Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Colombiana de Biotecnología
Print version ISSN 0123-3475
Rev. colomb. biotecnol vol.18 no.1 Bogotá Jan./June 2016
https://doi.org/10.15446/rev.colomb.biote.v18n1.57714
DOI: http://dx.doi.org/10.15446/rev.colomb.biote.v18n1.57714
ARTÍCULO DE INVESTIGACIÓN
Protein disulfide isomerase and heat shock cognate protein 70 interactions with rotavirus structural proteins using their purified recombinant versions
Interacciones de las proteínas disulfuro isomerasa y de choque térmico Hsc70 con proteínas estructurales recombinantes purificadas de rotavirus
Luz Y. Moreno*, Carlos A. Guerrero*, Orlando Acosta*,**
* Departamento de Ciencias Fisiológicas, Facultad de Medicina, Universidad Nacional de Colombia, Sede Bogotá, Colombia.
** Correspondence autor: C. A. Guerrero, caguerrerof@unal.edu.co
Recibido: marzo 15 de 2015 Aprobado: abril 15 de 2016
Abstract
Introduction. Rotavirus entry into cells seems to be mediated by sequential interactions between viral structural proteins and some cell surface molecules. However, the mechanisms by which rotavirus infects target cell are still not well understood. There is some evidence showing that rotavirus structural proteins VP5* and VP8* interact with some cell surface molecules. The availability of recombinant rotavirus structural proteins in sufficient quantity has become very important for the identification of the specific virus-cell receptor interactions during the early events of the infectious process.
Objective. The aim of the present work is to perform an analysis of the interactions between recombinant rotavirus structural proteins VP5*, VP8* and VP6, and cellular proteins Hsc70 and PDI using their purified recombinant versions.
Materials and methods. Rotavirus recombinant VP5* and VP8*, and cellular recombinant proteins Hsc70 and PDI were expressed in E. coli BL21(DE3) while VP6 was expressed in recombinant vaccinia virus-transfected MA104 cells. The interaction between rotavirus and cellular proteins was studied using ELISA, co-immunoprecipitation and SDS-PAGE/Western blotting analysis.
Results. The optimal conditions for expression of recombinant proteins were determined and antibodies were raised against them. The findings suggested that viral proteins rVP5* and rVP6 interact with Hsc70 and PDI in vitro. These viral recombinant proteins were also found to interact with raft-associated Hsc70 in a cell culture system. The treatment of cells with either rVP6 or DLPs produced significantly inhibition of rotavirus infection.
Conclusion. The results allow us to conclude that rVP5* and rVP6 interact with Hsc70 and PDI during the rotavirus infection process.
Key words: rotavirus; recombinant proteins; protein disulfide isomerase; heat shock cognate protein 70.
Resumen
Introducción. La entrada de rotavirus a las células parece estar mediado por interacciones secuenciales entre las proteínas estructurales virales y algunas moléculas de la superficie celular. Sin embargo, los mecanismos por los cuales el rotavirus infecta la célula diana aún no se comprenden bien. Existe alguna evidencia que muestra que las proteínas estructurales de rotavirus VP5* y VP8* interactúan con algunas moléculas de la superficie celular. La disponibilidad de las proteínas estructurales de rotavirus recombinantes en cantidad suficiente se ha convertido en un aspecto importante para la identificación de las interacciones específicas de los receptores virus-célula durante los eventos tempranos del proceso infeccioso.
Objetivo. El propósito del presente trabajo es realizar un análisis de las interacciones entre las proteínas estructurales de rotavirus recombinante VP5*, VP8* y VP6, y las proteínas celulares Hsc70 y PDI utilizando sus versiones recombinantes purificadas.
Materiales y métodos. Las proteínas recombinantes de rotavirus VP5* y VP8* y las proteínas recombinantes celulares Hsc70 y PDI se expresaron en E. coli BL21 (DE3), mientras que VP6 se expresó en células MA104 con virus vaccinia recombinante transfectada. La interacción entre el rotavirus y las proteínas celulares se estudió mediante ELISA, co-inmunoprecipitación y SDS-PAGE/ Western.
Resultados. Las condiciones óptimas para la expresión de proteínas recombinantes se determinaron y se generaron anticuerpos contra ellas. Los resultados sugirieron que las proteínas virales rVP5* y rVP6 interactúan con Hsc70 y PDI in vitro. También se encontró que éstas proteínas virales recombinantes interactúan con Hsc70 en las balsas lipídicas ("Rafts”) en un cultivo celular. El tratamiento de las células, ya sea con DLP o rVP6 produjo significativamente la inhibición de la infección por rotavirus.
Conclusión. Los resultados permiten concluir que rVP5 * y rVP6 interactúan con Hsc70 y PDI durante el proceso de la infección por rotavirus.
Palabras clave: rotavirus; proteínas recombinantes; proteína disulfuro isomerasa; proteína de choque térmico Hsc70.
Introduction
Rotavirus infection has been recognized as the leading cause of severe dehydrating diarrhea in infants and young children worldwide (Parashar et al., 2006; Esposito et al., 2011 ), being responsible for more than half a million annual deaths globally (Parashar et al., 2006). Rotaviruses are non-enveloped viruses containing an 11-segmented double-stranded (ds) RNA genome enclosed by a triple-layered protein capsid (Pesavento et al., 2006; Estes et al., 2007). The outermost protein layer is composed by VP4 and VP7 while the intermediate layer consists of VP6. The innermost layer is built from VP2 which encloses the dsRNA segmented genome, and VP1 and VP3. VP4 requires to be cleaved into two trypsin proteolytic products, VP5* and VP8*, for the virus becomes infectious (Estes et al., 1981; Benureau et al., 2005).
Rotaviruses exhibit a remarkable tropism for polarized mature enterocytes from the small intestinal villus epithelium (Ramig 2004). However, primary small intestinal epithelial cell (IEC) cultures have been little used for rotavirus infection purposes (Kaushik et al., 2008; Coulson et al., 2002; Macartney et al., 2000); most studies about rotavirus infection mechanism have been conducted in cultured cell systems such as MA104 cells, a non-differentiated kidney epithelial cell line (Guerrero et al., 2000; Londrigan et al., 2000; Halasz et al., 2008). The polarized epithelial cell line Caco-2 derived from a human colon adenocarcinoma (Fogh et al., 1977) has been also used as a model for studying the mechanisms by which rotaviruses affect intestinal functions (Servin et al., 2003; De Marco et al., 2009; Beau et al., 2007). It has been shown that rotavirus can replicate in Caco-2 cells being then released without destroying them (Jourdan et al., 1997).
The mechanisms by which rotavirus infects target cell are still not well understood. Rotavirus entry seems to be mediated by sequential interactions between viral structural proteins and some cell surface molecules (Lopez et al., 2006). There is evidence showing that sialic acid (SA) interacts with VP8*, the smaller trypsin cleavage product of VP4, during the initial virus binding to cell surface (Isa et al., 2006; Haselhorst et al., 2009). Integrin α2β1 has been also found to mediate binding to cells for some rotavirus strains through direct interaction with VP5*, the larger trypsin proteolytic product of VP4 (Méndez et al., 1993; Zárate et al., 2000). Rotavirus interaction with other integrins such as αxβ2 and αvβ3 through VP7 has been demonstrated during post-binding events (Graham et al., 2003; Guerrero et al., 2000; Zárate et al., 2004). Cell surface heat shock cognate protein 70 (Hsc70) has been shown to interact with VP5 and VP6 during rotavirus entry (Jolly et al., 2001; Guerrero et al., 2000; Zárate et al., 2004; Gualtero et al., 2007). VP5* implication in rotavirus entry has been further extended by proposing that this viral protein could assume fusion-inducing conformational changes similar to those characterized in fusion proteins from enveloped viruses, leading to lipid bilayer perforation (Dormitzer et al., 2004; Trask et al., 2010). Rotavirus interaction with cell surface protein disulfide isomerase (PDI) has been suggested based on the sensitivity of rotavirus infection to DTNB [5,5-dithio-bis-(2-nitrobenzoic acid)], bacitracin, and anti-PDI antibody treatment (Wolf et al., 2011). However, the identity of the rotavirus cell surface receptor and entry mechanisms continuous to be a controversial issue (Trask et al., 2010; Wolf et al., 2011; Ruiz, 2009; Arias, 2002).
Although the presence of Hsc70 and PDI has been documented in MA104 cells and their implication in rotavirus infection has been suggested by different strategies including the use of synthetic peptides derived from rotavirus proteins (Jolly et al., 2001; Guerrero et al., 2002; Zárate et al., 2003; Calderon et al., 2012) and antibodies against these cellular proteins (Calderon et al., 2012), a study of rotavirus-cell interaction using the entire rotavirus structural proteins will further provide important insights into how rotavirus enters into the host cell. The aim of the present work is to perform an analysis of the interactions between recombinant rotavirus structural proteins VP5*, VP8* and VP6, and cellular proteins Hsc70 and PDI in the context of rotavirus entry. Interactions between viral proteins and cellular proteins were followed by ELISA and SDS-PAGE/Western blotting analysis of the immunoprecipitated complexes. Our findings confirm the usefulness of recombinant proteins in the identification of protein interactions during virus entry into the host cell. We show that addition of recombinant VP6 to MA104 cells led to the inhibition of rotavirus infection, suggesting its participation in the virus entry process. The results also extend the knowledge about rotavirus VP5* and VP6 interaction with the cellular proteins Hsc70 and PDI which have been found to be implicated in virus entry into the host cell.
Materials and methods
Expression of recombinant VP5*, VP8*, Hsc70 and PDI
Recombinant VP5* and VP8* fused with glutathione-transferase (GST) were expressed in E. coli BL21(DE3) that had been transfected with pGEX-4T-VP5* and pGEX-4T-VP8* (Pharmacia, Stockholm, Sweden), respectively. Plasmids were kindly donated by Dr. C. F. Arias (Instituto de Biotecnología, Universidad Nacional Autónoma de México - UNAM). VP5* and VP8* are encoded by RRV (G3 P5B [3]) VP4 sequences spanning nucleotides 1 to 750, and 749 to 2347, respectively (Zárate et al., 2000; Isa et al., 1997). Conditions of transfection, expression and purification were previously described (Zhang et al., 2010). The purified VP5*-GST and VP8*-GST in Tris buffer (50 mM Tris-HCl, pH 8.0, 10 mM CaCl2) were treated with thrombin (CleanCleave kit, Sigma, St. Louis, MO, USA) to cleave the GST tag from the fusion protein.
Recombinant Hsc70 and PDI were expressed in E. coli BL21(DE3) which had been transfected with pET-28a(+)-Hsc70 or pET-28a(+)-PDI expression vector (kindly donated by Dr. C. F. Arias, Instituto de Biotecnologia, UNAM, Cuernavaca, Mexico). A cDNA for human Hsc70 in plasmid pETHSC (GenBank accession No. M11717) (kindly provided by R. T. Morimoto, Northwestern University) containing the complete sequence of Hsc70 protein was subcloned into the pET28 expression vector (Novagene) to generate a fusion protein with a six-histidine tail at its COOH-terminus (Vargas et al., 2006). The rat liver PDI cDNA, taken as a TthIII-SalI fragment from pMAL5.1, and kindly donated by Dr. R.T. Raines (Department of Chemistry, University of Wisconsin-Madison) was subcloned into the NdeI-SalI sites of pET28a (Novagen) (after blunting the NdeI site) to produce a fusion protein with a tail of six histidines at its NH2-terminus (Edman et al., 1985; Laboissiere et al., 1995).
BL21(DE3) competent E. coli cells were prepared as previously described (Chung et al., 1989; Moreno et al., 2013) with some modifications. An E. coli colony was resuspended in LB medium and grown overnight at 37 °C with orbital shaking (150 rpm). An aliquot (1 ml) of the culture was inoculated in LB medium (100 ml) to reach an OD 600 nm of 0.3 - 0.4 and then centrifuged (12000 g) for 10 min to collect the cells which were resuspended in 5 ml of freezing medium [10 % polyethylenglicol (PEG) 3350, 5 % dimethyl sulfoxide (DMSO), 100 mM NaCl, 10 mM MgCl2, 10 mM MgSO4, and 10 % glycerol in 60 % LB medium, pH 6.7 - 7.0] and aliquots were kept on ice for 5 min before being stored in liquid nitrogen. An aliquot was mixed with 10 ng of pET-28a(+)-Hsc70 or pET-28a(+)-PDI expression vector dissolved in 100 ml of a solution containing 100 mM KCl, 30 mM CaCl2 and 50 mM MgCl2. The suspension containing bacteria and plasmid DNA was gently agitated and incubated on ice for 20 min before heat shock treatment at 42 °C for 2 min. The transfected bacteria were seeded in LB-agar plates with 50 µg/ml kanamicin and incubated for 14 h at 37 °C.
A colony from bacteria transfected with pET-28a(+)-Hsc70 was inoculated in 25 ml of LB medium containing 50 µg/ml kanamicin and incubated for 12 h at 37 °C with orbital shaking (150 rpm). An aliquot (5 ml) from this culture was inoculated into 500 ml of LB medium supplement with 50 µg/ml kanamicin and 2 % glucose followed by incubation at 37 °C with orbital shaking (150 rpm) until the OD 600 nm profile indicated that the exponential growth phase was reached. Isopropyl thiogalactoside (IPTG) (0.5 to 2 mM final concentration) was added to the culture at this growth phase followed by further incubation for 2, 4, 6 or 8 h. In the case of bacteria transfected with pET-28a(+)-PDI, the procedures were as those described for Hsc70 expression vector, except that 2XYT medium was used. Recombinant protein expression was assessed by SDS-PAGE/Western blotting analysis using non-transfected BL21(DE3) cells and transfected BL21(DE3) cells without IPTG treatment as a control. The total protein loaded into each gel well was spectrophotometrically quantified at 280 nm using a BSA standard curve.
Solubilization and purification of recombinant Hsc70 and PDI
Solubilization of rHsc70 and rPDI from BL21(DE3) lysates was performed following the procedure reported (Frangioni et al.,1993) with some modifications. Bacteria were collected by centrifugation at 8000 g for 10 min and washed twice with PBS. Cell pellets were resuspended in 50 ml PBS, treated with 100 µg/ml of lysozyme for 15 min at 4 °C with constant agitation. The suspension was made 5 mM in EDTA, 1% in sodium N-lauroylsarcosine and sonicated for 4 cycles of 15 sec at 20% amplitude before centrifugation at 12000 g for 15 min at 4 °C. Triton X-100 at 1% was added and the presence of rHsc70 and rPDI was analyzed by SDS-PAGE and Western blotting.
Recombinant proteins were purified using a 10 ml His Trap (BioRad) affinity column. The column was washed with 5 volumes of 50 mM phosphate buffer containing 300 mM NaCl and equilibrated with 5 volumes of the same buffer added with 10 mM imidazole. Cell lysates were also added with 10 mM imidazole before applying on the column. Following washing with phosphate buffer containing 10 mM imidazole until no protein was detected with Bradford's method, recombinant proteins were eluted from the column using increasing imidazole concentrations (0.25 - 1 M) in phosphate buffer. The column was finally washed with 10 volumes of H2O and 5 volumes of 30% ethanol.
Expression and purification of recombinant VP6
Recombinant VP6 was expressed in MA104 cells that had been infected with a recombinant vaccinia virus containing the rotavirus SA11 (G3 P5B (Esposito et al., 2011) gene encoding structural VP6 (kindly donated by Dr. M. Franco, Instituto de Genetica, Universidad Javeriana). Recombinant VP6 containing 397 amino acids is encoded by a VP6 sequence spanning nucleotides 1 to 1362 (Tang et al., 1997). Cells were cultured in Eagle Dulbecco's modified medium (DMEM) supplemented with 2% fetal bovine serum (FBS) and 2 mM L-glutamine at 37 °C in a 5% CO2 incubator. Cells at 80 - 100% confluence were washed twice with PBS and infected with the recombinant vaccinia virus present in a MA104 cell lysate for 1 h at 37 °C. The remaining cell lysate containing the virus was removed and cells were incubated in Eagle minimal essential medium (MEM) for 24 h at 37 °C.
To identify the subcellular fraction in which VP6 is located, the vaccinia virus-infected cells were frozen and thawed 3 times at - 70 °C, sonicated for 2 cycles of 15 sec at 20% amplitude and centrifuged at 15000 g for 15 min at 4 °C. The soluble fraction (SF) was kept and the insoluble fraction (IF) washed twice with Tris buffer (20 mM Tris-HCl, pH 8.0, 150 mM KCl). The IF was treated with 1% Triton X-100 and 8 M urea before SDS-PAGE/Western blotting analysis. Solubilization of VP6 from the previously Tris buffer-washed IF was attempted by resuspension in 1 ml RIPA buffer (10 mM Tris-HCl, pH 7.4, 0.1% SDS, 1% Triton X-100, 1% sodium deoxycholate and 150 mM NaCl 150) and incubation for 1 h at 4 °C before centrifugation at 12000 g for 10 min at 4 °C. The precipitated fraction was treated with 500 µl of Tris buffer containing 1% N-lauroylsarcosine, CHAPS, β-octylglucoside, Triton X-100, Tween 20, Nonidet P40 or sodium deoxycholate. The solubilized pellets were incubated overnight at 4 °C and then centrifuged at 12000 g for 10 min at 4 °C. The remaining pellet was further treated with 500 µl of 8 M urea dissolved in buffer Tris (20 mM Tris-HCl, pH 8.0, 150 KCl, 1% sodium lauroyl sarcosine). Proteins in each fraction were separated by SDS-PAGE, analyzed by Western blotting and compared with samples from uninfected cells.
Purification of rVP6 was also attempted by a sequential treatment of the IF from recombinant vaccinia virus-infected cell lysates. The IF was sequentially treated with RIPA buffer followed by treatment with 1% β-octylglucoside, 8 M urea, and 8 M urea plus 1% N-lauroylsarcosine in Tris buffer. The sequential pellets were obtained by centrifugation at 12000 g for 10 min at 4 °C. The different solubilized fractions were analyzed by SDS-PAGE/Western blotting using rabbit polyclonal antibodies against rotavirus particles. The solubilized fractions containing rVP6 were dialyzed against a decreasing gradient of urea in PBS, diluted 10 times in PBS and finally concentrated using Amicon Ultra - 4 (cut-off 10 kD; Millipore, Billerica, MA, USA).
Production of antibodies against rVP6 and rHsc70
Purified rHsc70 or partially purified rVP6 were separated in preparative 12% SDS-PAGE. Proteins in a gel lane were transferred onto a PVDF membrane and rHsc70 or rVP6 were identified by Western blotting using rabbit polyclonal antibodies against either of these proteins, HRP-conjugated secondary goat anti-rabbit antibodies and 3-amino-9-ethylcarbazole (AEC) substrate. Gels were stained with Coomassie blue and the identified proteins bands were excised from gels and distained in methanol-acetic (50% - 12.5%) solution before being equilibrated in PBS. Gel pieces were crushed in PBS and the slurry passed several times through an 18-, 21- and then 23-gauge needle. Three month-old New Zealand rabbits were subcutaneously inoculated in the back with 750 µg of either recombinant protein emulsified in complete Freund's adjuvant. Each rabbit received 3 injections spaced at 20 days intervals. Rabbits were bled 10 days after the final immunization and sera were obtained and mixed (1:1; v/v) with glycerol before storing at - 20 °C.
The antibodies raised against recombinant proteins were characterized by ELISA, immunochemistry and SDS-PAGE/Western blotting. ELISA plates were coated with growing concentrations (2.5 - 20 µg/ml) of rVP6 in PBS, blocked with 1% BSA and reacted with rabbit antibodies against rVP6 diluted (1: 1000) in PBS containing 0.05% Tween 20 and 0.1% BSA. Detection was performed with HRP-conjugated goat anti-rabbit antibodies and AEC in 50 mM Na-acetate buffer, pH 5.0, and 0.04% H2O2. Immunocytochemistry analysis was carried out by infecting MA104 cell monolayers in 96-well culture plates with purified trypsin activated RRV triple layered particles (TLPs) (MOI of 0.05). Cells were incubated for 1 h at 37 °C, washed twice with MEM and further incubated in MEM for 12 h at 37 °C. Cells were fixed in ice-cold methanol for 40 min and treated with dilutions (1:250 - 1:2000) of rabbit anti-rVP6 antibodies in PBS for 1 h at 37 °C. HRP-conjugated secondary antibody and AEC substrate in 50 mM Na-acetate buffer, pH 5.0, and 0.04% H2O2 were used for developing the reaction. Western blotting analysis of rabbit antibodies to rVP6 and rHsc70 was conducted using as antigen both purified recombinant proteins and cell lysates containing the recombinant proteins. Purified RRV TLPs (500 ng/well) were also used as antigen to test anti-rVP6 antibodies. Lysates (25 - 100 µg protein/well) from non transfected cell were used as a control. Rabbit antibodies diluted (1:1000) in PBS containing 0.05% Tween 20 and 0.1% skimmed milk were added to membranes. Detection limit for anti-rVP6 serum was determined using dilutions (1:250 - 1:1000) of this serum to detect 250 ng/well of purified rVP6. HRP activity present in the secondary antibody used was visualized as described above for the immunochemistry assay.
Interaction between rotavirus structural proteins and cellular Hsc70 and PDI
Rotavirus structural proteins rVP5*, rVP8* and rVP6 were tested for their interaction with cellular rHsc70 and rPDI. ELISA was performed by coating 96-well plates with 40 μg/ml of rHsc70 or rPDI in PBS at 4 °C overnight and blocking with 1% BSA in PBS at 37 °C for 2 h. After washing with PBS, increasing concentrations (1 - 20 µg/ml) of either rotavirus recombinant protein in the same buffer were added and incubated at 37 °C for 2 h. Wells were washed with PBS before addition of rabbit polyclonal antibodies (1:1000) against rotavirus proteins in blocking buffer and incubation at 37 °C for 2 h. Antibodies against rVP5* y rVP8* were produced in our animal facilities (Zhang et al., 2010). Reaction was detected using HRP-conjugated goat anti-rabbit antibodies and ortho-phenylenediamine dihydrochloride (OPD) in substrate buffer (25 mM citric acid, 50 mm Na2HPO4, pH 5.0, containing 0.03% H2O2). For negative controls, 1% BSA was used instead of antibodies or rotaviral and cellular proteins. Data were expressed as mean of two independent experiments performed with duplicate samples. The plate readings were done at 492 nm and the ΔOD values were calculated by subtracting mean values of control samples containing 1% BSA instead of antibodies or rotaviral and cellular recombinant proteins.
Co-immunoprecipitation analysis for interactions between rotavirus proteins and cellular proteins was conducted by incubating equal volumes (100 µl) of 2.5 µg/ml of rVP5*, rVP8* or rVP6 with 10 µg/ml of rHsc70 or rPDI in the presence of 0.1% BSA. Each mix of proteins was maintained at 4 °C overnight prior addition of rabbit policlonal antibodies (1:200) against rotavirus proteins and incubation for 1 h at 37 °C. BSA (4%)-treated Affi-Gel protein A-agarose (10 µl) (Bio-Rad, Hercules, CA, USA) was added to each mix (200 µl) of antibody-treated proteins followed by incubation for 1 h at room temperature, according to the manufacturer's instructions. After incubation at room temperature for 1 h, the resin was washed 3 times with PBS-Tween 20 (0.05%) and collected by centrifugation at 2000 g for 10 min. Protein in the precipitated complex was analyzed by SDS-PAGE/Western blotting using primary anti-Hsc70 or anti-PDI goat polyclonal antibodies followed by addition of HRP-conjugated rabbit anti-goat antibodies and AEC substrate. rHsc70 or rPDI were incubated with antibodies against rVP5*, rVP8* or rVP6 and then immunoprecipitated to be used as a control. Conversely, the mix of viral and cellular proteins was immunoprecipitated with goat polyclonal antibodies (1 µg/ml, Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA) against either Hsc70 or PDI and the Western blotting analysis was performed using rabbit primary antibodies against rVP5*, rVP8* or rVP6. These rotavirus proteins were incubated with antibodies against Hsc70 or PDI and then immunoprecipitated to be used as a control.
Interaction of VP5*, VP6 or VP8* with raft-associated Hsc70
The interaction between rotavirus proteins and Hsc70 in lipid microdomains (rafts) was determined by mixing a MA104 cell suspension (9.2 x 106) (600 µl) with 100 µl of rVP5* (15 µg/ml), rVP8* (15 µg/ml) or rVP6 (1.5 µg/ml) followed by incubation for 1 h at room temperature. The cell viability was assessed using the Tripan blue exclusion test after treatment with recombinant proteins. Cells that had not been treated with rotavirus proteins were used as a control. Cells were washed twice with PBS and incubated with 120 µl of lysis buffer (25 mM Tris-HCl, pH 7.5, 150 mM NaCl, 1 mM CaCl2, 1 mM DTT, 1 % Triton X-100, 1 mM PMSF) during 30 min at 4 °C. The lysate was mixed (1:1; v/v) with 80% sucrose in buffer TNC (25 mM Tris-HCl, pH 7.5, 150 mM NaCl; 1 mM CaCl2) in a centrifuge tube. A discontinuous sucrose gradient (30 - 5%) in TNC was layered onto the lysate and then centrifuged (Sorvall TST 60.4 at 170000 g for 8 h at 4 °C. The gradient was fractionated and fractions were mixed with β-cyclodextrin (10 mM final concentration) and octylglucoside (0.8% final concentration) and incubated at 37 °C for 1 h. The fractions were incubated with goat polyclonal antibodies against Hsc70 (Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA) at 37 °C for 1 h before addition of 30 µl Affi-Gel protein A-agarose and incubation for 30 min at room temperature. Proteins in the immunoprecipitated fractions were analyzed using Western blotting and the separated proteins detected with rabbit polyclonal antibodies against rVP5*, rVP8* or rVP6.
Infectivity inhibition assay
The effect of rVP5*, VP8* or rVP6 on rotavirus infectivity was assessed by adding either protein (1.2 - 40 µg/ml) to a MA104 cell monolayer in 96-well culture plates and incubating for 1 h at 37 °C. Following the treatment of cells with the recombinant proteins, cell viability was assed using the Tripan blue exclusion test. RRV TLPs (50 µl) containing 5 x 104 FFU/ml were inoculated into cells followed by incubation for 1 h at 37 °C. Cell monolayers were washed with PBS and incubated in MEM for an additional 12 h at 37 °C. Infectivity was determined by fixing cells in ice-cold methanol for 40 min and incubating them with rabbit anti-RRV antibodies (1:1000) in PBS for 1 h at 37 °C. HRP-conjugated goat anti-rabbit secondary antibody (Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA) was used at 0.133 µg/ml and developed with AEC substrate. Infectivity was expressed as mean percentage of infected cells.
Results
Expression and purification of rHsc70, rPDI and rVP6
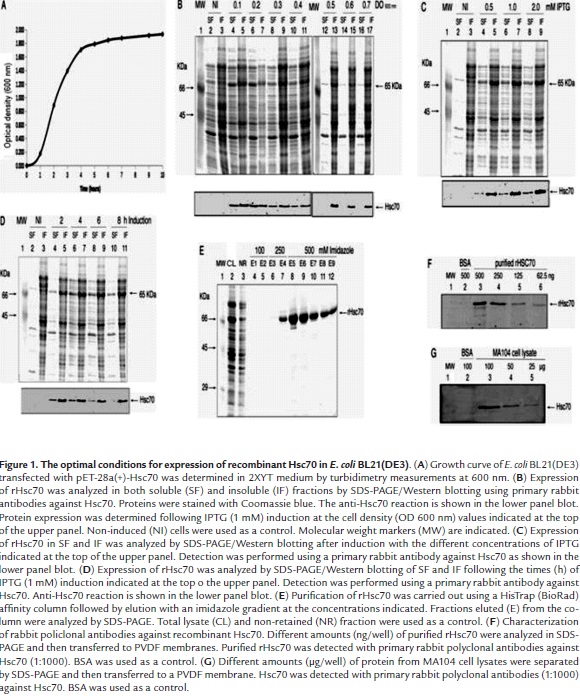
The optimal conditions for expression of pET28a-Hsc70 in E. coli BL21(DE3) were determined. These conditions included bacterial concentration (OD at 600 nm) before induction, induction time, and inductor concentration. Induction assays were conducted in bacterial cultures showing OD 600 nm values that ranged from 0.1 to 0.7 which encompassed the exponential growth phase and the starting of the stationary growth phase. A typical bacterial growth curve is shown in Fig. 1A. Expression of the rHsc70 was followed by SDS-PAGE and Western blotting analysis using rabbit anti-Hsc70 antibodies and taking into account the expected molecular mass (65 kDa) for this protein. In the absence of IPTG, no expression for rHsc70 was found either in the soluble or in insoluble fraction (Fig. 1B, lanes 2, 3). Addition of IPTG (1 mM final concentration) at OD 600 nm 0.1 - 0.4 for 4 h resulted in induction of basal expression of rHsc70 which was detected as a Western blot band present in either the soluble or insoluble fractions (Fig. 1B, lanes 4 - 11). IPTG induction at OD 600 nm 0.5 - 0.7 for 4 h led to rHsc70 expression only in the insoluble fraction (Fig. 1B, lanes 13, 15, 17). The highest amount of rHsc70 expression was obtained when the bacterial culture at OD 600 nm 0.5 was induced with IPTG (1 mM) for 4 h. In this case, rHsc70 represented 1.02% of the total insoluble protein loaded in SDS-PAGE wells (Fig. 1B, lane 13).
Recombinant Hsc70 was found in both the soluble and insoluble fractions when cultures were induced with IPTG at concentrations that ranged from 0.5 to 2 mM (Fig. 1C, lanes 4 - 9) when the culture was induced at OD 600 nm 0.5. However, a higher proportion of rHsc70 was always observed in the lysate insoluble fraction from cultures irrespective of the IPTG concentration used (Fig. 1C, lanes 5, 7, 9). Cultures induced with 1 - 2 mM IPTG showed the highest total accumulation of rHsc70 (1.15%) relative to the total protein content in both fractions (Fig. 1C, lane 6 and 7). Variations in the IPTG concentration produced slight changes in the rate of rHsc70 accumulation and its location. In order to increase the rHsc70 accumulation and its location in the soluble fraction, the induction time effect was assessed between 2 and 8 h by treating the culture at OD 600 nm 0.5 with 1 mM IPTG. The recombinant protein was identified in all lysate fractions from cells induced during the times indicated (Fig. 1D, lanes 4 -11), but a higher accumulation of rHcs70 continued to be observed in the insoluble fraction. However, the highest rHsc70 accumulation (1.39%) was detected in the insoluble fraction relative to the total protein present in both lysate fractions when induction with 1 mM IPTG was initiated at OD 600 nm 0.5 and maintained during 4 at 37 °C. The rHsc70 contained in the insoluble fraction was solubilized by treatment with PBS containing N-lauroylsarcosine and DTT but without EDTA to avoid Ni2+ chelating which interferes with rHsc70 binding to the resin during its purification. Additional treatment with 1% Triton X-100 was performed for refolding the recombinant protein and reducing nonspecific interaction between bacterial proteins and the resin used for rHsc70 purification.
The rHsc70 was efficiently purified as demonstrated by the comparison of the electrophoretic profiles corresponding to the proteins present in the total lysate and the eluted fractions from the resin (Fig. 1E, lanes 2, 3). The analysis of fractions eluted from the resin with an imidazole gradient is shown in figure 1E (lanes 6 - 12). Fractions analyzed in lanes 8, 9 and 10 show the presence of some non-specific protein bands located both in the high and low molecular weight region. Western blotting analysis using rabbit polyclonal antibodies (1:1000) against Hsc70 showed that these antibodies specifically recognized the purified rHsc70 at amounts as low as 62.5 ng/well (Fig. 1F, lane 6), whereas rHsc70 was detected in cell lysates only when samples contained at least 50 µg/well of total protein (Fig. 1G).
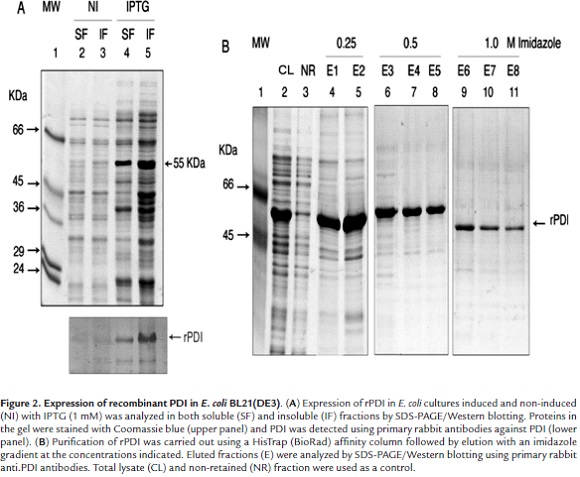
PDI expression was efficiently induced with 0.5 mM IPTG during 4 h at 37 °C. The SDS-PAGE/Western blotting analysis of lysates from the induced cells showed a 55 kDa protein band which was identified as being PDI (Fig. 2A, lanes 4, 5). The PDI purification was carried out by treating cell lysates with PBS containing DTT and sodium N-lauroylsarcosine before passing them through a Ni2+ column. The profile of the fractions eluted with the imidazole gradient (0.25 - 1 M) is shown in Fig. 2B. The highest amount of PDI eluted with 250 mM imidazole although some non-specific protein bands showing higher and lower molecular weight than PDI were observed (Fig. 2B, lanes 4, 5). Lower amounts of PDI were obtained at higher imidazole concentrations (Fig. 2B, lanes 6 - 11).
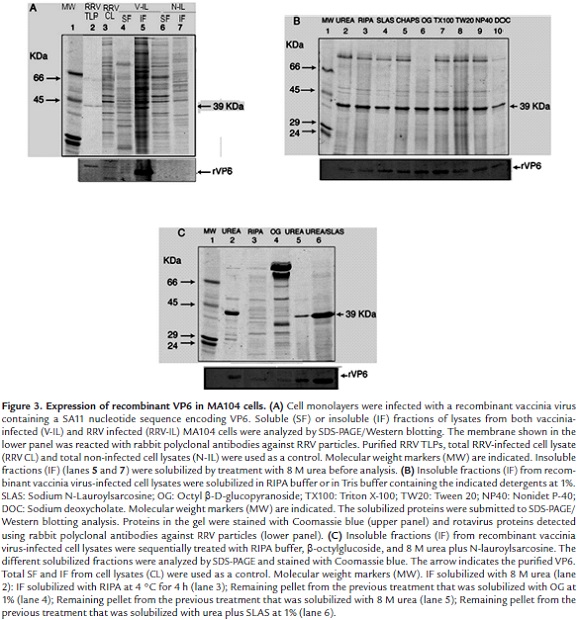
Recombinant VP6 was expressed in MA104 cells after the infection with a recombinant vaccinia virus vector. The Western blotting analysis indicated that a protein band of 39 KDa corresponding to rVP6 was identified predominantly in the IF from the vaccinia virus-infected cell lysates (V-IL) (Fig. 3A, lane 5). Less rVP6 accumulation was detected in the soluble fraction (Fig. 3A, lane 4). This protein band was absent from non-infected cell lysates (N-IL) (Fig. 3A, lanes 6 and 7). Solubilization of rVP6 from the IF was attempted by treatment with RIPA buffer followed by the recovery of a secondary insoluble fraction which was further treated with 1% sodium N-lauroylsarcosine, CHAPS, β-octylglucoside, Triton X-100, Tween 20, Nonidet P40 or sodium deoxycholate. The treatment of the remaining pellets from these detergent treatments with 8 M urea in Tris buffer showed that, despite some contaminant proteins, all the urea-solubilized fractions were enriched in rVP6 (Fig. 3B). In order to eliminate additional contaminant proteins, the standard procedure for rVP6 purification started with RIPA treatment to solubilize some contaminant proteins from the cell lysate insoluble fraction (IF) (Fig. 3C, lane 3), followed by treatment of the resultant pellet with 1% β-octylglucoside (Fig. 3C, lane 4). Then, the remaining pellet was treated with 8 M urea (Fig. 3C, lane 5) and finally with 8 M urea plus 1% sodium N-lauroylsarcosine (Fig. 3C, lane 6). Urea treatment of the IF in the absence of other treatments (Fig. 3C, lane 2) showed that the previous detergent treatments of the IF contributed to decrease contaminant proteins. The solubilized VP6 was dialyzed against PBS and concentrated by ultrafiltration (Amicon Ultra 4, Millipore).
Characterización of antibodies against VP6 and Hsc70
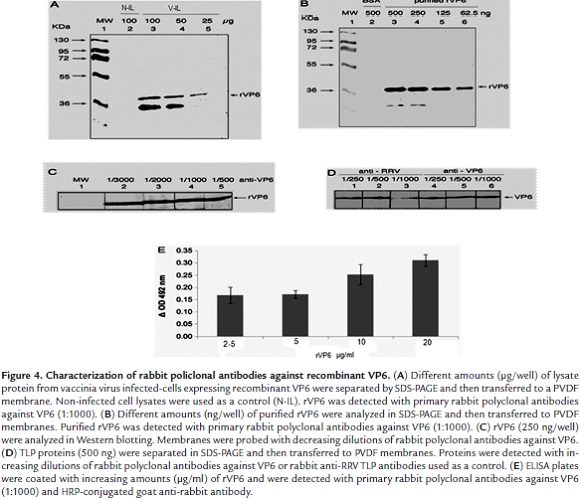
Western blotting analysis of anti-VP6 antibodies showed that these antibodies recognized the corresponding viral antigen in lysates from vaccinia virus-infected cells expressing recombinant VP6 when gel wells were loaded with 25 - 100 µg of total protein (Fig. 4A, lanes 3 - 5). Purified rVP6 (500 - 62.5 ng/well) was also recognized in this analysis (Fig. 4B, lanes 3 - 6). However, a positive reaction corresponding to a lower molecular weight band was seen which could be explained in terms of a degradation product from rVP6 (Fig.4A and B, lanes 3 and 4). Partially purified rVP6 (250 ng) was detected by the highest dilution (1:3000) of the antiserum against rVP6 (Fig. 4C, lanes 2 - 5). VP6 from purified TLPs (500 ng/well) was also detected by Western blotting using anti-VP6 antibodies at dilutions ranging from 1:250 to 1:1000 (Fig. 4D, lanes 4 - 6). ELISA showed increasing absorbance values for samples containing rVP6 (2.5 - 20 µg/ml) (Fig. 4E). Anti-VP6 antibodies at a dilution of 1:2000 showed to be useful for detecting RRV TLP-infected MA104 cells that had been inoculated with a MOI of 0.8 since more than 50% of cells were found to be positive to the viral antigen as detected by the immunochemistry assay (Data not shown).
In vitro interaction of VP5*, VP8* and VP6 with Hsc70 and PDI
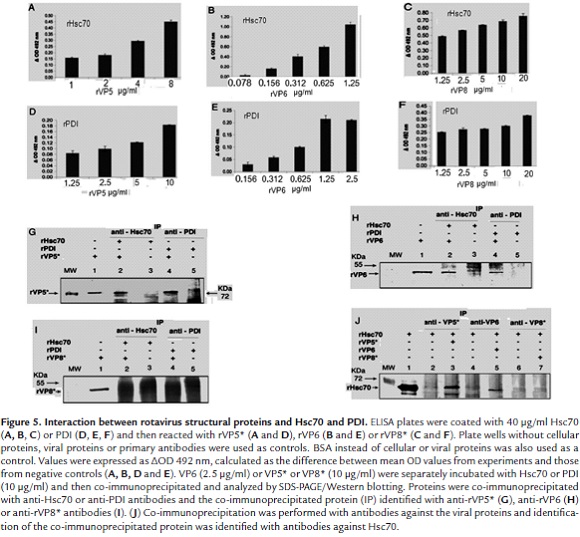
To assess the interaction between rotaviral proteins (VP5*, VP8* or VP6) and cellular proteins (Hsc70 or PDI), ELISA plates were coated with either of the latter proteins (40 µg/ml) and reacted with either rotaviral proteins at the concentrations indicated. Absorbance values showed an increasing profile which was directly proportional to the growing rVP5* concentrations (1 - 8 µg/ml) added to rHsc70 coated wells (Fig. 5A). A similar interaction profile was observed when rHsc70 coated wells were reacted with increasing rVP6 concentrations (0.078 - 1.25 µg/ml) (Fig. 5B). These findings suggest that viral rVP5* and rVP6 interact with Hsc70 at least in the in vitro conditions used. When rPDI coated ELISA wells were reacted with increasing rVP5* concentrations (1.25 - 10 µg/ml), and also with different rVP6 concentrations (0.156 - 2.5 µg/ml), a positive linear correlation with the absorbance values was found (Fig. 5D and 5E). Although absorbance values were relatively low mainly for VP5* (less than 0.2), these results suggest that rVP5* and rVP6 are directly interacting with PDI in vitro. ELISA results for rVP8* interactions with Hsc70 or PDI were negative (Fig. 5C and 5F).
An immunoprecipitation assay was conducted to confirm the interactions found by ELISA. rPDI or rHsc70 (10 µg/ml) were separately incubated with either rVP6 (2.5 µg/ml), rVP5* or rVP8* (10 µg/ml). Immunoprecipitation was performed using specific antibodies against either cellular protein. Western blotting analysis of the immunoprecipitated proteins showed the presence of recombinant viral proteins rVP5* and rVP6 which were detected with the respective polyclonal antibodies (Fig. 5G and 5H). The immunoprecipitation results were not conclusive for VP8* since its molecular weight was shared with that of immunoglobulin heavy chains which were recognized by the secondary antibody used in this assay (Fig. 5I).
Reciprocal immunoprecipitation of the viral and cellular protein mix was also conducted using antibodies specifically directed against viral proteins. As shown in Fig. 5J (lanes 3 and 5), Hsc70 co-immunoprecipitated with rVP5* and rVP6 but no with rVP8* (lanes 7). Hsc70 was not bound to the immunoprecipitation resin in the absence of viral protein addition (Fig. 5J, lanes 2, 4 and 6). These results confirm the interaction between viral proteins (VP5* and VP6) and cellular proteins Hsc70 and PDI. In the absence of antibody addition, no viral proteins were bound to the resin used in the immunoprecipitation assay (Data not shown).
Interactions between cell surface Hsc70 and viral proteins rVP5*, rVP8* and rVP6
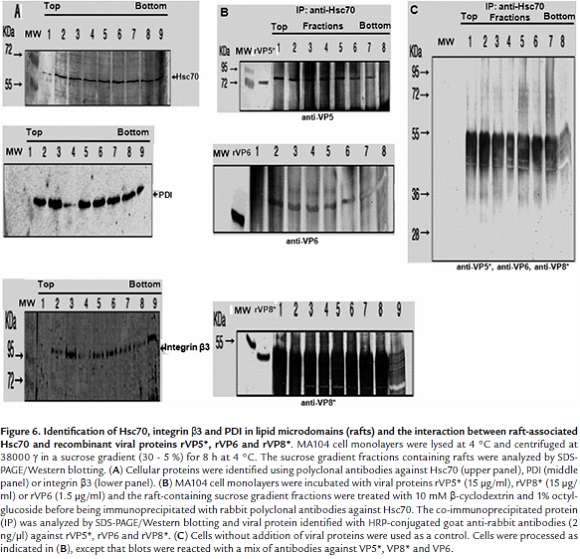
To test interactions between viral proteins and Hsc70, MA104 cell monolayer were separately incubated with rVP5*, rVP8* (15µg/ml) or rVP6 (1.5 µg/ml). Cells were lysed with Triton X-100 at 4 °C and the lipid microdomains (rafts) fractionated by centrifugation through a sucrose gradient. After treatment of gradient fractions with β-clyclodextrin and β-octylglucoside, fractions were immunoprecipitated with antibodies against Hsc70 and analyzed by SDS-PAGE/Western blotting. Viral proteins were detected with specific antibodies against them. Cellular proteins Hsc70 (Fig. 6A, upper panel), PDI (Fig. 6A, middle panel) and integrin β3 (Fig. 6A, lower panel) were detected in lipid raft fractions from control cells that had not been treated with viral proteins. These cellular proteins have previously been found to be associated in rafts from MA104 cells and also involved in rotavirus infection (Calderon et al., 2012). It was also found that viral proteins VP5* and VP6 co-immunoprecipitated with cellular Hsc70. These results show that rVP5* (Fig. 6B, upper panel) and rVP6 (Fig. 6B, middle panel) were located in lipid rafts interacting directly or indirectly with Hsc70. Again, identification of rVP8* resulted difficult as its molecular weight was similar to that from antibody heavy chains which reacted with the secondary antibody used (Fig. 6B, lower panel). Gradient fractions from cells that had not been treated with viral proteins, were not reactive with antibodies against VP5* or VP6 after immunoprecipitation with anti-Hsc70 antibodies (Fig. 6C). This result excludes any non-specific reaction of antibodies against viral proteins.
Inhibitory effect of VP5*, VP8* or VP6 on RRV infection of MA104 cells
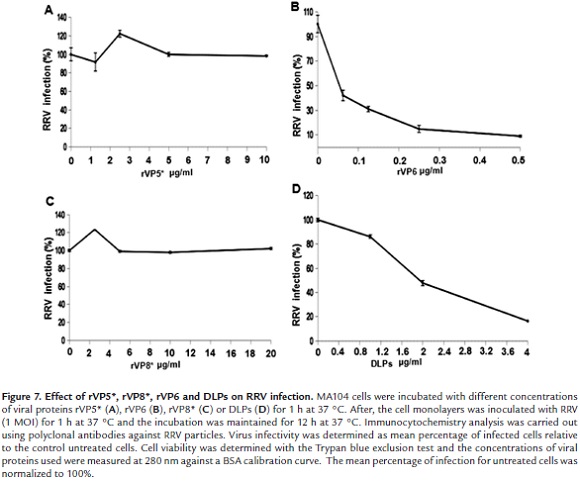
Previous work showed that MA104 cell infection with RRV TLPs was inhibited by treatment of cells with DLPs (double layered particles) (Dormitzer 2004). This suggested that VP6 located on the surface of DLPs is involved in rotavirus penetration to host cell. In order to confirm this hypothesis, MA104 cell were incubated with viral proteins rVP5*, rVP8* or rVP6 for 1 h at 37 °C and then infected with RRV TLPs. Immunochemistry detection after 12 h infection showed that infection was decreased by more than 90% when cells were previously incubated with rVP6 (0.5 µg/ml) (Fig. 7B). rVP5* or rVP8* treatment of cells was unable to interfere with virus infection even at much higher concentrations than those used for rVP6 (Fig. 7A and 7C). Increasing DLP concentrations led to inhibition of virus infection when cells were DLP-treated before TLP infection (Fig. 7D). Trypan blue exclusion assay showed that cell viability was not affected by DLP treatment. These results confirm the implication of VP6 in the rotavirus infection process in MA1|04 cells.
Discussion
Protein expression in heterologous systems has been a widely used strategy for investigating the molecular mechanisms underlying protein interactions. Expression vectors have been very useful in allowing uptake of genetic foreign material followed by its transcription and translation in heterologous systems (Sorensen et al., 2005; Sahdev et al., 2008). However, proteins expressed in heterologous systems usually are accumulated forming insoluble inclusion bodies and their solubilization requires detergent treatments and procedures aimed at their in vitro refolding (Sahdev et al., 2008). Insolubilization of VP5* and VP8* in heterologous environments has been shown to depend on the expression systems used (Frangioni et al., 1993; Tang et al., 1997). In the present study, the conditions for expression of recombinant Hsc70 and PDI were compared in terms of bacterial concentration at the time of induction, time of induction and inductor concentration. Therefore, the optimum conditions for expression of the cellular proteins Hsc70 and PDI were established in an E. coli system. Optimum conditions for vaccinia virus expressing recombinant VP6 in the MA104 cell system were also defined. The urea and detergents assayed for solubilization of recombinant proteins in combination with the 6-his-tagged expression of Hsc70 and PDI allowed their efficient purification after using His Trap affinity columns. On the other hand, the specificity of the rabbit antibodies raised against recombinant Hsc70 and VP6 was demonstrated by Western blotting analysis.
The mechanisms associated with the rotavirus entry into the host cell continue to be a very controversial subject. The high level of purity of the recombinant proteins obtained from the expression and purification procedures described in the present work allowed their use to gain further insight into rotavirus-cell interaction mechanisms. It had been previously shown that RRV DLPs were able to interfere with rotavirus infection in MA104 cells (Gualtero et al., 2007). This finding suggested that VP6 might be involved in rotavirus entry. The results shown here indicated that addition of rVP6 to MA104 cells resulted in more than 90% inhibition of virus infection. This new result gives further support to the hypothesis that this most abundant rotavirus structural protein could be implicated in rotavirus entry. Llama-derived single-chain antibody fragments directed to rotavirus VP6 have been found to possess broad neutralizing activity both in vitro and in vivo against rotavirus infection (Garaicoechea et al., 2008). The inhibition of rotavirus infection by either DLPs or VP6 could be explained by their binding to cell surface Hsc70 since this protein was found to interact with free or DLP-associated VP6 in ELISA (Gualtero et al., 2007) or co-immunoprecipitation assay. Differential binding strength of Hsc70 to TLPs and DLPs has been previously reported (Guerrero et al., 2002). This finding could be explained by the apparent existence of two exposed Hsc70 binding sites in TLPs which are represented by the VP5* domains 642-658 (Zárate et al., 2003) and 531- 554 (Gualtero et al., 2007). In contrast, the DLP-associated VP6 seems to have less Hsc70 binding sites (Gualtero et al., 2007).
Hsc70 has been shown to interact with VP5* and DLPs in previous inhibition and competition studies (Guerrero et al., 2000; Zárate et al., 2004; Gualtero et al., 2007), and cell surface PDI has been found to be needed for rotavirus infection as thiol/disulfide inhibitors and antibodies to PDI interfere with virus infection (Calderon et al., 2012). In the present work, ELISA and co-immunoprecipitation analysis led to concluded that Hsc70 or PDI certainly interact physically with VP5* and VP6. The inhibitory binding of VP6 to Hsc70 following incubation of VP6 with MA104 cells, as evidenced in the co-immunoprecipitation assay, suggests that VP6 binds to Hsc70 during the infectious process which results in a competition for rotavirus binding to cell surface Hsc70. Similarly, these interactions were also found in raft-associated Hsc70 or PDI, although whether these interactions are direct or are mediated by an intermediary protein remains to be established. We propose that rotavirus entry involves the cell surface PDI reducing activity which probably generates thiol groups in VP5* and/or VP6 that contribute either to virion disassembly or priming of viral proteins to interact with other cell surface receptors. The endoplasmic reticulum (ER)-associated oxidant PDI has been reported to interact with VP7 to generate the disulfide bridges which are needed for the proper VP7 folding during its assembly in TLPs (Svensson et al., 1994; Maruri-Avidal et al., 2008). Despite VP8* and immunoglobulin heavy chains could not be resolved by Western blotting analysis, and interaction between VP8* and Hsc70 or PDI remains to be elucidated. The treatment of Ma104 cells with VP5* or VP8* did not affect rotavirus infection. This finding would suggest that the binding of these proteins to the cell surface is dependent of conformational changes that are only produced when these proteins are associated to viral particles and interact with cell surface molecules during the infectious process. VP5* and VP8* failed to compete with viral particles probably because they lacked the conformational changes required to interact with cellular receptors. Our results give support to the hypothesis that conformational rearrangements in rotavirus particle proteins occur during the entry process which are similar to those generated in the HIV gp120 envelope glycoprotein conformation (Markovic et al., 2004) or in the non-enveloped reovirus (Madren et al., 2012) or poliovirus (Brandenburg et al., 2007) structural proteins in order to expose critical proteins to cell receptors.
Taken together, the results presented here confirm the usefulness of recombinant proteins in the identification of protein interactions during virus entry into the host cell. However the sequence in which rotavirus VP5* and VP6 interact with Hsc70, PDI or other cellular proteins during entry remains to be determined in more detail.
Acknowledgements
This study was supported by a grant (No. 201010018595) awarded by Universidad Nacional de Colombia to support the strengthening of research and artistic creation groups.
References
Arias, CF, Isa P, Guerrero, CA, Méndez, E, Zárate, S, López, T, Espinosa, R, Romero, P, López S. (2002). Molecular biology of rotavirus cell entry. Arch Med Res., 33 (4), 356-61. [ Links ]
Beau, I, Cotte-Laffitte, J, Géniteau-Legendre, M, Estes, MK, Servin, AL. (2007). An NSP4-dependant mechanism by which rotavirus impairs lactase enzymatic activity in brush border of human enterocyte-like Caco-2 cells. Cell Microbiol., 9, 2254-66. [ Links ]
Benureau, Y, Huet, JC, Charpilienne, A, Poncet, D, Cohen, J. (2005). Trypsin is associated with the rotavirus capsid and is activated by solubilization of outer capsid proteins. J Gen Virol., 86, 3143-51. [ Links ]
Brandenburg, B, Lee, LY, Lakadamyali, M, Rust, MJ, Zhuang, X, Hogle, JM. (2007). Imaging poliovirus entry in live cells. PLoS Biol., 5(7), e183. [ Links ]
Calderon, MN, Guerrero, CA, Acosta, O, Lopez, S, Arias, CF. (2012). Inhibiting rotavirus infection by membrane-impermeant thiol/disulfide exchange blockers and antibodies against protein disulfide isomerase. Intervirology, 55, 451-64. [ Links ]
Calderon, MN, Guzmán, F, Acosta, O, Guerrero, CA. (2012). Rotavirus VP4 and VP7-Derived Synthetic Peptides as Potential Substrates of Protein Disulfide Isomerase Lead to Inhibition of Rotavirus Infection. Int J Pept Res Ther., 18(4), 373-382. [ Links ]
Coulson, B. S., Witterick, P. D., Tan, Y., Hewish, M. J., Mountford, J. N., Harrison, L. C., & Honeyman, M. C. (2002). Growth of rotaviruses in primary pancreatic cells. Journal of virology, 76(18), 9537-9544. [ Links ]
Chung, CT, Niemela, SL, Miller, RH. (1989). One-step preparation of competent Escherichia coli: transformation and storage of bacterial cells in the same solution. Proc Natl Acad Sci U S A., 86(7), 2172-5. [ Links ]
De Marco, G., Bracale, I., Buccigrossi, V., Bruzzese, E., Canani, R. B., Polito, G., ... & Guarino, A. (2009). Rotavirus induces a biphasic enterotoxic and cytotoxic response in human-derived intestinal enterocytes, which is inhibited by human immunoglobulins. Journal of Infectious Diseases, 200(5), 813-819. [ Links ]
Dormitzer, PR, Nason, EB, Prasad, BV, Harrison, SC. (2004). Structural rearrangements in the membrane penetration protein of a non-enveloped virus. Nature, 430, 1053 - 1058. [ Links ]
Edman, J, Ellis, L, Blacher, R, Roth, R, Rutter, W. (1985). Sequence of protein disulphide isomerase and implications of its relationship to thioredoxin. Nature, 317(19), 267 - 270. [ Links ]
Esposito, D. H., Holman, R. C., Haberling, D. L., Tate, J. E., Podewils, L. J., Glass, R. I., & Parashar, U. (2011). Baseline estimates of diarrhea-associated mortality among United States children before rotavirus vaccine introduction. The Pediatric infectious disease journal, 30(11), 942-947. [ Links ]
Estes, MK, Kapikian, AZ. (2007). Rotaviruses and their replication. In Fields Virology. Knipe, D.M. & Howley, P.M (ed). 5th edition; Philadelphia, PA; Lipincott -Williams and Wilkins. [ Links ]
Estes, MK, Graham, DY, Mason, BB. (1981). Proteolytic enhancement of rotavirus infectivity: molecular mechanisms. J Virol., 39, 879-888. [ Links ]
Fogh, J, Fogh, JM, Orfeo, T. (1977). One hundred and twenty seven cultured human tumor cell lines producing tumor in nude mice. J Natl Cancer Inst., 59, 221-6. [ Links ]
Frangioni, JV, Neel, BG. (1993). Solubilization and purification of enzymatically active glutathione S-transferase (pGEX) fusion proteins. Anal Biochem., 210(1), 179-87. [ Links ]
Garaicoechea, L., Olichon, A., Marcoppido, G., Wigdorovitz, A., Mozgovoj, M., Saif, L., ... & Parreno, V. (2008). Llama-derived single-chain antibody fragments directed to rotavirus VP6 protein possess broad neutralizing activity in vitro and confer protection against diarrhea in mice. Journal of virology, 82(19), 9753-9764. [ Links ]
Graham, K. L., Halasz, P., Tan, Y., Hewish, M. J., Takada, Y., Mackow, E. R., ... & Coulson, B. S. (2003). Integrin-using rotaviruses bind α2β1 integrin α2 I domain via VP4 DGE sequence and recognize αXβ2 and αVβ3 by using VP7 during cell entry. Journal of virology, 77(18), 9969-9978. [ Links ]
Gualtero, DF, Guzmán, F, Acosta, O, Guerrero, CA. (2007). Amino acid domains 280-297 of VP6 and 531-554 of VP4 are implicated in heat shock cognate protein hsc70-mediated rotavirus infection. Arch Virol., 152, 2183-96. [ Links ]
Guerrero, CA, Zárate, S, Corkidi, G, López, S, Arias, CF. (2000). Biochemical characterization of rotavirus receptors in MA104 cells. J Virol., 74, 9362-71. [ Links ]
Guerrero, CA, Méndez, E, Zárate, S, Isa, P, López, S, Arias, CF. (2000). Integrin αvβ3 mediates rotavirus cell entry. Proc Natl Acad Sci U S A., 97, 14644-9. [ Links ]
Guerrero, C. A., Bouyssounade, D., Zárate, S., Iša, P., López, T., Espinosa, R., ... & Arias, C. F. (2002). Heat shock cognate protein 70 is involved in rotavirus cell entry. Journal of virology, 76(8), 4096-4102. [ Links ]
Halasz, P, Holloway, G, Turner, SJ, Coulson, BS. (2008). Rotavirus replication in intestinal cells differentially regulates integrin expression by a phosphatidylinositol 3-kinase-dependent pathway, resulting in increased cell adhesion and virus yield. J Virol., 82, 148-60. [ Links ]
Haselhorst, T., Fleming, F. E., Dyason, J. C., Hartnell, R. D., Yu, X., Holloway, G., ... & von Itzstein, M. (2009). Sialic acid dependence in rotavirus host cell invasion. Nature chemical biology, 5(2), 91-93. [ Links ]
Isa, P, Arias, CF, López, S. (2006). Role of sialic acids in rotavirus infection. Glycoconj J., 23, 27-37. [ Links ]
Isa, P, López, S, Segovia, L, Arias, CF. (1997). Functional and structural analysis of the sialic acid-binding domain of rotaviruses. J Virol., 71(9), 6749-6756. [ Links ]
Jolly, CL, Beisner, BM, Ozser, E, Holmes, IH. (2001). Non-lytic extraction and characterisation of receptors for multiple strains of rotavirus. Arch Virol., 146, 1307-23. [ Links ]
Jourdan, N, Maurice, M, Delautier, D, Quero, AM, Servin, AL. (1997). Trugnan G. Rotavirus is released from the apical surface of cultured human intestinal cells through nonconventional vesicular transport that bypasses the Golgi apparatus. J Virol., 71, 8268-78. [ Links ]
Kaushik, R. S., Begg, A. A., Wilson, H. L., Aich, P., Abrahamsen, M. S., Potter, A., Griebel, P. (2008). Establishment of fetal bovine intestinal epithelial cell cultures susceptible to bovine rotavirus infection. Journal of virological methods, 148(1), 182-196. [ Links ]
Laboissiere, M, Chivers, P, Raines, R. (1995). Production of rat protein disulfide isomerase in Saccharomyces cerevisiae. Protein Expr Purif. 6(5), 700-706. [ Links ]
Lopez S, Arias CF. 2006. Early steps in rotavirus cell entry. Curr Top Microbiol Immunol. 309, 39-66. [ Links ]
Londrigan, SL, Hewish, MJ, Thomson, MJ, Sanders, GM, Mustafa, H, Coulson, BS. (2000). Growth of rotaviruses in continuous human and monkey cell lines that vary in their expression of integrins. J Gen Virol., 81, 2203-13. [ Links ]
Macartney, KK, Baumgart, DC, Carding, SR, Brubaker, JO, Offit, PA. (2000). Primary murine small intestinal epithelial cells, maintained in long-term culture, are susceptible to rotavirus infection. J Virol., 74, 5597-603. [ Links ]
Madren, JA, Sarkar, P, Danthi, P. (2012). Cell entry-associated conformational changes in reovirus particles are controlled by host protease activity. J Virol., 86(7), 3466-73. [ Links ]
Markovic, I., Stantchev, T. S., Fields, K. H., Tiffany, L. J., Tomiç, M., Weiss, C. D., ... & Clouse, K. A. (2004). Thiol/disulfide exchange is a prerequisite for CXCR4-tropic HIV-1 envelope-mediated T-cell fusion during viral entry. Blood, 103(5), 1586-1594. [ Links ]
Maruri-Avidal, L, López, S, Arias, CF. (2008). Endoplasmic reticulum chaperones are involved in the morphogenesis of rotavirus infectious particles. J Virol., 82(11), 5368-80. [ Links ]
Méndez, E, Arias, CF, López, S. (1993). Binding to sialic acids is not an essential step for the entry of animal rotaviruses to epithelial cells in culture. J Virol., 67, 5253-9. [ Links ]
Moreno, L. Y., Guerrero, C. A., & Acosta, O. (2013). Expression and purification of rotavirus structural proteins VP5* and VP8* in bacteria E. coli BL21 (DE3). Revista Colombiana de Biotecnología, 15(1), 82-97. [ Links ]
Parashar, UD, Gibson, CJ, Bresee, JS, Glass, RI. (2006). Rotavirus and severe childhood diarrhea. Emerg Infect Dis. 12, 304-6. [ Links ]
Pesavento, JB, Crawford, SE, Estes, MK, Prasad, BV. (2006). Rotavirus proteins: structure and assembly. Curr Top Microbiol Immunol., 309, 189-219. [ Links ]
Ramig, RF. (2004). Pathogenesis of Intestinal and Systemic Rotavirus Infection. J Virol., 78, 10213-10220. [ Links ]
Ruiz, MC, Leon, T, Diaz, Y, Michelangeli, F. (2009). Molecular biology of rotavirus entry and replication. Scientific World Journal., 9, 1476-97. [ Links ]
Sahdev, S, Khattar, SK, Saini, KS. (2008). Production of active eukaryotic proteins through bacterial expression systems: a review of the existing biotechnology strategies. Mol Cell Biochem., 307 (1-2), 249-64. [ Links ]
Servin, AL. (2003). Effects of rotavirus infection on the structure and functions of intestinal cells, p. 237-254. In U. Desselberger and J Gray (ed.), Viral gastroenteritis. Amsterdam, Netherlands; Elsevier Science BV. [ Links ]
Sorensen, HP, Mortensen, KK. (2005). Advanced genetic strategies for recombinant protein expression in Escherichia coli. J Biotechnol., 115(2), 113-28. [ Links ]
Svensson, L, Dormitzer, PR, von Bonsdorff, CH, Maunula, L, Greenberg, HB. (1994). Intracellular manipulation of disulfide bond formation in rotavirus proteins during assembly. J Virol., 68(8), 5204-15. [ Links ]
Tang, B, Gilbert, JM, Matsui, SM, Greenberg, HB. (1997). Comparison of the rotavirus gene 6 from different species by sequence analysis and localization of subgroup-specific epitopes using site-directed mutagenesis. Virology, 237(1), 89-96. [ Links ]
Trask, SD, Kim, IS, Harrison, SC, Dormitzer, PR. (2010). A rotavirus spike protein conformational intermediate binds lipid bilayers. J Virol., 84, 1764-70. [ Links ]
Vargas, J, Romero, P, López, S, Arias, C. (2006). The Peptide-Binding and ATPase domains of recombinant hsc70 are required to interact with rotavirus and reduce its infectivity. J Virol., 80(7), 3322-3331. [ Links ]
Wolf, M, Vo, PT, Greenberg, HB. (2011). Rhesus rotavirus entry into a polarized epithelium is endocytosis dependent and involves sequential VP4 conformational changes. J Virol., 85, 2492-503. [ Links ]
Zárate, S, Espinosa, R, Romero, P, Méndez, E, Arias, CF, López, S. (2000). The VP5 domain of VP4 can mediate attachment of rotaviruses to cells. J Virol., 74, 593-9. [ Links ]
Zárate, S, Romero, P, Espinosa, R, Arias, CF, López, S. (2004). VP7 mediates the interaction of rotaviruses with integrin αvβ3 through a novel integrin-binding site. J Virol., 78, 10839 - 47. [ Links ]
Zárate, S., Cuadras, M. A., Espinosa, R., Romero, P., Juárez, K. O., Camacho-Nuez, M., ... & López, S. (2003). Interaction of rotaviruses with Hsc70 during cell entry is mediated by VP5. Journal of virology, 77(13), 7254-7260. [ Links ]
Zhang, Y, Wen, Y, Wei, H, Li, Ch, Fan, Y, Chen, Y. 2010. Expression of VP5* and VP8* of group A rotavirus and immunological characteristics. China Biotechnology, 30 (2), 32-38. [ Links ]