1. Introduction
Solar heterogeneous photocatalysis, based on TiO2, has showed to be an effective treatment for the removal of many emerging contaminants from industrial wastewater 1,2. Pharmaceutical residues are considered as emerging pollutants and they are resistant to conventional biological treatments, so the solar heterogeneous photocatalysis has been used as an alternative to remove these compounds and their metabolites 3,4.
Pharmaceutical contamination has become a relevant topic, due to the damage that these compounds may cause to aquatic ecosystems. In some cases, it was observed that there was feminization of some fish species that have been exposed to estrogen concentrations lower than 10 ppm 5. Also, the appearance of drug-resistant bacterial strains, specifically to antibiotics, has been associated with residues that have appeared in water surfaces 6. Anthelmintic drug residues, have been found in chicken eggs 7, and may have antiangiogenic favorable for cancer treatment, but harmful to gestation stage because they inhibit the growth of blood vessels in fetus 8. Although, there are no public reports about health problems related to contamination of water surface bodies with pharmaceutical residues, some isolated cases whose origin may be the exposure or ingestion of pharmaceutical residues from drinking water has been reported 9.
Several studies have reported promising results about the performance of solar heterogeneous photocatalysis as an alternative treatment of these contaminants. Some drugs, such as naproxen or antihistamines, have been submitted to photocatalytic degradation using polycrystalline TiO2, with very acceptable results. Furthermore, the photocatalytic degradation of ibuprofen has been analyzed in previous studies 10,11. However, all these studies were carried out at lab scale and the applicability of solar heterogeneous photocatalysis of pharmaceutical compounds at larger scales depends on appropriate process modeling and simulation in order to determine the physical requirements of a full-scale facility.
One of the main challenges of photocatalysis, from a commercially applicable perspective, is the lack of kinetic models that allow scaling up photocatalytic reactors properly. The difficulty in studying this kind of systems is that the photonic component must be coupled with the reaction kinetics law. Some mathematical approaches have been proposed to solve this problem. The rigorous approach, with the numerical solution of the radiative transfer equation (RTE), was proposed as a very good approximation to the behavior of photoreactors 12. However, this approach requires considerable computing time and it has been validated under highly controlled experimental conditions. This becomes a very limiting aspect regarding to the model applicability in several types of photocatalytic reactors, especially those that use solar radiation for promoting redox reactions 13,1.
Li Puma et al.14 proposed a simplified approach for calculating the photonic component by the SFM and coupling it to a L-H kinetic law. The semi-empirical model fitted satisfactorily to experimental data from lab scale photoreactors with arjpgicial UV radiation. Colina-Marquez et al.15 adapted this approach for fitting to experimental data obtained with pilot-scale CPC solar reactors used for degrading a mixture of commercial pesticides. A more recent study developed by Mueses et al. (16 proposed a model that introduced the concept of the effective radiant field, which was successfully validated with experimental data reported in previous works, such as: dichloracetic acid, 17-β estradiol and 4-chlorophenol.
This study used the approach adapted by Colina-Marquez et al.15,17 for describing the kinetics of the photocatalytic degradation of commercial albendazole in a pilot scale solar CPC reactor. The results obtained in this study showed a good fitting to the experimental data, regarding to TOC removal of the commercial drug and its oxidation byproducts degradation. In addition, an optimal catalyst load (0.21 g/L) was obtained from simulating the solar photoreactor.
2. Materials and Methods
2.1 Materials
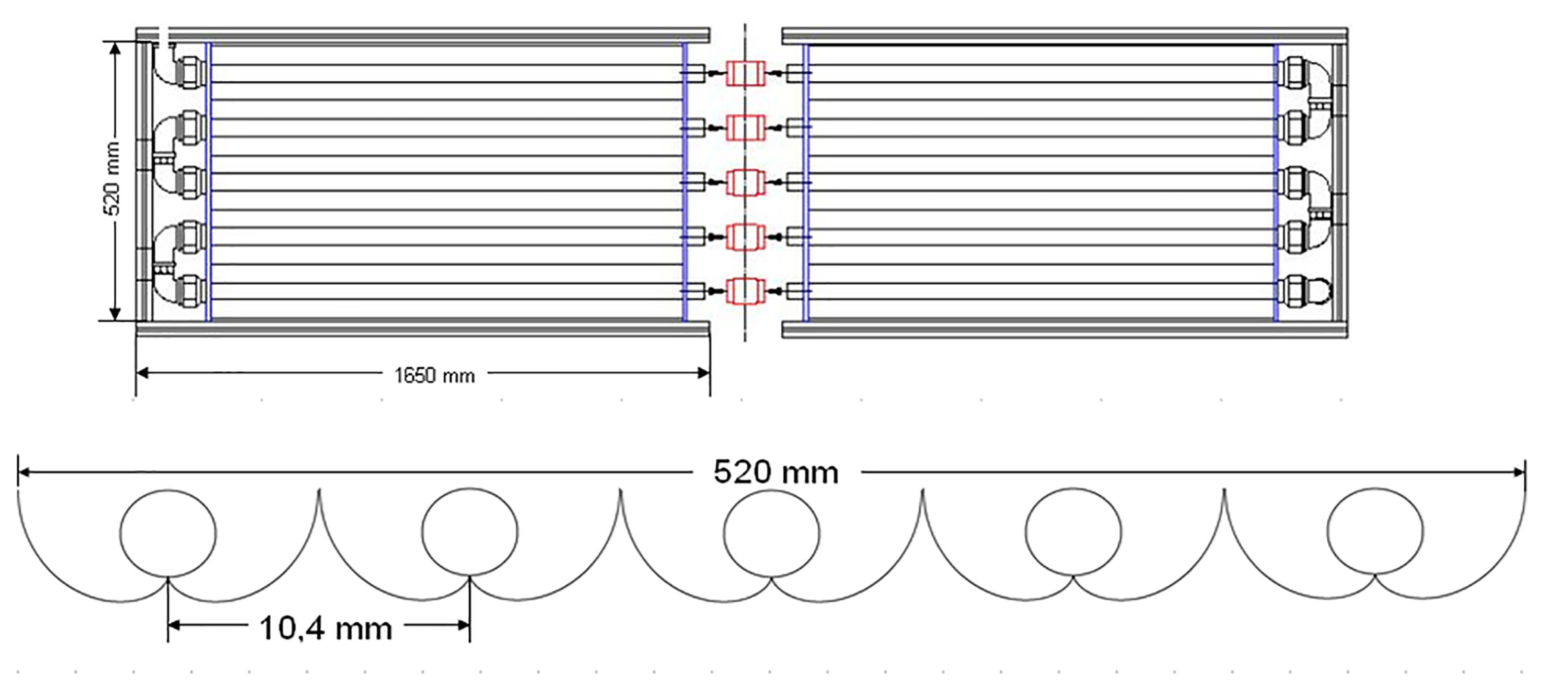
The pilot-scale CPC photoreactor used in this study is shown in Figure 1. It consisted of 10 glass tubes SCHOTT Duran ® of 32 mm of outside diameter, 1.4 mm of wall thickness and 1.27 m of length, organized in five rows onto compound parabolic collectors, made of aluminum of high reflectivity (85%) 18. This assembly was supported by a metal structure. The CPC reactor was located at the Laboratory for Solar Photocatalysis of the Universidad del Valle.
photoreactor: principal structure. (a) General Scheme, (b) Cross-section view 19
A 0.5 HP nominal power centrifugal pump delivered the recycling flow, from the feed tank and through the system, releasing a maximum flow-rate of 35 l·min-1 and assuring the turbulent regime. The flow-rate was kept at an average value of 30 l·min-1.
The solutions for pilot-scale tests were prepared with 20L of tap water and commercial preparation of albendazole (La Santé 400 mg/20 mL). The TOC initial concentrations used for this study were set at the following values: 159, 95, 75, 58 and 40 ppm, which correspond to the concentration range found in the wastewater of the Laboratory of Pharmaceutical Sciences in the Universidad de Cartagena.
The catalyst used in this study was the commercial TiO2 Evonik ® P-25 (particle size, 20-30 nm by TEM, surface area of 52 m2/g by BET and a composition of 78% anatase and 22% rutile by XRD). The catalyst loading used was 0.6 g/l and the initial pH was set at 5.0 by using HCl 0.1 N and NaOH 0.1 N solutions. Experimental tests were carried out in late September 2011, with sunny weather conditions. The intensity and the accumulated UV solar radiation (within the 295-380 nm interval) was measured with the radiometer UV Acadus S50.
The slurry with the catalyst was recirculated for five minutes before being exposed to solar radiation in order to guarantee the adsorption-desorption equilibrium. Then, the system was irradiated until the solar UV accumulated energy reached 35 W·h/m2. Samples were taken every 5 W·h/m2 and then, they were centrifuged. The clear liquid of the samples was analyzed with a Shimadzu TOC-VCPH analyzer for measuring the TOC content.
2.2. Photoreactor modeling for albendazole treatment
The mathematical approach used in this work includes combination of several sub-models such as: emission/incidence, absorption (SFM), kinetic and dynamic fluid models, as suggested by Colina-Marquez et al.20.
2.2.1. Six Flux Model
Six Flux Absorption Scattering Model allows analytical estimation of LVRPA in each point of the reactor, considering the photon is absorbed or scattered when it collides with a catalyst particle, following the route of one of the six directions of the Cartesian plane 21. This model contemplates uniform distribution of catalyst particles in suspension and invariant optical characteristics; negligible absorption of energy by fluid or reactor walls; solar UV radiation absorbed by the photocatalyst (TiO2) of wavelength between 295 and 384 nm (anatase band gap); constant reflectivity of the reflector surface (17,22.
Due to the average intensity of UV solar radiation varies with time of year, geographical location and atmospheric conditions. Photocatalytic treatment time was standardized based on a constant solar radiation intensity of 30 W/m2 (15, which corresponds to the average intensity achieved on a sunny day.
The most important optical parameter considered in the SFM is scattering albedo (ω) of the TiO2 suspension. This parameter depends on the wavelength of the energy spectrum and it has been reported for several types of TiO2 catalysts, including P-25 used in this study.
Equation 1 is the LVRPA estimated from the SFM in direction of the incident photon flux is
I 0 in the equation corresponds to the component of solar radiation reaching the reactor walls, (direct or diffuse radiation). ω 𝑐𝑜𝑟𝑟 is the corrected scattering albedo, λ ωcorr is the extinction length and r p is the distance of traveling of the incident photon flux.
Finally, LVRPA r,θ , at a specific location (r, θ) in the reaction space was the sum of the LVRPA estimated from each radiation component (direct and diffuse and reflected radiation by these two components) 15.
2.2.2. Reaction Kinetics
In most cases, the kinetic study concludes that degradation rate on the reaction depends on the concentration of pollutant adsorbed according to the Langmuir-Hinshelwood kinetics 21.
For heterogeneous photocatalytic system, the kinetic model based on the reaction mechanism and the absorption rate of radiant energy, is expressed as follows in the Equation 2:
Where k T, is the kinetic constant, and K is the binding constant associated to the adsorption of the drug onto the catalyst surface; m takes a value between 0.5 y 1 and depends on the efficiency of electron-hole formation and recombination at the catalyst’s surface 20.
2.2.3. Model fluid dynamics
Pilot-scale CPC photoreactors have operated TiO2 suspensions in turbulent flow regime (Re> 10000) 20,23. This avoids mass transfer limitations and catalyst sedimentation.
Some of the assumptions for the approach of the fluid dynamics within photocatalytic reactors are given below:
Liquid is considered as a Newtonian fluid with constant physical properties; steady state operation; catalyst particles are uniformly distributed in the liquid, constant physical and rheological properties of the suspension; the entrainment of bubbles is limited and the effect of velocity gradients in the particles radial distribution are not considered 21.
Literature describes the velocity profile of the fluid as indicated in the Equation 3 and Equation 4:
Where,
R is the reactor radius, 𝑣𝑧 𝑚𝑎𝑥 is the maximum fluid velocity into the reactor and n is a parameter which can be calculated as follows (Equation 5):
Where 𝑓 is the friction factor.
3. Discussion of results
3.1 Solar Radiation Parameters
Solar radiation parameters were taken from previous studies carried out under the same irradiation conditions and in the same reactor geometry.
3.2. Estimating LVRPA
Optical parameters were calculated from the optical properties of the catalyst, as well as the solar radiation spectrum within the reactor, and results are summarized on Table 1. These parameters allowed the determination of the reaction time at each point in photocatalytic reactor space, using a code in the Visual Basic program developed by Colina-Márquez 15 and modified it in this work to fit to the experimental conditions.
Table 1 Optical Parameters
| Parameters | Unit | Value |
|---|---|---|
| Specific mass absorption coefficient, κ | m2/kg | 174.74 |
| Specific mass scattering coefficient, σ | m2/kg | 1295.7 |
| Scattering albedo, ω | dimensionless | 0.88 |
| SFM parameter, a | dimensionless | 0.87 |
| SFM parameter, b | dimensionless | 0.66 |
| Scattering corrected albedo, ω corr | dimensionless | 0.75 |
| Extinction length, | m | 1.98×10-3 |
Results obtained, showed the importance of solar collectors, which concentrate solar radiation within the reactor, permitting a better photonic distribution and consequently, improving photonic availability in the reactor. Similar works have analyzed the presence of the collectors in the CPC solar reactors. Although it is evident that the presence of the reflective screen improves the usage of the UV photons 24-26, there is an interesting approach where the use of other distribution of the tubes (without collectors) can achieve better results than the CPC reactors with the same footprint 27. Inherently, more conclusive results are needed for getting a clearer idea about the need of solar collectors for photocatalytic applications.
3.3. Kinetic parameters
The initial velocity method, allowed finding kinetic parameters. The equation resulting from the combination of reaction rate law and the material balance in the reactor is Equation 6, written as follows
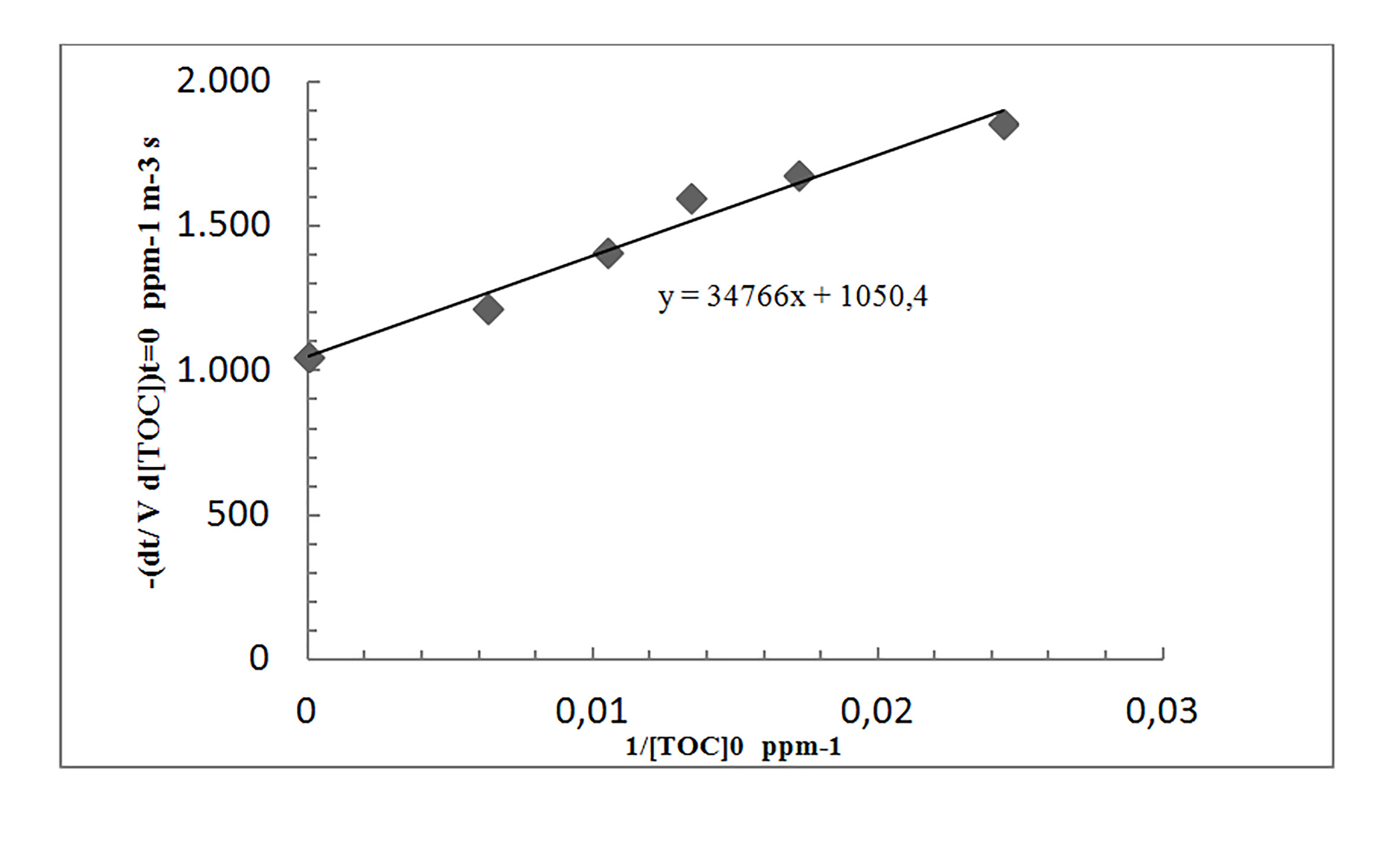
From a plot of the inverse initial rate versus the inverse pollutant concentration, the kinetic parameter obtained were K 1 and k T , as the intercept and slope, respectively. The values obtained are summarized in Table 2.
Table 2 Experimental parameters of L-H equation
| m | b | K 1 | k t | K 1 /12000 | k t /12000 | |
|---|---|---|---|---|---|---|
| Unit | s/m3 | s/ppm m3 | 1/ppm | ppm m1,5/s W0,5 | L/mol | mol m1,5/L s W0,5 |
| Value | 34,766 | 1050.4 | 3.02×10-2 | 9.28×10-4 | 368.22 | 7.74×10-8 |
Figure 2 shows the graph of the inverse initial concentration and inverse initial rate, according to experimental conditions described above. Linear regression revealed a correlation factor of 0.973, showing a good fitting of the data.
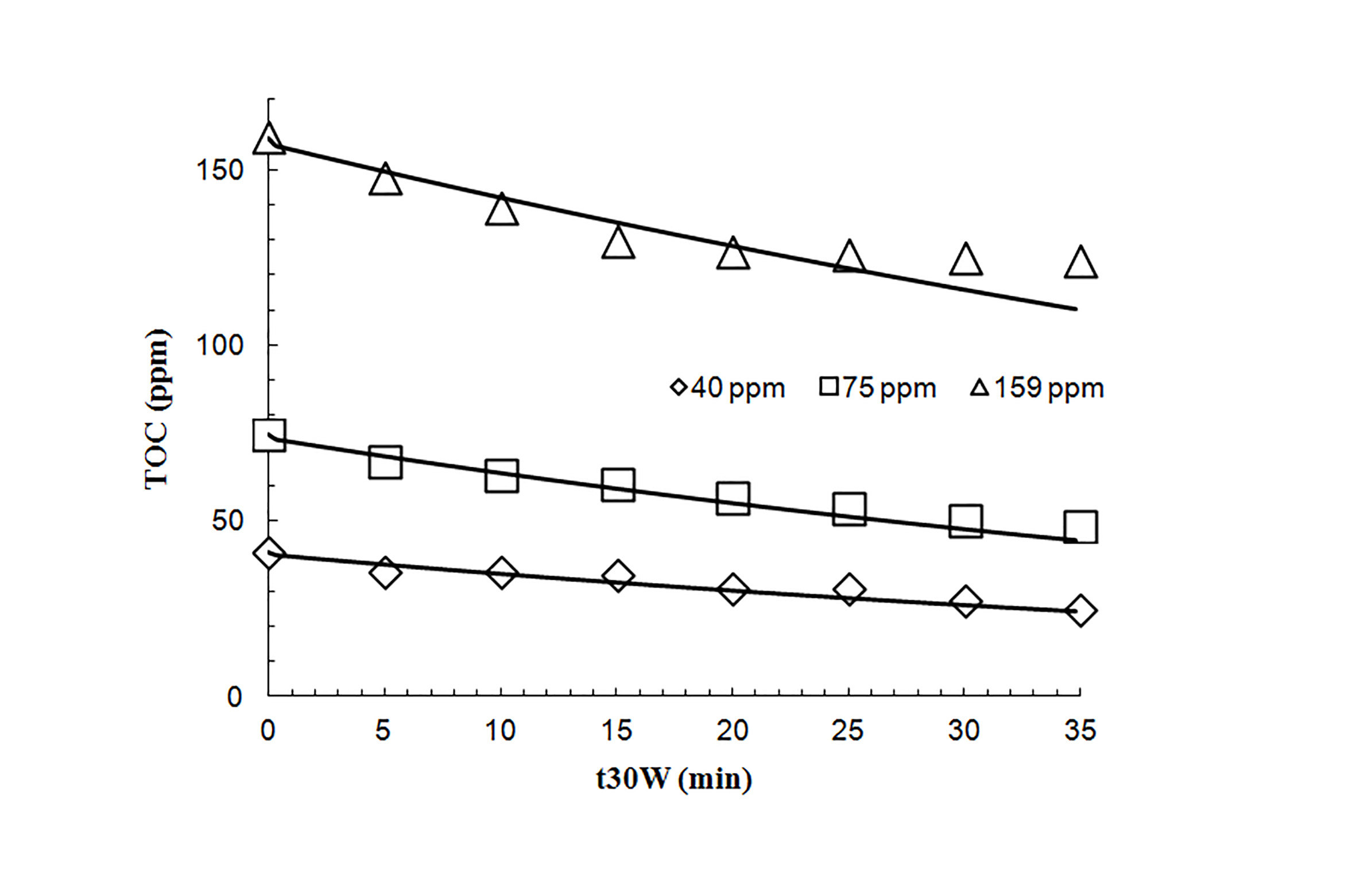
Theoretical and experimental degradation percentages are showed in Table 3. The maximum theoretical degradation percentage was 40%, with the lowest concentration of the contaminant. Solar photocatalysis performs correctly at low pollutant concentrations as demonstrated by experimental degradation percentages. An increase of initial concentration of albendazole, causes a decrease of degradation rate. Probably, what happens is that at high concentrations, pollutant molecules cover the catalyst surface and they block the electron-hole pair formation, which is important when starting the substance degradation.
Table 3 Theoretical and experimental degradation percentage
| TOC o (ppm) | |||
|---|---|---|---|
| 40 | 75 | 159 | |
| % degradation (Exp) | 39.36 | 34.77 | 22.16 |
| % degradation (Model) | 40.52 | 40.52 | 30.52 |
Figure 3 shows a satisfactory settlement obtained from theoretical model and experimental data. The solid line on this figure represents values obtained from the model, while, the data points were obtained experimentally.
Kinetic parameters obtained theoretically from the experimental results, are independent of the radiation field within the reactor, and they can be used in the future for scaling up applications or photocatalytic reactor design, for the same conditions and energy radiation spectrum used in this study, and for the initial pollutant concentrations and radiation intensity accumulated, that is, 35 Wh/m2.
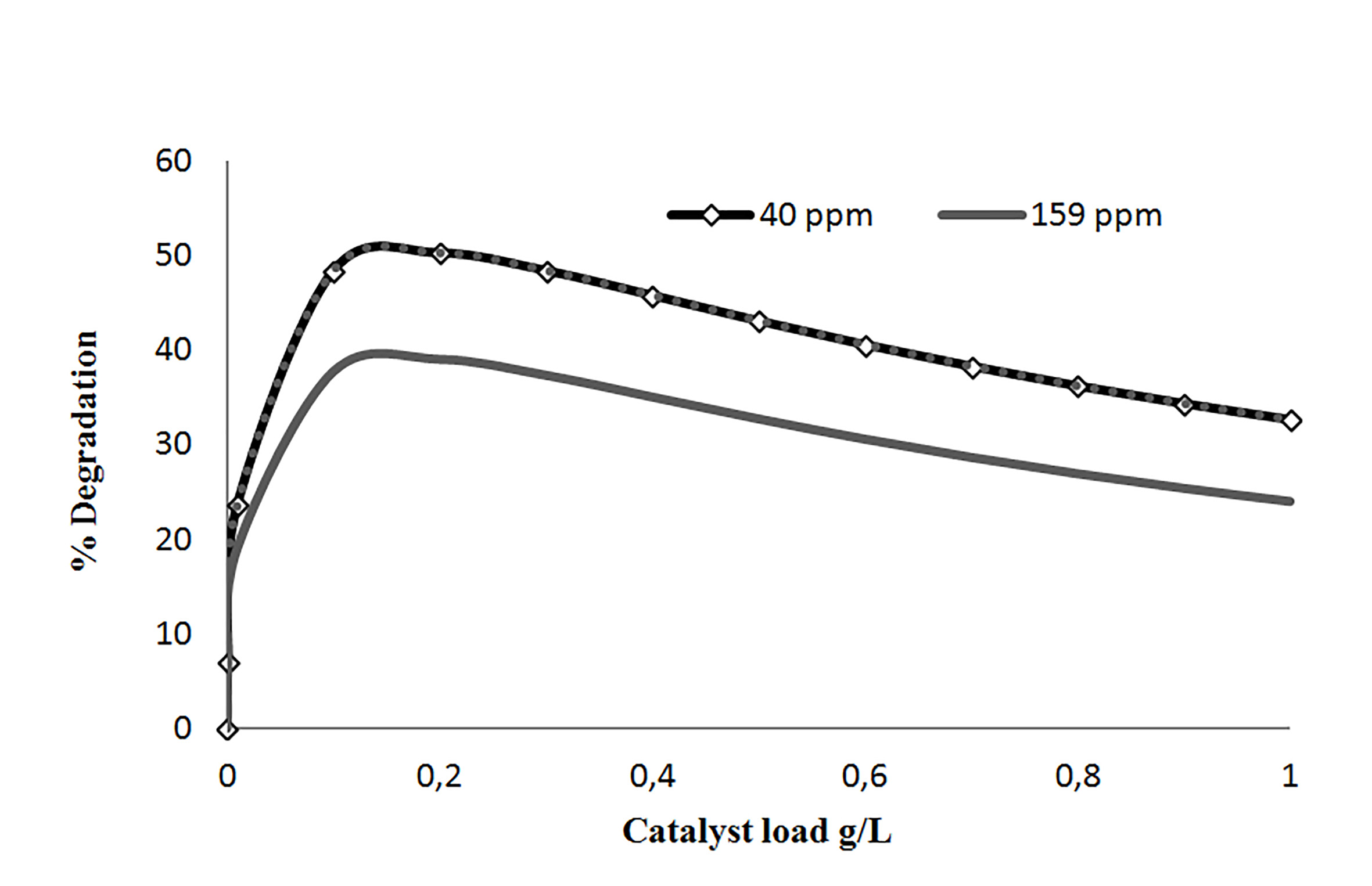
3.4. Optimal catalyst loading
It was found that in using 0.2 g/L catalyst loading, the highest degradation percentage were obtained for concentrations pollutants ranges used in this study. Other research concluded that the catalyst loading to optimize the degradation percentage, optimizes the absorption of photons too, which corresponds to 0.15 - 0.2 g/L interval 28. Figure 4, shows the results obtained about catalyst concentration.
An increase of the amount of titanium dioxide, favored the electron-hole pair formation. However, experimental data showed that a high catalyst loading promotes overlap, and only the upper layers receive photons directly, while the lower ones are not. As a result there is an amount of titanium dioxide that contributes to the electron-hole formation and this is directly linked with the loading value that optimizes the degradation percentage.
4. Conclusions
SFM and ray tracing articulation were accounted satisfactorily for albendazole photocatalytic degradation, as in the LVRPA was described appropriately along the reactor with a low catalyst loading and low contaminant initial concentration, radiant field complex geometry CPC.
Model described adequately Hilshenwood-Langmuir law degradation kinetics of albendazole. Adsorption constant (K 1 ) was greater than the rate constant (k T ), ensuring that limitations are not for adsorption kinetics, degradation is limitated by the reaction in the catalyst surface.
Kinetic parameters derived from fitting the model to the experimental results were independent of the radiation field in the reactor and therefore, it is found to be a suitable model for scaling photocatalytic reactors.