Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Boletín de Investigaciones Marinas y Costeras - INVEMAR
Print version ISSN 0122-9761
Bol. Invest. Mar. Cost. vol.42 no.2 Santa Marta July/Dec. 2013
NOTES ON THE ECOLOGY OF THE COLOMBIAN LEAFTOED GECKO (PHYLLODACTYLUS TRANSVERSALIS), ENDEMIC TO MALPELO ISLAND
NOTAS SOBRE LA ECOLOGÍA DEL GECO PHYLLODACTYLUS TRANSVERSALIS, ENDÉMICO DE LA ISLA MALPELO
Mateo López-Victoria1, Matthias Jurczyk2 and Volkmar Wolters2
1 Pontificia Universidad Javeriana Cali, Facultad de Ingeniería, Departamento de Ciencias Naturales y Matemáticas. Calle 18 No. 118-250, Cali, Colombia. malov@javerianacali.edu.co.
2 Justus-Liebig University, Heinrich-Buff-Ring, Department of Animal Ecology, 26-32, 35392, Giessen, Germany.
ABSTRACT
The Colombian leaf-toed gecko (Phyllodactylus transversalis) is the least known reptile species of Malpelo Island (Eastern Tropical Pacific). In the 37 years since its original description, no single study strictly dedicated to this species has been conducted. With the goal of providing information on the ecology and population size of this gecko, data collected during two visits to the island are presented. Behavior and reproduction were studied by following individuals during the night and by searching for hidden individuals and eggs during the day. Population's density and habitat preferences were estimated by classifying the island's surfaces into four types and counting geckos inside those habitats within band transects. General morphological measurements of geckos were performed as reference points for future comparisons. Phyllodactylus transversalis lays its eggs in narrow crevices of rocks, apparently communally. Reproduction seems to take place during most of the year, probably associated with the peak breeding season of seabirds. Although conspecifics shared shelters during the day, densities during the night suggest no apparent habitat preference for foraging. Population size of this species was estimated at approximately 114000 individuals.
KEY WORDS: Phyllodactylus transversalis, behavior, reproduction, habitat preference, population size.
RESUMEN
El geco de Malpelo (Phyllodactylus transversalis) es la especie de reptil menos conocida de esa isla (Pacífico Oriental Tropical). Después de 37 años de su descripción original, ningún estudio, estrictamente dedicado a esta especie ha sido realizado. Con el propósito de suministrar información sobre la ecología y reproducción de este geco, se presentan datos colectados durante dos visitas a Malpelo. El comportamiento y la reproducción fueron estudiados siguiendo individuos durante la noche, y buscando huevos e individuos escondidos durante el día. La densidad poblacional y la preferencia de hábitat fueron estimadas clasificando las superficies de la isla en cuatro hábitats y contando los gecos al interior de esos hábitats, usando transectos de banda. Medidas morfológicas generales de los gecos fueron tomadas, como punto de referencia para comparaciones futuras. Phyllodactylus transversalis pone sus huevos en grietas estrechas de las rocas, aparentemente en forma comunal. La reproducción parece tener lugar durante la mayor parte del año, probablemente sincronizada con la época reproductiva mayor de las aves marinas. Aunque los refugios fueron compartidos entre gecos durante el día, las densidades estimadas durante la noche sugieren que no hay una preferencia de hábitat aparente para el forrajeo. El tamaño de la población de esta especie es cercano a 114000 individuos.
PALABRAS CLAVE: Phyllodactylus transversalis, comportamiento, reproducción, preferencia de hábitat, tamaño de población.
INTRODUCTION
Due of its late discovery and nocturnal activity, Phyllodactylus transversalis Huey, 1975 is the less studied reptile species on the volcanic island of Malpelo, Colombia. The island is remote, difficult to reach and climb, and remained almost unexplored until late 20th century (Gorman and Chorba, 1975). Only two studies dealt directly with this species: the first study, besides describing the species, adds little about its ecology: that this gecko must be scansorial and that feeds on ants and crickets (Huey, 1975). The second one confirmed its feeding habits and provided a first estimation of its population size (López-Victoria, 2006). Other studies dealing with general ecology of the island and interactions among the terrestrial fauna pointed out the relationships between this gecko and other organisms (Wolda, 1975; López-Victoria et al., 2009, 2011).
After its original description as a new species, based on preserved material (Huey, 1975), only few pictures of this endemic gecko have appeared in books, papers and unpublished reports. Furthermore, there have been no studies specifically focused on this unique species. The present study is the first one strictly dedicated to this gecko. The main objective of this study was to provide first-hand information on its ecology, habitat preferences, population size, and morphology.
STUDY AREA
Malpelo is a volcanic island surrounded by 11 islets, located about 385 km off the continental coast of Colombia. Total surface (three-dimensional) of the islands is close to 1.2 km2, with a maximum height of approx. 300 m. The grounds of the island are uneven and rough, covered with rocks and small and isolated patches of soil. Margins of the main island and the islets are steep and permanently splashed by the sea (Stead, 1975; López-Victoria and Rozo, 2006).
Malpelo is inhabited by breeding sea birds, mostly Nazca-boobies (Sula granti), an endemic land crab (Johngarthia malpilensis), two more endemic reptiles, the dotted galliwasp (Diploglossus millepunctatus) and an anole (Anolis agassizi), and over 70 macroinvertebrate species (Wolda, 1975; Calero et al., 2011). Vegetation is scarce, mainly composed by lichens and microalgae, and scattered patches of grass and ferns (Wolda, 1975; López-Victoria and Rozo, 2006). Human presence is restricted to the eastern side of the island, where two Colombian Navy cabins (occupied by six soldiers) function as garrison since 1986. Sporadically, some tourists and scientists visit the island. Information of this study comes mainly from a 30-day and a 4-day visit to Malpelo conducted during May-June 2006 and October 2009, respectively.
MATERIALS AND METHODS
During nighttime observations were conducted wearing head lamps and doing random walks in search of geckos. Behavior was monitored by following 55 marked and unmarked individuals, trying to include all sizes (ages). During daytime, the search for geckos, their shelters and eggs consisted of randomly exploring dens and clefts and, whenever possible, lifting rocks and widening the shelters to look inside.
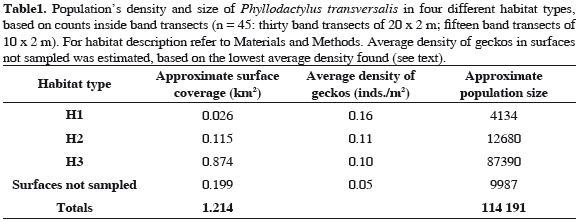
Population's density and habitat preference were estimated in three different types of substrates, defined according to their structural composition: H1: surfaces with moderate slopes covered by big and medium-sized rocks (ranging from 50 cm in diameter to >1 m); H2: surfaces with moderate slopes covered by small and mediumsized rocks (ranging from few to 50 cm in diameter), and H3: rocky bottoms with moderate to steep slopes covered by few and small stones (<10 cm in diameter). The rest of the island, consisting of even surfaces from steep cliffs and walls were inaccessible and not sampled. Average density of geckos in these unreachable surfaces was set as being the half of the lowest average density found among the other habitat types; this habitat type was not considered for statistical comparisons or the average density value. Geckos were simultaneously counted during the night, inside 45 band transects deployed according to substrate's type availability, two people walking together each carrying a 1-m plastic tube for reference (thirty 20 x 2 m transects, and fifteen 10 x 2 m transects). Detailed search for geckos (including inside crevices and under loose rocks) was performed to minimize differences in their detectability among habitats. Differences between densities among habitat types were tested using a Kruskal-Wallis test. Average density from each habitat was extrapolated to the approximate surface covered by each habitat type to estimate overall population's size. These surfaces were extracted from a digital elevation model of the island, considering a combination of surface slope and bottom type (see López-Victoria and Rozo, 2006).
General morphology of the gecko was studied based on 55 individuals (30 in 2006, 25 in 2009) of all sizes, collected by hand, which were measured and photographed in detail. Measurements included snout-vent length (SVL), tail length (TL), total length (ToL), and weight.
RESULTS
Phyllodactylus transversalis is rigorously nocturnal and hides in clefts and holes during the day. During the night geckos of all ages were spotted on all existing substrates showing no noticeable preference to a particular microhabitat (but see habitat preference below). All man-made structures were frequented as well. Main activity observed during the nighttime was search for food; during the day just resting. Examination of the fecal samples and direct observations confirm previous observations on their diet, feeding mostly on ants and crickets.
Several geckos could often be found within close distance (< 20 cm), but without clear signs of territorial display. Ten out of the 55 geckos marked were spotted close to the place of first catch, three of them up to two nights after being marked. Social interactions were never witnessed in the field during the night, with no apparent signs of intentional aggregation or avoidance (but the light of the head lamps could have altered their behavior). Proximity of geckos to asleep Anolis, galliwasps, or boobies was usual; only crabs were avoided.
No gravid females or copulations were observed. Nevertheless, inside a stone crevice remains of eggs were found (Figure 1a-b): six eggshells or remains of formerly affixed eggs were inside this den. Their differing condition (i.e., texture, quantity of eggshell remaining, color) suggest they were of different ages, indicating the recurrent use of that den as a laying spot. While some eggshells were almost completely intact, others could only be identified by the chalk remains on the stone. The crevice had a longish opening with the narrowest access being 4 mm and the largest 7 mm wide. All of the eggs' leftovers were in an area of about 10 x 10 cm, whereas the crevice's total area was about 30 x 40 cm. The size of the best preserved eggshell was 14 mm in length and 8 mm in width, with oval shape (Figure 1c). The eggshell was compact but very thin; two adult geckos were resting in the crevice.
Several young, recently hatched geckos were spotted and some of them were measured (both in May-June and October). The smallest gecko found had a SVL of 23.0 mm, a TL of 24.0 mm, and weighed 0.6 g; it still had some vestiges of the shell membrane at the extremities. Three more young geckos were measured: 60.7, 72.1, and 72.3 mm ToL, and 0.7, 1.3, and 1 g weight, respectively. Those three juveniles also had complete tails.
The largest gecko found was 76.6 mm SVL, 69.0 mm TL, and 10.5 g weight. Average values (± 1 SE) for adult individuals were 60.22 mm SVL (± 1.22), 53.84 mm TL (± 1.60), and 5.28 g (± 0.29). Around half (56%) of the inspected animals showed a morphological characteristic exhibited by species of this genus (see Kluge, 1982): these individuals had a pair of slits at the tail base, corresponding to the openings of the cloacal sacs (Figure 1d). In general, these openings are present in both females and males, but are more conspicuous in the latter, suggesting that the sex ratio of the population is close to 1:1.
Kruskal-Wallis test showed no significant differences between average densities between the three main habitat types (H(2,N=45) = 4.44, p = 0.11), suggesting that this gecko has no evident habitat preference for foraging. A slightly higher concentration of geckos was found in H1 (big and medium-sized rocks; Table 1). Combining all habitats, Malpelo has an average concentration of 0.12 (± 0.01) geckos m-2. Extrapolating the corresponding average density values inside each one of the habitats to the surface they represent in Malpelo, the total population of P. transversalis is close to 114000 individuals (Table 1).
DISCUSSION
Like other species of Phyllodactylus, Colombian leaf-toed gecko is rigorously nocturnal (Dixon and Huey, 1970), a condition most likely inherited from its ancestors. Being nocturnal certainly facilitated its adaptation to the particular conditions on Malpelo, where a high pressure of competition and predation exists, since availability of resources is spatially and temporally limited (Wolda, 1975; López-Victoria and Werding, 2008; López-Victoria et al., 2009). As soon as a source of nutrition is available at daytime, several galliwasps, Anolis, and crabs immediately gather around to consume it, as fast as possible (Kiester, 1975; López-Victoria and Werding, 2008; López-Victoria et al., 2009). The gecko, being nocturnal, avoids predation and food competition in such situations, and preys upon nocturnal invertebrate species that are less available to the other diurnal lizards (Kiester, 1975; Wolda, 1975; López-Victoria et al., 2009).
Despite the small size and homogeneous environment of Malpelo, there is a diverse fauna of invertebrates (Calero et al., 2011). Most of the species live under stones and in clefts at daytime, but become more active and leave their shelters at night, representing an ideal food source for the geckos. Concerning the size of the invertebrates, all of the species found fit the prey spectrum of an adult gecko. Therefore, even freshly hatched young geckos have a large spectrum of species available. However, ants and crickets might be the main nutrition source as they are numerous, something that makes them also the most frequent invertebrates eaten by Anolis agassizi (Rand et al., 1975). Crabs might also be a part of the geckos' spectrum of prey, but only during their early stages of life (as freshly recruited megalops or as juveniles; López-Victoria and Werding, 2008).
Since the gecko is by far the smallest squamate vertebrate on Malpelo, the risk of predation is supposedly high. The galliwasp, the land crab, and the anole take advantage of ill, wounded, or careless animals (M. L. V. and M. J. pers. obs.). Intraspecific aggression seems to result in a high percentage of tail regenerates found in all three reptile species. Intra-specific aggression or territorial behavior do not seem to be the main cause of this phenomenon, because none of the three species really shows hierarchical, tight structured social systems (Kiester, 1975; Rand et al., 1975; López-Victoria et al., 2011). The four geckos' gatherings we found during daytime (inside crevices and cracks of boulders) were most likely based on the mutual usage of suitable dens.
The eggshells of different ages indicate a multiple usage of the laying place and a joint laying of eggs. Although Malpelo offers a lot of clefts and gap systems, the predation pressure is quite high. Therefore, an extensive use of appropriate dens for laying seems plausible (i.e., crevices with ideal conditions, like temperature and humidity). The remains of the eggs were not in a double egg clutch but separately. This is a typical behavior for the genus Phyllodactylus (Dixon and Huey, 1970), as well as for other representatives of the family Phyllodactylidae (Gamble et al., 2008).
Considering the size and external aspect of adult geckos, the juveniles we found were between a few days and weeks old. Previous personal observations and our findings during this study suggest active breeding during most of the year, coupled with the major breeding season of the Nazca Booby. The reproduction of the other two endemic lizards from Malpelo seems also to be temporally coordinated with the breeding season of the birds (Kiester, 1975; Rand et al., 1975). A linking between birds' and lizards' reproduction is reasonable, because during the breeding season the birds produce the largest amount of excrements, food remains, eggs, and dead squabs (López-Victoria et al., 2009). Consequently, the nutrient input during this time is significantly higher than outside the breeding seasons, having at least two positive effects on the geckos (i.e., all the lizards): the availability of nutrition has its peak at this time, because the invertebrates probably achieve their highest number of individuals, and predation and competition pressures are likely to decrease because of the availability of more and easier prey.
The fact that gecko densities were not statistically different among habitats suggests that geckos are more or less evenly distributed, and that they wander around randomly during the night in search for food and/or potential mates. If the prey (i.e., ants and crickets) is randomly distributed or is not associated to a particular habitat type, then this would result in a habitat-independent foraging behavior of the geckos, spreading over all four habitats. During the day narrow clefts and gaps are of pivotal importance as shelter for rest and to avoid predation.
Most of the species inhabiting Malpelo achieve higher densities and sizes where the boobies breed (close to their nests), as shown by studies on the population of the land crab (López-Victoria and Werding, 2008), and population estimation of the two other endemic lizards (Kiester, 1975; Rand et al., 1975; López-Victoria, 2006; López- Victoria et al., 2011). Our new estimation of around 114 000 individuals is higher than the single previous report (López-Victoria, 2006), but further studies with more accurate techniques (i.e., mark and recapture estimates) should provide more accurate numbers.
ACKNOWLEDGEMENTS
This investigation was partially supported by the Marine and Coastal Research Institute (Invemar), the Department of Animal Ecology at the Justus- Liebig-University of Giessen (JLU), the Just'us Program (Junior Teaching and Science Units) from the JLU, the Malpelo Foundation, the National Natural Parks System of Colombia, and the Colombian Navy. We are grateful to S. Bessudo, G. Soler, P. Herrón, M. Rodríguez, and J. C. Botello for their logistic support and their assistance in the field.
LITERATURE CITED
1 Calero, D., M. López-Victoria and P. Chacón de Ulloa. 2011. Composición y estructura trófica de los macroinvertebrados terrestres de la isla Malpelo, Pacífico colombiano. Bol. Invest. Mar. Cost., 40 (Supl. Esp.): 155-173. [ Links ]
2 Dixon, J. R. and R. B. Huey. 1970. Systematics of the lizards of the gekkonid genus Phyllodactylus of mainland South America. Los Angeles County Mus. Contrib. Sci., 192: 1-78. [ Links ]
3 Gamble, T., A. Bauer, E. Greenbaum and T. Jackman. 2008. Out of the blue: a novel, trans-Atlantic clade of geckos (Gekkota, Squamata). Zoologica Scripta, 37 (4): 355-366. [ Links ]
4 Gorman, G. C. and T. L. Chorba. 1975. Terrestrial biology of Malpelo Island: a historical review. Smithson. Contrib. Zool., 176: 9-12. [ Links ]
5 Huey, R. B. 1975. A new gecko from Malpelo Island (Sauria: Gekkonidae: Phyllodactylus). Smithson. Contrib. Zool., 176: 44-46. [ Links ]
6 Kiester, A. R. 1975. Notes on the natural history of Diploglossus millepunctatus (Sauria: Anguidae). Smithson. Contrib. Zool., 176: 39-43. [ Links ]
7 Kluge, A. G. 1982. Cloacal bones and sacs as evidence of gekkonoid lizard relationships. Herpetologica, 38 (3): 348-355. [ Links ]
8 López-Victoria, M. 2006. The lizards of Malpelo (Colombia): some topics on their ecology and threats. Caldasia, 28: 129-134. [ Links ]
9 López-Victoria, M. and D. M. Rozo. 2006. Model-based geomorphology of Malpelo Island and spatial distribution of breeding seabirds. Bol. Invest. Mar. Cost., 35: 111-131. [ Links ]
10 López-Victoria, M. and B. Werding. 2008. Ecology of the endemic land crab Johngarthia malpilensis (Decapoda: Brachyura: Gecarcinidae), a poorly known species from the Tropical Eastern Pacific. Pac. Sci., 62: 483-493. [ Links ]
11 López-Victoria, M., V. Wolters and B. Werding. 2009. Nazca Booby (Sula granti) inputs maintain the terrestrial food web of Malpelo Island. J. Ornithol., 150 (4): 865-870. [ Links ]
12 López-Victoria, M., P. A. Herrón and J. C. Botello. 2011. Notes on the ecology of the lizards from Malpelo Island. Bol. Invest. Mar. Cost., 40 (Supl. Esp.): 79-89. [ Links ]
13 Rand, A. S., G. C. Gorman and W. M. Rand. 1975. Natural history, behavior and ecology of Anolis agassizi. Smithson. Contrib. Zool., 176: 27-38. [ Links ]
14 Stead, J. A. 1975. Field observations on the geology of Malpelo Island. Smithson. Contrib. Zool., 176: 17-20. [ Links ]
15 Wolda, H. 1975. The ecosystem of Malpelo Island. Smithson. Contrib. Zool., 176: 21-26. [ Links ]
FECHA DE RECEPCIÓN: 19/04/2013 FECHA DE ACEPTACIÓN: 04/07/2013