Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Boletín de Investigaciones Marinas y Costeras - INVEMAR
Print version ISSN 0122-9761
Bol. Invest. Mar. Cost. vol.42 no.2 Santa Marta July/Dec. 2013
MICROANATOMY OF THE DIGESTIVE SYSTEM OF ENTEROCTOPUS MEGALOCYATHUS (CEPHALOPODA, OCTOPODA) OF THE SOUTHWEST ATLANTIC
MICROANATOMÍA DEL SISTEMA DIGESTIVO DE ENTEROCTOPUS MEGALOCYATHUS (CEPHALOPODA, OCTOPODA) EN EL ATLÁNTICO SUROCCIDENTAL
Rosana Griselda Garri1 and Lilia Lauria de Cidre2
1 Universidad Nacional de la Patagonia "San Juan Bosco", Facultad de Ciencias Naturales, Trelew, Argentina. rossanna_g@hotmail.com.
2 Universidad de Buenos Aires (UBA), Facultad de Ciencias Exactas y Naturales, Departamento de Biodiversidad y Biología Experimental, Laboratorio de Histología Animal. Pabellón II, Piso 4, Lab. 26, Ciudad Universitaria (C1428EHA), Ciudad Autónoma de Buenos Aires, Argentina. lilia_lauria@yahoo.com.ar.
ABSTRACT
A detailed description of the microanatomy of the digestive system of Enteroctopus megalocyathus is given, and this revealed some differences between this octopod species and other species of Octopodidae previously described. These differences are mainly related to tissues lining the lumen of digestive organs. Unlike descriptions in some octopods mucosa, in E. megalocyathus a pseudostratified epithelium is found covering the lumen of oesophagus, crop, stomach, intestine and caecum. Another major finding is that in the posterior salivary glands, two cell types are present in the same tubular adenomere; they do not constitute independent glandular tubules with only one type of glandular cells. The tubules of these glands are lined by two distinct epithelial cells, distributed from the distal to the proximal zones: type A, in the proximal zone of the glandular tubule, with cylindrical cells and globular mucous cells; and type B in the distal zone, cylindrical, with a weakly eosinophilic cytoplasm. The secretion of the cells was detected applying PAS and alcian blue at different pH. In the octopuses maintained without food in the aquarium for two days, histological structure of the epithelium of the caecum, intestine and digestive gland showed histological changes specially related with the mucosa. Taken together, the results allow to set the histological structure of the digestive tract of E. megalocyathus in their natural habitat and to provide preliminary evidence that this system responds rapidly (~2 days) to food deprivation, and give baseline data for future studies investigating the impact of environmental changes on the gut.
KEY WORDS: Histology, digestive organs, cephalopods, Southwest Atlantic, Enteroctopus megalocyathus.
RESUMEN
Se presenta la descripción de la anatomía microscópica del sistema digestivo de Enteroctopus megalocyathus, observándose algunas diferencias entre este octópodo, y otras especies de Octopodidae descritas previamente. Estas diferencias están principalmente relacionadas con los tejidos que revisten el lumen de los órganos digestivos. Contrariamente a las descripciones de la mucosa de algunos octópodos, en E. megalocyathus el lumen del esófago, buche, estómago, intestino y ciego está revestido por un epitelio pseudoestratificado. Otro hallazgo notable es que en las glándulas salivales posteriores están presentes dos tipos celulares en el mismo adenómero tubular: las del tipo A, en la zona proximal del túbulo glandular, con células cilíndricas y células mucosas globulares; y las de tipo B en la zona distal, cilíndricas con citoplasma débilmente eosinofílico. La secreción de estas células se detectó con las técnicas de PAS y alcian blue a distintos pH. En los pulpos mantenidos sin alimento en el acuario durante dos días, la estructura histológica del epitelio del ciego, intestino y de la glándula digestiva mostró cambios histológicos con respecto a los de la mucosa de los pulpos procesados inmediatamente luego de su captura. En conjunto, estos resultados permitieron caracterizar histológicamente los órganos del sistema digestivo de E. megalocyathus en su hábitat natural y establecer observaciones preliminares respecto de la respuesta rápida (~2 días) a la privación de alimento, y brindó datos de base para futuros estudios que investiguen el impacto de cambios ambientales en el tracto digestivo.
PALABRAS CLAVE: Histología, órganos digestivos, cefalópodos, Atlántico suroccidental, Enteroctopus megalocyathus.
INTRODUCTION
Enteroctopus megalocyathus (Gould) is a coastal benthic Octopodidae, which distributes along the Southwest Atlantic Ocean from the San Matías Gulf (41°30'S, 64°40'W) to the Beagle Channel (54°53'S, 67°50'W), the Falkland (Malvinas) Islands (51°50'S, 59°40'W) and Burdwood Bank (56°10'S, 54°20'W) (Ré, 1998a,1998b), and the range extends from Chiloé Island (42°S) in the Pacific Ocean (Rocha, 1997). This species is of great commercial importance in the regional economies of the Northern Patagonian coast, Argentina (Ré, 1998a, 1998b). Ré (1998b) reported that E. megalocyathus can be found in hard-bottom habitats from the lower intertidal zone to 140 m depth.
There are only a few histological studies of the digestive system in species of the family Octopidade (Suborder Incirrata), for example Eledone cirrhosaa (Boucher-Rodoni, 1976) and Octopus vulgaris (Bidder 1957, 1976; Boucaud-Camou et al., 1976; Boucher-Rodoni and Mangold, 1977; Andrews and Tansey, 1983). These previous studies analyzed in detail the microanatomy of the digestive organs and the cellular changes during digestion processes in the octopuses.
However we have no knowledge of the histology of the digestive tract of the Octopodidae from the coastal waters of the Argentina Patagonian. Particularly, there are no histological studies on the digestive system in the genus Enteroctopus, although the study by Garri and Ré (2002) described in detail the gross morphology of these organs. The aim of the present study is to determine the histological structure of the digestive organs in E. megalocyathus in their natural habitat and to analyze possible alterations of the digestive tract of animals maintained for two days in sea water without food.
MATERIALS AND METHODS
Sampling conditions
Six octopuses of the species E. megalocyathus were collected alive with artificial shelters at 10, 15 and 20 m deep in Punta Cuevas (42º46'S, 65º02'W) and by scuba-diving in Punta Flecha (42º38'S, 64º58'W) in waters of Nuevo Gulf (North Patagonian Coast, Argentina), in February and July 1998 respectively. In the Commercial Fish and Shellfish Laboratory of the National Patagonian Center (Lapemar-Cenpat), sex determination was performed and the total weight (TW, g) and dorsal mantle length [DML in mm, according to Roper and Voss (1983)] were recorded. The information is summarized in Table 1.
The octopuses named A, B and C, considered wild or freshly caught specimens, were immediately anaesthetized with 2% ethanol/seawater and decapitated. Those from the group D, E and F were maintained in the laboratory in 200 L aquarium containing filtered recirculating seawater. They were at room temperature without food, during two days. Animals from each group, were decapitated under anesthesia and the organs of the digestive system (oesophagus, crop, stomach, caecum, proximal, medial and distal intestine, digestive gland, appendages of the digestive gland ducts and submandibular, anterior and posterior salivary glands), were removed and rinsed with seawater. The oesophagus and crop of wild octopuses had mainly a moderately solid content of food, where fragments of arthropods exoskeleton were seen, while those from animals maintained without food were empty or had only a scarce liquid material. Current guidelines for laboratory animal care were followed.
Histology
The digestive organs of each animal from both groups were processed for histology. They were fixed by immersion in 4% (v/v) formaldehyde in seawater. Each organ was embedded in paraffin and five slides with fifteen serial sections (5-7 µm) were made. They were deparaffinised, rehydrated and stained with haematoxylin-eosin to evaluate general histological architecture. To detect neutral and sulphated glycosaminoglycans, PAS and Mowry techniques were employed respectively (Martoja and Martoja-Pierson, 1970). Samples were coded and examined blindly by two independent observers, under a light microscope. Histological examination of the samples and micrographs were done with a Zeiss Photomikroskop Primo Star and Digital Camera Canon Powershot A640 in the Laboratory of Animal Histology of the Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires. Measurements of different structures were done by means of an ocular scale calibrated for X60 and X100, in five slices of every slide from the same organ of the three animals.
RESULTS
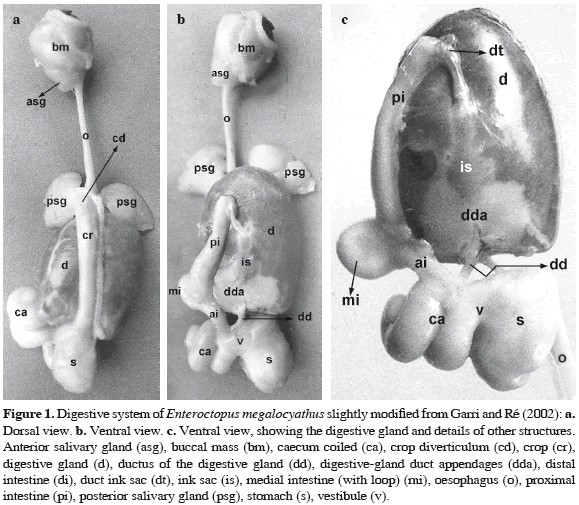
The digestive system of E. megalocyathus presents the typical morphology in U shape of the Cephalopoda Coleoidea. The dorsal descendant part of the digestive tract follows the buccal mass, and includes the oesophagus (Figures 1a, b) that widens forming a crop with a prominent anterior diverticulum. The crop has a diameter approximately four times greater than the oesophagus (Figure 1a). After the crop, the stomach bipartite and the caecum with two coils are linked by the vestibule. The stomach and caecum spiral are approximately equal in size. The intestine originates here, first as the ascendant branch which is comprised by a proximal, medial (with a loop) and distal part, ending in the anus covered by two anal flaps. The ink sac, large, is completely embedded on the ventral face of digestive gland. The ink sac duct leads ventrally in the rectum (Figures 1b, c). The most conspicuous organ of the digestive tract is the digestive gland (Figure 1), with the appendages of the digestive gland enclosed at its distal end. In previous studies, the digestive gland and the appendages of the digestive glands were referred to as "liver" and "pancreas" respectively (Bidder, 1976) (Figures 1b, c). The digestive gland, occupies most of the visceral mass, and has two separated ducts which enter the caecum (Figure 1c). There are three salivary glands annexed to the digestive system: 1) The anterior salivary glands are moderately large, paired and located externally in the dorsal part of the posterior wall of the mouth bulb. Their ducts end at the lateral lobes level. 2) The large posterior salivary glands are triangular in shape, paired and located externally at the anterior and dorsal region of the crop (Figures 1a, b). The ducts of the anterior and posterior salivary glands open into the buccal mass. 3) The submandibular gland is single and included at its distal end of the buccal mass.
Histological observations
Animals studied directly from the sea
The three octopuses studied directly from the sea (A, B and C), considered wild or freshly caught specimens, are first described.
Oesophagus
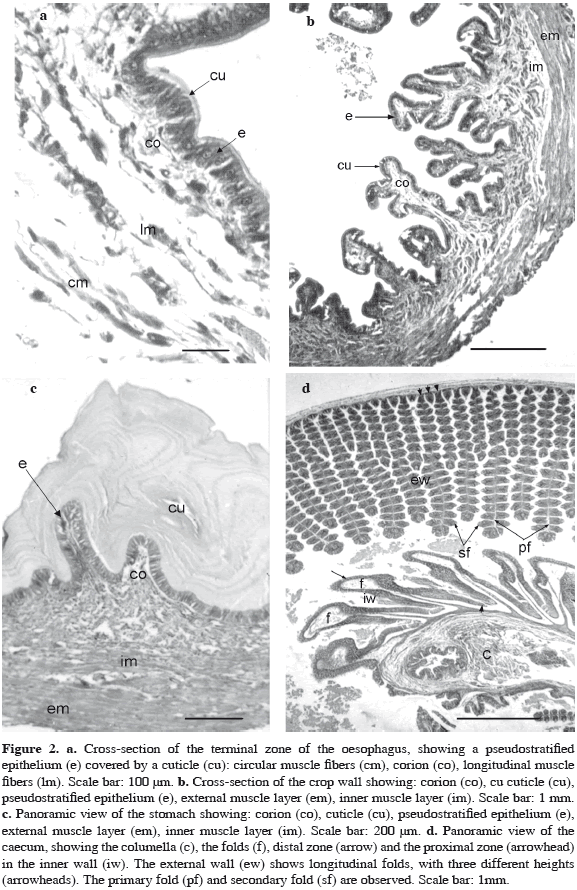
Is the first organ following the buccal mass. The wall forms longitudinal folds. In the anterior part, the wall is covered by a simple epithelium whose cells vary from cuboidal to cylindrical. Between them scarce PAS (+) goblet cells are seen.
Towards the terminal zone, near to the connection with the crop, the epithelium turns into pseudostratified. The oesophageal lumen is covered by a cuticle with a thickness between 12.5 and 26 µm. The epithelium lies on a loose stratum of connective tissue. Under it, fascicles of longitudinal muscular fibers are observed, as well as, circular muscular fibers which run circularly around the organ. All these fascicles are loosely distributed (Figure 2a).
Crop
The oesophagus widens in a crop, with its wall folding longitudinally allowing the enlargement of two orders which allows the enlargement in volume of this organ. The mucosa is constituted by the pseudostratified epithelium and numerous PAS (+) goblet cells are easily seen. The epithelium secretes a cuticle with a thickness between 10 and 16 µm, and lies on a loose connective tissue (corion) containing abundant connective cells and blood vessels. The tunica muscularis consists of two layers of muscle fibers: the inner one with longitudinal, and the external with circular muscle fibers; disposition of nerve fibers are usually observed between the longitudinal and circular muscular layers (Figure 2b).
Stomach
The wall is less folded than the oesophagus and crop and is lined by a pseudostratified epithelium consisting of tall cells with a nucleus with sparse chromatin and a conspicuous nucleolus. In the apical cytoplasm a PAS (+) reaction is seen. No goblet cells are present. The stomach lumen is covered by a wide cuticle PAS (+) and with a thickness between 165 and 205 µm. It presents numerous well defined and visible strata. The epithelium is located on a loose connective tissue with numerous blood vessels and slender fascicles of longitudinal muscle fibers or even isolated ones. These isolated fibers are not, in any case, like the muscular mucous layer of some vertebrates digestive system organs. The muscle coat is well developed and consists of two layers of muscles fibers: an internal longitudinal layer and an external oblique and circular one (Figure 2c).
Caecum
The caecum is a tubular organ spiralled around an axis or columella. The wall near the axis develops ample folds (Figure 2d). At the distal zone each fold is covered by a ciliated pseudostratified epithelium with PAS (+) goblet cells. Other mucous cells reacted positively with alcian blue. At the proximal zone, near to the columella, the mucosa is composed by a simple cylindrical epithelium without cilia and each cell presents an apical membrane protruding towards the lumen (Figure 2d). The nucleus, with a well developed nucleolus, is located near the medial zone of the cell. The cytoplasm is basophilic and shows a vacuolated aspect.
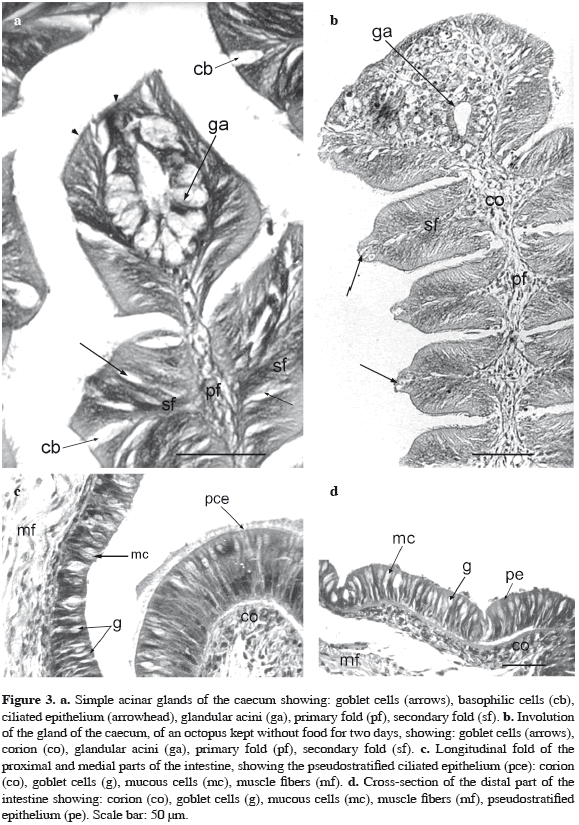
The external wall of the caecum shows longitudinal folds, with three different heights ranging from 500 to 2000 µm. They protrude into the lumen, enlarging the surface area of the organ. These primary folds give origin to secondary ones about 100 µm of height (Figure 2d). They are all lined by a simple, cylindrical and ciliated epithelium with PAS (+) vesicles in the cytoplasm and with a large number of goblet cells scattered between them. In all the secondary folds in a subterminal position (Figure 3a) basophilic cylindrical cells are present. These cells possess a vacuolated cytoplasm, lack cilia and present protrusions of the apical membrane. These characteristics are similar to those observed in the epithelium lining the columella. All the folds at the zone of the columella and in the external wall of the caecum have a central axis of loose connective tissue (or corion) with high vascularization. In the apical zone of every primary fold, there are simple acinar glands, also called caecal gland, that open directly into the lumen of the organ (Figure 3a). These glands are formed by secreting cells with a basal flattened nucleus and a light vacuolated cytoplasm. The vesicles are PAS (+) and Alcian Blue pH = 3.5 (-), showing a secretion of neutral carbohydrates. The mucosa is underlined by scarce connective tissue and thin fascicles of longitudinal muscles fibers.
Intestine
It is divided in 1-proximal, 2-medial and 3-distal zones. The proximal and medial zones of the intestine show longitudinal folds protruding into the lumen. The epithelium is tall, pseudostratified and ciliated, with three rows of nuclei; there are two types of mucous cells intercalated among them. One of them are the PAS (+) goblet cells and the other are mucous cells that react positively with alcian blue pH = 3.5. When PAS and alcian blue techniques were applied together in one stain, results demonstrate that the secretion of these cells consists mainly of neutral and acid proteoglycans. The terminal zone of the folds has cells with well developed cilia which decrease in height and disappear in the epithelium between the folds. The epithelium lies on a loose connective tissue highly vascularized and with isolated muscle fibers (Figure 3c). At the distal zone of the intestine, the folds reduce their height. The epithelium continues being pseudostratified but its thickness is reduced compared to the proximal zone and lacks cilia. The epithelium is characterized by the presence of two rows of nuclei. There are scarce goblet and mucous cells and they react positively with PAS (+) and alcian blue pH=3.5 (+) respectively. The muscular wall of the intestine is thin and comprises an external circular layer and an internal longitudinal one (Figure 3d).
Digestive gland ("liver") and appendages of the digestive gland ducts ("pancreas")
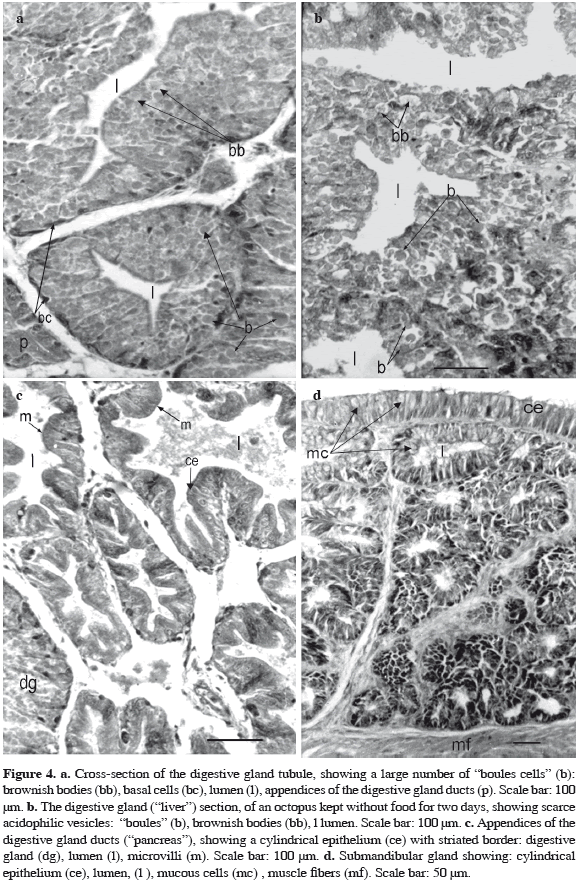
The digestive gland ("liver") and the appendages of the digestive gland ducts ("pancreas") are branched tubular organs enclosed in a capsule of loose connective tissue with scarce smooth muscular fibers. In the digestive gland (Figure 4a) the lining epithelium comprises three main cellular types: 1) Thin cells, extending from the basal lamina to the lumen, with an ovoid nucleus and condensed chromatin. 2) Basal cells, in differentiation, that do not reach the lumen of the organ and commonly present a basophilic cytoplasm and a basal nucleus with a developed nucleolus. Occasionally, these cells show weakly acidophilic vesicles. 3) Digestive cells, "boules cells" which are the most abundant. They extend from the basal lamina to the lumen of the organ. In these cells, the nucleus is spherical in the middle or basal part of the cell, with a well developed nucleolus. Most of these cells, but not all of them, show apical microvilli forming a discontinuous brush border around the lumen of the tubule. The cytoplasm contains three kinds of vesicles, which vary in number and size: a) abundant acidophilic and PAS (+) vesicles ("boules"), 8 µm of medium size diameter; b) weakly acidophilic vesicles 7 µm diameter, containing a granular substance, and c) less abundant vesicles, of great size up to 15 µm diameter, with a light brownish material (brownish bodies), located at the basal zone or frequently at the apical cytoplasm. The size of the vesicles is summarized in Table 2.
The appendages of the digestive gland ducts (Figure 4c) have a lumen lined by cylindrical epithelial cells of variable height; the nucleus is located at the basal half of the cell and present vacuolated chromatin and a developed nucleolus. Short microvilli are observed in the apical domain of the cell forming a striated border. In the cytoplasm few PAS (+) granules are observed, and a PAS reaction is shown in the apical cell membrane at the microvilli level. The epithelium lies on a PAS (+) basal lamina that is underlain by loose connective tissue and isolated muscular fibers.
Salivary glands
Submandibular gland. It is a simple branched tubular gland consisting of lobes invested by loose connective tissue which emits thin trabeculae surrounding each secretory unit (Figure 4d). The covering epithelium is simple and cylindrical with abundant mucous cells, which react positively with PAS and weakly with alcian blue pH = 3.5. Both stains show that the secretion of mucous cells consists mainly of neutral and acidic proteoglycans. These cells decrease in number towards the terminal zone of tubules. At this zone, cylindrical cells are lower and show subnuclear basophilic cytoplasm. Externally, the gland is surrounded by an inner circular muscular layer and an outer longitudinal one. These muscular layers continue surrounding the mouth cavity towards the ventrolateral region.
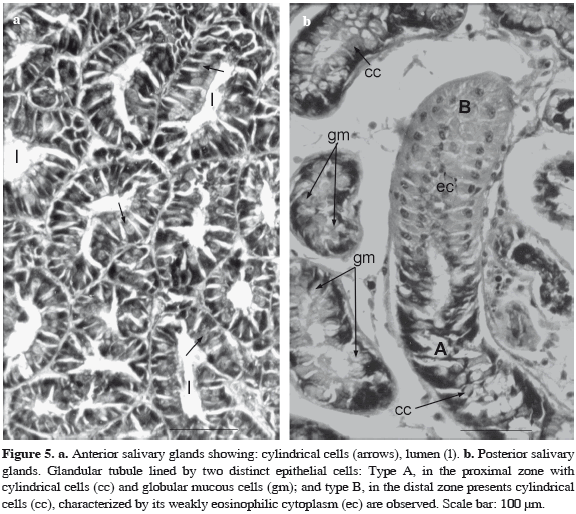
Anterior salivary glands. They are simple branched tubular glands surrounded by a thin capsule of lax connective tissue, with scarce muscular fibers. Each glandular tubule is constituted by cylindrical cells with a spherical nucleus and basophilic cytoplasm. At the apical zone, light vesicles are observed with haematoxylin-eosin (Figure 5a). The alcian blue (+) pH = 3.5 and PAS (+) techniques evidenced vesicles containing neutral and acid glycosaminoglycans.
Posterior salivary glands. As the gland described above, posterior salivary glands are simple branched tubular glands. They are covered by a connective capsule with muscular fibers surrounding the glandular tubules like thin connections. Each tubule presents two zones formed by different cells. The first or proximal zone (A) consists of globular mucous cells with flattened basal nuclei (Figure 5b). The supranuclear cytoplasm contains high level of PAS (+) and alcian blue (+) pH = 3.5, vesicles denoting the presence of weakly sulphated glycosaminoglycans content. Cylindrical cells with a basal spherical nucleus surrounded by a basophilic cytoplasm are also seen. The second or distal zone (B) present cylindrical cells characterized by its weakly eosinophilic cytoplasm. The nucleus with basal or apical location is observed (Figure 5b). The PAS (+) and Alcian Blue (-) pH = 3.5 reaction in the cytoplasm, suggests the presence of neutral carbohydrates.
Animals kept in the aquarium without food for two days
Octopuses belonging to the group D, E and F were kept in the aquarium without food for two days. We observed that the oesophagus, crop, stomach and appendages of the digestive gland ducts of these animals presented the same description as those named A, B and C or freshly caught specimens. Some differences with respect to freshly caught specimens were registered in the following organs:
Caecum
The epithelium of the columella does not show differences when compared to the one observed in freshly caught specimens. Nevertheless at the level of the primary folds of the external wall, an involution of the glandular acini or caecal gland of the terminal zone is observed (Figure 3b). These cells are reduced in height. The nucleus turns spherical with sparse chromatin and it occupies a central position in the cell. No vesicles with secretion are observed. The loose connective tissue that forms the axis of the primary fold is well developed and well vascularized at the terminal zone. A conspicuous reduction in goblet cells number is observed (Figure 3b).
Intestine
In octopuses kept without food during two days in the laboratory, the animals presented in the intestine epithelium numerous goblet cells and scarce mucous cells.
Digestive gland ("liver")
In the digestive gland we saw the same three types of cells observed in the freshly caught specimens. The differences appeared in the "boules cells" at the cell granules level. In animals maintained without food (Figure 4b), the tubular lumen is wide and digestive cells show a less number of acidophilic vesicles but bigger (13 µm in diameter) than in freshly caught specimens. They are located at the apical zone of the cell. The weakly acidophilic vesicles increase in size up to 11 µm diameter. The light "brownish bodies", decrease in size (11 µm in diameter) and number, and are generally located towards the subapical zone of the cell. These observations were similar in the three animals deprived of food for two days (Table 2).
Salivary glands
The same morphology of the salivary glands in both groups seems to tell that all the salivary glands remain morphologically prepared for secretion, in spite of deprivation of food during two days.
DISCUSSION
The structure of the digestive organs in E. megalocyathus, an octopus common in Argentina coasts, was studied here with the aim to define its histological constitution. The alimentary canal shows general features similar to those described in other Octopoda Incirrata of the family Octopodidae. Along its entire length, its mucosa presents numerous longitudinal folds with intervening grooves that cause the lumen to appear to be obstructed. However, when the oesophagus is distended the folds disappear and the lumen becomes patent.
Several authors, Boucher-Rodoni (1976) and Boucher-Rodoni and Mangold (1977) between others, observed that in some Octopodidae the epithelium of the oesophagus, crop, stomach, caecum and intestine was simple and cylindrical. However, as a distinctive feature, in E. megalocyathus a pseudostratified epithelium exists from the terminal part of the oesophagus up to the middle part of the intestine.
We observed in the lumen of the oesophagus and crop of wild octopuses material containing little arthropods exoskeleton fragments. As it was referred by Ré (1980, 1998a), these octopuses feed mainly on crabs, such as Ovalipes trimaculatus, Peltarion spinosulum and Leurocyclus tuberculosus. Coincidently, in the oesophagus, crop and stomach, the mucosa presented a prominent cuticle. It is thinner in the oesophagus and crop but enough to protect the mucosa of these organs from the food ingested as the crustacean carapace. In the stomach, cuticle is very thick (about 200 µm) and the striation parallel to the epithelial surface suggests that the cuticle secretion is discontinuous. The presence of cuticle in the three organs, its special thickness in the stomach, and the fact that the mucosa houses no digestive glands, constitute three morphological points that allow us to affirm, in agreement with Boucher-Rodoni and Mangold (1977), that the digestion begins in the stomach, and that the absorption of food seems not to be carried there.
Hematoxylin and eosin stain and histochemical techniques show that cuticle of the oesophagus, crop and stomach is constituted by proteoglycans secreted by the epithelial mucosa. A PAS (+) reaction detected in the goblet cells of oesophagus and crop speaks about neutral proteoglycans presence in the cuticle, where this reaction was also observed. The muscular wall of all the digestive organs is thin, and especially in the oesophagus and crop which have numerous longitudinal folds along their entire length. When the volume of the ingestion is big, the lumen is distended, the folds disappear and the lumen becomes patent without the muscle coat interfering. The exception is the stomach, where the muscle is well developed in relation to the mechanical crush of food ingested.
Capurro (1961) mentioned the presence of stereocilias between the epithelium and the cuticle in the oesophagus and the stomach of O. vulgaris. Besides, Boucher-Rodoni (1976) observed in E. cirrhosa a thin striation between the cuticle and the epithelium in both organs, which she named "formative fibrillar layer". In contrast, in E. megalocyathus neither the presence of stereocilias nor a formative fibrillar layer were observed in the oesophagus and the stomach epithelium beneath the cuticle. Only thin protoplasmic projections were observed at the apical zone of the cells, penetrating the cuticle in some sections where there was a cell retraction during the histological process. We think that Capurro (1961) and Boucher-Rodoni (1976) saw the same structure than us. The name stereocilias is not correct, due that stereocilias are very long microvilli found in the apical cell domain, leading to a free space, and "formative fibrillar layer" is a name not necessary for the relation between the apical folding of the cell membrane and its own secretion. Furthermore, this is a biological model highly repeated in different organs and zoological groups without an own name. We believe that this zone will be better defined with the aid of a transmission electron microscope.
The histological characteristics described for the spiralled caecum and the intestine of E. megalocyathus are coincident with those described for other Octopoda Incirrata as E. cirrhosa (Boucher-Rodoni, 1976) and O. vulgaris (Bidder, 1966; Boucaud-Camou et al., 1976; Boucher-Rodoni and Mangold, 1977; Boucaud-Camou and Boucher-Rodoni, 1983; Boucher-Rodoni et al., 1987; Mangold and Bidder, 1989). The caecum is the receiver of the crushed food from the stomach and the very little particles and enzymes from the digestive gland. In this complex organ, interesting structures as primary and secondary folds, the coating ciliated epithelium and the mucous cells, both secreting neutral carbohydrates, appear together. This association speaks about the function of this organ in the sense of being a place for the sticking of food and enzymes from the digestive gland and the caecum. The observations and interpretations we have just given are shared with those of Boucher-Rodoni and Mangold (1977) and Boucaud-Camou and Boucher-Rodoni (1983).
The intestine is the recipient of digested food included in a mucous band to be conducted to the anus. It contributes to the process producing neutral and slightly acid mucus. Cilias, irregularly distributed, are relevant in this process of evacuation by making ways to conduct the intestine content to the anus. We did not observe any other secretion.
The digestive gland ("liver") is an interesting organ related to the digestion process supplying most of the digestive enzymes. We have identified three cellular types in the digestive gland in E. megalocyathus. However, in other Octopodidae such as O. vulgaris (Bidder, 1957; Boucher-Rodoni and Mangold, 1977) and E. cirrhosa (Boucher-Rodoni, 1976), a fourth cellular type was identified in the digestive gland: "the grey cells", whose function is associated with the elimination of debris. These cells were not observed in the present study. The first cell type we saw is a thin cell extending from the basal lamina to the lumen of the organ. We did not see any cytoplasm differentiation. These cells are similar to those that Boucher-Rodoni (1976) considered cells of replacement. The second type is the basal cell. It is also described by Boucher-Rodoni (1977) in O. vulgaris. The author considered that no connection with the digestion could be shown. Looking at the microscopic images of the basal cells, we can say that they do not reach the lumen of the organ, so it is not probable to have an absorptive role. Otherwise, considering that this is a pseudostratified epithelium, basal cells in this tissue may be the stem cells which could differentiate into the other cells of this organ. This is a hypothesis that must be confirmed with the appropriate methodology. The last type of cells we found in the digestive gland, the "boule cells", are characterized, like in other species of the Octopodidae, by the presence of acidophilic vesicles ("boules") containing enzymes for the extracellular digestion, and light brownish vesicles consisting of residual bodies which would be exocytosed (Boucher-Rodoni and Mangold, 1977). Other big vesicles, weakly acidophilic, could represent vesicles storing enzymes in a process of maturation or in a process of condensation of debris for a posterior transformation into light brownish vesicles (Bidder, 1957, 1966; Boucaud-Camou et al., 1976; Boucher-Rodoni and Mangold, 1977). From the present study and according to the observations of Bidder (1957), Boucher-Rodoni (1976) and Boucher-Rodoni and Mangold (1977), it can be assumed that the acidophilic and the light brownish vesicles develop in accordance to the digestive process. In animals maintained without food for two days, the lumen is bigger than that of freshly caught specimens, due to the diminishing of cell height. The digestive gland cell affected is the "boules cell". We have seen that the boules are around 5 µm bigger than in freshly caught specimens and their number macroscopically lesser. The diminished number may be due to the enzyme secretion during the last digestion. This was also observed by Boucher-Rodoni (1976) in E. cirrhosa and she concluded that the number of boules varies during the digestion process. The increased size, although not documented in this work, may be the result of no exocited vesicle fusion. Brownish bodies diminish in number and size. They are vesicles present in several cells where waist material is confined to be discarded to the lumen of the organ or to the extracellular matrix, depending of the cell. In the octopuses maintained without food, although reduced in number, they were not totally released. With respect to the diminished size, based in Wells and Wells (1989) opinion that brown bodies production may be for four days after the last meal, we think that two days without food may be considered a time of permanency of brown bodies produced in the last ingest. After identifying the main cellular types in this organ, we share with Boucher-Rodoni and Mangold (1977) that this is a place for nutrients absorption and storage of reserves.
The appendages of the digestive gland ducts ("pancreas") in E. megalocyathus share the general characteristics observed in O. vulgaris (Bidder, 1966; Boucher-Rodoni and Mangold, 1977), and E. cirrhosa (Boucher-Rodoni, 1976; Mangold and Bidder, 1989). As in other species, in E. megalocyathus brown bodies were not observed. Mangold and Young (1998) mentioned that in the "pancreas" of different species of cephalopods studied, their absence may serve to differentiate histologically the "pancreas" from the "liver". We think that in the species we studied this consideration is not necessary. Boucher-Rodoni (1976) indicated that cells of the "pancreas" of E. cirrhosa do not seem to vary during the digestion process in the octopus without food. In the species studied, there was no histological change in individuals deprived of food for two days.
In relation with the description of the salivary glands in this paper, we found characteristics coincident with those observed in the rest of the Octopoda Incirrata as mentioned by Boucher-Rodoni and Mangold (1977) and Mangold and Young (1998). One difference has been seen in the posterior salivary glands, where we identified two cellular types distributed in the same tubular gland but in different zones as observed in Figure 5b. We saw that cells secreting neutral proteoglycans are found in the distal part of the tubule, while those cells secreting neutral and acid proteoglycans are in the proximal one. This gland cells distribution along the glandular tubule is similar in glands from organs of other species (Lauria de Cidre et al., 2006). Apical cells secreting neutral proteoglycans in distal part of the tubule may migrate to the apical zone while secretions mature acquiring, for example, sulphated radicals in neutral proteoglycans just synthesized. The results showed above about the kind of sulphated glycosaminoglycans present in the caecum, intestine, submandibular gland and anterior and posterior salivary glands, are coincident with those described by Boucher-Rodoni (1976) and Boucher-Rodoni and Mangold (1977).
With regard to the two days food deprived octopuses in the laboratory, the organs that showed histological changes were: 1) the caecum, where there were scarce goblet cells in the secondary folds and there was a reduction of the glands in the apex of the primary folds. 2) The intestine, which shows an increase of goblet cells and a reduction of the mucous cells. The reduction of acini glands and mucous cells in the caecum and the intestine, respectively, demonstrates the active functional role of these organs, with respect to the feeding stimuli. 3) Histological changes were also observed in the digestive gland, where there was a large reduction in number of the acidophilic and the refringent brownish vesicles when compared to the one observed in freshly caught specimens. The reduction of the refringent brownish vesicles suggests that they are implicated in the storing of substances that will be then exocytosed. The histological characteristics observed in the food deprivation animals of E. megalocyathus, are similar to those cited in the experimental studies carried out in O. vulgaris (Bidder, 1957; Boucher-Rodoni and Mangold, 1977) and E. cirrhosa (Boucher-Rodoni, 1976).
Our results evidence that the histological structure of the digestive organs of E. megalocyathus, described for the first time in the genus Enteroctopus, shares the general histological characteristics mentioned in other Octopoda Incirrata, but with slight differences, as described above. On this baseline, we could detect high sensitiveness of some organs of the digestive system to changes of the environment.
ACKNOWLEDGEMENTS
The authors are very grateful to Dr. M. E. Ré (Commercial Fish and Shellfish Laboratory of the National Patagonian Center, Lapemar-Cenpat) for generous provision of samples in the Nuevo Gulf and maintenance of the octopuses in the laboratory, as well as, provide valuable knowledge on the cephalopods; and O. Weller for their valuable assistance in the collection of the octopuses for study. A special thank to N. Del Luján for their valuable technical assistance (Cenpat). We also acknowledge to Dr. H. Groba for their help with the microphotographs (Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires). This histological research was developed in the University of Buenos Aires (Facultad de Ciencias Exactas y Naturales), Argentina. We also thank two reviewers, Dr. P. Andrews and Dr. M. Vecchione, for their helpful comments.
LITERATURE CITED
1 Andrews, P. L. R. and E. Tansey. 1983. The digestive tract of Octopus vulgaris: the anatomy, physiology and pharmacology of the upper tract. J. Mar. Biol. Ass. UK, 63: 109-134. [ Links ]
2 Bidder, A. M. 1957. Evidence for an absorptive function in the "liver" of Octopus vulgaris. Lam. Pubbl. Staz. Zool., 29: 139-150. [ Links ]
3 Bidder, A. M. 1966. Feeding and digestion in cephalopods. 97-125. In: Wilbur, K. M. and C. M. Yonge (Eds.). Physiology of mollusca, Volume II. Academic Press, New York. 645 p. [ Links ]
4 Bidder, A. M. 1976. New names for old: the cephalopods "midgut gland". J. Zool., 180: 441-443. [ Links ]
5 Boucaud-Camou, E. and R. Boucher-Rodoni. 1983. Feeding and digestion in cephalopods. 149-187. In: Saleuddin, A. S. M. and K. M. Wilbur (Eds.). The mollusca, Volume V: Physiology, Part 2. Academic Press, New York. 500 p. [ Links ]
6 Boucaud-Camou, E., R. Boucher-Rodoni and K. Mangold. 1976. Digestive absorption in Octopus vulgaris (Cephalopoda: Octopoda). J. Zool., 179: 261-271. [ Links ]
7 Boucher-Rodoni, R. 1976. Ëtude histologique du tube digestive de deux Céphalopodes; Eledone cirrhosa (Octopoda) et Illex illecebrosus (Teuthoidea), au cours de la digestion. Cah. Biol. Mar., 17: 254-260. [ Links ]
8 Boucher-Rodoni, R. and K. Mangold. 1977. Experimental study of digestion in Octopus vulgaris (Cephalopoda: Octopoda). J. Zool., 183: 505-515. [ Links ]
9 Boucher-Rodoni, R., E. Boucaud-Camou and K. Mangold. 1987. Feeding and digestion. 85-108. In: Boyle, P. R. (Ed.). Cephalopod life cycles, Volume II: Comparative reviews. Academic Press, New York. 441 p. [ Links ]
10 Capurro, S. 1961. Ricerche istochimiche sui mucopolysaccharidi degli epiteli del canale alimentare di Octopus vulgaris. Ann. Musc. Civ. Stor. Nat., 72: 249-284. [ Links ]
11 Garri, R. and M. E. Ré. 2002. Morfología del aparato digestivo de Enteroctopus megalocyathus y Loligo sanpaulensis (Mollusca, Cephalopoda). Iheringia, Sér. Zool., 92 (2): 81-91. [ Links ]
12 Lauria de Cidre, L., F. A. Crespo and H. J. Aldana Marcos. 2006. Morphologic, histochemical and immunohistochemical characterization of tracheobronchial respiratory glands in South American Sea Lion Otaria Flavescens (Pinnipedia, Mammalia). J Histotechnol., 29 (1): 29-35. [ Links ]
13 Mangold, K. and A. M. Bidder. 1989. L'appareil digestif et la digestion. 321-373. In: Grassé, P. P. (Ed.). Volume V: Traité de zoologie, anatomie, systématique, biologie, Part 4. Masson, Paris. 804 p. [ Links ]
14 Mangold, K. and R. E. Young. 1998. The systematic value of the digestive organs. 21-30. In: Voss, N. A., M. Vecchione, R. B. Toll and M. J. Sweeney (Eds.). Systematic and biogeography of cephalopods, Volume I. Smithsonian, Washington. 586 p. [ Links ]
15 Martoja, R. and M. Martoja Pierson. 1970. Técnicas de histología animal. Toray-Masson, Barcelona. 350 p. [ Links ]
16 Ré, M. E. 1980. Estudio taxonómico de Enteroctopus megalocyathus (Gould, 1852) (Cephalopoda, Octopoda) con notas sobre su biología y pesca. Centr. Nac. Patag., 53: 1-34. [ Links ]
17 Ré, M. E. 1998a. Pulpos octopódidos (Cephalopoda: Octopodidae). 69-98. In: Boschi, E. E. (Ed.). El mar argentino y sus recursos pesqueros. Tomo 2: Los moluscos de interés pesquero. Cultivos y estrategias reproductivas de bivalvos y equinoideos. Instituto Nacional de Investigación y Desarrollo Pesquero, Mar del Plata. 35 p. [ Links ]
18 Ré, M. E. 1998b. Pesquerías de pulpos. 99-114. In: Boschi, E. E. (Ed.). El mar argentino y sus recursos pesqueros. Tomo 2: Los moluscos de interés pesquero. Cultivos y estrategias reproductivas de bivalvos y equinoideos. Instituto Nacional de Investigación y Desarrollo Pesquero, Mar del Plata. 35 p. [ Links ]
19 Rocha, F. 1997. Cephalopods in Chilean waters, a review. Malacol. Rev., 30: 101-113. [ Links ]
20 Roper, C. F. E. and G. L. Voss. 1983. Guidelines for taxonomic descriptions of cephalopod species. Mem. Natl. Mus., 44: 49-63. [ Links ]
21 Wells, M. J. and J. Wells. 1989. Water uptake in a cephalopod and the function of the so-called "pancreas". J. Exp. Biol., 145: 215-226. [ Links ]
FECHA DE RECEPCIÓN: 05/09/2011 FECHA DE ACEPTACIÓN: 25/04/2013