Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Boletín de Investigaciones Marinas y Costeras - INVEMAR
Print version ISSN 0122-9761
Bol. Invest. Mar. Cost. vol.44 no.1 Santa Marta Jan./June 2015
NOTES ON THE MARINE ALGAE OF THE INTERNATIONAL BIOSPHERE RESERVE SEAFLOWER, CARIBBEAN COLOMBIA V: FIRST STUDY OF THE ALGAL FLORA OF QUITASUEÑO BANK
NOTAS SOBRE LAS ALGAS MARINAS DE LA RESERVA INTERNACIONAL DE BIOSFERA SEAFLOWER, CARIBE COLOMBIANO V: PRIMER ESTUDIO PRELIMINAR SOBRE LA FLORA MACROALGAL DEL BANCO QUITASUEÑO
Brigitte Gavio1,2, Maria Alejandra Cifuentes-Ossa3 and Michael J. Wynne4
1 Universidad Nacional de Colombia sede Caribe. Circunvalar San Luis Free Town No. 52-44, San Andrés Isla, Colombia. bgavio@unal.edu.co.
2 Universidad Nacional de Colombia sede Bogotá, Departamento de Biología, Ciudad Universitaria, Bogotá, Colombia.
3 Universidad Nacional de Colombia Sede Palmira. Carrera 32 No. 12-00 Chapinero, Vía Candelaria, Palmira, Valle del Cauca, Colombia. macifuenteso@unal.edu.
4 University of Michigan Herbarium, Ann Arbor, MI 48108, USA. mwynne@umich.edu.
ABSTRACT
During a biodiversity expedition to the northern cays of the Archipelago of San Andrés, Old Providence and Saint Catalina, we collected macroalgal samples along Quitasueño Bank, a submerged 60 km-long coralline bank. This is the first phycological study on the bank. We present a preliminary list of 76 macroalgae, including ten species of cyanobacteria. Fifteen of these taxa are new records for Colombia, and an additional nine are new records for the Archipelago. With this preliminary study, we increase the number of taxa in the macroalgal flora of this region of Colombia by 10.1%.
KEYWORDS: Biodiversity, Colombia, Marine algae, New records, Quitasueño.
RESUMEN
En el marco de una expedición científica a los cayos del norte del Archipiélago de San Andrés, Providencia y Santa Catalina se colectaron muestras de macroalgas en el banco Quitasueño, un banco coralino sumergido de 60 km de longitud. Este es el primer estudio ficológico del banco. Se presenta una lista preliminar de 75 macroalgas, incluyendo 10 especies de cianobacterias. Quince de estas especies son nuevos registros para Colombia, y nueve especies adicionales son nuevos registros para el archipiélago. Con este estudio preliminar, se aumenta en 10.1% la biodiversidad de macroalgas de este departamento de Colombia.
PALABRAS CLAVE: Biodiversidad, Colombia, Algas marinas, Nuevos registros, Quitasueño.
INTRODUCTION
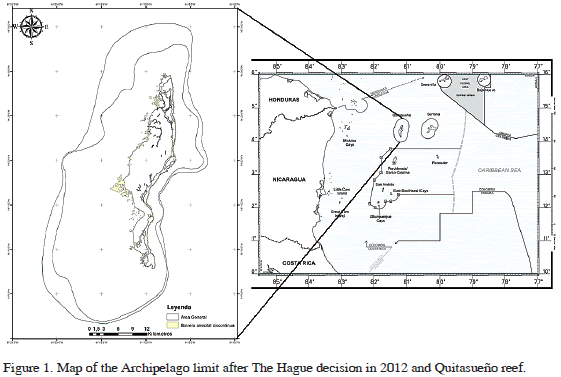
The Archipelago of San Andrés, Old Providence and Saint Catalina is one of the most pristine marine regions of the Caribbean (Friedlander et al., 2003) and harbors a largely unexplored biodiversity (e.g. Albis-Salas and Gavio, 2011; Ortiz and Gavio, 2012; Reyes-Gómez et al., 2013). It is situated in the southwestern Caribbean Sea, off the continental shelf of Nicaragua, and since 1822 has been part of the Republic of Colombia (Díaz et al., 1996). It encompasses three inhabited islands and several cays, banks and atolls for a total marine extension of almost 350,000 km2. In 2000 this region was declared the International Biosphere Reserve Seaflower, and it is now the 10th largest marine reserve in the world. The International Court of Justice in The Hague assigned approximately 53.6% of the marine territory of the Archipelago to Nicaragua (Bolaño and Acosta, 2013), in November 19th 2012, after an 11-year-long dispute, leaving two of the three northern cays, Quitasueño Bank and Roncador Bank, to Colombia, surrounded by Nicaraguan waters (Figure 1). The ultimate fate of the Reserve is uncertain because it cannot be predicted how those isolated cays will be managed. Quitasueño Reef (14° 28'N, 81° 07'W) (also called Queena reef by the raizal population of the Archipelago), located about 70 km north of Old Providence Island, is the largest reef complex of the Archipelago. The bank is about 60 km long in a NNE direction and 10-20 km wide (Díaz et al., 1996). There is no emerged cay near the bank, although there is a lighthouse close to the northern end of the bank. Despite the intense fishing activity historically undertaken in the area, this bank has been the subject of little scientific exploration, with its biodiversity not well known. Sánchez et al. (2005) reported high coral cover in Quitasueño, comparable to the best conserved coral habitats in the Caribbean. To date, no phycological studies have been undertaken on this bank. In September 2011, the Universidad Nacional de Colombia, sede Caribe, Coralina, and the regional government unified efforts to undertake a scientific exploration of the northern Cays, Quitasueño, Serrana, and Roncador. We herein present preliminary results of the macroalgal collection taken from Quitasueño.
MATERIALS AND METHODS
Quitasueño is located 70 km north of Old Providence Island. With a length of 60 km and a width of 10-20 km, it is the largest reef complex of the Archipelago, with no conspicuous emerged area. The fore reef, where most of the wave energy is discharged, extends for about 40 km in a north-south direction. The coral reef habitat is dominated by the corals Palythoa and Millepora, and the coralline red alga Porolithon. Due to the shallow depth, the wrecks of several boats still persist there. The reef is not continuous but presents deep and shallow channels towards its center. In the reef lagoon, where water is calmer, the depth is between 5-12 m, and there are abundant colonies of Montastrea (Díaz et al., 1996).
All the specimens were collected by scuba diving, at depths ranging from 1 to 30 m. The algae were collected by hand and preserved in a 4% formalin/ seawater solution. In the laboratory, algae were identified using an Olympus BX 51 compound light microscope and with appropriate reference books for species identification (e.g. Littler and Littler, 2000; Komárek and Anagnostidis, 2005; Dawes and Mathieson, 2008). Slide material was mounted in 50% glycerin, after staining in a dilute, acidified aniline-blue solution. Voucher specimens were deposited in the Herbarium of the Universidad Nacional de Colombia sede Caribe (HJBSAI-UN).
RESULTS AND DISCUSSION
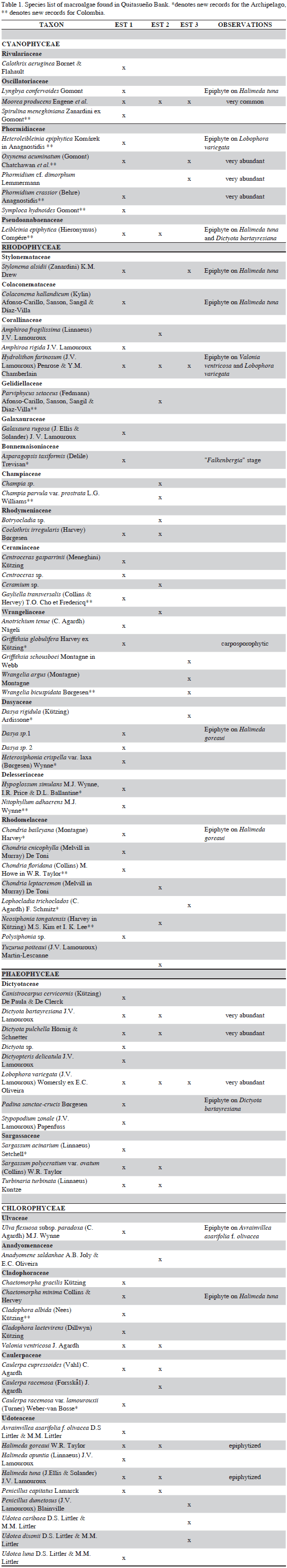
We report a total of 75 species: 10 species of Cyanophyta, 35 Rhodophyta, 11 Phaeophyceae and 19 Chlorophyta (Table 1). The families with the highest species number were Udoteaceae (9), Rhodomelaceae (8) and Dictyotaceae (8). The most common species were Moorea producens, which was very abundant as an epiphyte on other algae, Lobophora variegata, and Hydrolithon farinosum. Of these taxa, 9 species are new records for the Archipelago and an additional 15 are new records for Colombia; 12% of the taxa recorded in the present paper are new additions for the local marine flora, while 20% are new records for Colombia. With this preliminary study, we increase the marine flora biodiversity of the archipelago by 10.1%. Díaz-Pulido and Bula-Meyer (1997) reported a total of 171 taxa from the other atolls and cays of the Archipelago. Despite the scarcity of studies, the marine flora of the Archipelago appears to be much richer than previously recorded (Albis-Salas and Gavio, 2011; Ortiz and Gavio, 2012; Gavio et al., 2013; Reyes-Gómez et al., 2013), and can be compared to other well studied Caribbean reef complexes (Díaz-Pulido and Bula-Meyer, 1997). Particular attention should be given to the cyanobacterial flora of the bank. Some of these species, such as Heteroleibleinia epiphytica, Oxynema acuminatum, Phormidium crassior, and Oscillatoria meneghiniana, are reported for the first time in the Caribbean Sea. Although molecular analysis is necessary to determine any cryptic species, the specimens that we observed fit well the morphological descriptions of the mentioned taxa. In the field, we observed extensive mats covering all substrates, from sand to coral and macroalgae. The generic name Moorea Engene et al. (2012) is a later homonym and thus illegitimate; because the two previous uses of this name are not in current usage [the original one having been rejected], Wynne (2013) has formally proposed that the name Moorea Engene et al. be conserved.
We wish to recognize the var. laxa of Heterosiphonia crispella because we see the taxonomic value of accepting Børgesen's (1919) original arguments for treating this infraspecific taxon as worth accepting because of the difference from the nominate variety in terms of the number of pericentral cells (4 vs. 5-6) and its more slender and softer habit in contrast to the squarrose habit of var. crispella. Lipkin and Silva (2002) did not recognize var. laxa because they regarded it as a "shaded-habitat ecophene". Rull Lluch (2002) observed var. laxa to grow rather deep and in sheltered places and var. crispella to occur in exposed sites and near the surface. He also said that they showed the same ecology on the Namibian coast and because they are "difficult to distinguish", he thought that they should be considered a single taxon. But others, such as Schneider and Searles (1991) and Fernández-García et al. (2011), continue to accept var. laxa.
Coral cover in Quitasueño was rather low (Abril and Arango, 2012), especially if compared to former studies (Sánchez et al., 2005), suggesting a deterioration of the bank ecosystem. Considering that cyanobacteria are becoming increasingly prominent in declining reefs, that they may actively inhibit coral recruitment (Chapry et al., 2012), and that cyanobacterial mats may be toxic to scleractinian corals and are able to kill live coral tissue (Titlyanov et al., 2007), the extensive mats observed in Quitasueño may be an indicator of coral demise. However, it is necessary to carry out monitoring studies to establish the seasonality, distribution and extension of these cyanobacterial mats, in order to understand better the ecosystem dynamics of this reef.
ACKNOWLEDGEMENTS
The authors are grateful to all the people participating in the cruise: we wish to thank Captain Spencer Chow and all his crew, especially Monroy Hooker; Margarita Albis-Salas and Elizabeth Galeano, members of the algae group; Martha Manrique, Maria Fernanda Maya, Daniele Florio, Heins Bent, Alfredo Abril, Leonardo Arango, Arturo Acero, Anthony Rojas, Trisha Forbes, Jairo Lasso, and Samuel Ward. We acknowledge Anthony Rojas for providing the map of Quitasueño reef. This research was funded by the Convenio Interadministrativo 027 de 2011, Universidad Nacional de Colombia, Sede Caribe (UNC), Coralina and Gobernación Departamental de San Andrés, Providencia y Santa Catalina. Additional financial support was provided by UNC through the projects Hermes # 12617 and # 12386. Financial support to MACO was provided through the program of Movilidad Intersedes de la Sede Caribe, financed through the project Quipú No. 20101003960.
LITERATURE CITED
1 Abril, A. and L. Arango. 2012. Evaluacion de la comunidad coralina de Serrana, Quitasueño y Roncador, zona norte del AMP. pp. 39-49. In: Gavio, B. (Ed.). Informe final Convenio Interadministrativo No. 027 de 2011, celebrado entre el Departamento Archipiélago de San Andrés, Providencia y Santa Catalina, la Corporación para el Desarrollo Sostenible del Archipiélago de San Andrés, Providencia y Santa Catalina -CORALINA-, y la Universidad Nacional de Colombia sede Caribe, San Andrés. 59 p. [ Links ]
2 Albis-Salas, M. and B. Gavio. 2011. Notes on marine algae of the International Biosphere Reserve Seaflower, Caribbean Colombian I: New records of macroalgal epiphytes on the seagrass Thalassia testudinum. Bot. Mar., 54: 537-543. [ Links ]
3 Bolaño, N. and A. Acosta. 2013. ¿Qué perdió Colombia con el fallo de La Haya? Rev. Javeriana, 792: 56-62. [ Links ]
4 Børgesen, F. 1919. The marine algae of the Danish West Indies. Part 3. Rhodophyceae (5). Dansk Botanisk Arkiv, 3: 305-368. [ Links ]
5 Charpy, L., B.E. Casareto, M.J. Langlade and Y. Suzuki. 2012. Cyanobacteria in coral reef ecosystems: a review. J. Mar. Biol., 2012 Article ID 259571, 9 p. doi:10.1155/2012/259571. [ Links ]
6 Dawes, C.J. and A.C. Mathieson. 2008. The seaweeds of Florida. University Press of Florida, Gainesville, USA. 656 p. [ Links ]
7 Díaz, J.M., G. Díaz-Pulido, J. Garzón-Ferreira, J. Geister, J.A. Sánchez and S. Zea. 1996. Atlas de los arrecifes coralinos del Caribe colombiano. I. Complejos arrecifales oceánicos. Serie Publicaciones Especiales Invemar, 2. 83 p. [ Links ]
8 Díaz-Pulido, G. and G. Bula-Meyer. 1997. Marine algae from oceanic atolls in the southwestern Caribbean (Albuquerque Cays, Courtown Cays, Serrana Bank and Roncador Bank). Atoll Res. Bull., 448: 1-18. [ Links ]
9 Engene, N., E.C. Rottacker, J. Kastovsky, T. Byrum, H. Choi, M.H. Ellisman, J. Komárek and W.H. Gerwick. 2012. Moorea producens gen. nov., sp. nov. and Moorea bouilonii comb. nov., tropical marine cyanobacteria rich in bioactive secondary metabolites. Internatl. J. System. Evol. Microbiol., 62: 1171-1178. [ Links ]
10 Fernández-García, C., R. Riosmena-Rodríguez, B. Wysor, O.L. Tejada and J. Cortés. 2011. Checklist of the Pacific marine macroalgae of Central America. Bot. Mar., 54: 53-73. [ Links ]
11 Friedlander, A., J. Sladek Nowlis, J.A. Sánchez, R. Appeldoorn, P. Usseglio, C. McCormick, S. Bejarano and A. Mitchell-Chui. 2003. Designing effective marine protected areas in Seaflower Biosphere Reserve, Colombia, based on biological and sociological information. Conserv. Biol., 17: 1769-1784. [ Links ]
12 Gavio, B., V.P. Reyes-Gómez and M.J. Wynne. 2013. Crouania pumila sp. nov. (Callithamniaceae: Rhodophyta), a new species of marine red algae from the Seaflower International Biosphere Reserve, Caribbean Colombia. Rev. Biol. Trop., 61: 1015-1023. [ Links ]
13 Komárek, J. and K. Anagnostidis. 2005. Süsswasserflora von Mitteleuropa. Cyanoproparyota: 2. Teil/2nd Part: Oscillatoriales. Vol. 19. Elsevier Spektrum Akademischer Verlag, Munich. 759 p. [ Links ]
14 Lipkin, Y. and P.C. Silva. 2002. Marine algae and seagrasses of the Dahlak Archipelago, southern Red Sea. Nova Hedw., 75: 1-90. [ Links ]
15 Littler, D.S. and M.M. Littler. 2000. Caribbean reef plants. OffShore Graphics, Washington D.C. 542 p. [ Links ]
16 Ortiz, J.F. and B. Gavio. 2012. Notes on the marine algae of the International Biosphere Reserve Seaflower, Caribbean Colombia II: Diversity of drift algae in San Andres Island, Caribbean Colombia. Caribb. J. Sci., 46: 313-321. [ Links ]
17 Reyes-Gómez, V., B. Gavio and H. Velásquez. 2013. Notes on the marine algae of the international biosphere reserve Seaflower, Caribbean Colombia III. New records of Cyanophyta. Nova Hedw., 97: 349-360. [ Links ]
18 Rull Lluch, J. 2002. Marine benthic algae of Namibia. Scientia Mar., 66(Suppl. 3): 5-256. [ Links ]
19 Sánchez, J.A., V. Pizarro, A.R. Acosta-de Sánchez, P.A. Castillo, P. Herrón, J.C. Martínez, P. Montoya and C. Orozco. 2005. Evaluating coral reef benthic communities in remote Caribbean atolls (Quitasueño, Serrana, and Roncador banks) to recommend marine-protected areas for the Seaflower Biosphere Reserve. Atoll Res. Bull., 531: 1-66. [ Links ]
20 Schneider, C.W. and R.B. Searles. 1991. Seaweeds of the southeastern United States, Cape Hatteras to Cape Canaveral. Duke University Press, Durham and London. IV + 553 p. [ Links ]
21 Titlyanov, E.A., I.M. Yakovleva and T.V. Titlyanova. 2007. Interaction between benthic algae (Lyngbya bouillonii, Dictyota dichotoma) and scleractinian coral Porites lutea in direct contact. J. Exper. Mar. Biol. Ecol., 342: 282-291. [ Links ]
22 Wynne, M.J. 2013. (2175) Proposal to conserve the name Moorea Engene and al. (Cyanophyeae). Taxon, 62: 830. [ Links ]
FECHA DE RECEPCIÓN: 01/08/2013 FECHA DE ACEPTACIÓN: 21/04/2015