The lionfish Pterois volitans (Linnaeus) is a scorpionfish native to the Indo-Pacific that was introduced in the Western Atlantic (WA) more than three decades ago. The first known capture of the species in the WA took place in Florida, USA, in 1985, and in the following decade additional records from locations in the USA and Bermuda were known (Schofield, 2009). By 2010 the lionfish was already considered an exotic invasive species across most of the Greater Caribbean, including Colombia (González et al., 2009; Schofield, 2010). Showing a remarkable dispersal capacity, in less than 30 years the invader made presence in most of the WA, from North Carolina on the east coast of USA, Gulf of Mexico, and the Caribbean (USGS-NAS, 2020). With an occupied area of almost 7.3 million km2 (Côté et al., 2013), lionfish invasion is now recognized as the fastest invasion by a marine fish in history (Hamner et al., 2007; Schofield, 2010; Betancur-R et al, 2011). There is even a report of a lionfish caught in 2014 as far south as Rio de Janeiro, Brazil (Ferreira et al., 2015).
The lionfish has a set of biological characteristics that determine its success as an invader and turn it on a serious threat to the WA native biota: a rapid growth (Pusack et al., 2016) and high reproductive output (Gardner et al.,, 2015; Fogg et al., 2017), as well as generalist traits in habitat and diet (Muñoz et al., 2011; Claydon et al., 2012; Cure et al., 2014; Acero et al., 2019); in addition to a series of physiological, morphological and behavioral characteristics make it a mighty hunter and a difficult prey (Morris, 2013; Díaz-Ferguson and Hunter, 2019; Green et al., 2019; Rojas et al., 2019). The apparent everywhereness and abundance of the species in the invaded area synergistically with its character as a voracious and opportunistic predator, give it a great potential to catastrophically affect the native fish communities (Côté et al., 2013; Ballew et al., 2016; Ingeman, 2016). Coral reefs fish populations are particularly in risk of reduction by lionfish, putting local fishermen's economy in peril (Albins and Hixon, 2008; Green et al., 2012) and even triggering cascading effects on the benthic community's structure (Lesser and Slattery, 2011).
The first recorded sighting of lionfish in Colombia dates of December 2008 at the Manta City dive site, on the island of Providencia, at 5 m depth (Schofield, 2009). Five months later the species was recorded in the Tayrona National Natural Park (NNP) at 16 m depth (González et al., 2009), and since then a lot of records have been received in the INVEMAR database for the entire Colombian Caribbean (http://invasoresmarinos.invemar.org.co/). Since the first record, research on the invasion has been carried out in the country in several topics, from the characterization of the species (Hernández-Abello et al., 2015; González et al., 2011) to its trophic ecology (e.g.Muñoz-Escobar and Gil-Agudelo, 2012; Pabón and Acero, 2015; Acero et al., 2019). Some studies have estimated lionfish abundance on specific locations, such as Tayrona NNP and Santa Marta (Bayraktarov et al., 2014; Coronado-Carrascal et al., 2015; García-Urueña et al., 2015), and San Andrés island (González-Corredor et al., 2016); but until date, just one published work exists in which distant locations were evaluated (Rojas-Vélez et al., 2019). So far, there are no reports of this species abundance with standardized methodologies and in a number of locations as to make comparisons on a broader spatial scale. In that sense, this work offers information on the distribution and abundance of the lionfish on a regional level in Colombia, contributing to the information pool on this biological invasion in the country.
Between August-October 2015 and May-September 2016, the six localities with the most significant extension of coral formations throughout the Colombian continental Caribbean were visited to verify the presence and estimate the abundance of lionfish (Figure 1; Table 1). Lionfish census were made between 7-17 h following the Green (2013) methodology, widely used in the WA. Through SCUBA diving, an intensive search of lionfish in transects of 25 x 10 m was carried out, taking care of searching under the overhangs and among reef cavities and cracks.
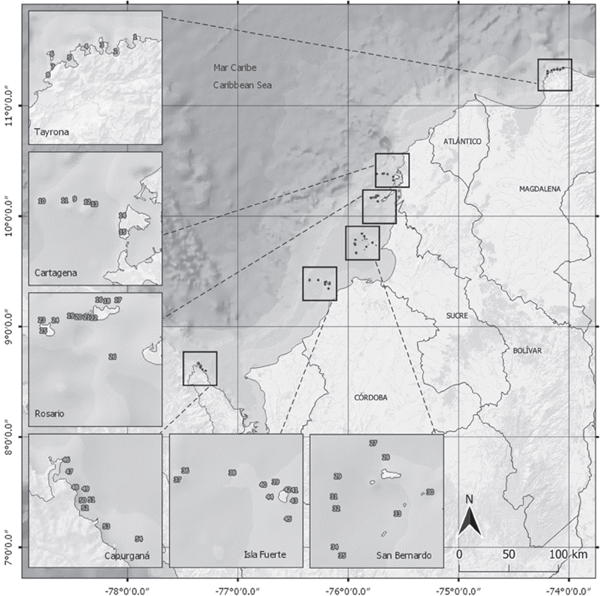
Figure 1 Sampling sites in six localities from the Colombian Caribbean coast where lionfish abundance was evaluated in 2015 and 2016. The names of the sites corresponding to each number in the localities boxes are in Table 1.
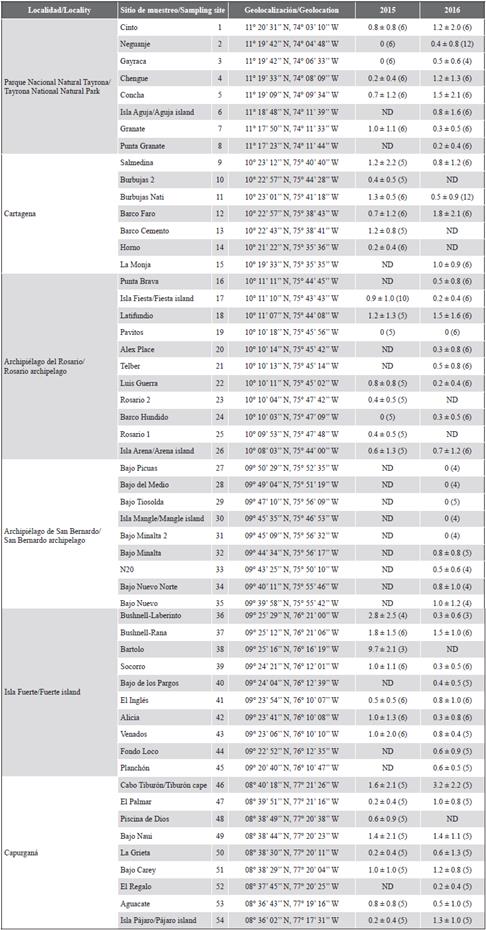
Table 1 Lionfish abundance in the Colombian Caribbean coast in 2015 and 2016. Average density (individuals/250 m2) ± 1 standard deviation with the number of censuses per sampling site in parentheses. ND means the site was not evaluated that year.
Density values were estimated in individuals per 250 m2, averaged by site and locality and extrapolated to hectares for comparison with other studies. The number of census vary between sites (3 to 12) (Table 1) because time to evaluate each transect changed according to physical traits of the sampling sites (e.g. greater/lesser structural complexity, depth, visibility); as well as with diving time in line to decompression limits of scuba diving with air. To assess for differences in density among years, between locations, and related to depth, the statistical test of Mood for the medians was used. Previously, the normality and homoscedasticity of the data were analyzed with the Shapiro-Wilk and Bartlett tests. For the analyzes depth were ranked as follows: 1: 1-10 m, 2: > 10-20 m, 3: > 20-30 m, 4: > 30-40 m. All analyzes were done with Statgraphics Centurion XVI.
A total of 452 censuses were conducted in 54 sites in the six localities during the two years (191 in 2015; 261 in 2016), with a sampled area of 113,000 m2 between 4.5 and 44 m deep. The search in each transect lasted 17.3 min on average for a whole sampling time of around 130 h, during which 350 lionfishes were observed in 185 transects from 48 sites. From a total of 85 dives, the invader was observed in 75 for a sighting frequency of 88 %, which allows considering the species as frequent in the locations and years of sampling (> 70 %) according to the standards of Schmitt and Sullivan (1996). The sites where the species was not registered during the censuses were Pavitos in the Rosario archipelago, which was visited both years (n = 11); and Bajo Picuas (n = 4), Bajo del Medio (n = 4), Bajo Tiosolda (n = 4), Mangle island (n = 4) and Bajo Minalta 2 (n = 4), which are in the archipelago of San Bernardo and which were visited only in 2016 (Table 1).
Density values between 0 to 12 ind/250 m2 (480 ind/ha) were recorded during the study, with a general average of 0.8 ± 1.3 ind/250 m2 (32 ind/ha). In 2015 the average density was 0.9 ± 1.6, and in 2016 it was 0.7 ± 1.1 ind/250 m2. The highest average density per site was 9.7 ± 2.1 ind/250 m2 (388 ind/ha) and was registered in Bartolo (Fuerte island) in October 2015. The locality with the highest average density in the two years was Fuerte island with 1.2 ± 2.8 ind/250 m2 (48 ind/ha), followed by Capurganá and Cartagena with 0.9 ± 1.3 ind/250 m2 and 0.9 ± 1.2 ind/250 m2 respectively. When extrapolated to hectares the highest average density per site is similar to the maximum density of lionfish reported in Colombia by González-Corredor et al. (2016), that is of 379.03 ind/ha during 2012 in San Andrés island. Both, San Andrés and Bajo Bartolo’s lionfish density values are near to some of the highest densities reported for the species on the invaded area by studies that used similar methods (Table 2).
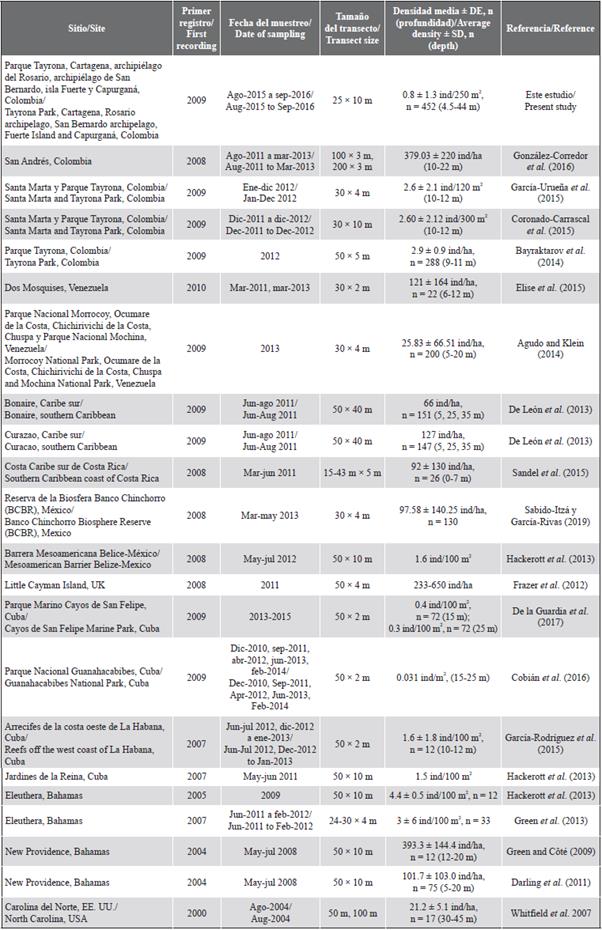
Table 2 Lionfish densities estimated through visual censuses in studies carried out in several places of the Western Atlantic. SD: Standard deviation; n: number of transects.
Bajo Bartolo is a seafloor elevation located about 10 km northwest of Fuerte island, which shallow part is around 35 m deep, making the site unsuitable for spearfishing or recreational diving so that lionfish removal has been unusual or absent before this work. Other places with significant densities of lionfish were Bushnell-Laberinto (2.8 ± 2.5 ind/250 m2), also on Fuerte island, and Tiburón cape in the locality of Capurganá (3.2 ± 2.2 ind/250 m2). In general terms, these are remote sites, where actions to control the invasion are infrequent or null due to logistical issues for diving and a limited presence of environmental authorities because they are not real protected areas. The lowest average density of lionfish was recorded in San Bernardo archipelago with just 13.7 ind/ha (0.3 ± 0.7 ind/250 m2), being even absent in five out of nine sampled sites at this locality (Table 1).
Assuming that the average density throughout sites in the two years (0.8 ± 1.3 ind/250 m2) (32 ind/ha) was representative of lionfish abundance on the Colombian continental Caribbean, then we can say that this value was low compared to densities reported in works with similar methods (i.e. UVC in transects) in other places in the Caribbean, such as in the Bahamas, Cayman islands, Cuba, Belize, Mexico, Costa Rica, Venezuela, and Curacao (Table 2). In Colombia, much higher densities have been recorded in local surveys, like that by González-Corredor et al. (2016) in San Andrés, and the study by García-Urueña et al. (2015) in Santa Marta (Table 2). However, when making comparisons on lionfish density among studies, a suite of factors must be considered, such as depth, date of sampling, the time elapsed since the arrival of the invader, transect size (length and width), and spatial scale of the survey, among others.
When considering only densities reported by studies including multiple locations at a regional scale (e.g. greater than 100 km), we found similar values to the average density reported here. For example, Agudo and Klein (2014) sampled five localities distributed over more than 400 km of the Venezuelan coast in 2013 and reported 25.83 ind/ha; Whitfield et al. (2007) scan sites along 160 km off the North Carolina coast in 2004 and reported densities of 21.2 ind/ha. Therefore, it seems like lionfish densities obtained by studies with several localities scattered in large areas can be more diagnostic of this biological invasion on a national scale. Furthermore, such studies allow direct comparisons to be made among places by the consistency in sampling methods, highlighting the importance of establishing standardized monitoring programs in the country.
Studies on a broad spatial scale evaluating lionfish abundance through underwater visual censuses in transects do not exist in Colombia. Recently, Rojas-Vélez et al. (2019) evaluating a biological resistance hypothesis counted P. volitans through roving visual censuses and reported densities equivalent to 84 ind/ha (0.84 ± 0.89 ind/100 m2). Differences in methods, sampling intensity, and sites preclude a direct comparison with our results; however, despite lower sampling effort (44 censuses in 24 sites versus 452 transects in 54 sites), their average density is three-fold than the one herein reported, which could be explained by methodological differences and their counts in San Andrés island, where highest lionfish densities in Colombia have been recorded (González-Corredor et al., 2016).
The only one location in Colombia with several publications on lionfish abundance estimated through visual censuses in transects is Tayrona NNP, and the variability in density values are remarkable, ranging from 2.9 ind/ha (Bayraktarov et al., 2014) up to 216.7 ind/ha (García-Urueña et al., 2015). The estimated abundances in this study for that locality (Table 1) are more similar to those in Coronado-Carrascal et al. (2015), who reported values equivalent to 68, 63.7, and 37 ind/ha for Cinto, Chengue, and Aguja island. It cannot be ruled out that all this variability in lionfish abundance estimates, even in one location, can be a reflection of highly dynamic populations due to variations in reproductive effort and recruitment success versus natural mortality, plus fishing mortality caused by differential control efforts in each locality. However, in the absence of results from a long-term monitoring with standard methods, it is easier to attribute that variability to differences in methods and sampling effort among the different studies. Hence the importance to establish a national monitoring program for lionfish invasion.
Lionfish densities at the majority of surveyed sites were similar between years; however, some extreme values were present, with a few sites showing densities much higher than average and others where the presence of lionfish was not recorded (Figure 2). The Mood median test showed that there were no significant differences in lionfish densities between the sampled years (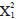 = 1.70; p = 0.19), neither among localities (
= 1.70; p = 0.19), neither among localities (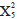 = 5.93; p = 0.31) and nor between sampling depths (
= 5.93; p = 0.31) and nor between sampling depths (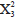 = 1.47; p = 0.68).
= 1.47; p = 0.68).
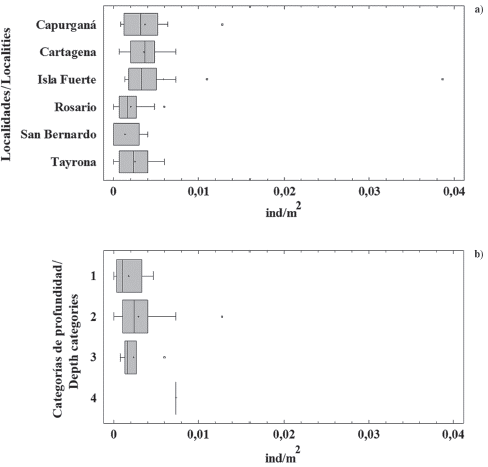
Figure 2 a) Lionfish density in six localities from the Colombian Caribbean coast: Capurganá (n = 77), Cartagena (n = 63), Fuerte island (n = 84), Rosario (n = 99), San Bernardo (n = 38), Tayrona (n = 88). b) Lionfish density at different depths: 1 (1-10 m, n = 25), 2 (> 10-20 m, n = 108), 3 (> 20-30 m, n = 34), 4 (> 30-40 m, n = 3). The line inside boxes represents the median, the limits of the boxes are the 25th and 75th percentiles, the whiskers mark minimum and maximum values, and points outside the boxes are outliers.
Variations in lionfish density between localities could be due to differences in the regime of control actions, mainly because lionfish derbies and frequent culling by diving schools are happening just in some areas. For example, the localities Rosario, San Bernardo, and Tayrona, are protected areas into the National System of Natural Parks and have control actions of the invasion since 2010 (e.g. Resolution MAVDT 0132 of 2010). Furthermore, due to the proximity of touristic cities to these areas, lionfish culling by fishers and divers are frequent (L. Chasqui pers. obs.). On the contrary, Capurganá and Fuerte island are remote places without protection status and where the presence of divers is scarce; therefore, control actions of the invader populations have been practically non existent. Different studies in the WA stand out the need of controlling lionfish numbers through constant culling and derbies, which are maybe the only actions available until date to face the biological invasion by P. volitans (Barbour et al., 2011; Frazer et al., 2012; De León et al., 2103; Green et al, 2017).
Variations in lionfish abundance (and size) have been previously documented in different locations on the invaded area, often increasing with depth (e.g.Claydon et al., 2012; González et al., 2016; Hunt et al, 2019). Among the explanations, a highest fishing pressure in shallow waters has been mentioned. However, fishing pressure decreases with depth until being virtually non-existent in environments outside the reach of recreational diving (e.g. mesophotic coral ecosystems - MCE). In this regard, Andradi-Brown et al. (2017) studying lionfish populations in a depth gradient found high densities over 30 m depth, as well as larger sizes and more fertile females, which may be undermining control efforts in shallow waters. They point out the need to understand and consider the importance of MCE in the life cycle of the species, and to seek strategies to extend control actions of lionfish invasion to those environments.
Multiple factors, acting alone or synergistically, can influence the distribution and abundance of lionfish, and can be determinant on the observed differences in density among sampling sites; for example, the structural complexity of the bottom, availability of shelters, the spatial connectivity of habitat patches, and prey abundance, among others (Biggs and Olden, 2011; Hernández-Abello et al, 2015; Rivera-Aponte et al., 2019; Díaz-Ferguson and Hunter, 2019).











 text in
text in 


