencia). Aciconula sp., Caprella cf. penantis Leach, 1814, Caprella danilevskii Czerniavskii, 1868, Deutella caribensis n.sp., D. incerta (Mayer, 1903), D. mayeri Stebbing, 1895, Hemiaegina minuta Mayer, 1890, Paracaprella pusilla Mayer, 1890, Pseudaeginella colombiensis n.sp. y Tritella chibcha n.sp. Todas las especies se ilustran con detalle en el presente trabajo y se aporta una clave taxonómica a nivel de especie.
PALABRAS CLAVE: Amphipoda, Caprellidae, Taxonomía, Nuevas especies, Caribe, Colombia.
INTRODUCTION
There is a lack of studies dealing with the caprellid amphipods of Central and South America. In fact, McCain and Steinberg (1970) pointed out that these coasts are virtually unstudied and that undoubtedly many new records and species would be expected in these areas. However, there are recent attempts to improve the knowledge on caprellids along these coasts: Chile (Guerra-García, 2001a; Guerra-García and Thiel, 2001; Thiel et al., 2003), Brazil (Wakabara et al., 1991; Wakabara and Serejo, 1998), the Gulf of Mexico (Ortíz et al., 2002; Escobar-Briones and Winfield, 2003; Winfield et al., 2006), Venezuela (Díaz et al., 2005). These studies are necessary to fill the gap in the knowledge of the biodiversity of caprellids in Central and South America, to properly address future biogeographical and ecological studies.
In general, the amphipods from Colombia are poorly studied, as pointed out by González and Watling (2003). The taxonomical studies dealing with peracaridean crustaceans from Colombia have been more focused on isopods (Müller, 1992, 1993a, 1993b; Kensley and Schotte, 1994) than in other groups such as amphipods. In fact, the present study represents the first comprehensive work about caprellids from Colombia.
MATERIAL AND METHODS
Sampling area
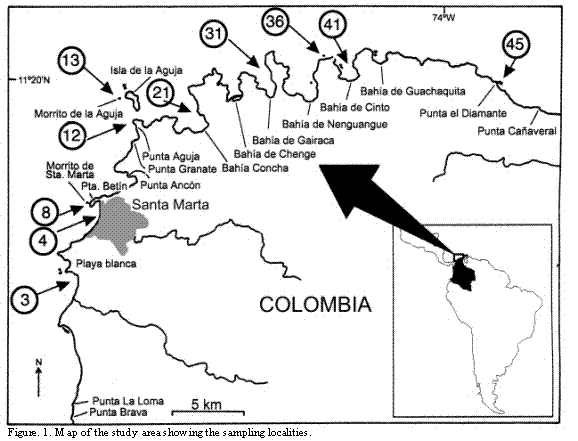
Samples were taken along the about 70 km long coast of Santa Marta, on the Caribbean coast of Colombia (11o 15' N - 74o 13' W) (Figure 1). The geomorphology is influenced by the Sierra Nevada de Santa Marta (SNSM), the highest coastal mountain range. In the West area, the rivers bring a rather estuarine character. The Tayrona National Natural Park in the North-East area has deep bays with a much less intense freshwater influence, except during the rainy period (between August and early December). According to measurements in Bahía de Santa Marta the salinity sinks in this time from 38 to 30 (Müller, 1979) and the water gets cloudy due to suspended particles of the rivers. During the dry period, between late December and May, a b wind called "brisa" predominates, coming from the SNSM (Herrmann 1971), which drives away the warm surface waters into the open sea, and cooler waters from the depth surge in areas near the coast. At the end of the rainy period the seawater temperature drops from 29oC to 25oC in February within short time (Salzwedel and Müller, 1983). Thus temperature, salinity and light-transparency of the sea water are changing drastically throughout the year.
Collection data
Collecting was carried out at the Instituto de Investigaciones Marinas de Punta de Betín (nowadays Instituto de Investigaciones Marinas y Costeras-INVEMAR) in
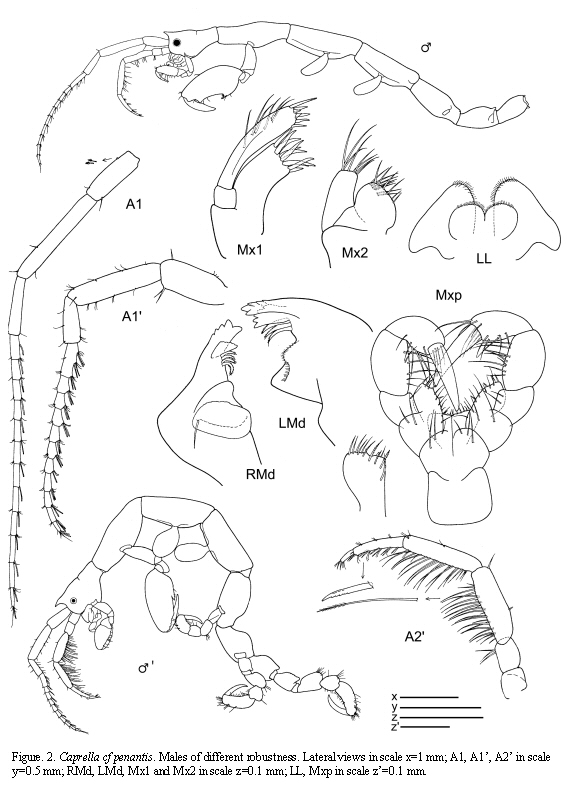
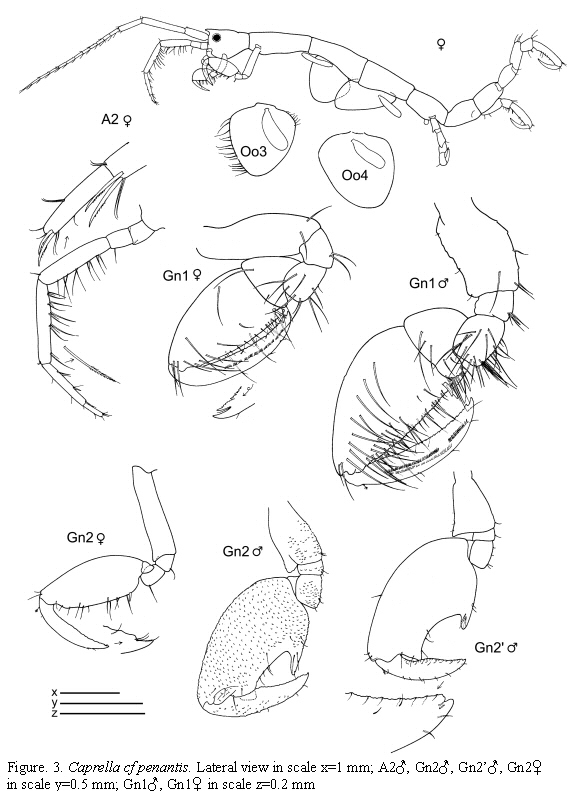
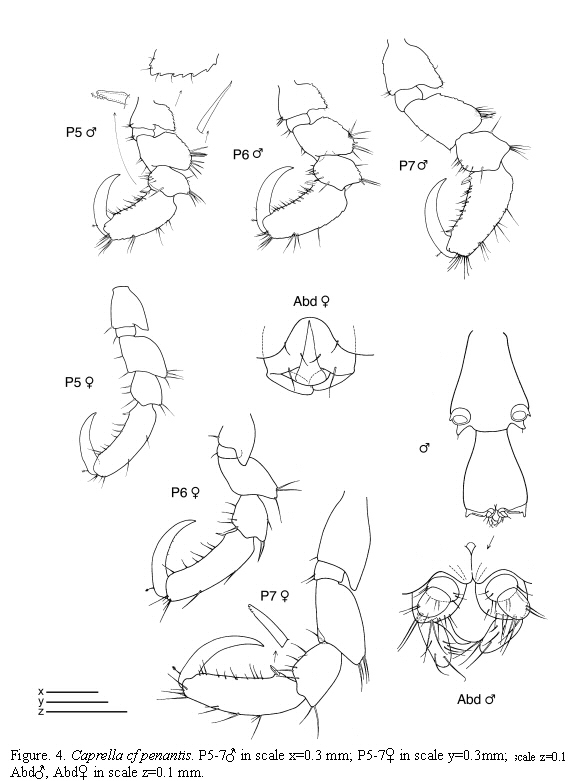
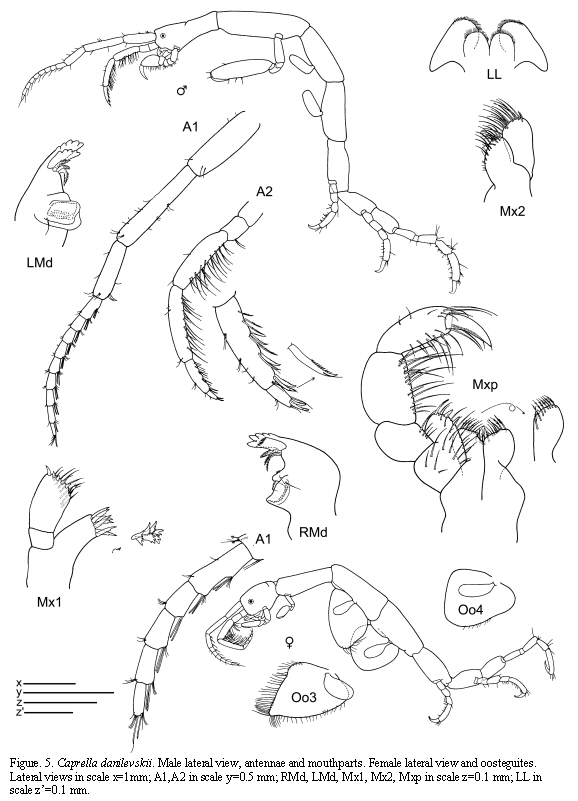
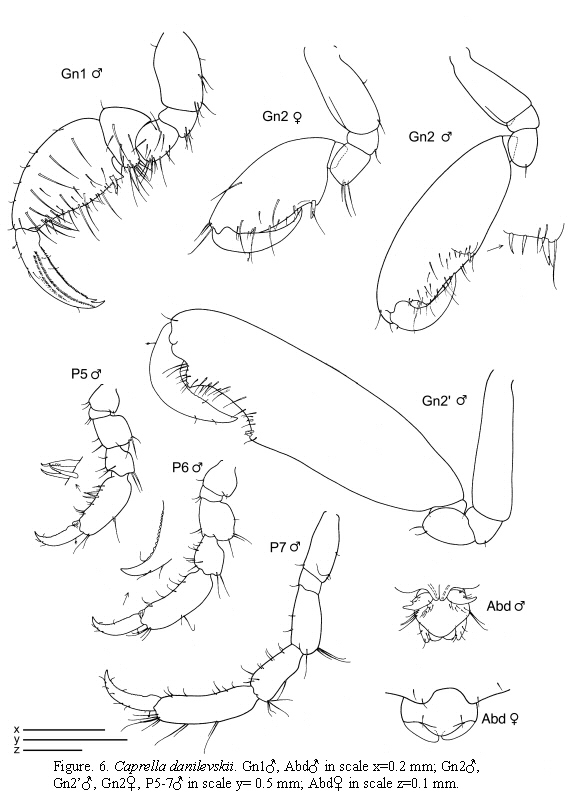
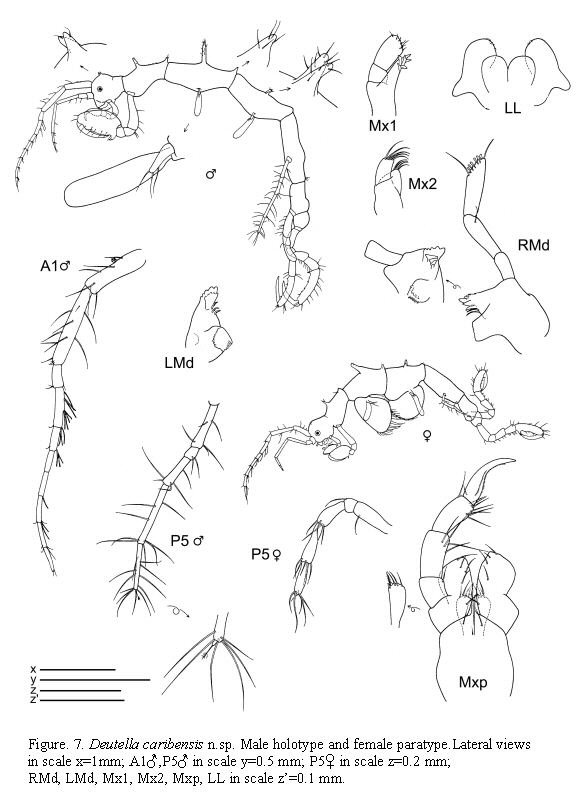
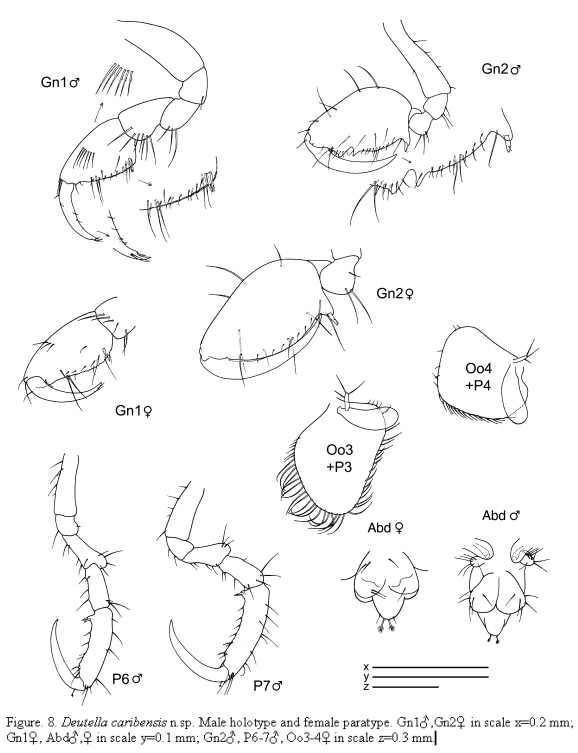
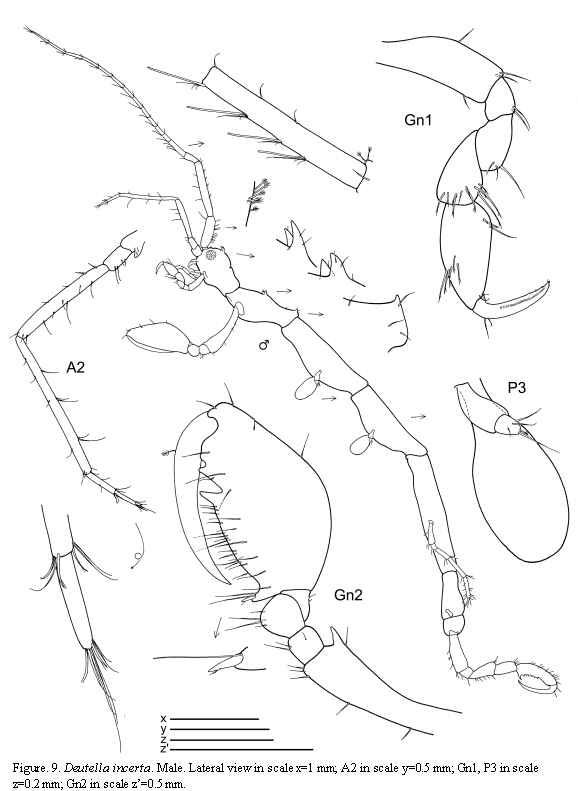
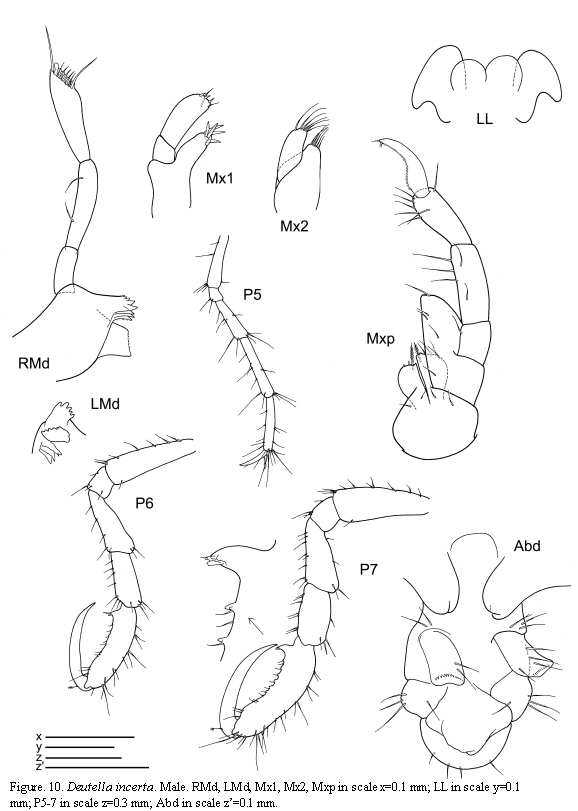
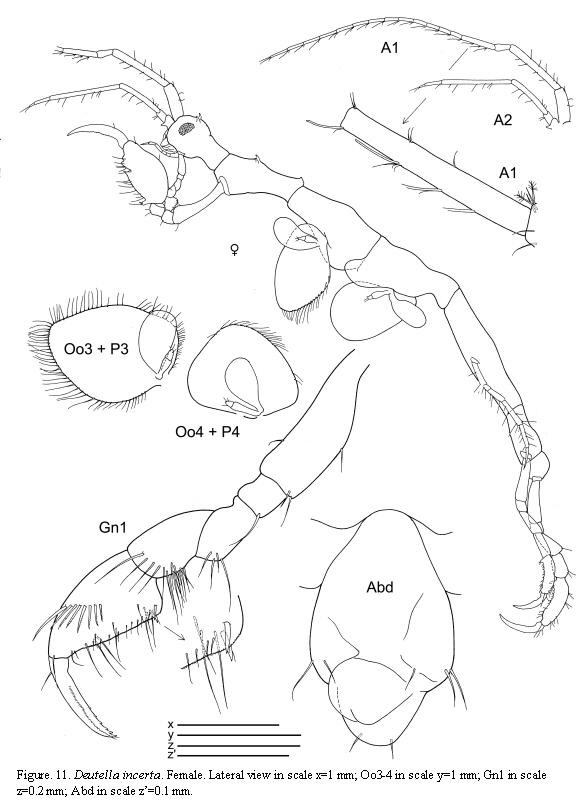
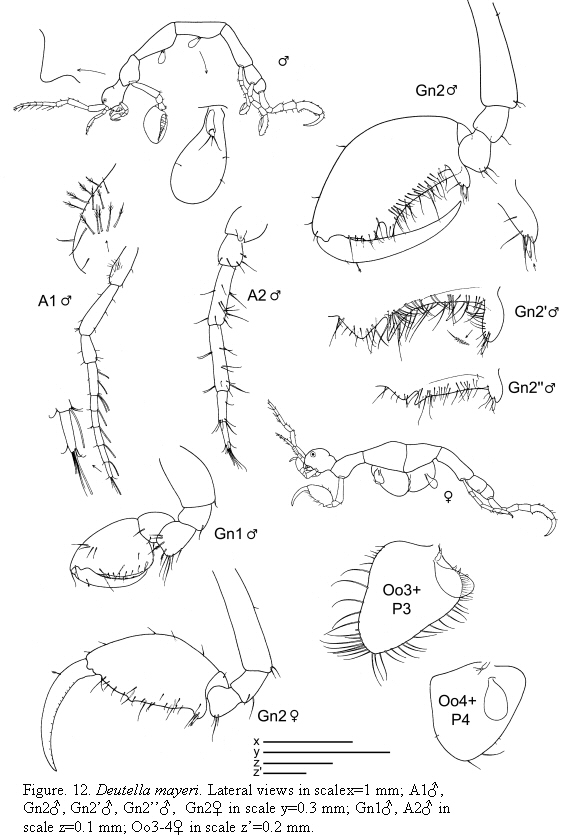
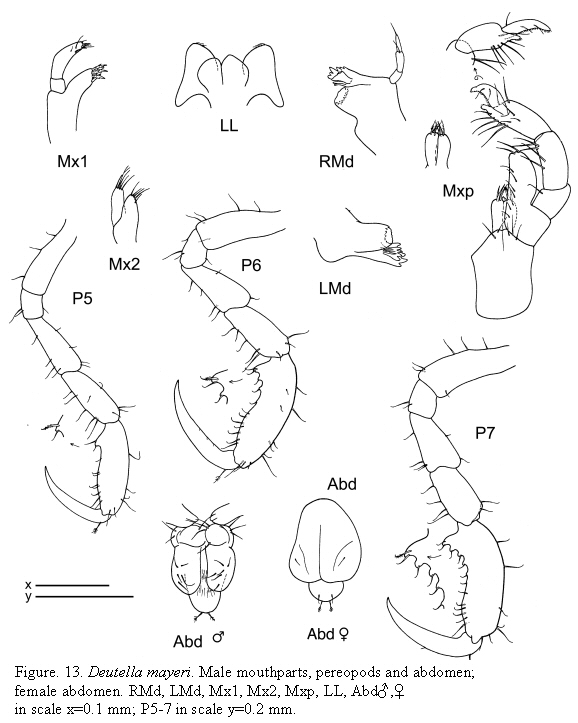
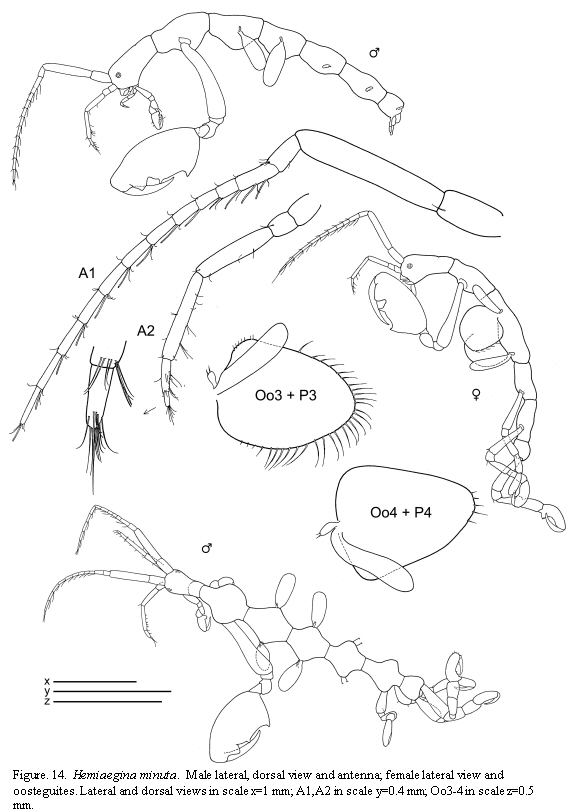
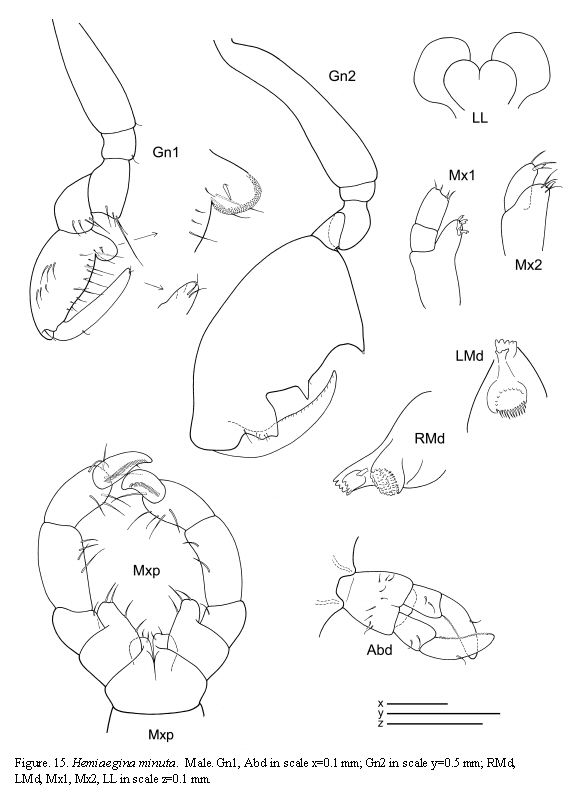
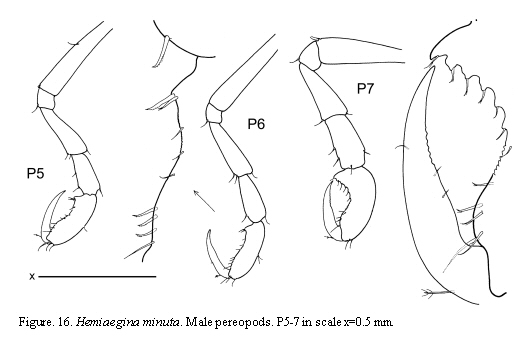
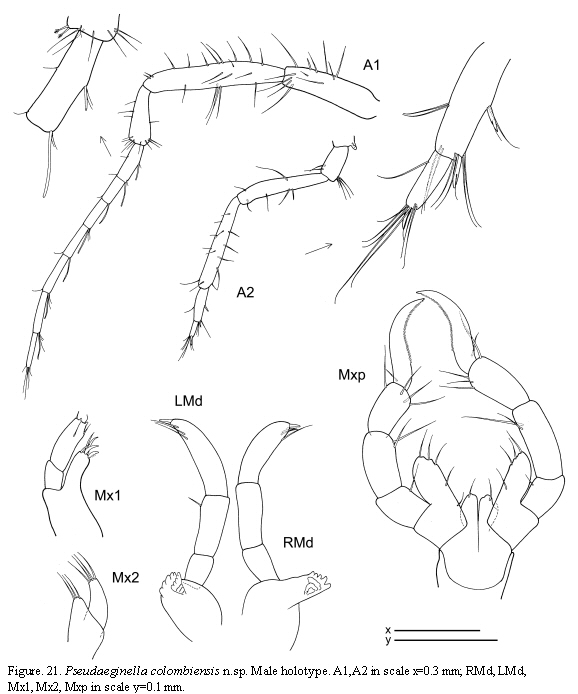
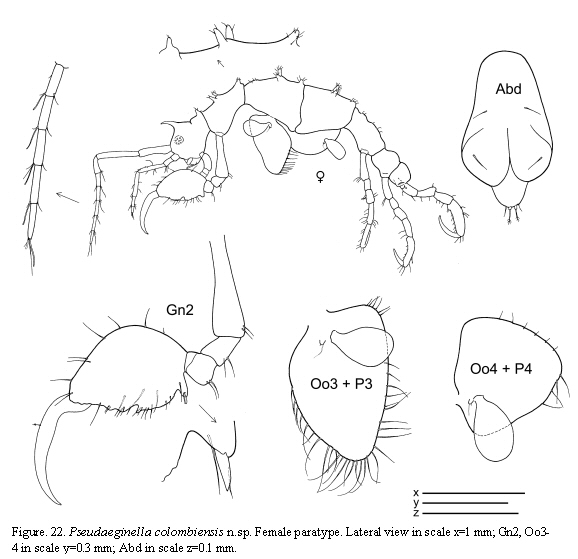
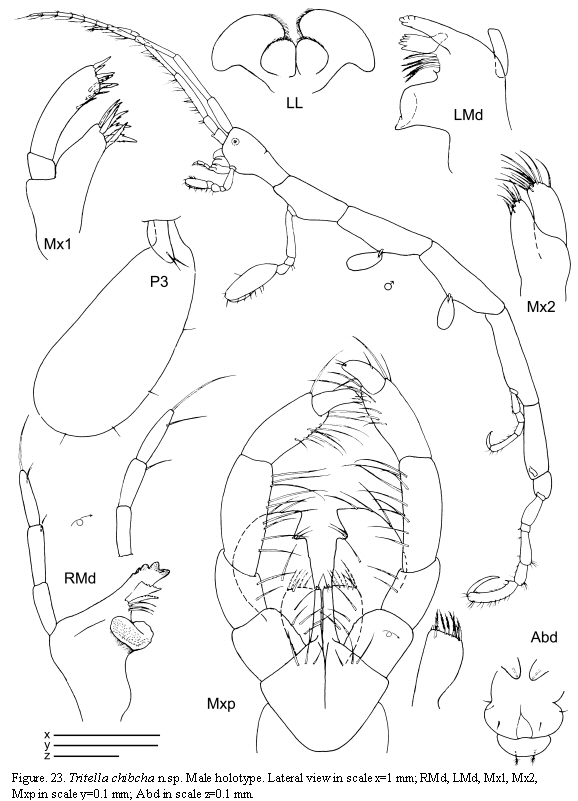
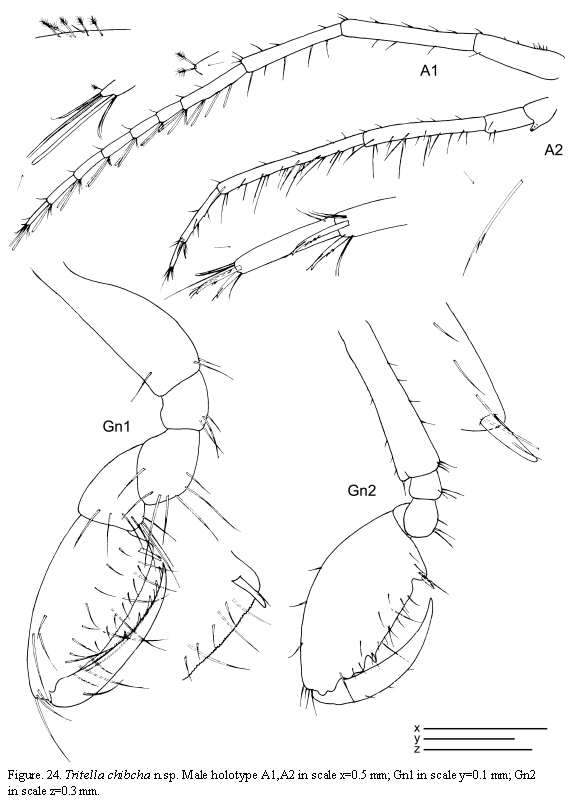
The examined material for the present study is deposited partly at the Instituto de Investigaciones Marinas y Costeras (INVEMAR) in Santa Marta, Colombia, partly at the Museo Civico di Storia Naturale di Verona, Italy. The symbols used in plates are (in alphabetic order): A1, 2 = Antenna 1, 2; Abd = Abdomen; Gn1, 2 = Gnathopod 1, 2; LL = Lower lip = labium; LMd = Left mandible; Mx1, 2 = Maxilla 1, 2; Mxp = Maxilliped; Oo3-4 = Oosteguite 3, 4; P3-7 = Pereopods 3-7; RMd = Right mandible; UL = Upper lip = labrum.
Although the phylogeny and higher classification of the caprellids is still under debate (Laubitz, 1993; Takeuchi, 1993), Myers and Lowry (2003) have recently proposed a new phylogeny and classification for the suborder Corophiidea Leach, 1814, which is divided into two infraorders, the Corophiida and the Caprellida, based on a hypothesis of the evolution of different feeding strategies. In their new classification, the superfamily Caprelloidea contains five families: Caprellidae, Caprogammaridae, Cyamidae, Dulichiidae and Podoceridae. The Caprellidae are subdivided into three subfamilies: Caprellinae, Paracercopinae and Phtisicinae. In the present paper we have adopted this classification and have focused our study on members of the family Caprellidae.
Station list and caprellids found:
Station 3: Playa Blanca, near El Rodadero/Santa Marta. Under stones covered with algae, 0.5-1 m. 30.XII.1985. Deutella mayeri.
Station 4: Bahía de Santa Marta, Isla Morro Grande de Santa Marta, North side. Algae on rocks, coral rubble, porifera, and hydroids, 0-30 m. 24.V.1985, 18.IX.1985, 9.X.1985, 9. XII.1985, 12.XII.1985, 10.III.1986. Caprella cf. penantis, Deutella incerta, Hemiaegina minuta, Tritella chibcha n.sp.
Station 8: Punta Betín. Epiphytes on shadowy or light-exposed concrete piers; detritus, corallinacea, porifera, hydroids (mainly Cnidoscyphus), bryozoa, and coral rubble, 0-28 m. 24.IV.1985, 25.IV.1985, 4.V.1985, 8.V.1985, 10.V.1985, 12.V.1985, 14.V.1985, 16.V.1985, 18.V.1985, 19.V.1985, 27.V.1985, 3.VI.1985, 17.VI.1985, 19.VI.1985, 21.VI. 1985, 22.VI.1985, 1.VII.1985, 1.VIII.1985, 4.VIII.1985, 3.IX. 1985, 15.IX.1985, 21.IX.1985, 1.XII.1985, 24.XII. 1985, 29.XII.1985, 29.I.1986, 10.II.1986, 14.III.1986. Deutella incerta, Deutella mayeri, Hemiaegina minuta, Paracaprella pusilla.
Station 12: Punta Aguja. Coral rubble, porifera, hydroids, and bryozoans, 11-20 m. 16.IX.1985, 2.X.1985. Aciconula sp., Caprella cf. penantis, Deutella incerta.
Station 13: Isla Aguja. Coral rubble, 30 m, 18.II.86. Deutella incerta.
Station 21: Bahía Concha. Brown-algae Digenia simplex and Sargassum cymosum on dead Porites - reef; quantitative sample of 5 l each, cutting off the entire thallus; mesolitoral. Thalassia and Halocordyle and Cnidoscyphus hydroids. 0-1 m. 22.V.1985, 7.VI.1985, 27.VI.1985, 5.VII.1985, 8.VII.1985, 15.VII.1985, 13.VIII.1985, 9.IX.1985, 2.X.1985, 28.X.1985, 8.XI.1985, 7.XII.1985, 12.II.1986, 3.III.1986, 2.IV.1986, 3.IV.1986. Caprella danilevskii, Caprella cf. penantis, Hemiaegina minuta, Deutella mayeri, Deutella caribensis n.sp.
Station 31: Eastern beach near Punta Gayraca. Under stones on sand and from algae like Sargassum or seagrass Thalassia, mesolitoral, 6.VI.1985, 12.VII.1985, 17.I.1986. Caprella danilevskii, Deutella mayeri, Paracaprella pusilla.
Sample 36: Bahía Nenguangue, Roca Ahogada NE of Punta del Esbarrancado. Brown algae such as Cladophyllum schnetteri, hydroids, and Thalassia, 0.5-2 m. 27.V.1985, 30.VII.1985, 20.XII.1985, 17.I.1986. Caprella danilevskii, Deutella mayeri, Hemiaegina minuta.
Station 41: Bahía Cinto, southern beach. Pure sand with little detritus, Syringodium-weed, 0-3 m. 20.XII.1985, 17.III.1986, 14.IV.1986. Deutella mayeri, Tritella chibcha n.sp.
Station 45: Punta El Diamante (Arrecifes/Cañaverales). Algae, hydroids, and bryozoans on rocky slope, 1-10 m. 26.IX.1985, 25.II.1986. Deutella incerta, Deutella mayeri, Hemiaegina minuta, Pseudaeginella colombiensis n.sp.
Additional sample1: Isla de Providencia, Thalassia, 5.XII.80, leg. INVEMAR: Deutella mayeri.
Additional sample2: From stomach of Acanthurus coeruleus; Sta. Marta area, 5 m, leg. Sergio Duarte, 1.VII.85: Hemiaegina minuta.
SYSTEMATIC ACCOUNT
Family Caprellidae Leach, 1814
Subfamily Caprellinae Leach, 1814
Caprella cf. penantis Leach, 1814
(Figures 2, 3, 4)
Caprella Penantis Leach, 1814: 404.
Caprella acutifrons Mayer, 1882: 48; Mayer, 1890 (included f. neglecta, tabida, gibbosa, carolinensis, lusitanica, virginia): 50, pl. 2, figs. 36-37, 39-41, pl. 4, figs. 52-53, 55, 57-61, 65-69; Mayer, 1903: 79, pl. 3, figs. 4-28; pl. 7, figs. 62-65.
Caprella penantis McCain, 1968: 33, figs. 15-16; McCain and Steinberg, 1970: 33; Cavedini, 1982: 508; Krapp-Schickel, 1993: 791-793; Guerra-García and Takeuchi, 2002: 692-693, fig. 12; Guerra-García, 2004: 30, 32, fig. 26; Guerra-García and Takeuchi, 2004: 1013-1015, fig. 35; Krapp-Schickel and Guerra-García, 2005: 48-49, fig. 2.
Material examined: 187 specimens
St.4: 3 males, 3 females, hydroids, 11-19 m, 9.X.1985. St.12: 1 male, hydroids and bryozoans, 11-15 m, 2.X.1985. St.21: 1 male, 2 females, from hydroids, 0-0.5 m, 7.VI.1985; 11 males, 10 females (1 ov.), 5 juveniles, from intertidal hydroids, 5.VII.1985; 8 males, 7 females (2 ov.), from hydroids under overhanging rocks, intertidal, 8.VII.1985; 26 males, 18 females (13 ov.), 17 juveniles, from hydroids, intertidal, 15.VII.1985; 2 males, 5 females (1 ov.), from hydroids, lower intertidal, 2.X.1985; 36 males, 31 females (25 ov.), 2 juveniles, from hydroids, lower intertidal, 8.XI.1985.
Figured specimens are from St. 21, 15.VII.1985 (males) and St. 4, 9.X.1985 (female).
Remarks
The specimens collected from Colombian waters are close to C. penantis. At the moment, we have considered this species as C. cf. penantis since we have not been able to find constant morphological differences between the Colombian specimens and the Mediterranean and Atlantic C. penantis (Krapp-Schickel, 1993; Guerra-García and Takeuchi, 2002; Riera et al., 2003; Guerra-García et al., 2006.). Nevertheless, the present material from Colombia show high degree of morphological variation in connection with the robustness of antennae and body, number and length of swimming setae in antenna 2, and shape of the gills. Some specimens even presented the gnathopod 2 provided with tiny tubercles. Recently, Díaz et al. (2005) figured C. penantis based on a specimen collected from the Caribbean coast of Venezuela and it is very similar to the material collected from the Caribbean coast of Colombia, probably belonging to the same species.
Caprella penantis has been recorded under several specific or subspecific names from temperate regions worldwide (McCain and Steinberg, 1970) and further morphological and molecular studies are required to evaluate the respective status of this species around the world (McCain, 1968; Laubitz, 1972; Takeuchi and Hirano, 1995). In this sense, Guerra-García et al. (2006) have used a preliminary molecular approach (RAPD analysis) to explore intraspecific variation among populations of C. penantis from the Strait of Gibraltar, showing that the different morphs of C. penantis in that biogeographical area seem to be ecotypes rather than different species.
Habitat
This species was found nearly exclusively in Bahía Concha, and mainly on hydroid Cnidoscyphus from the intertidal area.
Caprella penantis has been found living on red and brown algae, Posidonia, hydroids, Alcyonaria, Zoantharia, Bryozoa, sponges, Arbacia (Echinodermata) and Libinia (Decapoda) (Krapp-Schickel, 1993). Guerra-García (2001b) found the species in intertidal exposed areas and in infralittoral areas of high hydrodynamics, clinging onto different species of algae and hydroids. The species has been also found in sponges, ascidians, spirorbids, gorgonaceans and Caulerpa beds (Guerra-García, 2004), and among mussels (Díaz et al., 2005)
Distribution
Type locality: Devonshire Coast, England (McCain and Steinberg, 1970). Other records: Atlantic Ocean, Indian Ocean, Pacific Ocean and Mediterranean Sea (Krapp-Schickel and Guerra-García, 2005). Doubtfully considered cosmopolitan.
Caprella danilevskii Czerniavskii, 1868
(Figures 5 - 6)
Caprella Danilevskii Czerniavskii, 1868: 92, pl. 6, figs. 21-34; Mayer, 1890: 58, pl. 5, fig. 44; pl. 7, figs. 12-13.
Caprella inermis Haswell, 1879: 319-351.
Caprella Danilewskii Chevreux and Fage, 1925: 454, fig. 432.
Caprella danilevskii McCain, 1968: 22, figs. 10-11; McCain and Steinberg, 1970: 16; Cavedini, 1982: 499; Krapp-Schickel, 1993: 779, fig. 531; Guerra-García and Takeuchi, 2002: 683-684, fig. 6; Guerra-García, 2004: 28-30, fig. 24; Guerra-García and Takeuchi, 2004: 1005-1007, figs. 28-29; Díaz et al., 2005: 3,4,12, fig.3; Guerra-García, 2006.
Material examined: 385 specimens
St. 21: 2 males, from hydroids, 0-0.5 m, 7.VI.1985; 6 males, 15 females, 14 juveniles, S. cymosum, 0-0.5 m, 7.VI.1985; 6 males, 2 females, 34 juveniles, hydroids, 0-0.5 m, 27.VI.1985; 5 males, 2 females (ov.), from intertidal hydroids, 5.VII.1985; 10 males, 6 females; from hydroids under overhanging rocks, 8.VII.1985; 18 males, 29 females (1ov.), 12 juveniles, from brown and red algae on rocky shore, intertidal, 15.VII.1985; 6 males, 1 ov.female, from s ? hydroids, intertidal, 15.VII.1985; 4 males, 6 females, 6 juveniles, from S. cymosum on dead Porites reef, 0-0.5m, 9.IX.1985; 1 male, hydroids, lower intertidal, 8.XI.1985; 2 males, 4 females, 2 juveniles, S. cymosum, 0-0.5 m, 7.XII.1985; 6 males, 20 females (10 ov.), 8 juveniles, S. cymosum on dead Porites reef, 0-0.5 m, 8.VII.1985; 1 male, 3 females (1 ov.), 1 juvenile, Digenia simplex, intertidal, 12.II.1986; 22 males, 20 females (4 ov.), 4 juveniles, S. cymosum, 0-0.5m, 3.IV.1986. St. 31: 17 females (5ov.), from Sargassum on rocky shore, lower intertidal, 12.VII.1985. St. 36: 18 males, 32 females, 40 juveniles, Punta Embestidero, from brown algae (C. schnetteri), 0.5 m, 23.IX.85.
Male and female figured from St. 21, 8.XI.1985.
Remarks
The present specimens of C. danilevskii collected from Colombia are in agreement with other material distributed worldwide (Guerra-García and Takeuchi, 2002). The distinctive male abdomen, the absence of grasping spines in the pereopods and the short dactylus of male gnathopod 2 distinguish clearly this species from other species of Caprella. Nevertheless, this species also shows some intraspecific morphological variation; recently, Guerra-García and Takeuchi (2004) studied material from Tasmania and found some males showing very elongate antenna 1, pereonites 1 and 2, basis and propodus of gnathopod 2. As the distance to the type locality is quite long, it could well be that this Colombian material is another species, but morphologically it matches with the typical description.
Habitat
The present material has been found attached mainly to S. cymosum and to the hydroid Cnidoscyphus, widely distributed in shallow waters. In previous studies, the species has been found in Cystoseira, Sargasum, amongst Bugula (Krapp-Schickel, 1993). Guerra-García (2001b) collected specimens from different species of algae in areas of high hydrodynamics. In Australian waters, the species has been found on brown algae, seagrass, sponges and tunicates (Guerra-García, 2004). Recently, the species has also been found in Venezuela clinging to algae. Caprella danilevskii and also C. penantis appear to have acquired the ability of attaching to the substrate using gnathopod 1 in a "parallel" posture rather than in the "upright" posture used by species living in calmer
waters. These species feed mainly by scraping (Takeuchi and Hirano, 1995; Guerra-García et al., 2002) and can be mainly found in very exposed areas, with high hydrodynamics and low values of sedimentation (Guerra-García and García-Gómez, 2001). Caprella danilevskii is one of the most studied caprellid species in respect of life history, based on Japanese specimens (Takeuchi and Hirano, 1991; 1992).
Distribution
Type locality: Black Sea (McCain and Steinberg, 1970). Other records: Mediterranean, South Africa, South Arabian coast, Bermuda, Venezuela, Brazil, Hawaii, north eastern Pacific, and Australia (McCain and Steinberg, 1970; Krapp-Schickel, 1993; Arimoto, 1976; Guerra-García, 2004; Díaz et al., 2005). Doubtfully considered cosmopolitan.
Deutella caribensis n.sp
(Figures 7 - 8)
Type material (9 specimens)
St. 21: Holotype male 2 slides (INV-CRU 5783) from Thalassia, 1 m, 27.VI.1985; Paratype female slide (INV-CRU 5784); additional paratype material in alcohol (MVRCr 450): 2 males, 2 females, 1 juvenile from Thalassia, 0.5-3 m, 9.IX.1985 and 2 males from Thalassia, 2-3 m, 7.XII.1985.
Type locality
Bahía Concha, Colombia, 0.5-3 m, Thalassia beds.
Etymology
Named caribensis alluding to the Caribbean Sea.
Description
Holotype male
Lateral view (Figure 7): Head with a pair of dorsal projections. Pereonite 1 fused with head, suture present. Pereonites 3 and 4 with a medial and a distal projection dorsally. Pereonite 4 with two medial projections dorsally. Pereonites 3 and 4 subequal. Pereonite 5 the longest. Pereonite 7 the shortest.
Gills (Figure 7): Elongate, length about four times width.
Mouthparts (Figure 7): Mandibles with 3-articulate palp; distal article of palp with a setal formula 1-6-1; second article provided with a simple setae; mandibular molar robust; left mandible with incisor and lacinia mobilis 5-toothed followed by three plumose setae; incisor of right mandible 5-toothed, lacinia mobilis serrate followed by 3 small plates; molar flake absent. Lower lip with well-demarcated inner lobes; outer lobes provided with setulae on apical margin. Maxilla 1 outer lobe with 6 robust setae. Maxilla 2 inner lobe triangular with 4 setae distally; outer lobe slightly larger than inner lobe, rectangular with 5 apical setae. Maxilliped inner plate rectangular with four setae and two robust and short seta (like "teeth"); outer plate about 1.5 times as large as inner plate, with six setae; palp 4-articulate, penultimate article of the palp without a distal projection, dactylus with two rows of setulae.
Antennae (Figure 7): Antenna 1 about half of body length; flagellum 7-articulate. Antenna 2 with short setae (no swimming setae); basal article of the peduncle with a distal projection; flagellum 2-articulate.
Gnathopods (Figure 8): Gnathopod 1 basis as long as ischium, merus and carpus combined; propodus length about 1.5 times width, palm with two proximal grasping spines and denticulate margin. Gnathopod 2 inserted on the anterior half of pereonite 2; basis almost as long as pereonite 2;
ischium rectangular; merus rounded; carpus short and triangular; propodus elongate, about 1.2 times as long as the basis; palm with a proximal projection carrying one grasping spine and two more triangular projections distally; dactylus long.
Pereopods (Figures 7, 8): Pereopods 3 and 4 subequal, 1-articulate, length about 1/3 of gills. Pereopod 5 less robust than pereopods 6 and 7, 6-articulate, distal article extremely reduced with a plumose seta. Pereopod 6 and 7 similar in feature but increasing in size respectively, 6-articulate; propodus with 2 proximal grasping spines.
Penes(Figure 8) rounded, situated laterally.
Abdomen (Figure 8) with a pair of rounded appendages 1-articulate, a pair of lateral lobes and a single dorsal lobe with 2 plumose setae.
Paratype female
Similar to the male holotype apart from the following characters: flagellum of antenna 1 with 6 articles; gnathopod 2 propodus smaller than in male, pereonite 5 also reduced with a tiny distal article but more robust than in male; oostegites on pereonite 3 very setose, on pereonite 4 scarcely setose; abdomen without appendages.
Remarks
The genus Deutella was established by Mayer (1890) and has been recently reviewed (Guerra-García, 2002a, 2002b, 2003a). Eleven species of this genus have been described so far: Deutella antonbruuni Guerra-García, 2002 D. aspiducha Gable and Lazo-Wasem, 1987, D. californica Mayer, 1890, D. incerta (Mayer, 1903), D. indica Guerra-García, 2002, D. margaritae Guerra-García, 2002, D. mayeri Stebbing, 1895, D. philippinensis Guerra-García, 2002, D. schieckei Cavedini, 1981, D. vemae (McCain and Gray, 1971) and D. venenosa Mayer, 1890.
Deutella caribensis n. sp. can be distinguished from the remaining species of Deutella mainly by the combination of the following characteristics: the arrangement of dorsal projections (2-1,1-1,1-2), which is unique in the genus; pereopods 3 and 4 uniarticulate; pereopod 5 clearly less robust than pereopods 6 and 7 and provided with a tiny distal article which does not look like a dactylus; a pair of 1-articulate abdominal appendages in males.
Habitat
Probably preferring shallow waters with Thalassia beds.
Distribution
So far, the species is only known from the type locality.
Deutella incerta (Mayer, 1903)
(Figures 9, 10, 11)
Luconacia incerta Mayer, 1903: 49-50, pl.2, figs. 11-14; pl.6, figs. 73-75; pl.9, figs. 21, 40, 57; McCain, 1968: 53-54, 68-72, figs. 33-35; McCain and Steinberg, 1970: 53.
Protellopsis stebbingii Kunkel, 1910: 11-113, fig. 43.
Deutella incerta Steinberg and Dougherty, 1957: 281, 285,286; Gable and Lazo-Wasem, 1987: 635-636, fig. 4; Guerra-García 2003a: 1062-1065, fig.3; Díaz et al. 2005: 4,5,15, fig.6.
Material examined: 410 specimens
St.4: 15 males, 5 females, 7 juveniles, hydroids + sponges, 25-30 m, 18.IX.1985; 16 males, 18 females, 1 juv., hydroids, 11-19m, 9.X.1985; 6 males, 3 females, 9 juveniles, from hydroids, 21-22 m, 9.XII.1985; 8 males, 6 females, 6 juveniles, from coral rubble with hydroids and bryozoans, 19.III.1986. St.8: 1 male, from shaded pillars of mole in harbor,
5-7 m, debris, sponges, bryozoans, 27.V.1985; 1 male, 2 females, debris and hydroids on stones, 28m, 3.VI.1985; 15 males, 18 females, 21 juvenile, detritus, hydroids on coral rubble, 22-23m, 19.VI.1985; 8 males, 1 female, 24 juv., hydroids, 25-27m, 22. VI. 1985; 2 males, 7 females, 12 juvenile; coral rubble, 15m, 1.VII.1985; 1 male, Thalassia, 2 m,
1.VIII.1985; 8 males, 7 females (1 ov.), 2 juveniles, coral rubble with hydroids, 13-19 m, 4.VIII.1985; 22 males, 16 females, 12 juveniles, hydroids, 22-27 m, 15.IX.1985; 4 males, 3 females, 1 juvenile, hydroids, 20-28m, 21.IX.1985; 8 males, 4 females, 7 juveniles, coral rubble with hydroids, 22-27 m, 1.XII.1985; 2 males, coral rubble with hydroids and bryozoans, 30m, 10.II.1986. St. 12: 1 male, coral rubble, 16-18m, 16.IX.1985; 1 male, 7 females, 11 juvenile, from hydroids and octocorals, 16.IX.1985; 20 males, 22 females, 21 juvenile, from hydroids and bryozoans, 11-15m, 2.X.1985. St. 13:8 males, 5 females, 1 juvenile, coral rubble, 30m, 18.II.1986. St. 45: 3males, 1female; algae and hydroids on rocks, 6-10 m, 26.IX.1985.
The figured male and female are from St. 8, 1.XII.1985.
Remarks
A detailed discussion regarding the status of D. incerta is included in Guerra-García (2003a). McCain (1968) pointed out that this species varies considerably in the degree of body spination; larger individuals bear dorsal projections while these are lacking in the smaller ones. The specimens of the present study are provided with the typical dorsal projections on the head and pereonite 2 and are in agreement with the material described and figured by McCain (1968) collected from the Western North Atlantic. The species has been recently collected also from the Venezuelan coast by Díaz et al. (2005).
Habitat
The Colombian specimes have been collected from hydroids, sponges, bryozoans, detritus and coral rubble. The material was mainly found in greater depth, rarely in shallow water, distributed from Santa Marta in eastern direction to Cañaverales. Previously, Deutella incerta had been reported from mangrove roots, Sargassum sp., Thalassia sp., sponges, hydroids, alcyonarians and ascidians, sandy bottoms, and occasionally it has been taken in plankton tows (Guerra-García, 2003a; Díaz et al., 2005). It has been recently reported inhabiting soft bottoms within a depth range of 116 to 1470 m (Winfield et al. 2006).
Distribution
Type locality: off Mobile Bay, Alabama, 29o 24' N, 88o 04' W. Other records: Bermuda, east coast of North America from Woods Hole, Massachusetts to Straits of Florida; Gulf of Mexico from Cedar Keys, Florida to Yucatan; Virgin Islands; Barbuda; Barbados; Isla Margarita, Venezuela; Cuba (Guerra-García, 2003a; Ortiz and Lalana, 1998).
Deutella mayeri Stebbing, 1895
(Figures 12, 13)
Deutella mayeri Stebbing, 1895: 400-402, pl. 15a; Mayer, 1903: 44-45; McCain, 1968: 54-57, figs. 25,26,52; McCain and Steinberg, 1970: 48; Guerra-García, 2003a: 1070-1071, fig.8.
Material examined: 155 specimens
St. 3: 2 males, 4 females, 1juveniles, from stones covered with algae, 0.5-1 m, 30.XII.1985. St. 8: 6 males, 4 females (3 ov.), from short algae on rocks, 2-3 m, 24.XII.85; 7 males, 7 females, from algae on rocky shore, 0.5-1 m, 29.XII.1985; 4 males, 3 females, 4 juveniles, from algae on rocks, 0-1 m, 29.I.1986. St. 21: 16 males, 3 females (1 ov.), from Halocordyle-hydroids on rocks, 1 m , 23.VI.1985; 2 males, 1 female, Thalassia, 2-3 m, 7 XII.1985; 2 males, 5 females, 1 juvenile, Thalassia, 1-2 m, 2.IV.1986. St.31: 5
males, Thalassia, 2 m, 6.VI.1985. St. 36: 8 males, 5 females, 4 juveniles, Thalassia, 0.5 m, 27.V.85; 7 males, 5 females (1 ov.), 4 juveniles, Thalassia, 0.5 m, 20.XII.85; 2 males, 2 females, Thalassia, 0.5 m, 17.I.86. St. 41: 8 males, 8 females, from stones, intertidal-0.5 m, 17.III.86; 3 males, 9 females, from stones covered with algae, 0-0.5 m, 14.IV.86. St. 45: 7 males, 4 females, Thalassia, 1-1.5 m, 25.II.86. Isla de Providencia: 2 males, 1 female (ov.), Thalassia, 5.XII.80, leg. INVEMAR.
Remarks
Deutella mayeri was redescribed and illustrated in detail by McCain (1968). The material from Colombia matches material figured by McCain (1968). The gnathopod 2 of males is very characteristic; it has a triangular elongate process proximally, provided with a grasping spine.
Habitat
The present material was found in shallow waters at many of the sampling stations, mainly clinging on Thalassia, but also living on hydroids and algae. The extant ecological information about this species is very scarce (Guerra-García, 2003a).
Distribution
Type locality: Antigua (Caribbean Sea). Other records: Bahía Fosforescente, Puerto Rico (Guerra-García, 2003a).
Hemiaegina minuta Mayer, 1890
(Figures 14, 15, 16)
Hemiaegina minuta Mayer, 1890: 40, pl. 1, figs. 25-27, pl. 3, figs. 32-35, pl. 5, figs. 52-53, pl. 6, figs. 13, 33-34, pl. 7, fig. 4; McCain, 1968: 61-64, figs. 29-30; McCain and Steinberg, 1970: 51; Gable and Lazo-Wasem, 1987: 637; Müller, 1990: 836; Serejo, 1997: 630-632, fig. 1; Guerra-García, 2003b:105-106, fig. 10; Guerra-García 2003c: 6-7, fig. 3; Guerra-García, 2004: 39-40, fig. 32; Díaz et al., 2005: 5,6,18, fig.9; Krapp-Schickel and Guerra-García, 2005: 50,51, fig. 3.
Hemiaegina quadripunctata Sundara Raj, 1927, p. 126-127, pl. 18.
Hemiaegina costai Quitete, 1972: 165-168, pls. 1-2.
Material examined: 67 specimens
St. 4: 2 males, 1 female, from algae on rocks, intertidal, 24.V.1985. St. 8: 1 male, 1 juvenile, algae on rocks, lower intertidal, 12.V.1985; 1 male, 2 females, 2 juveniles, from Sargassum on rocky shore, 0.5 m, 15.IX.1985; 1 male; from algae on rocks, 0.5-1 m, 29.XII.1985. St. 21: 2 males, 1 female, from algae, intertidal, 22.V.1985; 1 male, Thalassia, 1 m, 27.VI.1985; 2 males, from D. simplex, lower intertidal, 8.VII.1985; 1 female, 2 juveniles, from D. simplex, lower intertidal, 13.VIII.1985; 1 female, S. cymosum, lower intertidal-0.5 m, 9.IX.1985; 1 male, Thalassia, 2-4 m, 28.X.1985; 2 males, 4 females (1ov.), 3 juveniles, from S. cymosum on dead Porites reef, 0-0.5 m, 8.XI.1985; 4 males, 5 females, 5 juveniles, S. cymosum on dead Porites reef, 0-0.5 m, 7.XII.1985; 4 males, 2 females, 1 juvenile; from D. simplex on dead Porites-reef, intertidal, 12.II.1986; 1 male, D. simplex, lower intertidal, 3.III.1986; 2 males, D. simplex, lower intertidal, 3.IV.1986; 1 male, 1 female, S. cymosum, 0-0.5 m, 3.IV.1986. St. 36: 1 male, 1 female, 5 juveniles, Playa del Muerto, Thalassia, hydroids and brown algae, 0.5-2 m, 30.VII.1985. St. 45: 2 males, from algae and hydroids on rocks, 6-10 m, 26.IX.1985.
Additional sample: 1 female, from stomach of Acanthurus coeruleus; Santa Marta area, 5 m, leg. Sergio Duarte, 1.VII.1985.
The figured male and female come from St. 21, 8.XI.1985.
Remarks
The material of Hemiaegina minuta from Colombian waters agrees with the descriptions of specimens from other areas of the world. This species shows very distinctive and clear diagnostic characters which facilitate identification: third article of antenna 1 short, antenna 2 without swimming setae, gnathopod 1 propodus with a round projection proximally, pereonites 3 and 4 rounded, small and abdomen provided with a very distinctive pair of two-articulate appendages. The pereopods 5-7, usually lost in material from previous studies, are fully figured here based on the material from Colombia.
Habitat
The Colombian specimens were collected mainly from algae, specially D. simplex and Sargassum. Hemiaegina minuta has been previously collected from Sargassum sp. and taken in plankton tows (McCain and Steinberg, 1970). Müller (1990) reported H. minuta as preferring more or less exposed reef locations. Guerra-García (2003b and 2003c) found the species associated to algae in Papua New Guinea and Mauritius. This species was also found associated to many different substrata in Queensland: green, brown and red algae, sponges, tunicates, seagrass,dead corals encrusted with algal turf, and under small boulders (Guerra-García, 2006). This species has also been found on Arca zebra (Díaz et al., 2005).
Distribution
Type locality: Off Amoy, China, 15-46 m. (McCain, 1968). Other records: West coast of United States, South Africa, Hawaii, Bora Bora, Japan, Papua New Guinea, Australia, India, Mauritius, Indonesia, Venezuela, South Arabian coast (McCain and Steinberg, 1970; Guerra-García, 2003b, 2003c, 2004, 2006; Díaz et al., 2005; Krapp-Schickel and Guerra-García, 2005). Hemiaegina minuta is widely distributed in tropical and temperate waters of the world oceans (McCain, 1968).
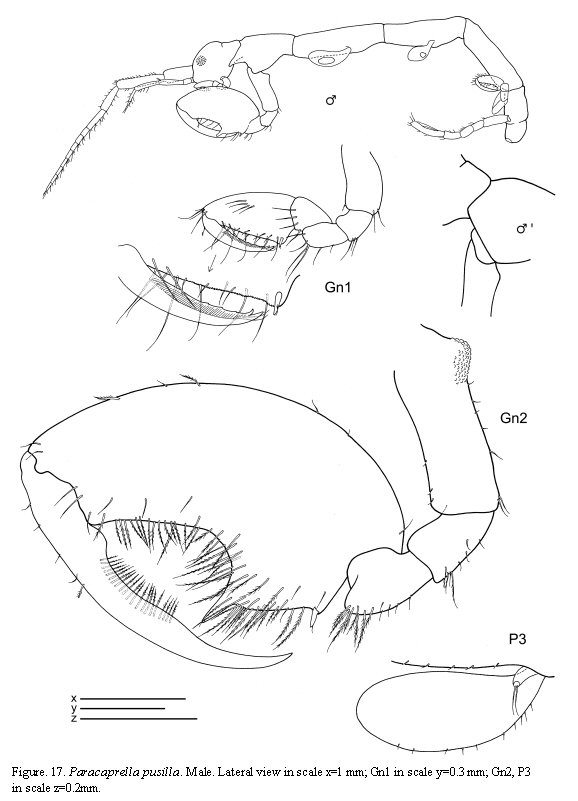
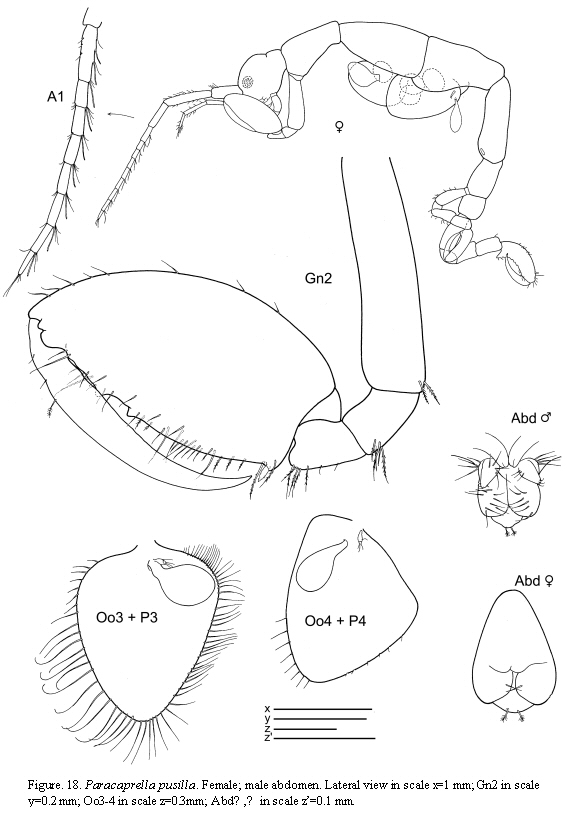
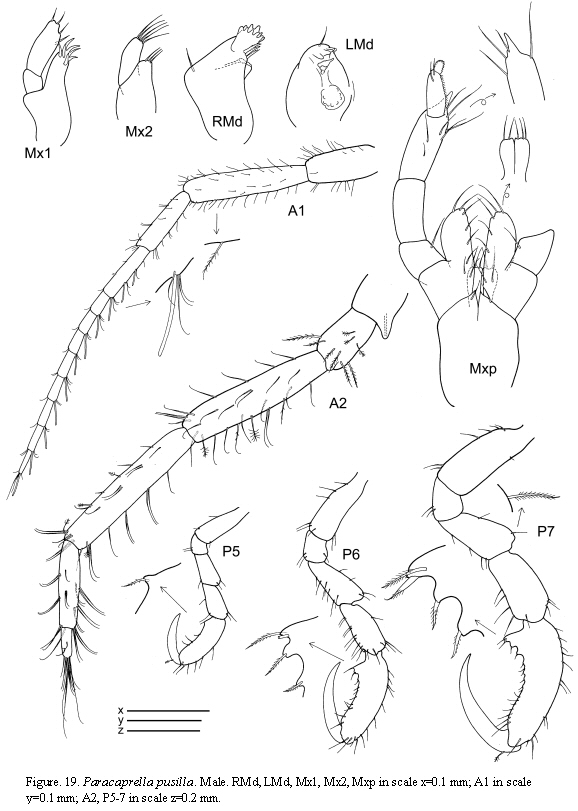
Paracaprella pusilla Mayer, 1890
(Figures 17, 18, 19)
Paracaprella pusilla Mayer, 1890: 41, pl. 1, figs. 28-30; pl. 3; figs. 45-47; pl. 5, figs. 48-49; pl. 6, fig. 10; 1903: 67, pl. 2, figs. 36-37; pl. 7, fig. 52; Steinberg and Dougherty, 1957: 283-284, figs. 16, 19, 24, 30; McCain, 1968: 82-86, figs. 41-42; Wakabara et al., 1991: 73; Camp, 1998: 132; Guerra-García and Thiel, 2001: 880, fig. 8; Díaz et al., 2005: 6,7,22, fig. 13.
Caprella nigra Reid, 1951: 283-284, 289, fig. 58.
Material examined: 454 specimens
St. 8: 4 males, 2 females (1ov.), 2 juveniles, sponges and hydroids on pillars, 0-1 m, 24.IV.1985; 22 males, 25 females (14 ov.), 8 juveniles, fouling on pilings, in darkness, 0-1 m, 25.IV.1985; 14 males, 19 females, 12 juveniles, sponges and hydroids on shaded mole pillars, 0-1 m, 4.V.1985; 3 males, 2 females, 5 juveniles, light-exposed mole pillars,
0-1 m, 8.V.1985; 11 males, 16 females (5 ov.), 9 juveniles, light-exposed pillars, from algae, sponges and hydroids, 0-1 m, 10.V.1985; 5 males, 2 females (1 ov.), 1 juvenile, sponges and hydroids on light-exposed mole pillars, 0-1 m, 14.V.1985; 19 males, 25 females, from sponges and hydroids on pilings, in darkness, 0-1 m, 16.V.1985; 2 males, 17 females (5 ov.), 6 juveniles, sponges and hydroids on pillars, 0-1 m, 18.V.1985;
7 males, 10 females (5 ov.), 7 juveniles, sponges and hydroids on pillars, 19.V.1985; 5 males, 2 females, 8 juveniles, pillars, from hydroids and bryozoans, 17.VI.1985; 31 males, 21 females (6 ov.), 7 juveniles, from hydroids and bryozoans on pilings, exposed to light, 0-1 m, 21.VI.1985; 27 males, 20 females (10 ov.), 26 juveniles, from sponges and hydroids on pilings, in darkness, 0-1 m, 21.VI.1985; 20 males, 12 females, fouling on pilings, exposed to light, 0.5 m, 3.IX.1985; 7 males, 6 females, sponges and hydroids on pillars, 14.III.1986. St. 31: 4 males, 3 females, Thalassia, 1-2m, 17.I.1986.
The figured male and female come from St. 31, 17.I.1986.
Remarks
The material of P. pusilla from Colombia agrees completely with previous descriptions of material from the Caribbean coasts (Díaz et al., 2005). Paracaprella pusilla is similar to P. tenuis Mayer, 1903; however, males of P. pusilla can be distinguished from those of P. tenuis by the large sharp-pointed projection on the anteroventral margin of pereonite 2, the proximal knob on the basis of gnathopod 2, and the presence of setae on the dactylus of gnathopod 2 (McCain, 1968). Large males of P. pusilla are very similar to large males of P. barnardi McCain, 1967 in that they both bear a small anterodorsal tubercle on pereonite 2; however, the tubercle is not as well developed in the former than in the later, and the ventrolateral projection on the anterior margin of pereonite 2 is much larger in P. pusilla (McCain, 1967).
Habitat
The present specimens were mostly found in shallow waters, mainly near INVEMAR. Paracaprella pusilla has traditionally been collected from mangrove roots, seagrasses, hydroids and ascidians (McCain, 1968), but also on gravel bottoms, ropes, mussels and oysters (Díaz et al., 2005).
Distribution
Type locality: Rio de Janeiro, Brazil (McCain, 1968). Other records: Western North Atlantic, Suez Canal, tropical west Africa, South Africa, Tanzania, Hawaii, China, Gulf of Mexico, Cuba, Venezuela (McCain, 1968; Wakabara et al., 1991; Ortiz and Lalana, 1998; Díaz et al., 2005; Winfield et al., 2006).
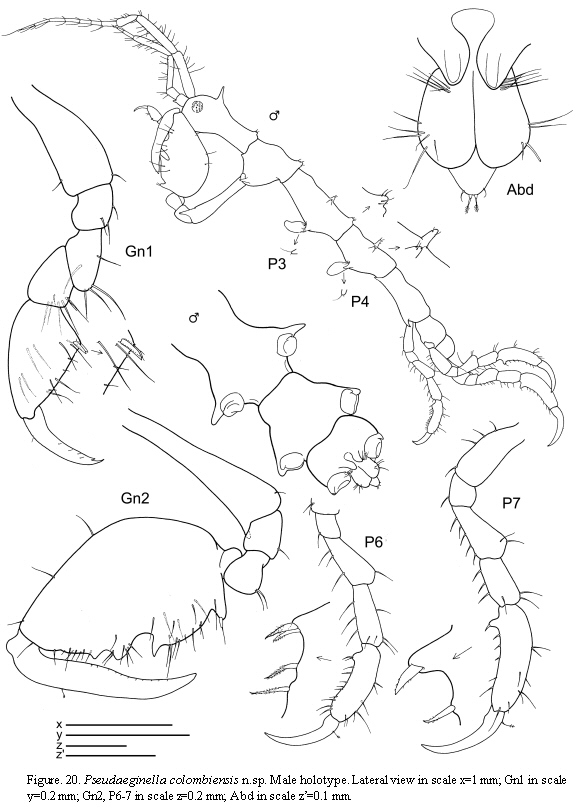
Pseudaeginella colombiensis n.sp
(Figures 20, 21, 22)
Type material (19 specimens)
St. 45: Holotype male 2 slides (INV-CRU 5785), from algae, hydroids and bryozoans on rocks, 6-10 m, 26.IX.1985; Paratype female (ov.) slide (INV-CRU 5786), collected together with the holotype; paratypes 10 males, 7 females in alcohol (MVRCr 449) from Thalassia, 1.5 m, 25.II.1986.
Type locality
Punta El Diamante (Arrecifes/Cañaverales), Colombia, 1.5-10 m, algae, hydroids and bryozoans on rocky slope.
Etymology
Named "colombiensis" after Colombia, the South American country where the specimens were collected.
Description
Holotype male
Lateral view (Figure 20): Head with a dorsal acute projection; suture between head and pereonite 1 non-marked. Pereonite 1 and pereonite 2 with a dorsal hump distally. Pereonite 2 with an apical projection laterally near the insertion of gnathopod 2. Pereonites 3 and 4 with a pair of dorsomedial projections and laterally acute projections near the insertion of the gills. Pereonite 5 with a lateroventral acute projection near the insertion of pereopod 5.
Gills (Figure 20): Present on pereonites 3 and 4, oval, length 2 times width.
Mouthparts (Figure 21): Mandibles with no sign of mandibular molar; left mandible with incisor 5-toothed, lacinia mobilis 5-toothed followed by three plates minutely serrate; right mandible with incisor 5-toothed, lacinia mobilis transformed into a plate, followed by two other plates; molar flake absent; palp three-articulate, second article with a single seta, distal article with a distal robust setae and a row of three setae. Maxilla 1 outer lobe carrying 5 robust seta; distal article of the palp with 4 setae. Maxilla 2 inner lobe triangular, with 3 setae; outer lobe rectangular, about 1.5 times as long as inner lobe, with 4 setae. Maxilliped inner plate small and rounded with a setae; outer plate elongate, 2.5 times as long as the inner plate, with 5 setae; palp 4-articulate, dactylus curved, with row of setulae.
Antennae (Figure 21): Antenna 1 about 2/3 of body length; flagellum 7-articulate. Antenna 2 without swimming setae but provided with abundant short setae; flagellum two-articulate.
Gnathopods (Figure 20): Gnathopod 1 basis as long as the combination of ischium, merus and carpus; palm of propodus non-serrate, provided with two grasping spines; dactylus margin smooth. Gnathopod 2 inserted on the middle of pereonite 2; basis 1.2 times as long as pereonite 2, with a projection distally; ischium rectangular; merus rounded; carpus triangular; propodus rounded, 1.5 as long as wide, provided with a grasping spine proximally and three triangular projections medially and distally respectively; dactylus with minute setae on the margin.
Pereopods (Figure 20): Pereopods 3 and 4 minuscule, 1-articulate, triangular, provided with two distal setae. Pereopods 5, 6 and 7 similar in feature but increasing in size respectively; palm of propodus with a group of robust plumose setae proximally.
Penes (Figure 20) large, length about 2 times width.
Abdomen (Figure 20) without appendages, with a pair of lateral lobes and a single dorsal lobe provided with a pair of plumose setae.
Paratype female
Similar to holotype male apart from the following characters: flagellum of antenna 1 with 5 articles; oostegites on pereonite 3 very setose, on pereonite 4 scarcely setose; abdomen without appendages.
Remarks
The genus Pseudaeginella was composed of nine species before this study: P. antiguae Barnard, 1932 from Antigua, P. biscaynensis (McCain, 1968) from Florida, P. cambellensis Guerra-García, 2003 from Subantarctica, P. inae Krapp-Schickel and Guerra-García, 2005 from Indonesia, P. montoucheti (Quitete, 1971) from Brazil, P. polynesica
(Müller, 1990) from Bora Bora and Moorea, Society Islands, P. sanctipauli Laubitz, 1995 from St Paul and Amsterdam Islands, P. tristanensis (Stebbing, 1888) from Tristan da Cunha, and P. vaderi Guerra-García, 2004 from the Indian Ocean. Laubitz (1995), after examination of specimens of P. tristanensis from Southern Indian Ocean, considered the genus Fallotritella synonymous with Pseudaeginella mainly based on the presence of minute pereopods 3 and 4 in Pseudaeginella. Consequently, the species P. byscaynensis, P. montoucheti and P. polynesica, previously included in Fallotritella, were transferred to Pseudaeginella. Species of Pseudaeginella are compared in Laubitz (1995).
Pseudaeginella colombiensis is very close to P. biscaynensis but these two species can be distinguished mainly by the arrangement of dorsal and lateral projections, the shape of gnathopod 2 and the structure of mandibular palp.Habitat
Probably preferring shallow waters. It is remarkable that it was found only in the eastern area of the studied region.
Distribution
So far, the species is only known from the type locality.
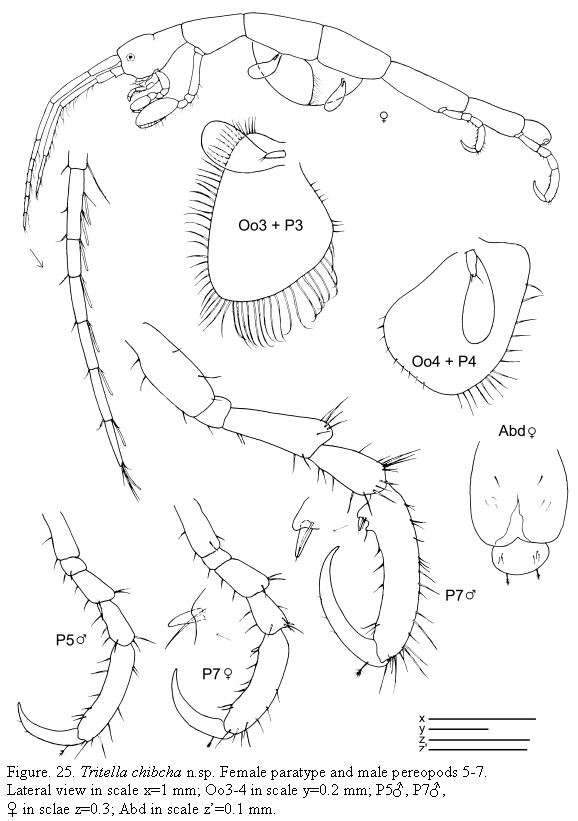
Tritella chibcha n.sp.
(Figures 23, 24, 25)
Type material (5 specimens)
St. 4: Holotype male 2 slides (INV-CRU 5787), from Syringodium, 3 m, 12.XII.1985; Paratype female slide (INV-CRU 5788), collected together with the holotype; 3 males paratypes (MVRCr 451) from the same locality.
Additional material
St. 41: 3 males, 1 female from Syringodium, 3 m, 20.XII.1985 (at MVR).
Type locality
Bahía de Santa Marta, Colombia, 3 m, Syringodium.
Etymology
When the Spanish conquered Colombia in the 15th century, the Chibcha culture was one of the three main civilisations in Colombia, together with Caribe and Arwac. Chibcha occupied the central part of the country and the Sierra Nevada de Santa Marta, the area in which this study was conducted.
Description
Holotype male
Lateral view (Figure 23): Body smooth. Pereonite 1 fused with head, suture present. Pereonites 3, 4 and 5 subequal in length. Pereonite 7 the shortest.
Gills (Figure 23): Oval, length about 2 times width.
Mouthparts (Figure 23): Mandibles with 3-articulate palp; distal article of palp with 2 setae; second article provided with a simple seta; mandibular molar robust; left mandible with incisor and lacinia mobilis 5-toothed followed by a row of plumose setae; incisor of right mandible 5-toothed, lacinia mobilis serrate followed by another plate and three more setae. Lower lip with well-demarcated inner lobes; inner and outer lobes provided with setulae on apical margin. Maxilla 1 outer lobe with 7 robust setae; palp with four distal b setae and 4 lateral setae. Maxilla 2 inner lobe triangular; outer lobe slightly larger than inner lobe, rectangular. Maxilliped inner plate rectangular with 7 plumose setae and 2 robust and short setae (like "teeth"); outer plate about 2 times as large as inner plate; palp 4-articulate, dactylus short and robust.
Antennae (Figure 24): Antenna 1 about half of body length; flagellum 7-articulate. Antenna 2 with short setae (no swimming setae); basal article of the peduncle with a distal projection; flagellum 2-articulate.
Gnathopods (Figure 24): Gnathopod 1 basis as long as ischium, merus and carpus combined; propodus length about 2 times width, palm with 1 proximal grasping
spines and denticulate margin. Gnathopod 2 inserted on the anterior half of pereonite 2; basis about 2/3 as long as pereonite 2; ischium rectangular; merus rounded; carpus short and triangular; propodus elongate, about 1.2 times as long as the basis; palm with a proximal projection carrying one grasping spine and other projection distally.
Pereopods (Figures 23, 25): Pereopods 3 and 4 subequal, 1-articulate, length about 1/5 of gills. Pereopod 5 less robust than pereopods 6 and 7, 6-articulate, propodus without grasping spines. Pereopod 6 and 7 similar in feature but increasing in size respectively, 6-articulate; propodus with 1 proximal grasping spines.
Penes(Figure 23) short and triangular, situated medially.
Abdomen (Figure 23) without appendages, a pair of lateral lobes and a single dorsal lobe with 2 plumose setae.
Paratype female
Similar to holotype male apart from the following characters: flagellum of antenna 1 with 6 articles; oostegites on pereonite 3 very setose, on pereonite 4 scarcely setose.
Remarks
Until now four species are described in the genus Tritella, distributed along the Pacific coast of North America: T. laevis Mayer, 1903, T. ornata Mayer, 1903, T. pilimana Mayer, 1890 and T. tenuissima Dougherty and Steinberg, 1953. Tritella laevis and T. pilimana have been fully redescribed by Laubitz (1970). Tritella chibcha clearly differs from these two species by the absence of lateral projections, the different shape of gnathopod 2, the different setal formula of the mandibular palp (being only 2 setae in T.chibcha and 1-x-1 in T. laevis and T. pilimana) and the swimming setae of antenna 2 being clearly shorter in T. chibcha. Tritella ornata can also be distinguished clearly from T. chibcha by the presence of abundant dorsal projections, which are lacking in T. chibcha. Tritella tenuissima seems to be the most similar species to T. chibcha, both species having an elongate and smooth body. However, in T. tenuissima the distal article of the mandibular palp has numerous setae, but only two are present in T. chibcha. Furthermore, a pair of grasping spines is present in pereopods 5,6 and 7 in T. tenuissima and only one grasping spine can be found in pereopods 6 and 7 of T. chibcha (pereopod 5 has no grasping spine). The insertion of pereopod 5 is posterior in T. chibcha and at midlength in T. tenuissima. Dougherty and Steinberg (1953) described T. tenuissima on the basis of the absence of swimming setae, pointing out that the genus Triliropus Mayer, 1903 appeared to be almost identical to Tritella, except by the lack of swimming setae on the second antenna and the questionable presence of a vestigial pair of abdominal appendages in the female. Although T. tenuissima lacks swimming setae, Dougherty and Steinberg (1953) felt that this species was indeed a member of Tritella and suggested that Triliropus could be certainly a synonym of Tritella. On the other hand, McCain (1968) remarked that T. tenuissima differs from the other species of Tritella in lacking swimming setae and in having pereopod 5 inserted near midlength on pereonite 5 and suggested that this species should be probably transferred to the genus Triliropus. The material from Colombia is characterized by intermediate characters, since the swimming setae are very reduced in T. chibcha, but the pereopod 5 is inserted posteriorly on pereonite 5. At the moment we have preferred to include this new species in Tritella instead of Triliropus. Future studies dealing with the genus Triliropus should be conducted to clarify its taxonomic status and validity.
Habitat
Syringodium is the preferred biotope of this species.
Distribution
Bahías de Santa Marta and Cinto, Colombia.
Incertae sedis
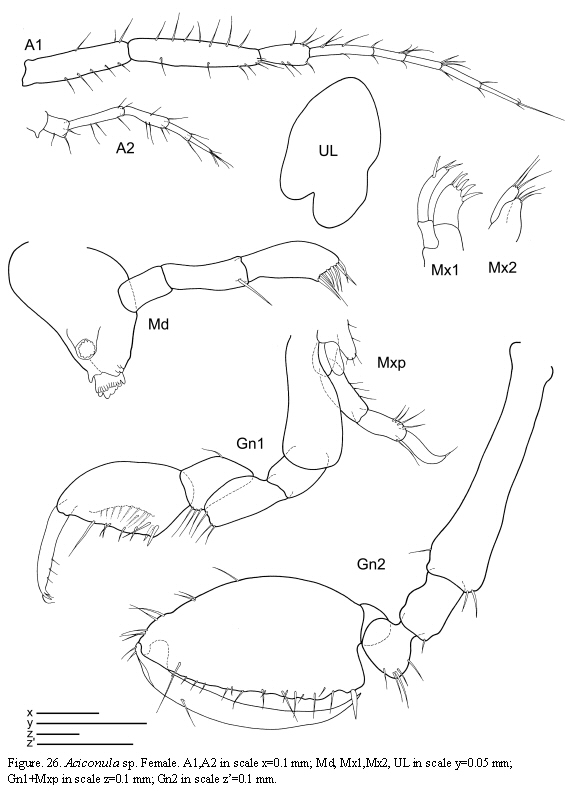
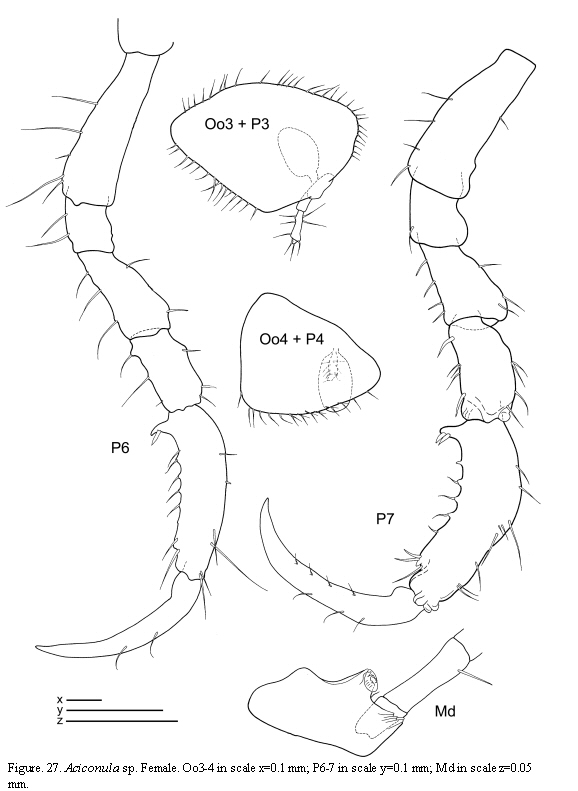
Aciconula sp.
(Figures 26 - 27)
Material examined: 1 specimen
St. 12: 1 female, hydroids and bryozoans, 2.X.1985.
Remarks
Three species of Aciconula have been reported so far: Aciconula miranda Mayer, 1903 from Malaysia and Thailand; A. acanthosoma Chess, 1989 from California, and A. australiensis Guerra-García,
Habitat
The specimen was found among hydroids and bryozoans.
KEY FOR COLOMBIAN CARIBBEAN CAPRELLIDS (based on adult specimens)
1.Pereopods 3 and 4 absent .................................................................................................................2
- Pereopods 3 and 4 present..................................................................................................................3
2.Head with rostrum. Pereopods provided with a pair of grasping spines...............................................
...........................................................................................................Caprella penantis (Figures 2, 3, 4)
-Head without rostrum. Pereopods without grasping spines............................................................
........................................................................................... Caprella danilevskii (Figures 5, 6)
3. Pereopods 3 and 4 1-articulate.................................................................................................................4
- Pereopods 3 and 4 at least 2-articulate.....................................................................................................7
4. Body smooth...............................................................................................................................................5
- Body with dorsal projections......................................................................................................................6
5. Abdomen with a pair of long 2-articulate appendages. Basis of gnathopod 2 longer than pereonite
2..............................................................................................................Hemiaegina minuta (Figures 14, 15, 16)
-Abdomen without appendages. Basis of gnathopods 2 shorther than pereonite 2................................
.............................................................................................................. Tritella chibcha (Figures 23, 24, 25)
6. Head with a dorsal projection. Lateral projectuions present. Pereopods 3 and 4 tiny, almost
undistinguishable............................................................Pseudaeginella colombiensis n.sp. (Figures 20, 21, 22)
-Head with 2 dorsal projections. Lateral projections absent. Pereopods 3 and 4 clearly
distinguishable..............................................................................................Deutella caribensis (Figures 7-8)
7. Pereopod 3 with 4 articles and pereopod 4 with 2 articles........................................................
...................................................................................................Aciconula sp. (Figures 26,27)
-Pereopods 3 and 4 with 2 articles.............................................................................................................8
8. Head and pereonite 2 with dorsal projections.........................................Deutella incerta (Figures 9, 10, 11)
-Body smooth...................................................................................................................................................9
9. Propodus of gnathopod 2 with an elongate projection proximally (see Gn2 in Figure 12).....................
.............................................................................................................. Deutella mayeri (Figures 12,13)
-Propodus of gnathopod 2 with trapezoidal projection proximally (see Gn 2 in Figure
17).........................................................................................................Paracaprella pusilla (Figures 17, 18, 19)
ACKNOWLEDGEMENTS
The third author wants to thank the staff of INVEMAR, for a stay of 14 months, supported by the DAAD (Deutscher Akademischer Austauschdienst). The late G. Bula Meyer kindly determined some of the algal species.
LITERATURE CITED
1. Arimoto, I. 1976. Taxonomic studies of caprellids (Crustacea, Amphipoda, Caprellidae) found in the Japanese and adjacent waters. Special Publications from the Seto Marine Biological Laboratory, Kyoto University, Series III, 1-229. [ Links ]
2. Camp, D. K. 1998. Checklist of shallow-water marine Malacostracan Crustacea of Florida. pp: 123-189 in: Camp, D. K., W.G. Lyons and T.H. Perkins (ed) Checklists of selected shallow-water marine invertebrates of Florida. Florida Marine Research Institute Technical Report TR-3, 238 p. [ Links ]
3. Cavedini, P. 1982. Contributo alla conoscenza dei Caprellidi del Mediterraneo (Crustacea, Amphipoda). Boll. Mus. Civ. Stor. Nat. Verona, 8: 493-531. [ Links ]
4. Chevreux, E. and L. Fage. 1925. Amphipodes. Faune de France (P. Lechevalier ed.), Paris. 488 p. [ Links ]
5. Czerniavskii, V. 1868. Materialia ad zoographiam Ponticam comparatam. Trav. Soc. Natural. St. Pétersbg., 1: 19-136. [ Links ]
6. Díaz, Y.J., J.M. Guerra-García and A. Martín. 2005. Caprellids (Crustacea: Amphipoda: Caprellidae) from shallow waters of the Caribbean coast of Venezuela. Org. Div. Evol., 10: 1-25. [ Links ]
7. Dougherty, E.C. and J.E. Steinberg. 1953. Notes on the skeleton shrimps (Crustacea: Caprellidae) of California. Proc. Biol. Soc. Wash., 66: 39-50. [ Links ]
8. Escobar-Briones, E. and I. Winfield. 2003. Checklist of the benthic Gammaridea and Caprellidea (Crustacea: Peracarida: Amphipoda) from the Gulf of Mexico continental shelf and slope. Belg. J. Zool., 133 (1): 37-44. [ Links ]
9. Gable, M.F. and E.A. Lazo-Wasem. 1987. The caprellids (Amphipoda: Caprellidea) of Bermuda: a survey of specimens collected from 1876-1987, including cave inhabitants, and the description of Deutella aspiducha, new species. Proc. Biol. Soc. Wash., 100: 629-639. [ Links ]
10. González, E.R. and L. Watling. 2003. A new species of Hyalella from Colombia, and the redescription of H. meinerti Stebbing, 1899 from Venezuela (Crustacea: Amphipoda). J. Nat. Hist., 37: 2095-2111. [ Links ]
11. Guerra-García, J.M. 2001a. Redescription of Caprellina longicollis (Nicolet, 1849) (Amphipoda, Caprellidea, Phtiscidae) from Chile, with notes on ontogenetic development and clinging behaviour. Crustaceana, 74(11): 1291-1303. [ Links ]
12. Guerra-García, J.M. 2001b. Habitat use of the Caprellidea (Crustacea: Amphipoda) from Ceuta, north Africa. Ophelia, 55(1): 27-38. [ Links ]
13. Guerra-García, J.M. 2002a. Littoral caprellids (Crustacea: Amphipoda: Caprellidea) from Philippines, with the description of a new species. Raffles Bull. Zool., 50(2): 395-406. [ Links ]
14. Guerra-García, J.M. 2002b. Two new species of Deutella Mayer, 1890 (Crustacea: Amphipoda: Caprellidea) collected by the R. V. "Anton Bruun" during the International Indian Ocean Expedition. Zootaxa,74:1-18. [ Links ]
15. Guerra-García, J.M. 2003a. Revision of the genus Deutella (Crustacea: Amphipoda: Caprellidea) with description of a new species, redescription of Deutella venenosa Mayer, 1890 and a key to the species of Deutella. J. Nat. Hist., 37: 1059-1084. [ Links ]
16. Guerra-García, J.M. 2003b. Caprellids (Crustacea: Amphipoda) from Papua New Guinea, with the description of a new species. Helg. Mar. Res., 57: 100-109. [ Links ]
17. Guerra-García, J.M. 2003c. The Caprellidea (Crustacea: Amphipoda) from Mauritius Island, Western Indian Ocean. Zootaxa, 232: 1-24. [ Links ]
18. Guerra-García, J.M. 2004. The Caprellidea (Crustacea, Amphipoda) from Western Australia and Northern Territory, Australia. Hydrobiologia, 522: 1-74. [ Links ]
19. Guerra-García, J.M. 2006. The Caprellidae (Crustacea: Amphipoda) from the Great Barrier Reef and adjacent localities. Rec. Aust. Mus., (In press). [ Links ]
20. Guerra-García, J.M. and J.C. García-Gómez. 2001. The spatial distribution of Caprellidea (Crustacea: Amphipoda): a stress bioindicator in Ceuta (North Africa, Gibraltar area). PSZNI Mar. Ecol., 22(4): 357-367. [ Links ]
21. Guerra-García, J.M. and I. Takeuchi. 2002. The Caprellidea (Crustacea: Amphipoda) from Ceuta, North Africa, with the description of three species of Caprella, a key to the species of Caprella, and biogeographical discussion. J. Nat. Hist., 36(6): 675. [ Links ]
22. Guerra-García, J.M. and I. Takeuchi. 2004. The Caprellidea (Crustacea: Amphipoda) from Tasmania. J. Nat. Hist., 38: 967-1044. [ Links ]
23. Guerra-García, J.M. and M. Thiel. 2001. The caprellid fauna (Crustacea: Amphipoda: Caprellidea) from the coast of Coquimbo, Northern-central Chile, with a taxonomic key for species identification. Rev. Chil. Hist. Nat., 74: 873-883. [ Links ]
24. Guerra-García, J.M., J. Corzo and J.C. García-Gómez. 2002. Clinging behaviour of the Caprellidea (Crustacea: Amphipoda) from the Strait of Gibraltar. Crustaceana, 75(1): 41-50. [ Links ]
25. Guerra-García, J.M., S. Redondo-Gómez, A.G. Espina, J.M. Castillo, T. Luque, J.C. García-Gómez and M.E. Figueroa. 2006. Caprella penantis Leach, 1814 and Caprella dilatata Kroyer, 1843 (Crustacea: Amphipoda) from the Strait of Gibraltar: a molecular approach to explore intra- and interspecific variation. Mar. Biol. Res., 2: 100-108. [ Links ]
26. Haswell, W.A. 1879. On some additional new genera and species of amphipods crustaceas. Proc. Linn. Soc. New South Wales, 4(3): 319-351. [ Links ]
27. Herrmann, R. 1971. Zur regionalhydrologischen Analyse und Gliederung der nordwestlichen Sierra Nevada (Kolumbien). Giessener geogr. Schr., 23: 1-88. [ Links ]
28. Kensley, B. and M. Schotte. 1994. Marine isopods from the Lesser Antilles and Colombia (Crustacea, Peracarida). Proc. Biol. Soc. Wash., 107: 482-510. [ Links ]
29. Krapp-Schickel, T. 1993. Suborder Caprellidea. Mémoires de l'Institute Oceanographique, Monaco, 13 (3): 773-809. [ Links ]
30. Krapp-Schickel, T. and J.M. Guerra-García. 2005. Littoral Caprellidae (Crustacea: Amphipoda) from Indonesia, with the description of a new species. Boll. Mus.Civ. St. Nat. Verona, 29: 47-62. [ Links ]
31. Kunkel, B.W. 1910. The Amphipoda of Bermuda. Transactions of the Connecticut Academy of Arts and Science, 16: 106-113. [ Links ]
32. Laubitz, D.R. 1970. Studies on the Caprellidae of the American North Pacific. Publications in Biological Oceanography, 1: 1-89. [ Links ]
33. Laubitz, D.R. 1972. The Caprellidae (Crustacea, Amphipoda) of Atlantic and Arctic Canada. National Museums of Canada, Publications in Biological Oceanography, 4: 1-82. [ Links ]
34. Laubitz, D.R. 1993. Caprellidea (Crustacea: Amphipoda): towards a new synthesis. J. Nat. Hist., 27: 965-976. [ Links ]
35. Laubitz, D.R. 1995. Caprellidea (Crustacea: Amphipoda) from the Southern and Western Indian Ocean. Mésogée, 54: 81-100. [ Links ]
36. Leach, W.E. 1814. Article Crustaceology: 429-437. In: The Edinburgh Encyclopaedia, Blackwood, Edinburgh. Vol. 7 [ Links ]
37. Mayer, P. 1882. Caprelliden. Fauna und Flora des Golfes von Neapel, 6: 201 p. [ Links ]
38. Mayer, P. 1890. Die Caprelliden des Golfes von Neapel und der angrenzenden Meeres-Abschnitte. Fauna und Flora des Golfes von Neapel, 17: 55 p. [ Links ]
39. Mayer, P. 1903. Die Caprelliden der Siboga-Expedition. Siboga Expeditie, 34: 1-160. [ Links ]
40. McCain, J.C. 1967. Paracaprella barnardi, a new species of caprellid (Crustacea; Amphipoda) from the west coast of Panamá. Proc. Biol. Soc. Wash., 80: 219-222. [ Links ]
41. McCain, J.C. 1968. The Caprellidea (Crustacea: Amphipoda) of the Western North Atlantic. US Nat. Mus. Bull., 278: 1-116. [ Links ]
42. McCain, J.C. and J.E. Steinberg. 1970. Amphipoda-I, Caprellidea-I. In: Crustaceorum Catalogus. Ed. Gruner, H. H. and L.B. Holthuis 2. The Hague, The Netherlands, 78 p. [ Links ]
43. Müller, H.G. 1990. New species and records of coral reef inhabiting Caprellidea from Bora Bora and Moorea, Society Islands (Crustacea: Amphipoda). Rev. Suisse Zool., 97(4): 827-842. [ Links ]
44. Müller, H.G. 1992. Anthuridae of the genera Amakusanthura, Cortezura and Mesanthura from the Caribbean Sea of Colombia (Crustacea, Isopoda). Rev. Suisse Zool., 99: 31-79. [ Links ]
45. Müller, H.G. 1993a. On the occurrence of the isopod Heteroserolis mgrayi (Menzies and Frankenberg, 1966) (Sphaeromatidea) in the Santa-Marta area, Caribbean Sea of Colombia, with notes on its variation. Zool. Anz., 230: 35-44. [ Links ]
46. Müller, H.G. 1993b. Cirolanidae of the genera Calyptolana, Cirolana, Neocircolana, Colopisthus and Excircolana from the Santa-Marta area, Caribbean Sea of Colombia (Crustacea, Isopoda, Cymothoida). Zool. Anz., 230: 191-225. [ Links ]
47. Müller, K. 1979. Interrelaciones entre salinidad y temperatura en la Bahía de Santa Marta. An. Inst. Inv. Mar. Punta de Betín, 11: 219-226. [ Links ]
48. Myers, A.A., and J.K. Lowry. 2003. A phylogeny and a new classification of the Corophiidea Leach, 1814 (Amphipoda). J. Crust. Biol., 23(2): 443-485. [ Links ]
49. Ortíz, M. and R. Lalana. 1998. Lista actualizada de los crustáceos no decápodos de Cuba. Rev. Inv. Mar., 19: 92-99. [ Links ]
50. Ortíz, M., F. Álvarez and I. Winfield. 2002. Caprellid amphipods (Amphipoda: Caprellidae) illustrated key for the genera and species from the Gulf of Mexico and the Caribbean Sea. Universidad Nacional Autónoma de México. Facultad de Estudios Superiores Iztacala. Tlalnepantla, México, 83 p. [ Links ]
51. Quitete, J.M.P.A. 1972. Hemiaegina costai, nova espécie de Caprellidae da costa brasileira (Crustacea: Amphipoda). Atas Soc. Biol. Rio de Janeiro, 15(3): 165-168. [ Links ]
52. Reid, D.M. 1951. Report on the Amphipoda (Gammaridea and Caprellidea) of the coast of tropical West Africa. Atlantide Rep., 2: 189-291 [ Links ]
53. Riera, R., J.M. Guerra-García, M.C. Brito and J. Núñez. 2003. Estudio de los caprélidos de Lanzarote, islas Canarias (Crustacea: Amphipoda: Caprellidea). Vierae, 31: 157-166. [ Links ]
54. Salzwedel, H. and K. Müller. 1983. A summary of meteorological and hydrological data from the Bay of Santa Marta, Colombian Caribbean. An. Inst. Inv. Mar. Punta de Betín, 13: 67-83. [ Links ]
55. Serejo, C.S. 1997. Hemiaegina costai Quitete, 1972 a synonym of Hemiaegina minuta Mayer, 1890 (Amphipoda, Caprellidae). Crustaceana, 70(5): 630-632. [ Links ]
56. Stebbing, T.R.T. 1895. Two new amphipods from the West Indies. Ann. Mag. Nat. Hist., 15: 397-403. [ Links ]
57. Steinberg, J.E. and E.C. Dougherty. 1957. The skeleton shrimps (Crustacea: Caprellidae) of the Gulf of Mexico. Tulane Studies in Zoology, 5: 267-288. [ Links ]
58. Sundara Raj, B. 1927. Suborder Caprellidea (Laemodipoda). The littoral fauna of Krusada Island in the Gulf of Manaar. Bull. Madras Gov. Mus. Nat. Hist., S. 1: 125-128. [ Links ]
59. Takeuchi, I. 1993. Is the Caprellidea a monophyletic group? J. Nat. Hist., 27: 947-964. [ Links ]
60. Takeuchi, I. and R. Hirano. 1991. Growth and reproduction of Caprella danilevskii (Crustacea: Amphipoda) reared in the laboratory. Mar. Biol., 110: 391-397. [ Links ]
61. Takeuchi, I. and R. Hirano. 1992. Duration and size of embryos in epifaunal amphipods Caprella danilevskii Czerniavski and C. okadai Arimoto (Crustacea: Amphipoda: Caprellidea). J. Exp. Mar. Biol. Ecol., 164: 161-169. [ Links ]
62. Takeuchi, I. and R. Hirano. 1995. Clinging behaviour of the epifaunal caprellids (Amphipoda) inhabiting the Sargassum zone on the Pacific Coast of Japan, with its evolutionary implications. J. Crust. Biol., 15 (3): 481-492. [ Links ]
63. Thiel, M., J.M. Guerra-García, D.A. Lancellotti, and N. Vásquez. 2003. The distribution of littoral caprellids (Crustacea: Amphipoda: Caprellidea) along the Pacific coast of continental Chile. Rev. Chil. Hist. Nat. 76: 297-312. [ Links ]
64. Wakabara, Y. and C.S. Serejo. 1998. Malacostraca - Peracarida. Amphipoda. Gammaridea and Caprellidae. pp: 561-594 in: Young, P.S. (ed) Catalogue of Crustacea of Brazil. Rio de Janeiro: Museu Nacional (Serie Livros 6). 717 p. [ Links ]
65. Wakabara, Y., A.S. Tararam and M.T. Valerio-Berardo, W. Duleba and F.P. Pereira. 1991. Gammaridean and caprellidean fauna from Brazil. Hydrobiologia, 223: 69-77. [ Links ]
66. Winfield, I., E. Escobar-Briones and J.J. Morrone. 2006. Updated checklist and identification of areas of endemism of benthic amphipods (Caprellidea and Gammaridea) from offshore habitats in the SW Gulf of Mexico. Sci. Mar., 70: 99-108. [ Links ]
DATE RECEIVED: 19/04/06 DATE ACCEPTED: 24/10/06