Introduction
The Calliphoridae, commonly known as “blowflies” are medium-sized dipterans, usually with green and blue metallic colors (Vargas & Wood, 2010). The family has approximately 1,000 species worldwide, 126 currently inhabit the neotropics (Pape, Wolff & Amat, 2004), and only 31 species of four subfamilies and 12 genera are known for Colombia (Wolff & Kosmann, 2016).
According to Prado & Guimarães (1982), members of the Calliphoridae family are excellent flyers with high dispersal abilities and feed on a variety of resources that can be found at considerably distances; although adults are floral visitors attracted by decomposition scents, and can be found in other resources such as feces, carrion, necrotic tissue or open wounds (Wolff, 2010) Some species have medical and veterinary importance either because of their positive effect on wound cleansing (Wolff et al., 2010). Several species are predators or parasites of earthworms (Pape et al., 2004), and others cause alterations in the health of birds and mammals since they carry pathogens, coupled with this, some species cause secondary myiasis or compulsory traumatic type in animals (Guimarães & Papavero, 1999).
The biology of the blowflies is diverse, with several synanthropic species living in a high degree of association with human settlements (Shewell, 1992). Due to their eating habits, mainly carrion, they play a key ecological role as carrion decomposers, used in the determination of the Postmortem Interval (PMI) (Ramos-Pastrana & Wolff, 2017; Ramos-Pastrana, Virgüez-Díaz & Wolff, 2018). These flies make up a large part of the cadaverous fauna and in addition to PMI dating, they provide evidence about the place of death and postmortem circumstances that can be used as fundamental information related to legal cases such as cases of abuse and neglect of children and the elderly (Arnaldos, Prado, López-Gallego & García et al., 2006; Ramos-Pastrana & Wolff, 2017), as well as in cases involving traffic accidents, origin or geographical origin of narcotic drugs and deaths caused by drug use and poisoning (Anderson & Sherah, 1996).
In Neotropical countries, such as in Brazil (Barros-Souza et al., 2012) and Colombia (Martínez et al., 2007), studies on Calliphoridae have focused mainly on cadaver decomposition rates and associated fauna. Few studies have been published on the Calliphoridae family in the Andean Amazon lowlands or high-altitude areas (Ramos-Pastrana et al., 2014, 2018, 2019); there is only one study involving human bodies (Ramos-Pastrana & Wolff, 2017). The main objective of this study was to identify the species of Calliphoridae that occur on the eastern slope of the Eastern Mountain Range of the Andean Amazon, analyzing their altitudinal distribution, abundance, richness, and diversity patterns.
Material and methods
The study was done in the municipality of Florencia, Caquetá in the Andean Amazon. This zone has an annual average rainfall of 3,840 mm, with a Low-Intensity Rainy season (LIR) from September to February, and a High-Intensity Rainy season (HIR) from March to August, where the average temperature tends to decrease (Instituto Geográfico Agustín Codazzi [IGAC], 2010).
Five stations located between 250 and 2,500 masl were sampled, with a minimum difference between sampling stations of 460 m (Table 1). Sampling was done for 12 days, non-consecutive, by climatic season (HIR: March 3, 4, 18, 19; April 14, 15, 29, 30; June 3, 4, 16, 17 and LIR: September 9, 10, 23, 24; November 10, 11, 23, 24; December 1, 2, 14, 15) for 24 sampling days per station. Nine Van SomerenRydon traps baited with decomposing fish were installed at each station, separated by a distance of approximately 50 meters from each other, and at an approximate height of 1.5 meters. Traps were active for 36-hour periods, for a total sampling effort of 1,620 hours. Traps were checked and specimens collected every 12 hours.
Table 1 Sampling stations in the eastern slope of the Eastern Mountain Range in the Colombian Andean Amazon.
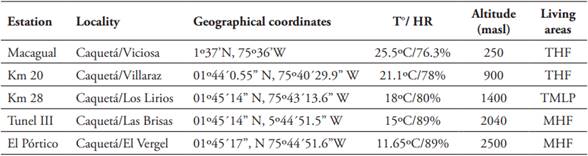
THF= Tropical Humid Forest; TMLP= Tropical Montane Lowland Pluvial; MHF= Montane Humid Forest.
Source: Own elaboration.
Specimens were identified in the Entomology Laboratory of the Universidad de la Amazonia, Campus Centro, using the taxonomic keys of Dear (1985); Amat et al. (2008); Vargas & Wood (2010) and Whitworth (2014) and deposited in the entomological collection of the Universidad Amazonia -LEUA-, (National Registry of Collections, number pending).
To evaluate the sampling effort, a species accumulation curve based on abundance was performed using the Chao 1, Chao 2, Jack 1 and Jack 2 estimators from the EstimateS version 8 program for Windows (Colwell, 2006). A rarefaction curve was performed to quantify species richness, based on the number of specimens, using the Biodiversity Pro version 2 program. An ANOVA with post-hoc Tukey HSD was used to test independently for significant differences in species richness and abundance between the two climatic seasons and the sampling areas, using the InfoStat version 2018 program (Di Rienzo et al., 2018). Species richness was measured as the number of species per station, abundance as the total number of specimens per station, alpha diversity was estimated using Shannon diversity and dominance was measured using Simpson dominance index (Álvarez et al., 2006). Factorial correspondence analysis (FCA) was performed to observe patterns in the data according to altitude, using XLSTAT 2012.5 software. Cluster analysis was then performed to group species using their abundance at each altitudinal range, in each sampling season, based on Euclidean distances using the statistical program InfoStat version 2019 (Di Rienzo et al., 2018). All altitudinal gradients and life zones were cataloged as proposed by Holdridge (1996).
Results
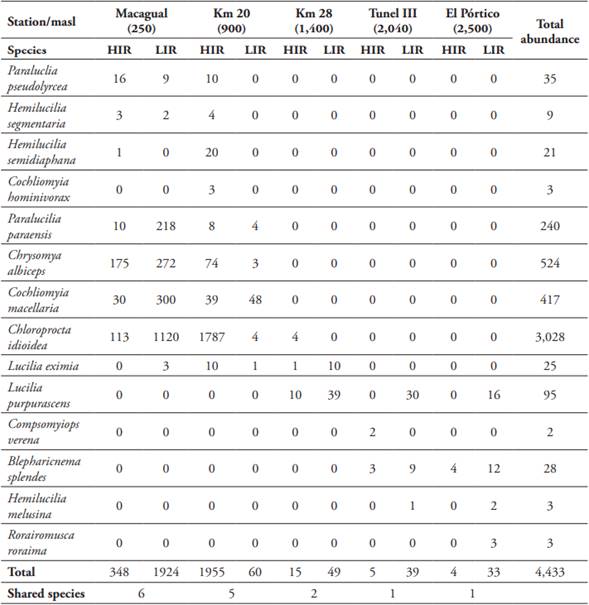
In total, 4,433 specimens of the family Calliphoridae were collected, distributed in nine genera and 14 species: Blepharicnema splendes Macquart, 1843; Chloroprocta idioidea (Robineau-Desvoidy, 1830); Chrysomya albiceps (Wiedemann, 1819); Cochliomyia hominivorax (Coquerel, 1858); Cochliomyia macellaria (Fabricius, 1775); Compsomyiops verena (Walker, 1849); Hemilucilia melusinaDear, 1985; Hemilucilia segmentaria (Fabricius, 1805); Hemilucilia semidiaphana (Rondani, 1850); Lucilia eximia (Wiedemann, 1819); Lucilia purpurascens (Walker, 1836); Paralucilia paraensis (Mello, 1969); Paralucilia pseudolyrcea (Mello, 1969) and Roraimomusca roraima Townsend, 1935 (Table 2) (Annex 1, fig. 7).
The most abundant species were C. idioidea with 3,028 specimens (68.24%), C. albiceps with 524 (11.81%), C. macellaria with 417 (9,40%) and P. paraensis with 240 (5.41%). Six species had intermediate abundances: L. purpurascens with 95 specimens (2.14%), P. pseudolyrcea with 35 (0.79%), B. splendes with 28 (0.63%), L. eximia with 25 (0.56%), H. semidiaphana with 21 (0.47%) and H. segmentaria with nine (0.20%).
H. melusina, R. roraima, and C. hominivorax had three specimens (0.07%) each, and C. verena only two (0.05%) (Table 2).
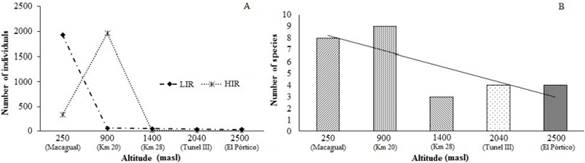
The rarefaction curve for the sampling areas shows that Km 20 and Macagual were the most diverse localities with nine and eight species respectively, while Km 28 and Tunel III were the least diverse localities, each with three species (Fig. 1). The species accumulation curve shows that the observed species correspond to 100% of those expected by the Chao 1 estimator, 98% of the species expected by the Chao 2 estimator, 91.5% of the species expected by Jack 1 and 89.1% of the species expected by the estimator Jack 2, additionally the curve tends to asymptote, indicating that the sampling was adequate (Fig. 2).

Source: Own elaboration.
Figure 1 Rarefaction curve of species of Calliphoridae in each altitudinal range in the Colombian Andean Amazon.

Source: Own elaboration.
Figure 2 Accumulation curve of species of Calliphoridae in all altitudinal ranges in the Colombia Andean Amazon.
HIR season had higher abundance values than the LIR season, 2,330 specimens (52.5%) in 12 species for the former, and 2,107 specimens (47.5%) in 11 species for the later, however, these did not have statistically significant differences (ANOVA: F = 0.86, p = 0.3; Tukey HSD test: p > 0.05). The sampling locations were not statistically different either (ANOVA: F = 3.72, p = 0.007; Tukey HSD test: p > 0.05); Macagual was the most abundant locality with 2,272 specimens (51.2%) and eight species (57.1%), followed by Km 20 with 2,015 specimens (45.45%) and nine species (64.2%), Km 28 with 64 specimens (1.44%) and three species (21,4%), finally the least abundant localities were Tunel III and El Pórtico with 44 (0.9%) and 37 specimens (0.83%) respectively and four species each (28.5 %) (Table 2; Fig. 3A).
Source: Own elaboration.
Figure 3 A: Abundance of species of Calliphoridae during climatic seasons estudied in the Colombian Andean Amazon. B: Richness of species of Calliphoridae in each altitudinal range.
Of the 14 species found, six are new records for the department of Caquetá: H. melusina, P. paraensis, C. hominivorax, C. verena, R. roraima, and L. purpurascens.
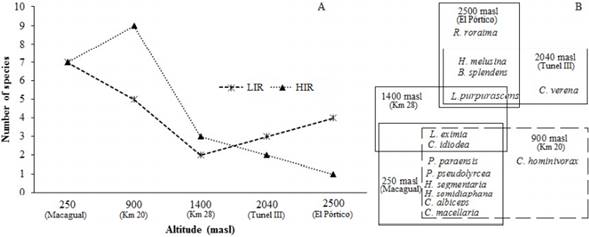
Species richness. The low altitudinal ranges 250 masl (Macagual) and 900 masl (Km 20), presented the highest species richness values, indicating an inverse relationship of the number of species conerning altitude (Fig. 3B). Highest richness values by altitudinal station were not the same during HIR and LIR seasons, the highest number of species during HIR was at 900 masl (Km 20), 250 masl (Macagual) and 1,400 masl (Km 28) with nine, seven and three species respectively, and at levels of 250 masl (Macagual), 900 masl (Km 20) and 2,500 masl (El Pórtico) with seven, five and four species respectively during LIR. The altitude level with the lowest species richness was 2,500 masl with one species during the HIR (Fig. 4A). H. semidiaphana, C. hominivorax, and C. verena were collected only during the HIR season, meanwhile, R. roraima and H. melusina were exclusive of the LIR season (Table 2).
Table 2 Abundance of Calliphoridae species in the eastern slope of the Eastern Cordillera in the Colombian Andean Amazon.
Source: Own elaboration.
Diversity. Diversity values were highest at lower altitude stations, the lowest station (Macagual) (H = 2.39) was the most diverse during both seasons. Although richness of species decreased linearly with altitude, diversity values did not, there was a second increase at middle altitudes during the HIR season at the altitude of 1,400 masl (Km 28) and 2,040 masl (Tunel III) for LIR, then the diversity for both seasons decreases to its minimum at 2,500 masl (El Pórtico) (Table 3). The highest Dominance values were at 1,400 masl (Km 28) (D = 0.67) during the LIR season and at 900 masl (Km 20) (D = 0.65) during the HIR season (Table 3).
Table 3 Index of diversity of Calliphoridae species in the eastern slope of the Eastern Cordillera in the Colombian Andean Amazon.
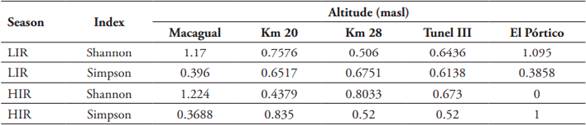
LIR: Low-Intesity Rain season, HIR: High-Intensity Rainy season.
Source: Own elaboration.
Altitudinal distribution and abundance at each altitude level. Station 1. “Macagual”, Vereda La Viciosa (250 m) - A total of 2,272 specimens of eight species were collected (Fig. 4B). The most abundant species for the two sampling seasons were C. idioidea with 1,233 specimens (54.27%), C. albiceps with 447 (19.67%), and C. macellaria with 330 (14.52%). The least abundant species for both seasons were P. pseudolyrcea, H. semidiaphana, H. segmentaria, P. paraensis and L. eximia, (Table 2). In LIR season a total of 1,924 specimens of six species were collected (43.36%). C. idioidea was the most abundant with 1,120 specimens (49.3%), followed by C. macellaria with 300 specimens (13.2%) and C. albiceps with 272 specimens (11.97%). In HIR season 348 specimens of six species were collected (7.84%), being the most abundant C. albiceps with 175 specimens (7.70%), followed by C. idioidea with 113 (4.97%) and C. macellaria with 30 (1.32%) (Table 2).
Source: Own elaboration.
Figure 4 A: Richness of species of Calliphoridae in each altitudinal range during the climatic seasons studied. B: Replacement of species of Calliphoridae in each altitudinal range in the Colombian Andean Amazon.
Station 2. “Km 20”, Vereda Villaraz (900 m) - A total of 2,018 specimens of nine species were collected (Fig. 4B). This altitudinal level shares the most abundant species with Macagual: C. idioidea with 1,791 specimens (88.75%). C. macellaria with 87 specimens (4.31%) and C. albiceps with 77 specimens (3.82%). The less abundant species were P. pseudolyrcea, H. semidiaphana, H. segmentaria, P. paraensis, L. eximia and C. hominivorax (Table 2). In LIR season a total of 1,958 specimens (97%) of eight species were collected. Again, the most abundant one was C. idioidea with 1,787 specimens (88.55%), followed by C. albiceps with 74 specimens (3.67%) and C. macellaria with 39 specimens (1.93%). During HIR season a significantly lower numbers were collected (60 specimens, 1.3%). C. macellaria was the most abundant species with 48 specimens (2.38%), followed by P. paraensis and C. idioidea with four specimens (0.2%) each (Table 2).
Station 3. “Km 28”, Vereda Los Lirios (1400 m) - A total of 64 specimens were collected, distributed in three species (Fig. 4B) L. purpurascens 49 specimens (76.5%), L. eximia 11 specimens (17.19%) and C. idioidea four specimens (6.25%) (Table 2). During the LIR season a total of 49 (76.5%) specimens were collected, L. purpurascens with 39 specimens (60.94%) was the most abundant, followed by L. eximia with 10 specimens (15.63%). During the HIR season 15 specimens (23.4%) were collected. The most abundant species were L. purpurascens (10 specimens (15.63%)), C. idioidea (four specimens (6.25%)) and L. eximia (one individual (1.56%)) (Table 2).
Station 4. “Tunel III”, Vereda Las Brisas (2040 m) - A total of 45 specimens of four species were collected (Fig. 4B): L. purpurascens with 30 specimens (66.67%), B. splendes with 12 specimens (26.67%), two specimens of C. verena (4.44%), and one H. melusina (Table 2). In the LIR season 40 specimens of three species were collected (88%), L. purpurascens with 30 specimens (66.67%) was the most abundant, followed by B. splendes with nine specimens (20%) and H. melusina with one individual (2.22%). In the HIR season five specimens were collected (11%), B. splendes with three specimens (6.67%) and C. verena with two specimens (4.44%) (Table 2).
Station 5. “El Pórtico” (2500 m) Vereda El Vergel -A total of 38 specimens were collected, distributed in four species (Fig. 4B) L. purpurascens with 17 specimens (44.74%), B. splendes with 16 specimens (42.11%), R. roraima with three specimens (7.89%) and H. melusina with two specimens (5.26%) (Table 2). In LIR 34 specimens were collected (89%), L. purpurascens with 17 specimens (44.74%), B. splendes with 12 specimens (31.58%), R. roraima with three specimens (7.89%), during the HIR four specimens were collected (10.53%), everyone B. splendes (Table 2).
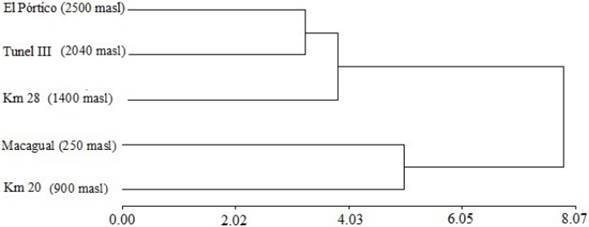
Station similarity and species altitudinal restrictions. The resulting dendrogram from the cluster test separates two main groups. The first one includes the highaltitude stations of Km 28, Tunel III, and El Pórtico. However, Km 28 is farther away from the other two stations due to the presence of low altitude species as C. idioidea. The second group comprises the Macagual and Km 20 stations (Fig. 5). The FCA shows the association of the species according to their abundance with each altitude sampled. C. idioidea, P. paraensis, C. albiceps, C. macellaria, P. pseudolyrcea, H. semidiaphana, and H. segmentaria inhabit areas between the 250 masl (Macagual) and 900 masl (Km 20) and C. hominivorax is restricted to 900 masl (Km 20). L. eximia and L. purpurascens prefer intermediate altitudes around 1,400 masl (Km 28), but L. purpurascens can also be found close to 2,040 masl (Tunel III) associated with C. verena, H. melusina, and B. splendes. R. roraima is the only species that prefer high altitude habitats near and above 2500 masl (El Pórtico) (Fig. 6).
Source: Own elaboration.
Figure. 5 Dendrogram generated of conglomerate analysis of species of Calliphoridae in each altitudinal range in the Colombian Andean Amazon using as measure Euclidean distance.
Discussion
The genera and species here reported double the number of genera and increases by three the number of species published by Baz, Cifrián, Díaz-Aranda & Martín-Vega (2007), but it is important to consider that there are significant geographical differences between the study sites than can explain this difference. Additionally, in that same paper, the authors captured almost five times more specimens than in this study (19,633 vs. 4,433).
Species richness values were negatively correlated with altitude, having higher values at lowland stations and lowest at highland stations, supporting similar research where the number of species decreases with an increase in altitude (Stevens, 1992). Decomposition rate at lower altitudes may influence these results, since baits are usually more attractive as a consequence of a faster decomposition due to higher temperatures and humidity, attracting more species.
All diversity values, as with species richness, were higher at the Macagual (250 masl), as a consequence of having the highest species richness and abundance as well as the fact that this is the most isolated and the least anthropized station. All the other sampling stations were located on the main road leading from Florencia to the municipality of Suaza, Huila. It is known that roads are linked to accelerated deforestation and fragmentation processes, inadequate land use, human settlings, decrease and pollution of water sources, the alteration of trophic chains, and soil degradation, causing loss of biodiversity (Instituto Amazónico de Investigaciones Científicas [SINCHI], 2011).
Chloroprocta idioidea is the only reported species of the genus for South America (Dear, 1985). It was found from 250 masl (Macagual) to 1,400 m (Km 28), agreeing with Pape et al. (2004) and Wolff & Vélez (2007), who report this species in tropical lowlands below 400 masl. However, Wolff (2010) reports it up to 1,900 m. Additionally, Baumgartner & Greenberg (1985) and Montoya et al., (2009) show that the main attractant for this species is rotting fish, the same bait as the one used in this study and possible cause for its dominance at the sampling stations. The habitat where it was collected is a well-preserved forest patch included in the Amazon Forest Reserve zone (SINCHI, 2011), coinciding with Montoya et al. (2009) regarding the preference of this species for forested areas.
Chrysomya albiceps is originally from Africa and Australia and was introduced to America in the 70s. Several studies show that it affects negatively the native communities of carrion feeders, displacing them from their original niches (Aguiar-Coelho et al., 1995). Ramos-Pastrana et al. (2014, 2018) indicate that C. albiceps and C. macellaria have similar behavior as early carrion colonizers. This study found that C. albiceps was more abundant than C. macellaria, probably because the C. albiceps is a tolerant generalist species that has adapted rapidly to different environments, including urban areas with a high synanthropic rate (Baumgartner & Greenberg, 1985; Paraluppi, 1996; Prado & Guimarães, 1982). According to Pape et al. (2004) and Wolff & Vélez (2007), C. albiceps is widely distributed and can be found between 0 and 2,700 masl. However, in this study it was only collected between 250 m (Macagual) and 900 m (Km 20), coinciding with the climate preferences proposed by Montoya et al. (2009), where the temperature and humidity are highest. In Colombia it is commonly found in rural and urban environments, behaving as an endophilic and exophilic species (Wolff & Vélez, 2007).
Cochliomiya macellaria is a native, usually dominant species in neotropical areas (Baumgartner & Greenberg, 1985). It was collected at 250 and 900 masl, similar to the altitudinal range found in Pape et al. (2004); Wolff & Vélez (2007), and Ramos-Pastrana et al. (2014). Ramos-Pastrana et al. (2018) claim that it is frequently found between 0 and 1,500 masl in the Peri-Caribbean Belt, Amazonia, and Norandina biogeographic provinces. Flies of this genus are of great sanitary importance because they are vectors of entomopathogens, such as viruses, bacteria, and helminths (Lima & Luz, 1991) and can cause myiasis in animals and humans (Guimarães et al., 1983). Additionally, Ramos-Pastrana & Wolff (2017); Ramos-Pastrana et al. (2014) and Ramos-Pastrana et al. (2018) report it as of great forensic interest for the Andean Amazon region.
Cochliomyia hominivorax is considered one of the main causes of human and animal myiasis. Its larvae, which feed gregariously, produce deep wounds characterized by liquefactive necrosis where hemorrhage can occur. Adult flies are attracted by body odors, dying animals, purulent material and wound secretions (Baumgartner & Greenberg, 1985), but despite these food preferences, Esposito et al. (2010) were able to collect only one individual in lowland areas at approximately 75 masl using decomposing animal material and according to the results obtained in this study, only three specimens were collected using rotting fish in the station located at 900 masl (Km 20).
Hemilucilia segmentaria has locality records for the departments of Amazonas, Caquetá, and Putumayo (Pape et al., 2004; Ramos & Wolff, 2011) between 300 and 1900 masl (Wolff & Vélez, 2007). In Peru, it was collected in lowland rain forests, with a strong preference for well-preserved forests (Baumgartner & Greenberg, 1985). Similar results were obtained in Antioquia where it was found exclusively forested areas (Montoya et al., 2009). H. segmentaria was collected at the lowest sampling station, which has the most extensive patch of tropical humid forest and at 900 masl (Km 20), station located on the edge Amazon Forest Reserves (SINCHI, 2011). This species is also of sanitary importance, according to Marinho et al. (2003) it is considered as mechanical vector of eggs of Dermatobia hominis (Linnaeus), another important myiasis causing agent in humans and animals. Although Hemilucilia semidiaphana has been found between 0 and 2,500 masl (Wolff & Kosmann, 2016), it was only collected in a narrow altitudinal range in the 250 masl (Macagual) and 900 masl (Km 20) stations, supporting the findings of Baumgartner & Greenberg (1985). Hemilucilia melusina is been previously found in the Andes region (Wolff & Kosmann, 2016), distribution supported by this study, where it was collected at 2,040 (Tunel III) and 2,500 masl (El Pórtico). Baugartner & Greenberg (1985) describe it as a rare asynchronous species, mainly attracted to decomposing fish, with a forensic value that can be used in case studies at these altitudinal levels.
Lucilia eximia was collected at 250 (Macagual), 900 (Km 20) and 1,400 masl (Km 28), as found by Wolff & Vélez (2007) who describe it as a generalist species, capable of inhabiting a wide range of bioclimatic zones, between 0 and 2,600 masl. It is found in rural and urban areas attracted to rotten fruit, garbage, human feces, and animal carcasses, reaching these substrates to feed and reproduce and oviposit (Madeira et al., 1989; Prado & Guimarães, 1982). Given its food resource preferences, it is considered as a pathogen vector, capable of causing secondary myiasis in humans and primary in animals (Madeira et al., 1989). It is considered of forensic importance in the Andean Amazon Region (Ramos-Pastrana et al., 2014, 2018). According to Wolff & Vélez (2007) Lucilia purpurascens may be found in Andean areas between 1,900 and 2,800 masl, associated with both rural and forest habitats with scarce human intervention. This species was collected at the 1,400 masl (Km 28), extending its known minimum altitude distribution.
Paralucilia paraensis is registered for Colombia in the departments of Cundinamarca (Wolff & Kosmann 2016) and Caquetá (Ramos-Pastrana et al., 2018). It is of forensic importance given its feeding preferences as an animal carrion feeder and can be used in studies cases that may occur at several altitudinal levels in the Andean Amazon.
Amat & Wolff (2007) consider that Blepharicnema splendes is a species restricted to high altitude areas in the Andes, usually above 2,500 masl, in relatively undisturbed habitats. This species was collected in the 2,040 (Tunel III) and 2,500 masl (El Pórtico), supporting these previous findings. B. splendes is a large species, compared to other carrion feeding flies, possibly an adaptation to the climatic stressors found at high altitudes including strong winds and low temperatures.
Compsomyiops veren is a synanthropic species that prefers altitudes near 1,500 masl (Baumgartner & Greenberg, 1985), however, Martínez et al. (2007) and Barrios & Wolff (2011) record it at 3,035 and 2,614 masl, respectively, using carrion in moderately to well-preserved areas in the Andes. Given its feeding habits, this species can be considered of forensic importance and can be used in case studies in this type of habitat.
Roraimomusca roraima is an Andean species that lives above 2,500 masl (Wolff, 2010). Little is known about the ecology of this fly, with few specimens captured since its description. Only three specimens were collected at the 2,500 masl (El Pórtico).
Paralucilia pseudolyrcea has been registered in Argentina (Mariluis & Scnack, 2002) and Colombia (Montoya et al., 2009) and it is considered as a synanthropic species, collected with decomposing animal material at altitudes near 600 masl. In this study it was collected at 250 (Macagual) and 900 masl (Km 20). It is also considered a rare species, with no forensic importance.
Conclusions
The distribution, diversity, and composition of Calliphoridae species along the eastern slope of the Eastern Mountain Range, in the Colombian Andean Amazon, is influenced by weather conditions. H. semidiaphana, C. hominivorax, and C. verena were species collected only in the HIR season, while R. roraima and H. melusina were exclusive to the LIR season. Although it was not evaluated directly, the degree of habitat disturbance can also be affecting this study results, and species presence or absence can be the result of habitat loss. Richness of species of the Calliphoridae on the eastern slope of the Eastern Andes is inversely proportional to the altitudinal level, with more species at low altitudes. Macagual and Km 20 are the lowest sites with set conditions that support a higher number of calliphorid species, mainly adequate temperature and humidity and accelerated decomposition rates.
















