Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Boletín de Investigaciones Marinas y Costeras - INVEMAR
Print version ISSN 0122-9761
Bol. Invest. Mar. Cost. vol.43 no.2 Santa Marta July/Dec. 2014
CAULLERIELLA PETERSENAE N. SP. AND TWO NEW RECORDS OF CIRRATULIDAE (ANNELIDA: POLYCHAETA) FROM VENEZUELA
CAULLERIELLA PETERSENAE N. SP. Y DOS NUEVOS REGISTROS DE CIRRATULIDAE (ANNELIDA: POLYCHAETA) PARA VENEZUELA
Oscar Díaz-Díaz1, Adibe Cárdenas-Oliva2 e Ildefonso Liñero-Arana1
1 Instituto Oceanográfico de Venezuela, Departamento de Biología Marina. Av. Universidad, Edif. IOV Piso 1, Cumaná, Venezuela. ofdiazd@gmail.com, ilineroarana@yahoo.es.
2 Instituto Oceanográfico de Venezuela, Posgrado en Ciencias Marinas. Av. Universidad, Edif. IOV Coordinación de Postgrado, Cumaná, Venezuela. adi_carol@yahoo.com.
ABSTRACT
A total of 295 specimens of cirratulids were collected at five localities from the northeastern coast of Venezuela. Caulleriella petersenae is described as a species new to science, which is characterized by the absence of eyes and the presence of the first notopodial hooks at the chaetiger 26. On the other hand, Monticellina cf. serratiseta and Timarete caribous were recorded for the first time from Venezuela waters. All these species have been found associated to macro-algae and coral reef.
KEYWORDS: Polychaetes, annelids, biodiversity, cirratulids, Great Caribbean region.
RESUMEN
Un total de 295 cirratúlidos fueron recolectados en cinco localidades de la costa nororiental de Venezuela. Caulleriella petersenae, nueva especie para la ciencia, se caracterizó por la ausencia de ojos y la presencia de ganchos notopodiales a partir del setígero 26. Por otro lado, Monticellina cf. serratiseta y Timarete caribous fueron registradas por primera vez para Venezuela. Todas estas especies fueron encontradas asociadas a macroalgas y formaciones coralinas.
PALABRAS CLAVES: Poliquetos, anélidos, biodiversidad, cirratúlidos, Gran Caribe.
INTRODUCTION
The family Cirratulidae Ryckholt 1851, is an important and common component of intertidal and sub-tidal benthic communities, due to its abundance and frequency. Gibbs (1971) reported that some species from deep waters, such as Aphelochaeta marioni (De Saint-Joseph, 1894), may reach densities of up to 1000 ind·m-2. Although it is a well-studied family in terms of its biology, physiology, and population dynamics, it is considered to be little known taxonomically as there are still many inconsistencies in its systematics (Rouse and Pleijel, 2001; Dean and Blake, 2007). Some of these arise with regard to the placing of multi-tentacled juveniles, which have different chaetae types and a branchial distribution unlike that observed in adults, thus making their identification difficult (Blake, 1996). Díaz-Díaz and Salazar-Vallejo (2009) have suggested that the low number of taxonomic characteristics used has led researchers to confuse juvenile specimens (or specimens at other stages of development) with adults, which has resulted in the wrong designation of several species and genera. A family revision and reordering is therefore critically necessary.
A total of 12 genera and more than 170 cirratulid species are recognized, although the latter has increased in recent years. Blake (1996) described 20 new species from the continental shelf and intertidal localities of the Western Pacific coasts, mainly from California. The most recent studies include those of Dean and Blake (2007), who described eight new species in Chaetozone (3) and Caulleriella (5), and Dean and Blake (2009), who described six new species of Monticellina, both from the Pacific Central American coast. As regards to the Atlantic Ocean, Cirratulidae have been reported by Chambers (2000) and Chambers and Woodham (2003) from the seas off the northeastern coast of North America; Doner and Blake (2006) reported four new species [Chaetozone (3) and Caulleriella (1)] from shallow waters off the coast of Massachusetts. In South American waters, Elías and Rivero (2008, 2009a, 2009b) described a total of five new species from the Argentine coast. Whereas in the Great Caribbean, Wolf (1984) recorded 16 species from the Gulf of Mexico and Díaz-Díaz and Liñero-Arana (2004, 2011) recorded ten species from the continental shelf off Venezuela.
Recently, an identification key for genera and species from both the Great Caribbean Sea and Mexican Pacific coasts have been published (Díaz-Díaz and Salazar-Vallejo, 2009). In this paper, three cirratulids species associated with macroalgae and coral reefs, Caulleriella petersenae n. sp, Monticellina cf. serratiseta (Banse and Hobson, 1968), and Timarete caribous (Grube, 1856), are described for the first time from the Venezuelan coast.
STUDY AREA
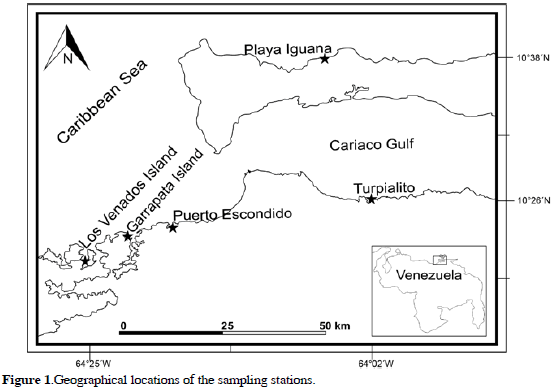
Biological material was collected from five localities along the northeastern coast of Venezuela (Figure 1): 1) Playa Iguana (10°38'10"N-64°04'57"W) is a rocky shore, with a Phragmatopoma sp. (Polychaeta: Sabellariidae) reef and Centroceras clavulatum population. 2) Garrapata Island (10°23'20"N-64°21'03"W) is also a rocky shore, with a great abundance of Halimeda opuntia and Caulerpa racemosa. 3) Los Venados Island (10°21'34"N-64°25'16"W) is made up of coralline sand and a reef dominated by Millepora alcicornis, Halimeda opuntia, Padina tetrastomatica and Ulva fasciata Delile, 1813, with associated macroalgae species. 4) Puerto Escondido (10°24'30"N-64°17'42"W) is characterized by coralline sand, and coral reef dominated by M. alcicornis and Acropora palmata. 5) Turpialito-Gulf of Cariaco (10°26'37"N-64°01'58"W) has coastal mangrove forests with a great abundance of Halimeda opuntia, C. racemosa, Sargassum vulgare and U. fasciata.
MATERIALS AND METHODS
Sampling was done between April 2007 and January 2009. Organisms were collected by hand with the aid of snorkeling equipment, between 0.5 and 3 m deep. Macroalgae were sampled using plastic bags, separating the algae from the substrate and introducing them into the bags carefully to prevent the escape of vagile polychaetes. Collected material was taken to the Polychaete Laboratory at the Instituto Oceanográfico de Venezuela. Polychaetes and algae were separated and fixed with 8 and 5% formaldehyde solution in seawater, respectively. Polychaetes were examined using compound and dissecting microscopes and structures with taxonomic value were extracted and mounted on slides for their study. Drawings of the complete specimens and particular morphological structures were done following Coleman (2006). Polychaetes were deposited in three collections: the Reference Collection at the Laboratorio de Biología de Poliquetos (LBP-FamilyNo.Cat./No.Specimens), Instituto Oceanográfico de Venezuela; Museo Oceanológico Hermano Benigno Roman, Margarita Island, Venezuela (MOBR-I-1174) 07/09/13 and Museo Marino de Cumaná (Universidad de Oriente) (MMUDO-IP-024) 27/07/13. Macroalgae were deposited in the reference collection of the Laboratorio de Ficología del Instituto Oceanográfico de Venezuela.
RESULTS AND DISCUSSION
In Venezuela, polychaete studies are scarce, most have been made in the northeastern coast (Díaz-Díaz et al., 2013). Currently, four hundred and six species in 40 families are recognized, and only ten cirratulids species are recorded from Venezuelan waters. With these results the knowledge of the family Cirratulidae increase to thirteen species recorded for the country.
Two hundred and ninety five cirratulid worms were examined and three species in three genera were described, Caulleriella petersenae n. sp., Monticellina cf. serratiseta (Banse and Hobson, 1968) and Timarete caribous (Grube, 1856). The latter two species are new records for Venezuela.
Caulleriella petersenae n. sp.
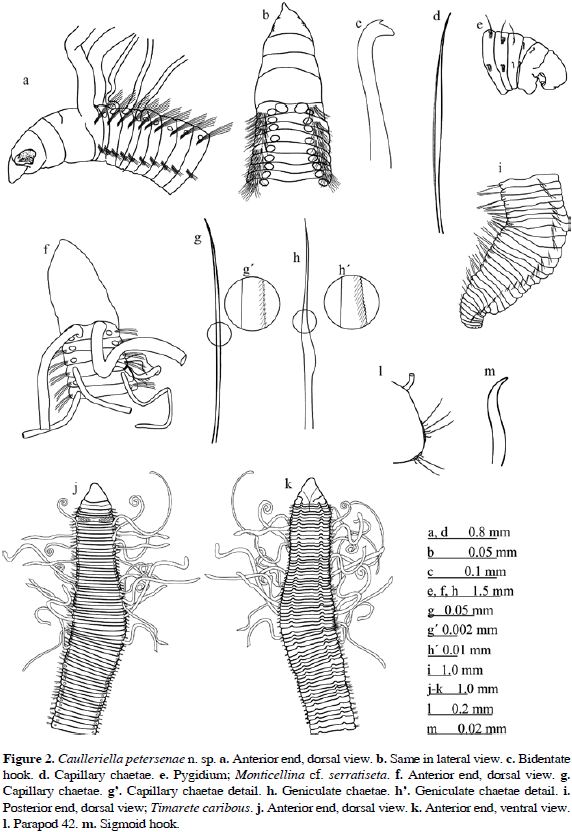
Figure 2a-e
Material examined. Nineteen specimens: Garrapata Is. LBP-Ci0100/1 (holotype). Paratype: LBP-Ci0101/1; LBP-Ci0102/1 LBP-Ci0103/1; LBP-Ci0105/1; LBP-Ci0108/1; Los Venados Is., LBP-Ci0106/1; Playa Iguana; LBP-Ci0104/1. Garrapata Is. LBP-Ci0109/8 (only anterior end fragments) LBP-Ci0110/3, Puerto Escondido, associated with Millepora alcicornis, 18/01/08.
Description. Complete specimen (holotype), 10 mm long, 0.4 mm wide, with 152 chaetigers. Prostomium conical, pointed (Figure 2a-b); eyes absent. The nuchal organs on the posterior margin of prostomium are dark and either semicircular or half-moon shaped, overlapping at the peristomial ridge. Peristomium triannulated; anterior ring covering posterior prostomium, dorsally and laterally; anterior and middle rings of similar length, posterior ring about twice the length of previous ones. Palps arising dorsally from the posterior margin of the peristomium. Branchiae from the first chaetiger emerging dorsally from the notopodial base. Capillaries (Figure 2d) on all notopodia. Bidentate hooks (Figure 2c) alternating with capillary chaetae from chaetiger 24; two hooks on chaetiger 24 and up to five on the mid body and mid-posterior chaetigers. One bidentate hook on posterior chaetigers. Capillaries and bidentate hooks from the first neuropodia. Pygidium rounded with four lobes ventrally, surrounding the anus (Figure 2e). No pattern with methyl green staining was observed, except for the pygidium which stained profusely.
Remarks. Caulleriella petersenae n. sp. is close to C. cucula Dean and Blake, 2007, and C. bremecae Elías and Rivero, 2008. Nevertheless, C. petersenae n. sp. is close to C. cucula Dean and Blake, 2007, differing in that:
- The notopodial hooks in C. cucula are present between chaetigers 43 and 92 (the latter corresponds to the holotype), while in C. petersenae n. sp. they are present between chaetigers 17 and 26 (24 in the holotype).
- The pygidium in C. cucula consists of a conical lobe, whereas in C. petersenae n. sp. it is cup-shaped, with a series of ventral lobes.
- The nuchal organ in C. cucula is L-shaped, whereas in C. petersenae n. sp. it is semicircular or half-moon-shaped and dark (preserved material without coloration).
- The type locality of C. cucula is the Gulf of Nicoya, Pacific coast of Costa Rica, where it is associated with soft sediments consisting mainly of mud, while C. petersenae n. sp. was found associated with macroalgae.
- Methyl green staining in C. cucula gave a uniform green stain over the body but with the prostomium unstained and the peristomial crest and mid-ventral band, extending over the length of the body, weakly stained.
Caulleriella petersenae n. sp. differs from C. bremecae in that:
- The nuchal organs in C. bremecae, described as eyespots by Elías and Rivero (2008), are located laterally, whereas in C. petersenae n. sp. they are found dorsally.
- The peristomium is not ringed in C. bremecae and the posterior margin is extended, forming a raised ridge that overlaps the mid-anterior portion of the first chaetiger, whereas in C. petersenae n. sp., the posterior margin is not extended and does not form a raised ridge.
- The type locality of C. bremecae is Mar del Plata, Argentina, where it is found associated with soft sediments retained by the beds of the mussel Brachidontes rodriguezii in areas moderately enriched by sewage discharge, whereas C. petersenae n. sp. was found associated with macroalgae.
- In C. bremecae the peristomium, palps, branchiae and the ventral region of the anterior chaetigers stain profusely with methyl green, whereas in C. petersenae n. sp. only the pygidium stains.
Three Caulleriella species -C. alata (Southern, 1914), C. bioculata (Keferstein, 1862), and C. zetlandica (McIntosh, 1911)- have been recorded for the Great Caribbean but their presence in the region is considered questionable (Díaz-Díaz and Salazar-Vallejo, 2009), mainly due to the type localities of these species (all in European waters). Caulleriella petersenae n. sp. differs from C. alata and C. bioculata by the absence of eyes and because the notopodial hooks start on chaetiger 24 instead of on chaetigers 20-21 and 12-23, respectively. Caulleriella petersenae n. sp. is different from C. zetlandica because in the latter the notopodial hooks are absent. Finally, C. petersenae n . sp. is the only species of this genus confirmed from the Great Caribbean region to date.
Variation. The paratype from Playa Iguana is 6.5 mm long, 0.3 mm wide and has 102 chaetigers, with notopodial hooks from chaetiger 21. Paratypes from Garrapata Is. are 3.5-9 mm in length, 0.2-0.4 mm in width and with 77-116 chaetigers. One paratype with 77 chaetigers has notopodial hooks from chaetiger 17, a specimen with 108 chaetigers has notopodial hooks from chaetiger 21, a specimen with 112 chaetigers has notopodial hooks from chaetiger 22, and specimens with 114 and 116 chaetigers, have hooks on chaetigers 26 and 24, respectively. Seven incomplete specimens have notopodial hooks on chaetigers 23 or 24. The smallest specimens have an inconspicuous constriction on the anterior peristomial ring, which gives a four-ringed appearance.
Etymology. The species is named in honor of Dr. Mary Petersen, a highly respected polychaete taxonomist, who specializes in the family Cirratullidae.
Distribution. Northeastern coast of Venezuela.
Monticellina cf. serratiseta (Banse and Hobson, 1968)
Figure 2f-i
Tharyx serratiseta Banse and Hobson, 1968: 37-39, Figs. 7k, l.
Aphelochaeta serratiseta, Blake, 1991: 28; Díaz-Díaz and Salazar-Vallejo, 2009: 137, Fig. 1G.
Monticellina serratiseta, Blake, 1996: 325, Fig. 8.25A-D; Dean and Blake, 2009: 122; Magalhães and Bailey-Brock, 2013: 96.
Material examined. LBP-Ci0020/1, Garrapata Is. (04/2007), associated with Caulerpa racemosa (Chlorophyta), collected by Díaz-Díaz and Cárdena-Oliva at a depth of 1.5 m.
Description. Complete specimen, 10 mm long, 0.4 mm wide with 121 chaetigers. Prostomium conical, pointed, eyes absent. Semicircular nuchal organs present. Peristomium triannulated (Figure 2f). Palps arising dorsally from the anterior margin of chaetiger 1. First pair of branchiae on chaetiger 1, dorsal to notopodial chaetal bundle and posterior to dorsal tentacles, one pair on each segment, continuing on subsequent chaetigers until near the end of the body, all branchiae longer than body width. Capillary chaetae on all parapodia; notochaetae (Figure 2g) half as long as the width of the segments, with weakly serrated margins (Figure 2g'); neurochaetae capillaries smooth on anterior segments; capillaries shorter, slightly geniculated, with expanded serrated blades, from chaetiger 12 (Figure 2h, h'). Chaetigers 90-111expanded (Figure 2i). Pygidium is a simple terminal lobe. Anus dorsal.
Remarks. Currently13 species of Monticellina are recognized (Blake, 1991,1996; Magalhães and Bailey-Brock, 2013); of these, only M. dorsobranchialis (Kirkeegard, 1959) has been recorded from the Great Caribbean, but its presence in the region is questionable (Díaz-Díaz and Salazar-Vallejo, 2009) since its type locality is Angola (Africa). Monticellina dorsobranchialis differs from the specimen examined in this study in that the capillary chaetae are serrated but not widened subdistally, the prostomium is short, the anterior branchiae are dorsally fused to the notopodia and from chaetiger 20 they are moved to the middle-dorsal region. Monticellina cf. serratiseta is somewhat similar to M. secunda (Banse and Hobson, 1968) and M. serratiseta (Banse and Hobson, 1968). However, it differs from the former species in presenting widened neurochaetae; in addition, M. secunda has chaetae from chaetiger 25 on both the notopodia and neuropodia, whereas the examined specimen has chaetae from chaetiger 12 but only on the neuropodia. It is, nevertheless, very similar to the original description, and agrees with its figure 7i (Banse and Hobson, 1968). Monticellina cf. serratiseta is close to M. serratiseta in that both show widened capillary neurochaetae with serrated margins; however, these start at chaetiger12 in the former and between chaetiger 75-100 in the latter. The specimen examined was identified as M. cf. serratiseta because 1) the second pair of branchiae emerge dorsally from the chaetal bundle on the second chaetiger; 2) the first chaetiger has no posterior margin (as in M. serratiseta); 3) the branchiae are longer than the width of the body; and 4) the type locality for M. serratiseta is Puget Sound, Washington. The revision of more specimens is required in order to confirm their identity or consider a new species.
Distribution. Northeastern coast of Venezuela.
Timarete caribous (Grube and Ørsted in Grube, 1859)
Figure 2j-m
Cirrhatulus caribous Grube and Oersted in Grube,1859: 106; Hartman, 1959: 403.
Timarete caribous, Díaz-Díaz and Salazar-Vallejo, 2009: 145, Fig. 6a.
Material examined. Two hundred and seventy five specimens. LBP-Ci0001/242, Venado Is. (08/2008), associated with Centroceras clavulatum; LBPCi0002/ 23, Turpialito (01/2009), associated with Padina tetrastomatica. Material collected by Díaz-Díaz and Cárdena-Oliva, between 1-3 m deep.
Description. Complete specimen with 80 chaetigers (26.7 mm long and 1.6 mm wide). Body elongated, thickened throughout, with short segments, tapering posteriorly (Figure 2j) and with a shallow ventral groove (Figure 2k). No color spots on either the dorsal or ventral body surfaces. Prostomium conical, broad, bluntly pointed on anterior margin, wider than long, without eyes. Peristomium short, about 1.5 times longer than the prostomium, with two distinct annulations. Tentacles cirriform from the fourth chaetiger arranged in two separate groups. Branchiae from chaetiger 1 to posterior segments, with variable positions; near the notopodia base on anterior segments and slightly displaced dorsally on the middle and subsequent chaetigers (Figure 2l). Parapodia poorly developed; notopodia and neuropodia widely separated. All chaetae simple, including the capillary chaetae in noto- and neuropodia all chaetigers. Acicular spines slightly sigmoid and pale brown (Figure 2m), with up to three spines from notopodium 11 and neuropodium 5. Last five neuropodia with 1-2 sigmoid spines, capillary chaetae absent. Pygidium rounded, lobe shaped, with terminal anus. Live specimens pale green.
Remarks. Only three species of the genus Timarete have been previously recorded for Venezuela: Timarete sp., T. punctata (Grube, 1859), and T. tentaculata (Montagu, 1808) (Díaz-Díaz and Liñero-Arana, 2004). Timarete tentaculata, from South Devon, England, differs from T. caribous because the former only has neuropodial hooks. Timarete caribous is, however, similar to T. punctata, from the Caribbean Sea, in that both species have hooks on both branches, but they differ in that T. caribous has a uniform color, while T. punctata has irregularly distributed dark spots on the body and branchial and tentacular filaments. It is likely that the material identified as Timarete sp. by Díaz-Díaz and Liñero-Arana (2004) corresponds to T. caribous.
Variation. Specimens 13.5-28 mm long, 0.7-1.4 mm wide, with 76-159 chaetigers; notopodial hooks from chaetigers 8-13; one specimen with 159 chaetigers has notopodial hooks from chaetiger 13 and one with 112 chaetigers has notopodial hooks from chaetiger 22. Neuropodial hooks from chaetigers 4-7. The smallest specimens have a smooth constriction on the peristomium, giving a four-ringed appearance. Some specimens with very short branchiae on the first four chaetigers.
Distribution. Great Caribbean region.
ACKNOWLEDGEMENTS
The authors would like to thank the Consejo de Investigación of the Universidad de Oriente for funding this study through the project "Poliquetos (Annelida: Polychaeta) de la costa nororiental" (CI-2-030601-1279/06), and the Instituto Oceanográfico de Venezuela for logistical support.
LITERATURE CITED
1 Banse, K. and K. D. Hobson. 1968. Benthic polychaetes from Puget Sound, Washington. Proc. U.S. Nat. Mus., 125 (3667): 1-53. [ Links ]
2 Blake, J. A. 1991. Revision of some genera and species of Cirratulidae (Polychaeta) from the western North Atlantic. Ophelia Suppl., 5: 17-30. [ Links ]
3 Blake, J. A. 1996. Family Cirratulidae Ryckholdt (sic), 1851, including a revision of the genera and species from the Eastern North Pacific. 263-384. In: Blake, J. A., B. Hilbig and P. H. Scott (Eds.). Taxonomic atlas of the benthic fauna of the Santa Maria Basin and western Santa Barbara Channel, Vol. 6. The Annelida, Part 3. Santa Barbara Museum of Natural History, Santa Barbara, USA. 418 p. [ Links ]
4 Chambers, S. J. 2000. A redescription of Chaetozone setosa Malmgren, 1867 including a definition of the genus, and a description of a new species of Chaetozone (Polychaeta: Cirratulidae) from the northeast Atlantic. Bull. Mar. Sci., 67: 587-596. [ Links ]
5 Chambers, S. J. and A. Woodham. 2003. A new species of Chaetozone (Polychaeta: Cirratulidae) from deep water in the northeast Atlantic, with comments on the diversity of the genus in cold northern waters. Hydrobiologia, 496: 41-48. [ Links ]
6 Coleman, C. O. 2006. Substituting time-consuming pencil drawings in arthropod taxonomy using stacks of digital photographs. Zootaxa, 1360: 61-68. [ Links ]
7 Dean, H. K. and J. A. Blake. 2007. Chaetozone and Caulleriella (Polychaeta: Cirratulidae) from the Pacific coast of Costa Rica, with description of eight new species. Zootaxa, 1451: 41-68. [ Links ]
8 Dean, H. K. and J. A. Blake. 2009. Monticellina (Polychaeta: Cirratulidae) from the Pacific coast of Costa Rica with descriptions of six new species. Zoosymposia, 2: 105-126. [ Links ]
9 Díaz-Díaz, O. and I. Liñero-Arana. 2004. Cirratulidae (Annelida: Polychaeta) de la región oriental de Venezuela. Bol. Inst. Oceanogr. Vzla., 43: 3-10. [ Links ]
10 Díaz-Díaz, O. and I. Liñero-Arana. 2011. Nuevas adiciones de poliquetos (Annelida: Polychaeta) para el Golfo de Cariaco, Venezuela II: Sedentarios. Bol. Inst. Oceanogr. Vzla., 50 (2): 183-208. [ Links ]
11 Díaz-Díaz, O. and S. I. Salazar-Vallejo. 2009. Cirratulidae. 131-147. In: De León-González, J. A., J. R. Bastida-Zavala, L. F. Carrera-Parra, M. E. García-Garza, A. Peña-Rivera, S. I. Salazar-Vallejo y V. Solís-Weiss (Eds.). Poliquetos (Annelida: Polychaeta) de México y América Tropical. Univ. Autón. Nuevo León, Monterrey. 737 p. [ Links ]
12 Díaz-Díaz, O., L. Troccoli and O. Díaz-Pérez. 2013. Estructura de la comunidad de poliquetos de fondos blandos en tres localidades de la bahía de Mochima, Venezuela. Scientia (Panamá), 23 (2): 65-88. [ Links ]
13 Doner, S. A. and J. A. Blake. 2006. New species of Cirratulidae (Polychaeta) from the northeastern United States. Scientia Mar., 70S3 (Suppl. 3): 65-73. [ Links ]
14 Elías, R. and M. S. Rivero. 2008. Two new species of Caulleriella (Polychaeta, Cirratulidae) from Argentina. Iheringia, Sér. Zool., 98 (2): 225-230. [ Links ]
15 Elías, R. and M. S. Rivero. 2009a. Two new species of Cirratulidae (Annelida: Polychaeta) from Mar del Plata, Argentina (SW Atlantic). Zoosymposia, 2: 139-148. [ Links ]
16 Elías, R. and M. S. Rivero. 2009b. First new Dodecaceria (Polychaeta: Cirratulidae) species from the SW Atlantic (38ºS-57ºW, Argentina). Rev. Biol. Mar. Oceanogr., 44 (1): 131-136. [ Links ]
17 Gibbs, P. E. 1971. A comparative study of reproductive cycle in four polychaetes belonging to the family Cirratulidae. J. Mar. Biol. Ass. UK, 51: 745-769. [ Links ]
18 Grube, A. E. 1859. Annulata Oerstediana. Enumeration Annulatorum, quae in itinere per Indiam occidentalum et American centralem annis 1845-1848 suscepto legit. d. A. S. Oersted, adjectis speciebus nonullis a d. H. Krøyer in itinere ad Americam meridionalem collectis. Pt. 3. Vidensk. Medd. dansk Naturh. Foren., 1858: 105-120. [ Links ]
19 Hartman, O. 1959. Catalogue of the polychaetous annelids of the world, parts 1-2. Allan Hancock Foun. Pub. Occas. Pap., 23:1-628. [ Links ]
20 Magalhães, W. F. and J. H. Bailey-Brock. 2013. Bitentaculate cirratulidae (Annelida: Polychaeta) from the northwestern Pacific Islands with description of nine new species. Zootaxa, 3630 (1): 080-116. [ Links ]
21 Rouse, G. and F. Pleijel. 2001. Cirratulidae. 208-211. In: Rouse, G. and F. Pleijel (Eds.). Polychaetes. Oxford Univ. Press, New York. 354 p. [ Links ]
22 Wolf, P. S. 1984. Family Cirratulidae Carus, 1863. 12.1-12.12. In: Uebelacker, J. M. and P. J. Johnson (Eds.). Taxonomic guide to the Polychaetes of the Northern Gulf of Mexico. Vol. 2. Barry A. Vittor and Associates. Mobile, USA. 251 p. [ Links ]
FECHA DE RECEPCIÓN: 07/10/2013 FECHA DE ACEPTACIÓN: 24/09/2014