Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Boletín Científico. Centro de Museos. Museo de Historia Natural
Print version ISSN 0123-3068
Bol. Cient. Mus. Hist. Nat. Univ. Caldas vol.17 no.2 Manizales July/Dec. 2013
USING FORAGING BEHAVIOR TO DETECT EDGE EFFECTS: A CASE STUDY WITH COLOMBIAN ANDEAN MAMMALS*
USO DEL COMPORTAMIENTO DE FORRAJEO PARA DETECTAR EFECTOS DE BORDE: UN ESTUDIO DE CASO CON MAMÍFEROS ANDINOS COLOMBIANOS
Ginna Narváez1 y Francisco Sánchez2**
* FR: 16-X-2012. FA: 7-IV-2013.
1 Departamento de Biología, Universidad de Nariño. San Juan de Pasto, Colombia. E-mail: ginnanar@gmail.com
2 Grupo Integrado de Investigaciones en Química y Biología (InQuiBio). Programa de Biología Aplicada, Universidad Militar Nueva Granada. Cajicá, km 3 vía Cajicá-Zipaquirá, Cajicá, Colombia. E-mail: fasbos@gmail.com
** A quien debe dirigirse la correspondencia.
Abstract
We compared the use of artificial food patches (giving-up density GUD technique) by small-granivorous mammals between forest interior and forest-pasture edge, strata (ground and understory), and across different moon illumination occurrence in a small, oak forest fragment in the Colombian Andes. Foraging costs for small mammals were higher near the edge than in the interior on the ground, but not at the understory. Small mammals preferred foraging on the ground than at the understory. Moon illumination had no effect. Our results indicate that forest destruction and its associated edge effect may reduce the area available for small mammals, consequently may affect the biotic interactions and the dynamics of Andean forest remnants, and this effect is modulated by the forest's vertical heterogeneity. Therefore, the use of behavioral indicators such as the food patch use, measured with the GUD technique, is an effective method to measure responses of Andean small granivorous mammals to human alterations.
Key words: Andes, giving-up density, oak forest, optimal patch use.
Resumen
Comparamos el uso de parches artificiales (técnica de densidades de abandono DDA) por pequeños mamíferos granívoros en el interior del bosque y el borde bosque-pastizal, estrato (suelo y sotobosque), en diferentes niveles de iluminación de la luna, en un pequeño fragmento de bosque de roble en los Andes colombianos. Los costos de forrajeo para los pequeños mamíferos fueron mayores cerca al borde que en el interior en el suelo, pero no en el sotobosque. Los pequeños mamíferos prefirieron forrajear en el suelo que en el sotobosque. La iluminación de la luna no tuvo efecto. Nuestros resultados indican que la destrucción del bosque y su asociado efecto de borde, reduce el área disponible para los pequeños mamíferos, consecuentemente puede afectar las interacciones bióticas y la dinámica de los remanentes de bosques andinos, y sus efectos son modulados por la heterogeneidad vertical del bosque. Por lo tanto, el uso de indicadores comportamentales como el uso de parches alimentarios, medidos con la técnica de DDA, es un método efectivo para medir las respuestas de los mamíferos granívoros andinos ante las alteraciones humanas.
Palabras clave: Andes, bosque de roble, densidades de abandono, uso óptimo de parches.
INTRODUCTION
Forest destruction and fragmentation usually imply replacing complex native forests with simpler ecosystems, and have been recognized as one of the main threats to wildlife in the Neotropics (WHITMORE & SAYER, 1992; MURCIA, 1995). Indeed, forest destruction and fragmentation are common forms of habitat degradation in Latin America, causing isolation amongst forest patches (PRIMACK, 2006), reducing core habitat availability and also increasing edge surface (EWERS & DIDHAM, 2007). In Colombia, high rates of forest destruction are mainly associated with agricultural activities; urban development and the exploitation of mineral resources, and some of the most threatened ecosystems are the Andean forests (KATTAN & ÁLVAREZ-LÓPEZ, 1996; ARMENTERAS et al., 2003). Indeed, CHAVES & ARANGO (1998) recognized that accumulated deforestation on Colombian mountains was around 73-90% at the end of the 20th century, and in particular, oak (Quercus humdoldtii) forests have suffered a considerable reduction in area and have been fragmented mainly due to the expansion of rural areas (SOLANO & VARGAS, 2006). Thus, since human-caused forest destruction and fragmentation are serious threats to the preservation of biological diversity in Colombia, and since these processes bring along the increase of anthropogenic edges, understanding edge effects or edge dynamics is important in order to design strategies to manage and monitor biodiversity in fragmented landscapes.
An ecological edge may be defined as the frontier between habitats that differ in quality (STRAYER et al., 2003), and therefore ecological processes near the edge generally differ from those at the core of the patch (DONOVAN et al., 1997). Edge effects are the result of the interaction between two adjacent patches separated by an abrupt transition that affects the performance of individuals (LIDICKER, 1999). These effects may be due to abiotic changes in the environment, e.g., illumination, air and soil temperature and/or caused by cover or vegetation structure changes or modifications in interspecific relationships associated to the ecological flow through the edge (MURCIA, 1995; FAGAN et al., 1999; CADENASSO et al., 2003). In small mammals, edge effects may affect species composition, abundance, habitat use, movement patterns or foraging behavior (MILLS, 1995; LIDICKER, 1999; ANDERSON & BOUTIN, 2002; RIES & SISK, 2004). However, different mammals may not react similarly to edges, in fact, edge effects may depend on species, and even individual characteristics such as sex and age (LIDICKER, 1999). For example, female voles Microtus canicaudus seem to prefer forest interior to borders, although the pattern of use of forest patch may vary with age (LIDICKER & PETERSON, 1999). In contrast, young red squirrels Tamiasciurus hudsonicus had higher survival rates at the edge than at the interior of a forest probably due to differences in food availability between habitats (ANDERSON & BOUTIN, 2002). HESKE (1995) reported no changes in the activity or abundance of small mammals between forest-farm edge and forest interiors in southern Illinois, USA. Compared to temperate zones, little is known about how forest edges may affect Neotropical mammals. In two Atlantic forest remnants in Brazil, species richness and diversity of small mammals decreased from the interior toward an edge with open areas dominated by grasses (STEVENS & HUSBAND, 1998). Also, experiments designed to examine possible edge effects on seed and nest predation probably by mammals show opposite trends (BURKEY, 1993; LÓPEZ-BARRERA et al., 2005), seed predation appears to decrease closer to the edge, whereas nest predation seems to have the opposite tendency (BURKEY, 1993; ESTRADA et al., 2002). In Colombia, OTÁLORA-ARDILA & LÓPEZ-ARÉVALO (2006) worked at an oak forest on the Eastern Cordillera of Colombia and found that bat species richness increased from the center of forest patches toward the edge and surrounding matrix.
In this study, we apply and advocate for the use of foraging behavior to study possible edge effects on Colombian, Andean terrestrial mammals. This is because reactions toward edges may depend on the perceptions animals have about the possible transition between habitats, and that perception should be reflected in their foraging decisions. Therefore, behavioral indicators should be useful tools for the conservation of mammals, more when studies that involve capturing wildlife in Colombia have become almost prohibited by current legislation (FERNÁNDEZ, 2011). Indeed, behaviors should be tightly linked to the individual's fitness, and because behaviors may change more quickly than for example population size, species composition or species diversity in response to environmental changes (LINDELL, 2008; MORRIS et al., 2009). Thus, different behaviors may be used as reliable indicators, that can be measured relatively easily, and that may even be used to forecast animal responses. In addition, the study of mammal behavior has a large body of theory and models to interpret the results. For example, theory on optimal patch use can be used to examine responses toward environmental changes, and we took advantage of this theory to examine the possible perceived edge effects by small mammals in an Andean forest fragment in the south-west of Colombia.
According to this theory, foragers should continue using a food patch until the benefits associated with feeding from it are greater than or equal to the costs associated with its exploitation (BROWN, 1988). On one hand, foraging costs include metabolic costs associated to the energy used exploiting the patch, the costs derived from the perceived risk of predation, and also missed opportunity costs. On the other hand, the benefits derive from the food obtained, expressed as harvest rates. Brown's model is an extension of Charnov's marginal value theorem that assumes that the forager experiences diminishing returns while foraging, and therefore the harvest rate depends on the amount of food at the food patch (CHARNOV, 1976). Thus, in a food patch with diminishing returns, the harvest rate at which the forager leaves the patch is related to the amount of food left by a forager after exploiting a patch, the giving-up density (GUD), and it therefore estimates the marginal value (MV) of the patch for the forager (BROWN, 1992).
According to the model, the more food left at the patch, i.e., the higher the GUD, the lower the MV. Also, the model predicts that two food patches with the same costs and benefits should be exploited by a forager to the same quitting harvest rate, i.e., they will have the same GUD. Therefore, this model can be used to examine how a particular background or habitat affects foraging behavior. For example, the model has been successfully applied to compare the foraging behavior of mule deer Odocoileus hemionus in forest interiors, edges and open areas, while feeding under the risk of being depredated by mountain lions Puma concolor (ALTENDORF et al., 2001). The theory has also been used to study the effect of environmental factors such us moon phase, air temperature and relative humidity on the foraging behavior of desert gerbils (KOTLER et al., 1993). For example, KOTLER et al. (1993) found that the brighter the moon was, the lower the marginal value of food patches to the gerbils, probably because they were more exposed to predators. Thus, we used Brown's model to answer the following questions about the foraging decisions of small mammals at a small, Andean, oak forest fragment: 1) do small mammals perceive the edge and interior of the forest remnant as different habitats?, i.e., will their foraging behavior differ between the two sites?; 2) are there differences in the use of food patches located at the ground level and understory?; and 3) does moon illumination affect the foraging behavior of small mammals?
MATERIALS AND METHODS
Study site and species
We studied the foraging behavior of small mammals at a forest fragment between April and July 2007 at the Chimayoy Environmental Park, located 4 km from the Pan-American Highway and north of the city of Pasto, Department of Nariño (1º31'17.9" N; 77º05'25.9" W). The fragment was approximately 223565 m2, located between 2670 and 2950 m alt., and surrounded by abandoned pastures, Andean secondary forest, subpáramo (transition ecosystem between Andean forest and paramo), pine (Pinus sp.) plantations, and secondary forest with selective logging. At the core of the fragment, the forest had three strata: herbaceous stratum up to 0.5 m, understory (2-8 m), and arboreal stratum (20-30 m). Oak trees of 15-22 m were abundant, and there were also trees of Clusia multiflora, Weinmannia sp., Freziera canescens, and Hyeronima macrocarpa. The selected edge for the study was the one with the highest contrast between vegetation covers, i.e., the limit between forest and abandoned pastures, because this edge is common in the landscape of the region due to agricultural activities. At the edge, herbs and shrubs were considerably more abundant than in the interior of the forest.
There have been no studies to determine species composition or abundance of mammals in the study area. Therefore, we did a short survey to have an idea of the composition of mammals and their relative abundance. We also used this survey to record footprints of the individuals captured, and to compare them with those obtained at feeding stations. We used 32 Sherman and 6 pitfall traps (50-cm deep buckets) for 16 nights; corn seeds were used as bait. Half the traps were placed at the interior of the forest, 100 m from the end of the forest vegetation (LÓPEZ-BARRERA et al., 2005), and half near the edge, 10 m from the end of the forest vegetation. The transect in the interior was located at approximately the center of the oak forest fragment. At both the interior and near the edge of the forest, half of the Sherman traps (8) where placed on the ground and half (8) in the understory, at heights of 2-4 m up on the trees. Adjacent traps were separated by 30-40 m, and their position, ground or understory, was alternated. Two of the captured animals of each species were prepared as collection specimens and were placed at the Museum PSO of Universidad de Nariño.
The foraging behavior of granivorous small mammals
We placed feeding stations both in the interior and the forest edge transects. Each transect consisted of 8 feeding stations: 4 on the ground and 4 in the understory, i.e., 2-4 m on an oak tree. Along a transect, the position of stations, ground or understory, was alternated, and there were 30-40 m between adjacent stations, i.e., each transect was ~280 m long. The feeding stations were plastic trays (28 × 14 × 6 cm) containing 3 L of sifted sand mixed with 3 g of popcorn grains (20-22 grains). Stations were protected from the rain with a plastic roof, and after a night of experiments, the sand from the stations was replaced with dry sand the next day to ensure that the foraging surface was not affected by changes in humidity. We chose popcorn grains after making preliminary trials during a habituation period with peanuts and birdseeds.
During 13 days we habituated small mammals to visit the feeding stations that contained sand mixed with earth from the forest and vanilla essence. After the habituation period all stations were visited by small mammals. During 30 days following the habitation we recorded GUDs, i.e., the mass of popcorn grains left at the stations after a night of foraging (BROWN, 1988). Thus, the total sampling effort was 16 stations × 30 nights. Feeding stations were prepared and set before sunset, ca. 1830-1900 h, and examined after sunrise, ca. 0530-0600 h. The preparation of stations included mixing the grains with the sand, and smoothing the surface to record footprints. During the measurements of foraging, we only used sand as non-edible substrate. The examination of the stations included first registering the tracks on the sand and then sifting the sand to remove the grains left. The grains retrieved from every station were kept in sealed plastic bags, marked with the number of the station, date, stratum (ground or understory) and site (edge or interior), and then weighted to obtain the GUD. We obtained percentage of moon illumination from the site http://www.timeanddate.com.
Statistical analysis
To examine the data, we used a general linear model where averaged GUD of the stations of a site per day entered as the dependent variable, whereas site and stratum were used as fixed factors, and percentage of moon illuminated was used as a covariate (ZAR, 1999). We used averaged GUD to prevent possible lack of independence among stations from the same transect. We used a simplification model technique (WILSON & HARDY, 2002) to work with a model containing only significant predictors, and used Tukey tests as a multiple comparisons procedure. We performed graphical analyses of residuals and found no deviations from the assumptions of normality and homogeneity of variance. We chose a = 0.05 as the significance level to evaluate statistical hypotheses.
RESULTS
Mammals at the study site
We captured with Sherman traps 5 montane colilargos Microryzomys minutus (Cricetidae, Rodentia) and 3 Mexican harvest mice Reithrodontomys mexicanus (Cricetidae, Rodentia); footprints on the sand of these two rodent species could not be told apart. Two M. minutus were captured at the edge-ground of the forest, whereas at the interior, 1 individual was captured on the ground and 2 were found in the understory. The R. mexicanus were only captured in the interior of the forest; 1 on the ground and 2 in the understory. Summing up, we captured 2 mice near the edge, 6 in the interior, 4 on the ground, and 4 at the understory. We captured no mammals with pitfall traps.
In addition, during our work we found a shrew Cryptotis sp. (Soricidae, Soricomorpha) dead at the forest edge. In the area we also observed the red-tailed squirrel Sciurus granatensis (Sciuridae, Rodentia), and a peasant captured a bare-tailed woolly mouse opossum Micoureus regina (Didelphidae, Didelphimorphia). We also observed local people who hunted red brockets Mazama rufina (Cervidae, Cetartiodactyla) and mountain pacas Cuniculus taczanowskii (Cuniculidae, Rodentia) in the vicinity of Chimayoy, about 10-20 km from the Park. Thus, in total we recorded six species of mammals for Chimayoy and its surroundings.
Foraging behavior of small mammals
Feeding stations were mainly visited by mice, but we also found stations with marsupial footprints that matched those of M. regina. Since footprints left at most stations could not be assigned to any of the possible species in the area and there were stations in which it was not possible to determine the last forager, GUDs were classified broadly as small mammal.
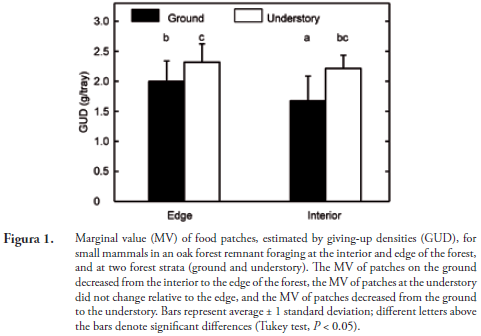
The simplified statistical model did not contain as significant factors the independent effects of site or moon illumination, or moon illumination in interaction with site or stratum (All P > 0.05). The only significant effects were those of stratum (Mean squares [MS] = 5.51, F1,116 = 51.79, P < 0.001) and site×stratum (MS = 0.85, F2,116 = 8.03, P < 0.001). Tukey tests showed that both at the interior and the edge of the forest, the GUD's were lower on the ground than at the understory (P < 0.05), i.e., they were treated them as different microhabitats (Figura 1). In addition, Tukey tests also showed that at the ground stratum, small mammals perceived edge and interior of the forest as different habitats, and preferred the interior (P < 0.05), whereas at the understory they did not perceive this effect (Figura 1). We interpret these results as an indication that small mammals perceived an edge effect, but this effect was modulated by the forest stratum.
DISCUSSION
Mammals at the study site
At the interior of the forest, M. minutus, R. mexicanus, and M. regina (tracks) were present both on the ground and the understory. Both M. regina and R. mexicanus are known to have arboreal habits and to feed upon seeds (EISENBERG, 1989; EMMONS, 1997; REID, 2009), whereas M. minutus is known to be mainly a ground-dwelling species that on the ground finds refuge, and forages on Renealmia sp. and palm seeds (Voss 1979, field notes cited in CARLETON & MUSSER, 1989; EISENBERG, 1989; DÍAZ DE PASCUAL, 1994). Thus, our preliminary results may suggest that at Chimayoy M. minutus is more arboreal than in other areas of its distribution. Another possibility is that, since feeding trays are more sensitive to measuring microhabitat use than are traps, in the previous studies could not be detected that M. mimulus regularly forages on trees. Also, although Cryptotis was registered in the study site, and other species of shrews have been captured in traps with corn on the Andes (SÁNCHEZ & ALVEAR, 2003), it is unlikely that shrews would be regular visitors of the stations given their predominantly invertebrate-feeding habits (EISENBERG, 1989; DÍAZ DE PASCUAL & DE ASCENÇAO, 2000).
Foraging behavior of small mammals
Our results indicate that small mammals perceived differences at two levels: microhabitat and habitat. At the microhabitat level, food patches on the ground had a higher marginal value than those at the understory level. At the habitat level, small mammals perceived an edge effect, and GUD's were lower in the interior than near the edge, but only at the ground level. Thus, horizontal and vertical heterogeneity of the forest patch affected food patch use by small mammals of small granivorous mammals in Chimayoy. Likewise, the results derived from the trapping are preliminary, but they complement the results about foraging behavior. We captured more rodents in the interior than at the edge of the forest, and we did not capture R. mexicanus near the forest edge. These results also suggest an edge effect, that probably affects more strongly R. mexicanus than M. minutus, but given the limited data set we cannot examine whether the abundance of rodents is also modulated by the interaction between horizontal and vertical heterogeneity of the patch.
To our knowledge, there have been no attempts to examine the possible join effects of edge and stratum on small Neotropical mammals. Nevertheless, the edge effect we found at the ground level is in general agreement with the reduction in species diversity and abundance found for small mammals in Atlantic forest remnants (STEVENS & HUSBAND, 1998). However, in a Neotropical area in Mexico, small mammals were more frequently captured near a forest-farm edge than in the forest interior (ESTRADA et al., 2002). Also in the same study, medium-sized and potential mammal predators were more frequently observed near the forest edge than in the interior of the forest. Hence, there seems to be different responses to forest edges by small Neotropical mammals, and it is likely that the response depends on the particular context of the site, including degree of fragmentation, altitude, and/or group of species present.
Nest predation experiments for several Neotropical sites suggest that predators are more efficient around the forest edges than in the interior (BURKEY, 1993; ESTRADA et al., 2002). Also, GUD's are usually associated to the perceived risk of predation or perception of security by foragers (KOTLER et al., 1994; ALTENDORF et al., 2001), since predation cost may be the most important cost of foraging (BROWN, 2000). Thus, the edge effect perceived by small mammals at Chimayoy may be a response toward an increased risk of predation reaching the forest border. Several species of Canidae, Felidae and Mustelidae have been reported for the mountains of the Nariño department, but unfortunately there is no information about their distribution in fragmented areas or detailed information about their feeding ecology to verify whether they are responsible for the observed edge effect.
The pattern of patch use observed through strata is similar to that of raccoons (Procyon lotor), which have higher GUD's while going higher on trees and this could be caused for an increased cost/benefit ratio of exploiting a patch derived from a higher risk of harm due to falling while trying to avoid predators or competitors (LIC, 2001). Cercopithecus monkeys also have higher GUD while exploiting fruit higher of the tree's crowns, and this may be due to an increased risk of being predated by carnivorous birds or to an associated risk to fall (HOULE et al., 2006). Also, it has been shown in red foxes that their patch use is affected by the risk of being injured (BERGER-TAL et al., 2009). Thus, the perceived risk of predation and the risk of an injury while foraging on the tree may be explaining the effect of vertical heterogeneity on the foraging behavior of small mammals.
Small mammals play important roles in the dynamics of tropical forests, since they are prey for multiple predators, as well as predators of nests, seeds, seedlings and invertebrates, and they may also disperse seeds and spores (FLEMING & SOSA, 1994; PÉREZ-HERNÁNDEZ et al., 1994; ASQUITH et al., 1997; ABRANCHES et al., 1998). For instance, in tropical oak forests small mammals feed upon oak acorns during years of high productivity affecting germination patterns (LÓPEZ-BARRERA & MANSON, 2006). Also, seedling predation by rabbits and rodents is an important cause of mortality for some plant species from oak forests (BONFIL, 2006). Our results show that the cost/benefit ratio of foraging increases from the interior toward the edge, and therefore, the strength of biotic interactions of small mammals should consequently decrease approaching the forest edge. Thus, one could expect that, since most small mammals visiting the stations were rodents, seed and seedling predators will have diminishing importance from the interior to the edge of the forest, as has been found in other Neotropical forests (BURKEY, 1993; LÓPEZ-BARRERA et al., 2005). Hence, forest edges may create enemy free zones (SCHMIDT, 2004), that should favor seeds and seedlings survival leading to higher forest regeneration and may help forests to recover more quickly from fragmentation. Thus, the study of the foraging behavior of one or few species important for the ecosystem function, such as the small mammals, can be important (sometimes more than just lists of presence/absence of species) if for example attempts of restoration or to increase patch connectivity are desired (LINDELL, 2008; MORRIS et al., 2009).
We showed that forest destruction and its resulting increase in edge surface may reduce the effective area available for small mammals, consequently may affect their biotic interactions, and therefore the dynamics of Andean forest remnants. Hence, we advocate for the use of behavioral indicators such as the food patch use measured with the GUD technique, that is a cost-effective method to study responses of Andean small mammals to human alterations. This technique allows examining how habitat quality changes along landscapes, and also if desired, experiments could be set to examine the effect of particular foraging costs, to identify resources which are critical for organisms or to examine the organization of communities (KOTLER & BLAUSTEIN, 1995; BROWN et al., 1997; BROWN, 1999; SCHMIDT, 2000). For example, these behavioral indicators could be used to compare the magnitude of the effect of different edges or to examine areas suitable for corridors, and therefore to give criteria to decide where more resources should be allocated for conservation purposes.
ACKNOWLEDGMENTS
We would like to thank Jhon Jairo Calderón for his support to this project, José y María for their help during the field work, and the Biology Department of Universidad de Nariño for their support to research. Dinesh Rao and Shomen Mukherjee made important comments that helped improving the manuscript. Also, Burt Kotler revised the manuscript and provided, as always, very insightful and helpful comments.
BIBLIOGRAPHY
ABRANCHES, J., VALENTE, P., NÓBREGA, H.N., FERNÁNDEZ, F.A.S., MENDOÇA-HAGLER, L.C. & HAGLER, A.N., 1998.- Yeast diversity and killer activity dispersed in fecal pellets from marsupials and rodents in a Brazilian tropical habitat mosaic. FEMS Microbiology Ecology, 26: 27-33. [ Links ]
ALTENDORF, K.B., LAUNDRE, J.W., GONZÁLEZ, C.A.L. & BROWN, J.S., 2001.- Assessing effects of predation risk on foraging behavior of mule deer. Journal of Mammalogy, 82: 430-439. [ Links ]
ANDERSON, E.M. & BOUTIN, S., 2002.- Edge effects on survival and behaviour of juvenile red squirrels (Tamiasciurus hudsonicus). Canadian Journal of Zoology, 80: 1038-1046. [ Links ]
ARMENTERAS, D., GAST, F. & VILLAREAL, H., 2003.- Andean forest fragmentation and the representativeness of protected natural areas in the eastern Andes, Colombia. Biological Conservation, 113: 245-256. [ Links ]
ASQUITH, N.M., WRIGHT, S.J. & CLAUSS, M.J., 1997.- Does mammal community composition control recruitment in Neotropical forests? Evidence from Panama. Ecology, 78: 941-946. [ Links ]
BERGER-TAL, O., MUKHERJEE, S., KOTLER, B.P. & BROWN, J.S., 2009.- Look before you leap: is risk of injury a foraging cost? Behav. Ecol. Sociobiol., 63: 1821-1827. [ Links ]
BONFIL, C., 2006.- Regeneration and population dynamics of Quercus rugosa at the Ajusco Volcano, Mexico: 155-163 (in) KAPPELLE, M. (ed.) Book title. Berlin: Springer-Verlag. [ Links ]
BROWN, J.S., 1988.- Patch use as an indicator of habitat preference, predation risk, and competition. Behavior, Ecology and Sociobiology, 22: 37-47. [ Links ]
________., 1992.- Patch use under predation risk: I. Models and predictions. Annales Zoologici Fennici 29: 301-309. [ Links ]
________., 1999.- Vigilance, patch use and habitat selection: Foraging under predation risk. Evolutionary Ecology Research, 1: 49-71. [ Links ]
________., 2000.- Foraging ecology of animals in response to heterogeneous environments: 181-215 (in) HUTCHINGS, J. & STEWART, A. (eds.) Book title. Oxford: Blackwell Scientific. [ Links ]
BROWN, J.S., KOTLER, B.P. & MITCHEL, W.A., 1997.- Competition between birds and mammals: a comparison of giving-up densitites between crested larks and gerbils. Evolutionary Ecology, 11: 757-771. [ Links ]
BURKEY, T.V., 1993.- Edge effects in seed and egg predation at two Neotropical rainforest sites. Biological Conservation, 66: 139-143. [ Links ]
CADENASSO, M.L., PICKETT, S.T.A., WEATHERS, K.C. & JONES, C.G., 2003.- A framework for a theory of ecology boundaries. BioScience, 53: 750-758. [ Links ]
CARLETON, M.D. & MUSSER, G.G., 1989.- Systematic studies of Oryzomyinae rodents (Muridae, Sigmodontinae): A synopsis of Microryzomys. Bulletin of the American Museum of Natural History, 191: 1-83. [ Links ]
CHARNOV, E.L., 1976.- Optimal foraging, the marginal value theorem. Theoretical Population Biology, 9: 129-136. [ Links ]
CHAVES, M.E. & ARANGO, N., 1998.- Informe nacional sobre el estado de la biodiversidad-Colombia. Bogotá, Colombia: Instituto de Investigación de Recursos Biológicos Alexander von Humboldt, PNUMA, Ministerio de Medio Ambiente. [ Links ]
DÍAZ DE PASCUAL, A., 1994.- The rodent community of the Venezuelan cloud forest, Mérida. Polish Ecological Studies, 20: 155-161. [ Links ]
DÍAZ DE PASCUAL, A. & DE ASCENÇAO, A., 2000.- Diet of the cloud forest shrew Cryptotis meridensis (Insectivora: Soricidae) in the Venezuelan Andes. Acta Theriologica, 45: 13-24. [ Links ]
DONOVAN, T.M., JONES, P.W., ANNAND, E.M. & THOMPSON, F.R., III. 1997.- Variation in local-scale edge effects: mechanisms and landscape context. Ecology, 78: 2064-2075. [ Links ]
EISENBERG, J.F., 1989.- Mammals of the Neotropics: The northern Neotropics. Chicago: The University of Chicago Press. [ Links ]
EMMONS, L.H., 1997.- Neotropical rainforest mammals. Chicago: The University of Chicago Press. [ Links ]
ESTRADA, A., RIVERA, A. & COATES-ESTRADA, R., 2002.- Predation of arti?cial nests in a fragmented landscape in the tropical region of Los Tuxtlas, Mexico. Biological Conservation, 106: 199-209. [ Links ]
EWERS, R.M. & DIDHAM, R.K., 2007.- The effect of fragment shape and species' sensitivity to habitat edges on animal population size. Conservation Biology, 21: 926-936. [ Links ]
FAGAN, W.F., CANTRELL, R.S. & COSNER, C., 1999.- How habitat edges change species interactions. American Naturalist, 153: 165-182. [ Links ]
FERNÁNDEZ, F., 2011.- The greatest impediment to the study of biodiversity in Colombia. Caldasia, 33 (2). [ Links ]
FLEMING, T.H. & SOSA, V.J., 1994.- Effects of nectarivorous and frugivorous mammals on reproductive success of plants. Journal of Mammalogy, 75: 845-851. [ Links ]
HESKE, E.J., 1995.- Mammalian abundances on forest-farm edges versus forest interiors in southern Illinois: is there an edge effect? Journal of Mammalogy, 76: 562-568. [ Links ]
HOULE, A., VICKERY, W.L. & CHAPMAN, C.A., 2006.- Testing mechanisms of coexistence among two species of frugivorous primates. Journal of Animal Ecology, 75: 1034-1044. [ Links ]
KATTAN, G.H. & ÁLVAREZ-LÓPEZ, H., 1996.- Preservation and management of biodiversity in fragmented landscapes in the Colombian Andes: 3-18 (in) SCHELHAS, J. & GREENBERG, R. (eds.) Book title. Washington, D.C.: Island Press. [ Links ]
KOTLER, B.P. & BLAUSTEIN, L., 1995.- Titrating food and safety in a heterogeneous environment: when are risky and safe patches of equal value? Oikos, 74: 251-258. [ Links ]
KOTLER, B.P., BROWN, J.S. & MITCHELL, W.A., 1993.- Environmental factors affecting patch use in two species of gerbilline rodents. Journal of Mammalogy, 74: 614-620. [ Links ]
KOTLER, B., GROSS, J.E. & MITCHEL, W.A., 1994.- Applying patch use to assess aspects of foraging behavior in Nubian ibex. Journal of Wildlife Management, 58: 299-307. [ Links ]
LIC, V., 2001.- Applying foraging theory to wildlife conservation: An application with the raccoon (Procyon lotor). Chicago: University of Illinois at Chicago. [ Links ]
LIDICKER, W.Z., Jr., 1999.- Responses of mammals to habitat edges: an overview. Landscape Ecology, 14: 333-343. [ Links ]
LIDICKER, W.Z., Jr. & PETERSON, J.A., 1999.- Responses of small mammals to habitat edges: 211-227 (in) BARRET, G.W. & PELES, J.D. (eds.) Book title. New York: Springer-Berlag. [ Links ]
LINDELL, C.A., 2008.- The value of animal behavior in evaluations of restoration success. Restoration Ecology, 16: 197-203. [ Links ]
LÓPEZ-BARRERA, F. & MANSON, R.H., 2006.- Ecology of acorn dispersal by small mammals in montane forests of Chiapas, Mexico: 165-176 (in) KAPPELLE, M. (ed.) Book title. Berlin: Springer-Verlag. [ Links ]
LÓPEZ-BARRERA, F., NEWTON, A. & MANSON, R., 2005.- Edge effects in a tropical montane forest mosaic: experimental tests of post-dispersal acorn removal. Ecological Research, 20: 31-40. [ Links ]
MILLS, L.S., 1995.- Edge effects and isolation: red-backed voles on forest remnants. Conservation Biology, 9: 395-403. [ Links ]
MORRIS, D.W., KOTLER, B.P., BROWN, J.S., SUNDARARAJ, V. & ALE, S.B., 2009.- Behavioral indicators for conserving mammal diversity. Annals of the New York Academy of Sciences, 1162: 334-356. [ Links ]
MURCIA, C., 1995.- Edge effects in fragmented forests: implications for conservation. TREE, 10: 58-62. [ Links ]
OTÁLORA-ARDILA, A. & LÓPEZ-ARÉVALO, H.F., 2006.- Incedencia de algunos elementos del paisaje fragmentado de Encino (Santander, Colombia) sobre la riqueza y diversidad de murciélagos: 83-94 (en) SOLANO, C. & VARGAS, N. (eds.) Book title. Bogotá: Fundación Natura-Pontificia Universidad Javeriana. [ Links ]
PÉREZ-HERNÁNDEZ, R., SORIANO, P. & LEW, D., 1994.- Marsupiales de Venezuela. Cuadernos Lagoven, Caracas, Venezuela. [ Links ]
PRIMACK, R.B., 2006.- Essentials of conservation biology. Sinauer Associates. [ Links ]
REID, F., 2009.- A field guide to the mammals of Central America and southeast Mexico. Oxford: Oxford University Press. [ Links ]
RIES, L. & SISK, T.D., 2004.- A predictive model of edge effects. Ecology, 85: 2917-2926. [ Links ]
SÁNCHEZ, F. & ALVEAR, M., 2003.- Comentarios sobre el uso de hábitat, dieta y conocimiento popular de los mamíferos en un bosque andino de Caldas, Colombia. Bol. Cient. Mus. Hist. Nat. U. de Caldas, 7: 121-144. [ Links ]
SCHMIDT, K.A., 2000.- Interactions between food chemistry and predation risk in fox squirrels. Ecology, 81: 2077-2085. [ Links ]
________., 2004.- Incidental predation, enemy-free space and the coexistence of incidental prey. Oikos, 106: 335-343. [ Links ]
SOLANO, C. & VARGAS, N., 2006.- Memorias del I Simposio Internacional de Robles y Ecosistemas Asociados. Bogotá: Fundación Natura-Pontificia Universidad Javeriana. [ Links ]
STEVENS, S.M. & HUSBAND, T.P., 1998.- The influence of edge on small mammals: evidence from Brazilian Atlantic forest fragments. Biological Conservation, 85: 1-8. [ Links ]
STRAYER, D.L., POWER, M.E., FAGAN, W.F., PICKETT, S.T.A. & BELNA, J., 2003.- A classification of ecological boundaries. BioScience, 53: 723-729. [ Links ]
WHITMORE, T.C. & SAYER, J.A., 1992.- Deforestation and species extinction in tropical moist forests: 1-14 (in) WHITMORE, T.C. & SAYER, J.A. (eds.) Book title. London: Chapman & Hall. [ Links ]
WILSON, K. & HARDY, I.C.W., 2002.- Statistical analysis of sex ratios: an introduction: 424 (in) HARDY, I.C.W. (ed.) Book title. Cambridge: Cambridge University Press. [ Links ]
ZAR, J.H., 1999.- Biostatistical analysis. Prentice Hall, Upper Saddle River, N. J. [ Links ]













