INTRODUCTION
Inappropriate use of medicine has given rise to pathogenic bacteria that are resistant to multiple drugs, which has become a threat to public health. It is estimated that this will be one of the main causes of death around the world by 2050 (WHO, 2014). Consequently, it is necessary to renovate the pharmaceutical arsenal against resistant pathogenic bacterial strains that are Gram-positive (e.g., Enterococcus sp., Mycobacterium tuberculosis (Zopf 1883) Lehmann and Neumann 1896, Staphylococcus aureus Rosenbach 1884) and Gram-negative (e.g., Acinetobacter baumannii Bouvet and Grimont 1986, Klebsiella pneumoniae (Schroeter 1886) Trevisan 1887, Pseudomonas aeruginosa (Schroeter 1872) Migula 1900) (WHO, 2014, 2015). In light of the above, marine organisms such as sponges have become an important source for obtaining biomolecules that allow designing new drugs (López et al., 2018).
Sponges (phylum Porifera) are aquatic, sessile, benthic, and filtering organisms. They are considered to be the oldest living animals, and they are estimated to have originated 760 million years ago (Brain et al., 2012). Their distribution is global; they inhabit freshwater, mixohyaline, and marine ecosystems, from the intertidal zone down to great depths (Hooper et al., 2002). They exhibit a simple level of organization, formed by cells of great motility and totipotency, which facilitates differentiation and transdifferentiation processes (Klautau, 2016). Most species are characterized by having a system of pores, channels, and choanocyte chambers (specialized filtering structures) known as aquiferous system, through which sponges feed and reproduce (with the exception of carnivorous sponges, Vacelet and Boury-Esnault, 1995). Moreover, they have a skeleton composed of spicules (mineral aggregations) made of silica or calcium carbonate and/or spongin organic fibers (Vos et al., 1991; Hooper et al., 2002).
These animals play an important role in aquatic ecosystems: they contribute to nutrient recycling (sponge loop, de Goeij et al., 2013), harbor many invertebrates (Pearse, 1932; Frith, 1976; Kunzmann, 1996), and form extensive aggregations known as sponge grounds of different types (Maldonado, 2016). On the other hand, they also stand out in the field of biotechnology, as studies have been conducted on their use as bioindicators of water quality, given their well-known filtering and bioaccumulation conditions, (Mahaut et al., 2013), the use of their spicules to elaborate nanomaterials (Kulchin et al., 2009), and the potential of their regeneration capacity for tissue engineering (Granito et al., 2016). In the field of blue biotechnology, they constitute the group of marine invertebrates that provides the largest number of natural compounds with pharmaceutical potential (Pierdacaris et al., 2013; Brinkmann et al., 2017; Santhanam et al., 2019), as more than 5,000 bioactive marine compounds have been isolated, out of which around 30 % come from sponges (Han et al., 2019). Natural compounds with antibacterial activity can be synthesized by sponges themselves or from symbiosis with other organisms (bacteria, yeasts, fungi) (Marinho et al., 2010; Anjum et al., 2016; McCauley et al., 2020).
In Peru, biotechnology is a unique opportunity to increase competitiveness and contribute to socioeconomic development. This approach is based on the search and conquest of new markets through the sustainable use of marine biodiversity. However, the problem lies in the fact that current biotechnology applications do not transcend academic efforts or remain as small-scale actions and/or initiatives, despite utilizing advanced technologies. Moreover, (Peruvian) companies still have technological and economic limitations regarding the application of biotechnology in the improvement of their products or processes, whose industrial-scale implementation is seen to be far away (Concytec, 2016).
In this context, the aim of this work was to provide an update on the state of knowledge on sponges in Peru and the perspectives of their antibacterial use, addressing three main axes: (1) scientific research, (2) species richness, and (3) antimicrobial potential. For the first axis, research projects and/or initiatives were compiled from repositories belonging to research centers, universities, blogs, and press releases through virtual search engines. Moreover, project leaders were consulted in order to understand the achievements made with regard to the training of human resources and scientific production. For the second axis, each of the records of sponge species (both marine and freshwater) registered for Peru in the World Porifera Database (http://www.marinespecies.org/porifera/) was reviewed, and their geographical and bathymetric distributions were verified. Considering the genera of sponges recorded for Peru, a list of congeneric species proven to have antimicrobial activity in other regions in the world was elaborated. To this effect, a search for scientific articles published in indexed journals was conducted in databases such as Scopus, JStor, ScienceDirect, SciELO, and Wiley Online Library, using the keywords “antibacterial activity”, “sponge”, and “Porifera” (in both English and Spanish), as well as the genera reported for Peru (in Latin). Finally, a brief comparative analysis of the state of knowledge and potential of sponges in Peru as a source of antimicrobial products is presented in relation to other countries in the region.
Research on the phylum Porifera in Peru
The study of sponges in Peru began with the record of the first species of the class Demospongiae, Dysidea ligneana (Hyatt, 1877) for the northern part of the country (Tumbes, Hyatt, 1877). Some years later, the first specimens of the class Hexactinellida were collected during the scientific expedition onboard the Albatross cruise (1904-1905) to the Tropical Pacific, which was led by Alexander Agassiz. The samples were obtained by throwing grabs to depths of 4,000 m, and they were identified by Austrian zoologist Robert von Lendenfeld (1915) from poorly preserved materials due to the collection methodology employed. Decades later, as a result of the review of the material registered in the Collection of the National Museum of Natural History (Smithsonian Institution, USNM), other Demospongiae species were identified (van Soest et al., 1991; Desqueyroux-Faúndez and van Soest, 1996). Van Soest et al. (1991) conducted a detailed biogeographical review of the genus Acarnus and described the first species of this genus for Peru, which was collected from an intertidal zone. In turn, Desqueyroux-Faúndez and van Soest (1996) described the first two Myxilla species, collected during the Programa Oceanográfico Biológico del Pacífico Este (East Pacific Oceanographical Biological Program, Sepbop) onboard the R.V. Anton Bruun in 1966.
In the last decades, initiatives have been carried out with the purpose to determine the richness of sponges in South America. The EsponjAS project, led by the Brazilian specialist Eduardo Hajdu (Museu Nacional-Universidade Federal do Rio de Janeiro, UFRJ), in cooperation with the Belgian biologist Philippe Willenz (Royal Belgium Institute of Natural Sciences, RBNSc), aimed to make an inventory of the sponge fauna in poorly explored areas of South America, with a particular interest in the coasts of Argentina and Chile. In 2008, this project expanded its study area to Peru. Almost in parallel, in 2007, the Peruvian-Belgian cooperation project ESPER (Esponjas del Perú) began, which was led by the biologist Yuri Hooker (Universidad Peruana Cayetano Heredia) and Philippe Willenz (RBNSc) and funded by the Global Biodiversity Information Facility (GBIF), with the purpose of identifying the sponges of Peru (Aguirre et al., 2011).
Within the framework of these projects (2007-2009), more than 800 sponge samples were collected along the Peruvian coast (ca. 90 localities) by manual collection in the intertidal zone and semi-autonomous diving in the subtidal zone (Aguirre et al., 2011; Azevedo et al., 2015). These projects included the participation of students from different universities and professionals related to the field of biology, who were trained in sponge collection and preservation techniques. In addition, some students did internships in the UFRJ, which were funded with grants provided by the National Council of Science, Technology, and Technological Innovation (Consejo Nacional de Ciencia, Tecnología e Innovación Tecnológica, Concytec) and the EsponjAS Project. As a result of these initiatives, 30 species have been identified and described, integrating morphological approaches that include spicule characterization aided by electronic microscopy (Aguirre et al., 2011, Hajdu et al., 2015, Recinos et al., 2020; Bispo et al., 2022) and molecular tools such as DNA sequencing (Azevedo et al., 2015, Cóndor-Luján et al., 2019). Identifying the collected material will be useful for the elaboration of a identification guide of sponges of Peru, which will be published later (Ph. Willenz, pers. comm. 01/2022).
Multiple efforts have been made in Peru to understand the ecological aspects of sponges, among which the determination of their associated macrofauna (Cóndor et al., 2010) and urchin-sponge interaction (Aguirre et al., 2011) can be highlighted. On the other hand, the research group of the Microbial Ecology Laboratory of Universidad Nacional Mayor de San Marcos (UNMSM), led by the microbiologist Jorge León, has conducted studies to determine the biological activity of actinomycetes associated with marine sponges and has obtained promising results (see León et al., 2015; Lino et al., 2016).
In 2019, Universidad Científica del Sur (Científica) continued its self-funded research on Porifera in Peru, starting with the determination of the sponges recorded in its Scientific Collection, within the framework of the project Evaluación de la diversidad de Demospongiae (Phylum Porifera) de las costas central y sur del Perú (Evaluation of the diversity of Demospongiae (Phylum Porifera) in the central and southern coasts of Peru), which involved Peruvian professors and students as well as international specialist researchers (Eduardo Hajdu and Philippe Willenz). In late 2019, Científica began to execute the subproject Bioprospección de esponjas marinas de las costas central y sur del Perú para obtención de principios activos (Bioprospection of marine sponges in the central and southern coasts of Peru to obtain active principles), with the purpose of understanding the richness and distribution of sponges with microbial potential in Peru’s central-southern coast. This subproject was co-funded by the World Bank through the National Program for Innovation in Fisheries and Aquiculture (PNIPA) of the Ministry of Production (Produce), as well as in collaboration with two social organizations of artisan fishermen (OSPAS): Cooperativa de Trabajadores Pesqueros Artesanales Algas Marinas (Cotrapalmar) and Asociación de Maricultores El Arca de Noé de Marcona.
During the last 15 years, nine research projects funded by various sources have been developed with Peruvian sponges. Among these, five correspond to Peruvian initiatives with an interest in identifying species and their biotechnological potential (Table S1). As a result of these initiatives, nine papers have been published in scientific journals, and ca. 26 presentations have been given at scientific events (Table S2), including Peruvian students (e.g., Aguirre et al., 2011; Azevedo et al, 2015; Lino et al., 2016; Arroyo et al., 2020).
Sponge richness in Peru
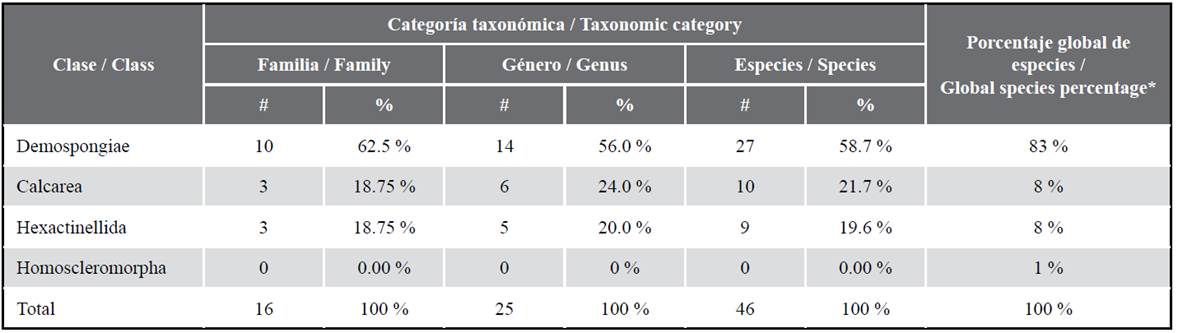
More than 9,000 sponge species are currently recognized, which are organized into four classes: Calcarea (8 %), Demospongiae (83 %), Hexactinellida (8 %), and Homoscleromorpha (1 %) (van Soest et al., 2012). Among them, only Calcarea includes species with a skeleton composed exclusively of calcium carbonate spicules, e.g., diactines, triactines, tetractines, and pentactines (Manuel et al., 2002; Rossi et al., 2006) and has the five types of aquiferous systems known for Porifera: asconoid, leuconoid, syconoid, sylleibid, and solenoid (Cavalcanti and Klautau, 2011). Unlike Calcarea, Demospongiae sponges have a diversity of colors and are adapted to freshwater environments, including some high Andean lagoons in Perú (Boury-Esnault and Volkmer-Ribeiro, 1991). Hexactinellida includes sponges characterized by having cells that form syncytial tissue, and most of them, present a distribution restricted to deep waters (200-6,000 m). Only Homoscleromorpha have basal membrane, flagellate pinacocytes, and silica spicules with tetraxonic symmetry (Gazave et al., 2012). Moreover, just as Calcarea, Homoscleromorpha sponges often inhabit places that are protected from light such as cavities in vertical walls or under rocks (Pérez et al., 2017). Furthermore, the class Archaecyatha is also recognized, which includes extinct species with an inverted cone shape and a calcium carbonate basal skeleton. These species formed reefs during the Cambrian period (Debrenne et al., 2002).
For Peru, 46 sponge species have been reported which are distributed in three out of the five classes known for this phylum (Table 1). Class Demospongiae leads the list with 10 families, 14 genera, and 27 species. To date, the genus Haliclona of the family Chalinidae (order Haplosclerida) harbors the greatest number of species (n = 10; Bispo et al., 2022). The genera Chalinula, Eurypon, Hamacantha, and Myxila include two species, while the remaining nine comprise only one species (Table S3). These sponges have been found in different habitats and at various depths. Among them, 21 have been found associated with hard substrate, at depths between 0 and 40 m; two of them were collected from sandy and muddy substrates more than 200 m depth (Desqueyroux-Faúndez and van Soest, 1996); and one of them belongs to the Titicaca Lake (3,812 masl, Boury-Esnault and Volkmer-Ribeiro, 1991). Recently, two species have been found on the roots of the mangroves in Tumbes (Bispo et al., 2022). Regarding their geographical distribution, 23 species have been recorded only for Peru, and the remaining four are related to neighboring localities. Acarnus peruanus van Soest, Hooper & Hiemstra, 1991 has been also found in the Galapagos Islands (Desqueyroux-Faúndez and van Soest, 1997) and in the Mexican Pacific (Aguilar-Camacho et al., 2013). Hamacantha (Zygherpe) hyaloderma (Laubenfels, 1932) and Johannesia reticulosa (Thiele, 1905) were previously described for California (de Laubenfels, 1932) and Chile (Thiele, 1905), respectively. Balliviaspongia wirrmanni Boury-Esnault and Volkmer-Ribeiro, 1991 is shared with Bolivia (Titicaca Lake).
As for the class Calcarea, three families, six genera, and 10 species have been recorded (Table S3), located at depths between the intertidal zone and 30 m, as well as in habitats protected from light (Azevedo et al., 2015; Cóndor-Luján et al., 2019). Seven species are preliminarily considered to be endemic to Peru, and, among these, Arturia spiralatta and Clathrina peruana show the greatest distribution. Clathrina aurea and Ernstia tetractina were first described for the Brazilian southeastern coast (Solé-Cava et al., 1991) and currently have an amphi-american distribution (Azevedo et al., 2015), including the Caribbean Sea for C. aurea (Fontana et al., 2018; Cóndor-Luján et al., 2021). Clathrina antofagastensis reports the greatest distribution in the Pacific coast, from the Mejillones Peninsula (Chile, Azevedo et al., 2009) to Sechura Bay (Perú, Azevedo et al., 2015).
The class Hexactinellida includes four families, five genera, and nine species (Table 1). The species were collected in localities far from the coast at depths greater than 4,000 m during the Albatross expedition (1904-1905). Four species were found only once during this expedition, and the remaining five have a greater distribution (von Lendenfeld, 1915; Lévi, 1964; Koltun, 1970; Kersken et al., 2018; O’Hara et al., 2020).
Antibacterial potential of sponges registered for Peru
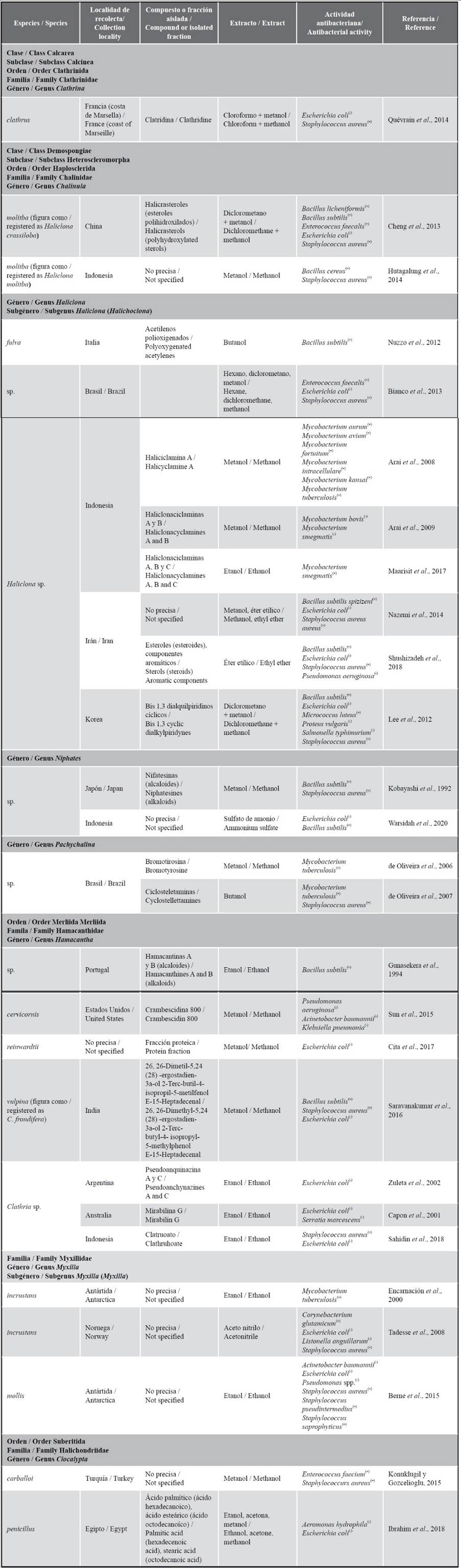
To date, the antimicrobial potential of sponges in Peru has not been yet object of study. However, considering the 25 genera reported for the country, 11 of these harbor at least 36 species that have shown antimicrobial activity. From the organic extracts of these species, collected in the Antarctic, Northern Atlantic (France, Ireland), Western Pacific (Indonesia), and other localities, several active principles have been isolated (Table 2).
Haliclona is the genus with the greatest number of studies on proven antibacterial activity (n = 13). Its effect against 20 Gram-positive and 20 Gram-negative bacteria has been demonstrated, which is attributed to the presence of sterols and alkaloids (Cheng et al., 2013; Viegelmann et al., 2014; Shushizadeh et al., 2018; Kaplan et al., 2021). Morevoer, some secondary metabolites have been obtained, such as Halicyclamine A (Arai et al., 2008), Haliclonacyclamines A and B (Arai et al., 2009), and Haliclonacyclamines A-C (Maarisit et al., 2017). Clathria is the second most studied genus, with 11 species recorded which show inhibitory action against five Gram-positive and eight Gram-negative bacteria. A significant number of compounds has been isolated, among which the following stand out: Mirabilin G (Capon et al., 2001), Pseudoanchynazines A and C (Zuleta et al., 2002), Microcionamides A and B (Davis et al., 2004), Araiosamines A and D (Wei et al., 2011), Clathrimides A and B (Gupta et al., 2012), Gombaspiroketals A-C, Phorone, Ansellone C, Saponin gombaside A (Woo et al., 2015), Crambescidin 800 (Sun et al., 2015), and Clathruhoate (Sahidin et al., 2018). In Dysidea, four species are recognized which have bactericidal action against 10 bacteria (Gram positive and negative), and, among them, two produce bromophenols against Escherichia coli (Migula 1895) Castellani and Chalmers 1919, Salmonella sp., and Staphylococcus aureus (Sun et al., 2015). Two Ciocalypta and Myxilla species have evidenced antibacterial activity, highlighting that of C. penicillus Bowerbank, 1862 against Aeromonas hydrophila (Chester 1901) Stanier 1943 (Ibrahim et al., 2018) and that of M. (Myxilla) incrustans (Johnston, 1842) against Mycobacterium tuberculosis (Encarnación et al., 2000), Listonella anguillarum corrig. (Bergman 1909) MacDonell and Colwell 1986 (Tadesse et al., 2008), S. pseudintermedius Devrieseet al. 2005, and S. saprophyticus (Fairbrother 1940) Shaw et al. 1951 (Berne et al., 2015). Niphates contains a group of pyridine alkaloids named Niphatesines, which act against Bacillus subtilis (Ehrenberg 1835) Cohn 1872 and S. aureus (Kobayashi et al.,1992). Furthermore, its extract inhibits the growth of E. coli and B. subtilis (Warsidah et al., 2020). Compounds such as Hamacanthines A and B and Acarnidines have been isolated, with effects against B. subtilis, from Hamacantha and Acarnus species, respectively (Gunasekera et al., 1994); and Bromotyrosine and Cyclostellettamines, with action against M. tuberculosis and S. aureus, from Pachychalina (de Oliveira et al., 2006, 2007). As for the class Calcarea, Clathrina is highlighted, with antibacterial activity against E. coli and S. aureus (Quévrain et al., 2014).
These results suggest the potential of the Peruvian congeneric species as a source of biocompounds or antibacterial metabolites. In the same way, it is likely that Peruvian species that are currently being described (Table 2) evidence an applicability of pharmaceutical interest, highlighting antimicrobial activity.
Table 2 Sponge species with proven antimicrobial activity from genera reported for Peru, with details (if possible) on the collection locality, the compound or isolated fraction, the composition of the extract, and the antibacterial activity. Gram-positive bacterium =(+), Gram-negative bacterium =(-).
In this context, the biotechnological potential of Peru is evidenced, where opportunities for research, innovation, and business for the fishing, aquaculture, and pharmaceutical industries can be identified. Nevertheless, the development of bio-businesses focused on the search for bio-products derived from aquatic species must involve technological plans accompanied by the development of capabilities and installed infrastructure, networking, and national and international agreements celebrated by the institutions and companies involved, which guide the utilization and valorization of resources, such as the marine sponges, under a sustainable perspective that protects biodiversity and contributes to the country’s socioeconomic development.
Research on Porifera in the South American Pacific
As in Peru, research on Porifera in other countries of the South American Pacific began with the descriptions of the specimens obtained in oceanographic campaigns e.g. H.M.S. Challenger (Ridley, 1881; Ridley and Dendy, 1887; Wilson, 1904; von Lendenfeld, 1910). However, no major studies were carried out until the 80s, when the researcher Ruth Desqueyroux thoroughly analyzed the material from Chile and Ecuador.
In Chile, studies have been predominantly oriented towards the knowledge of the diversity (richness and distribution) of species from the fjords (Thiele, 1905; Hajdu et al., 2013; Fernández et al., 2021). Currently, 190 species are known, including the classes Calcarea (n = 15), Demospongiae (n = 160), and Hexactinellida (n = 15). Among these, 140 have been registered for the fjords (Bertolino et al., 2020; Fernández et al., 2021). As a complement, research has been conducted on the biogeography, the ecology (macroalgae-sponge interaction), and the biotechnological potential of these organisms (e.g., Desqueyroux and Moyano, 1987; Hajdu and Desqueyroux-Faúndez, 2008; San Martin et al., 2011; Cárdenas et al., 2016).
In Ecuador, taxonomic efforts have been focused on the Galapagos Islands, with ca. 105 species described (van Soest et al., 2020; Sim-Smith et al., 2021), which were collected in both shallow and deep waters, while only four species have been registered for the Ecuadorian littoral (Jaramillo et al., 2021). On the other hand, only one study on the biotechnological potential of these animals is known (Calabro et al., 2018).
In recent years, the implementation of national and international projects in Chile and Ecuador have allowed not only the continuation of the research (collection of new samples and identification and description of new species), but also to contribute to the training of human resources in the region. As a result of these initiatives, scientific production (i.e., papers published in scientific journals, books or book chapters, and conference presentations) has increased, which is now led by Chile (Tables S4 and S5).
In the Colombian Pacific, there are few studies related to the identification and description of sponges, in comparison with those conducted in the Colombian Caribbean. Lizarazo et al. (2020a) consider the ecological contributions made by Narváez (1999) on four Demospongiae, an inventory that registers 21 species (Escobar, 2000), and the photoidentification guide by García-Suárez et al. (2012). Recently, a joint effort was made between the José Benito Vives de Andréis Institute for Marine and Coastal Research (Invemar) and the Institute for Studies on Sea Science (Cecimar), which managed to determine 24 sponge morphotypes and identify 12 species (Lizarazo et al., 2020b).
PERSPECTIVES
At a global level, sponges are a source of compounds with proven antimicrobial activity, as well as of active principles that have resulted in new medicines. However, in Peru, the development of research and innovation related to obtaining biomolecules and drugs from marine organisms not intended for direct or indirect human consumption is still incipient, as is the case of sponges. Despite this, Peruvian scientific production on sponges shows an increasing trend, mainly regarding species richness and distribution, conducted in academic university contexts and enriched by the existing international cooperation. The above demonstrates that there is a hydrobiological resource (sponges) in Peru with great biotechnological and economic potential, but that it requires the implementation of state and private programs that fund projects integrating research at different scales (taxonomic, ecological, and biotechnological), thus allowing the development of innovation and bio-business plans for the fishing, aquaculture, and pharmaceutical industries at national and international levels.











 texto en
texto en