INTRODUCTION
Lactic acid (LA) is an important naturally-occurring hydroxycarboxylic acid used in industry today (Castillo Martinez et al., 2013). It is valuable because of its versatile applications, mainly in food as a preservative and as an acidulating agent, and to a lesser degree in the pharmaceutical, cosmetic, and chemical industries. Due to its carboxylic and hydroxyl groups, LA is highly reactive and participates in numerous chemical reactions, yielding important products such as pyruvic acid, acrylic acid, lactide, and lactate ester (C. Gao, Ma, & Xu, 2011). It exhibits optical activity with L (+) and D (-) isomers. However, it is mainly found in the inactive L and D form made up of equimolar fractions of each isomer (Boonpan, Pivsa-art, Pongswat, Areesirisuk, & Sirisangsawang, 2013). Given that humans have only the L(+) lactate dehydrogenase enzyme, the L(+) LA isomer is preferred for commercial applications involving human consumption (Bishai, De, Adhikari, & Banerjee, 2013). Some years ago, the potential of LA as a precursor of polylactic acid, a biopolymer that is biodegradable and biocompatible, was discovered. This polymer has been used in medical applications, 3D printing, and the fabrication of plastics (RedCorn, Fatemi, & Engelberth, 2018). Because of this, the LA market has grown considerably, and the global demand is expected to increase by around 14% to 19% per year up to 2020, generating expectations for the improvement of current production processes in order to meet this demand (Hu, Kwan, Daoud, Sze, & Lin, 201 7). Presently, the global production of lactic acid is achieved mainly through fermentation (Abdel-Rahman, Tashiro, & Sonomoto, 2011). Nev-ertheless, a big difficulty for the fermentation process is the high cost of the raw materials used as sources of energy and carbon, mainly pure sugars, representing an operational cost between 30% and 60% of total produc-tion costs (Marques, Santos, Francisco, & Roseiro, 2008; Oh et al., 2005). Biotechnological processes can help to reduce the production costs by using organic waste from agricultural and food industries or from households (Bilanovic, Chang, Isobaev, & Welle, 2011). In addition to increasing the economic feasibility of the process, the use of waste in the fermentation is also an alternative that allows minimizing the impact caused by the dumping of waste into the environment (RedCorn & Engelberth, 2016). This review gives an overview of industrial and household wastes for utilization in the biotechnological production of lactic acid, aspects of fermentation for each waste, mainly in submerged culture, the advantages and disadvantages of different types of treatments used for the transformation of waste into suitable substrates, and some recent strategies for improving yields. In addition, specific recommendations are given for enhancing the value of these raw materials.
FERMENTATIVE PATHWAY AND LA-PRODUCING MICROOORGANISMS
LA can be produced by fermentation in one or two stages, according to the feedstock and type of microorganism used. The use of refined sugars, the addition of enzymes, co-culture systems, and microorganisms with the enzymatic ability to degrade complex substrates may allow carrying out the fermentation in a single step. The process with agroindustrial and household wastes can also occur in two steps. First, a pretreatment must be carried out in order to release sugars into the medium, which is generally enzymatic or thermochemical, and second, these sugars are used in the bioreactor for the fermentation and production of LA (John, Anisha, Nampoothiri, & Pandey, 2009). Lactic acid bacteria (LAB) and filamentous fungi have proven to be the most attractive microorganisms for LA production because of their high productivity and high yield (Nuttha, 2005).
Lactic acid bacteria
LAB is a large group of benign microorganisms that produce LA during anaerobic fermentation. This group can be divided into homofermentative and heterofermentative bacteria, according to the nature of the final products, as is shown in figure 1. The homofermentative bacteria metabolize hexoses via the Embden-Meyerhof pathway (glycolysis), generating two molecules of LA for each molecule of glucose, but they do not ferment pentoses or gluconate.
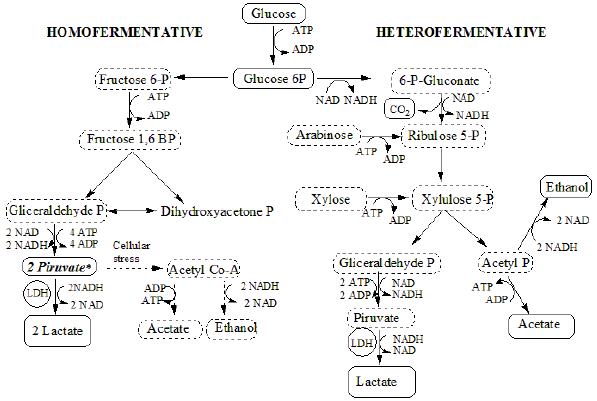
Figure 1 Metabolism of lactic acid bacteria - homofermentative and heterofermentative pathway. (Adapted from Hofvendahl & Hagerdal, 2000).
The heterofermentative lactic acid bacteria metabolize hexoses, pentoses such as xylose, and some other substances, following the phosphoketolase pathway (Panesar, Kennedy, Gandhi, & Bunko, 2007). The difference with the homofermentative route is the conversion of two of the six carbons of glucose to acetic acid, which limits yields and increases the costs of LA purification (Cock & Rodríguez, 2005). The proportion of the by -products is dependent on the redox potential of the system. In figure 1, it is shown that homofermentators can also generate mixed acids, but this only occurs when the microorganism is subjected to adverse culture conditions (Hofvendahl & Hagerdal, 2000).
Fungi
Filamentous fungi of the genus Rhizopus have been recognized for producing L(+) LA efficiently via aerobic metabolism and with lower nutritional requirements than LAB. These latter organisms need vitamin B complex, sev-eral aminoacids, and nucleotides for their growth (Abdel-Rahman, Tashiro, & Sonomoto, 2013; Watanabe et al., 2012). This fungus can produce other organic acids besides L(+) LA, such as fumaric acid, malic acid, and ethanol. Glucose is degraded to pyruvate through the Embden -Mayerhof-Parnas pathway, and it is used as a precursor for the different metabolites (Nuttha, 2005). The conversion of xylose to lactic acid has not been investigated as thoroughly as the conversion of glucose. However, it has been found that Rhizopus oryzae is also capable of metabolizing xylose via pentose phosphate to produce xylulose 5 phosphate, which subsequently can be metabolized via the Embden-Meyerhoff-Parnas pathway (Z. Y. Zhang, Jin, 6 Kelly, 2007).
These fungi can degrade more complex substrates than LAB, and their heterogeneous morphology enables easy separation of the biomass (Saito, Hasa, & Abe, 2012). Fungi in a submerged culture may take different physical forms: small pellet morphology is reported to be appropriate for LA fermentation, while the mycelial form causes problems of poor mass transfer and low production of LA (Liu, Liao, Liu, & Chen, 2006). Tools such as cell immobilization have been implemented in order to control the morphology and increase the production of LA (Wang, Wang, Yang, Wang, & Ren, 2010). In spite of the nutritional and separation advantages of LA in a bioprocess with fungi, we consider that the use of LAB on an industrial scale is more feasible, due to the easier operation of bioreactors. In addition, LAB have been more thoroughly studied, so they have relative advantages for increasing LA production through genetic transformations.
PRETREATMENT METHODS
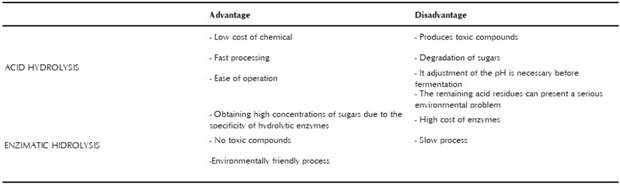
One of the biggest problems for the use of agroindustrial and household wastes for the production of lactic acid at the industrial level is the difficulty of achieving an integral and efficient use of all the nutrients of these raw materials, because they have a complex structure. Therefore, it is necessary to perform a pre-pretreatment of the raw material before the fermentation. The main objec-tive of the pretreatment of substrates such as starch or lignin is the gradual breakdown of the recalcitrant polysaccharides and matrices into low-weight molecules easily digestible by microorganisms. Currently, various pre-treatments have been used, among them physical ones, such as milling and grinding; chemical ones, such as acid and alkaline hydrolysis; physicalchemical ones, such as steam explosion and hydrotermolysis; and biological ones, such as the use of fungi, bacteria, or enzymes (Maria & Valencia, 2011). Complex materials usually need a physical treatment in order to reduce the size of the particles, so that the residue will be manageable and have better exposure to subsequent pretreatments. Acid and enzymatic hydrolysis are the most-reported treatments in studies of residue utilization for application in fermentative processes. Due to this, two types of pretreatment will be analyzed.
Acid hydrolysis
Acid hydrolysis is based on the use of acids (sulfuric, hydrochloric, trifluoroacetic, or hydrosulfuric) to perform catalysis at high temperatures and break complex matrices (Abdel-Rahman et al., 2011). The effectiveness of the waste degradation depends on three interdependent variables: acid concentration, temperature, and exposure time (Domínguez et al., 2011). Acidic conditions between 0.5% and 1.0% are considered low, and those above 1.5% strong; temperatures near 121 °C and 160 ° C are used. Nevertheless, in order to decrease the degradation of sugars such as fructose and sucrose, which occurs at 106 °C in acidic environments, temperatures below 120 °C are employed in the industry (Maria & Valencia, 2011). Hydrolyzed product may contain sug-ars such as glucose, xylose, and arabinose, but their pro-portion in the medium depends on the type of substrate used and the pretreatment conditions (M. Gao, Kaneko, Hirata, Toorisaka, & Hano, 2008; Laopaiboon, Thani, Leelavatcharamas, & Laopaiboon, 2010). In most cases, the different types of sugars released are not all consumed by a single organism, decreasing the product yield. Besides producing sugars in this process, some soluble materials such as lignin, acetic acid, furfural, 5 hydroxymethylfurfural (HMF), levulinic acid, and phenolic compounds can also be generated. These compounds are toxic and may inhibit both the growth and the sugar utilization capabilities of the microorganisms (Laopaiboon et al., 2010). The phenomenon of inhibition in LA fermenta-tion from acid hydrolyzates has been reported to occur with microorganisms such as L. casei subsp rhamnosus, L. pentosus, and E. coli (Guo, Jia, Li, & Chen, 2010). In order to reduce the inhibitory effect, the hydrolyzate is subjected to detoxification through methods that include, among others, neutralization with alkali or activated carbon and ion exchange (Woiciechowski, Soccol, Ramos, & Pandey, 1999). Woiciechowski et al. (1999), found that R. oryzae NRRL 395 has the ability to resist the inhibitory effect of toxic components in the hydrolyzate. Guo et al. (2010), isolated a microorganism identified as L. brevis (S3F4), which is able to tolerate potential inhibi-tors present in the hydrolyzate of corn stover. The use of acid hydrolysis with complex substrates also can destroy proteins and vitamins, which are essential for the growth and LA production of LAB.
Enzymatic hydrolysis
Enzymatic hydrolysis is the most promising method for obtaining a good yield of fermentable sugars from organic waste. Enzymes such as laccase, lignin peroxidase, and manganese peroxidase are used to degrade lignin; endoxylanases, acetyl xylan esterase, α-xylosidase, β-mannosidase, and α -L-arabinofuranosidase are utilized in the saccharification of hemicellulose; while cellobiohydrolases, β-glucosidases, and endoglucanases are employed for cellulose degradation. The use of individual enzymes involves limitations, while enzyme mixtures can exhibit a chain reaction that increases the effectiveness of the hydrolysis (Van Dyk & Pletschke, 2012). On the other hand, for hydrolyzed starch, the enzymes a-amylase, 0-amylase, and glucoamylase are used. These kinds of enzymes attack a-1.4 links but not a-1.6 links, which are hydrolyzed by isoamylases or dextrinases. The enzymatic process for degrading starch generally takes place in two consecutive stages: liquefaction and saccharification. Amylase acts during the first step, and glucoamylase acts during the second one. These processes are usually performed separately, because temperature conditions are different for each enzyme (Castaño & Mejia, 2008). In table 1, a comparison of the advantages and disadvantages of the use of acid and enzymatic pretreatment is shown. These two methods, acid and enzimatic hydrolysis, are equally effective when parameters such as percent of hydrolysis, time, cost of chemicals, and energy consumption are considered in one or another method. For example, in the hydrolysis process of 150 kg of cassava bagasse, US $34.27 was required for acid hydrolysis, while the cost of enzymatic hydrolysis was US $2470.99. Although acid pretreatment is less expensive and faster, it does not generate yields that are as high as does enzymatic hydrolysis (Woiciechowski, Nitsche, Pandey, & Ricardo, 2002). Therefore, the pretreatment selection depends on the substrate composition and the availability of resources. Presently, the American company NatureWorks LLC is the world leader in the production of lactic acid via biotechnology. The treatment of lignocellulosic raw materials by this company is done via the enzymatic method (NatureworksLLC, 2019).
INEXPENSIVE SUBSTRAES FOR LACTIC ACID PRODUCTION
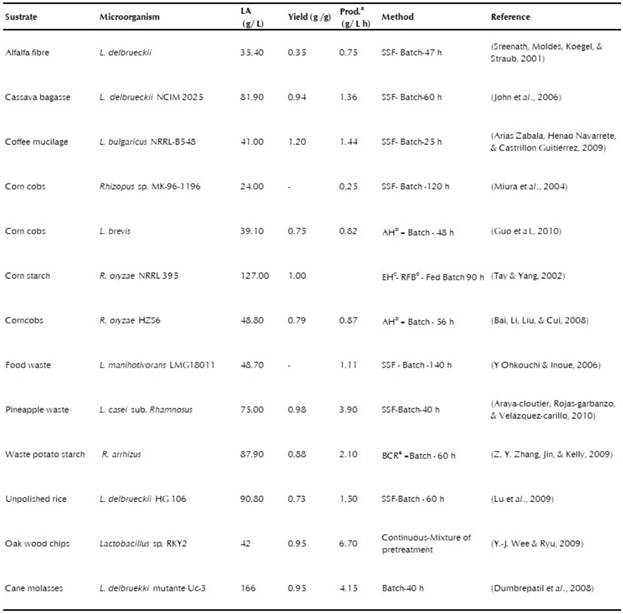
The highest concentration of LA is obtained when pure sugars are fermented. Glucose is considered to be the main raw material used in LA fermentations, because this sugar gives high yields and its assimilation is easy for the microorganisms. Nevertheless, the use of expensive substrates is a problem, since LA is a low-priced product (Oh et al., 2005). The utilization of cheap raw materials such as organic waste has emerged as an alternative that overcomes this problem. Globally, solid wastes have a negative environmental impact, caused by their improp-er disposal and the increasing rate at which they are generated, associated with industrial growth and human abuse of the environment (María & Echeverri, 2004; Pleissner et al., 2017). Alarm over high rates of pollution has led to a search for new processes that would take advantage of organic waste before its being discarded. Agroindustrial and household wastes have shown great potential for biotechnological processes on a large scale, because they are abundant, are low-priced, and have a high nutrient content that can be transformed into a val-uable product (Yousuf, Bastidas-Oyanedel, & Schmidt, 2018). Optimal raw materials for this purpose should have low levels of pollutants and toxic substances, be easily fermentable, need little or no treatment, and generate high levels of production of the metabolite of interest (Y.-J. Wee & Ryu, 2009). The residues used for LA production can be divided into three groups, according to their composition: lignocellulosic waste, starchy waste, and residues from dairy processes.
The production policy of NatureWorks LLC focused on the use of renewable and abundant raw materials such as corn, cassava, sugarcane, and beets. However, in 2016 they inaugurated a new laboratory in order to im-plement a fermentation technology that allows the transformation of methane (greenhouse gas) to lactic acid. Other companies, such as CORBION (formerly called PURAC), Galactic S.A, Hefei TNJ Chemical, and Shanghai Honghao Chemicals Co. are also recognized produc-ers of lactic acid through fermentation processes (Corbion, 2019; Galactic, 2019; NatureworksLLC, 2019).
Lignocellulosic material
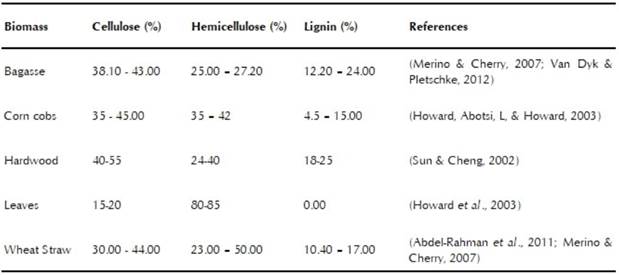
Lignocellulose is derived from organic materials and represents the most abundant global source of biomass that has not yet been effectively exploited. It is predominantly composed of a thoroughly mixed matrix of cellulose, hemicellulose, and lignin, as well as lesser amounts of minerals, oils, and other components (Doherty, Mousavioun, & Fellows, 2011). Each lignocellulosic material has a unique chemical composition. The composition in the matrix is variable, in accordance with species, age, growth stage, and environmental conditions (Cuervo, Folch, & Quiroz, 2001), as is shown in table 2.
Cellulose is the major component of plant biomass, which is constantly regenerated by photosynthesis. It is a rigid homopolysaccharide formed by linear units of P-D glucans. Hemicellulose is a heteropolysaccharide that is composed of two pentose sugars (xylose and arabinose) and three hexose sugars (glucose, mannose, and galac-tose). This polymer is more easily degraded by microorganisms than cellulose and lignin (Guo et al., 2010). Finally, lignin is a functional material that gives structural strength and rigidity to plants. Physically, it is a macromolecular aromatic polymer formed by the union of phenylpropane monomers that generates an extremely complex structure. Thus plants that have high amounts of lignin in their composition are more resistant to attacks by microorganisms and to some extreme conditions (Abdel-Rahman et al., 2011). Due to low bioavailability of sugars in lignocellulosic biomass, these materials are subjected to a pretreatment in order to liberate fermentable sugars before fermentation (Maas, Bakker, Eggink, & Weusthuis, 2006). Next, some work done on the production of LA with lignocellulose residues will be described.
Sugar cane. Since 2005, in Colombia five of the thirteen sugar manufacturers that exist in the country have distilleries dedicated to the production of fuel alcohol through biotechnological processes. In 2011, 337 million liters of bioethanol were produced from sugarcane. The growing use of this crop for the biofuel industry has diverted a high percentage of the raw material previously used for sugar production, entering into direct competition with the generation of this food, which is in high demand nationally. Characteristic residues of this gramineae have been investigated for use in bioprocess applications (Asocaña, 2012). Bagasse, which is generated after tritu-ration and extraction of juice from sugar cane, is considered to be the main by-product from sugar production centers (Maria & Valencia, 2011). This inert bagasse has been investigated along with cassava hydrolyzate for carrying out LA fermentation in the solid state (Rojan, Nampoothiri, Nair, & Pandey, 2005). In submerged cultures, Laopaiboon et al. (2010), obtained a LA concentration of 10 g/L, while Adsul et al. (2007), generated 67 g/L of the metabolite with a yield of 80% by using the genetically modified strain L. delbrueckii subsp delbrueckii mutant Uc-3. Cock and Rodriguez (2007) determined that a mixture of buds and leaf juice proved to be suitable as raw material for the growth of LAB, even without the addition of a nitrogen source. Molasses contains about 50% (w/w) of sugars; the most abundant of these is sucrose (Hofvendahl & Hagerdal, 2000). The high concentration of this sugar produces a viscous liquid that may bring on substrate inhibition. Also, molasses contains metals (iron, zinc, copper, etc.) that may affect the growth of the microorganisms and are implicated in the inactivation of the enzymes important for the biosynthesis of products (Y.-J. Wee, Kim, Yun, & Ryu, 2004). An advantage of this residue is its buffer capacity, which allows maintaining a pH above 5 during the fermentation of LA, avoiding the need to add neutralizers to the culture medium (Dumbrepatil, Adsul, Chaudhari, Khire, & Gokhale, 2008). Most of the results of LA production with molasses reported in the literature range between 40 and 166 g/L, with a volumetric productivity from 2.00 to 4.20 g/L h and a yield between 0.80 and 0.98 g/g (Cock & Rodríguez, 2005; C. Gao et al., 2011; Pal, Sikder, Roy, & Giorno, 2009). L delbrueckii is the most widely used microorganism for this substrate, although there are reports of the use of E. faecalis, R .oryzae, and E. coli (Dumbrepatil et al., 2008; Y. Wee, Kim, & Ryu, 2006).
Wheat bran and corn cobs. Wheat bran, in addition to being used as a carbon source, has also been shown to be an excellent source of nutrients containing nitrogen, with the possibility of replacing the expensive yeast ex-tract used to provide nutrients for bacterial growth and fermentation (Li, Han, Ji, Wang, & Tan, 2010; Y. Wee et al., 2006). However, some researchers suggest that the use of yeast extract in small amounts is necessary in or-der to avoid growth factor limitations if nutrients from waste materials are being used in fermentation with LAB. If no yeast extract is used, fermentation time increases, causing decreased productivity (K. Lee, 2005; Lu, Lu, He, & Yu, 2009). As a carbon source, it has been found that wheat bran is composed of lignocellulose and a considerable amount of starch; therefore, it is an interesting material for study. Investigations by Yun et al. (2004), show a LA production of 60.7 g/L from 300 g/L wheat bran. Garde et al. (2002), used wheat straw hydrolyzate and a co-culture of LAB to increase yields in the fermentation, while Bulut et al. (2004) reported that the use of wheat bran with Rhizopus oryzae is not sustainable if this substrate was used as the only carbon source. Similar to wheat bran, corn cobs are also an abundant agroindustrial waste rich in hemicellulose, cellulose, nitro-gen, and protein. They can be used as a complete source of carbon and energy, or, as in the case of corn steep liquor, as a source of nitrogen (Oh et al., 2005; Ruengruglikit & Hang, 2003). Miura et al., (2004) have reported the use of Rhizopus oryzae with an Acremonium cellulase preparation (10 U/g corncorb). The authors obtained a concentration of total sugars of 55 g/L, composed mainly of glucose (61%) and xylose (21%) from the enzymatic treatment of 100 g/L of raw corncob. The yield for the conversion of glucose to lactic acid was 82%.
Starchy material. Starch is a polysaccharide that exists mainly in tubers, such as potatoes and cassavas, and in seeds and grains such as wheat, corn, and rice. This is a glucose polymer that contains long linear and branched chains of amylose and amylopectin, respectively (Woiciechowski et al., 2002). This material must be hydrolyzed before fermentation, because its degradation is difficult, although it can also be fermented directly by LAB and fungi with amylolytic ability (Abdel-Rahman et al., 2013; Watanabe et al., 2012). LAB with amylolytic activity include L. fermentum, L. plantarum, L. manihotivorans, L. amylovorus, and L. amylophilus, among others (Reddy, Altaf, Naveena, Venkateshwar, & Kumar, 2008; Shibata, Flores, Kobayashi, & Sonomoto, 2007). In submerged fermentation with a different strain of L. amy lophilus, it has been reported that 90% of the soluble starch is efficiently converted to LA (Rojan et al., 2005; Vishnu, Seenayya, & Reddy, 2002). In most studies of fermentation by Rhizopus sp. of starchy materials, they were converted directly, without pretreatment or the addition of supplements (Oda, Saito, Yamauchi, & Mori, 2002). The maximum conversion factor of starch to LA using Rhizopus sp. was 1.11 g of LA/g of starch (Huang, Jin, & Lant, 2005). Next, some work done on the produc-tion of LA with starchy material will be described.
Cassava. Cassava is one of the most abundant starch crops in the world, and it is the third largest source of food for the inhabitants of tropical regions (Teixeira et al., 2012). This tuber grows in poor soils and contains more than 80% starch. Nonetheless, once collected, cassava deteriorates rapidly, which causes a great deal of reluctance to use it. Other disadvantages of this crop are its low protein content and its toxicity, due to the presence of cyanide in its peel (Teixeira et al., 2012). Cassava bagasse has a high starch content, around 52% dry weight (Woiciechowski et al., 2002). John et al. (2006), investigated the use of this residue with two LAB, L. casei NCIMB 3254 and L. delbruekki NCIM 2025, reaching a maximum concentration of 83.80 g/L and 81.90 g/L of LA respectively. Later, John et al. (2007) , employed a 1:1 mixture of the previously-mentioned strains to determine how their interactions affect the process, but only achieved a maximum product concentration of 81.00 g/L. With cassava powder (70% starch) as a carbon source, a yield of 0.85 g/g from 100.00 g/L of substrate was found. Similar results were obtained with glucose at 100.00 g/L and potato starch at the same concentration. Yin et al. (1997), found that 120.00 g/L is the optimum concentration of starch for fermentation by R. oryzae; high substrate concentrations exceeding 130 g/L result in low yields of LA.
Potatoes. Potatoes are one of the main starch crops in the world, with a production of 2x107 t in the United States and 9.5x107 t in China (RedCorn et al., 2018). A quarter of this raw material is discarded during industrial processing, generating large volumes of waste, such as peels and wastewater, with high starch content (Z. Zhang, 2008). Potato peel has been shown to be an agroindustrial residue with great potential for use in the production of lactic acid, because it is a very complete substrate in relation to its composition of macro- and micronutrients. In addition, the process of bioconversion to LA is fast and efficient (RedCorn et al., 2018). Bila-novic et al. (2011), made an economic assessment of the carbon sources used to produce LA. The price of potato waste is about 15 to 50 U.S. dollars/t, which is less than the cost of usual substrates in standard media for LA production such as molasses, starch, lactose, and glucose (54 to 317 U.S. dollars/t). The use of this substrate could generate a recovery of 5600 million USD/ year (RedCorn et al., 2018). Ping et al. (2005), utilized potato wastewater and the fungi R. oryzae and R. arrhizus for LA production with direct fermentation. Both fungi demonstrated a high rate of starch hydrolysis. The temperature of 30 °C, pH 6 and initial starch concentration of 20 g/L were the best growing condition for obtaining a product/substrate yield of 0.87g/g with R. oryzae 2062 and 0.97 g/g starch with R. arrhizus 36017 after 36 h of fermentation. Potato starch was used by Oda et al. (2002), to assess 38 strains of R. oryzae in the production of this acid. R. oryzae 4707 produced the highest final concentration of LA and was reported to exhibit a gradual production of this metabolite during the first 6 days. Since this fungus was able to secrete amylases, cellulases, hemicellulases, and pectinases from 33.1% initial starch, only 7.6% starch remained in the culture medium at the end of fermentation. Zhang (2008) also used this residue and obtained 88.70 g/L LA from 100 g/L of substrate in a bubble column bioreactor with R. arrhizus.
Rice. Nearly 650 Mt of rice were produced worldwide in 2007. This amount of rice could also generate a large amount of rice bran, which represents between 8 and 10% of the weight of the grain (Li, Tianwei, Jike, Zixin, & Lu, 2012). Depending on the separation process, rice bran can be classified as yellow or white. They differ in the content of starch and oils. For LA fermentation, oil-free bran is preferred (Li et al., 2012). Many researchers have worked on the feasibility of the production of LA from residues of rice production as a carbon source. However, some of them have also found considerable quantities of thiamine, riboflavin (B vitamins) and amino-acids in this raw material, and they have reported a positive result as a source of nitrogen and micronutrients (L. Gao et al., 2008; M. Gao et al., 2008). Watanabe et al. (2016), isolated a novel LAB (L. rhamnousus M23) able to produce extracellular protease with a high degree of activity. This enzyme can degrade complex substrates such as a non-sterilized mixture of the drainage from the washing of rice and rice bran, using them as the only source of nutrients for the growth and the production of LA. L. delbrueckii JCM1106, producer of D(-) LA, was used to transform the sugars present in the enzyme hydrolyzate from broken rice, achieving a high productivity of 3.59 g/Lh (Nakano, Ugwu, & Tokiwa, 2012). Lu et al. (2009), reported a yield of 73% in LA production using old untreated rice, which exceeds by 5.79% and 8.71% the production for fresh corn and processed rice, respectively. Most of the studies of LA production via fermentation consist of a single inoculation or a mixture of two microorganisms. Gao et al. (2008), focused on the utilization of a LAB consortium consisting of L. plantarum, L. fermentum, and L. paracasei to increase the production of acid using rice straw, since L. plantarum and L. paracasei are homofermentative, while L. fermentum is heterofermentative. Not only LA, but also acetic acid, is obtained, at a ratio of 3.88:1 (lactic acid:acetic acid).
Food Residues. Food residues have a high content of moisture, carbon such as starch, lipids, and protein (Yumiko Ohkouchi & Inoue, 2007). In the fermentation of food residues with natural microbiota, it has been identified that BAL such as L. plantarum and L. brevis are the dominant microorganisms, which favors the produc-tion of LA (RedCorn & Engelberth, 2016). The fermenta-tion of this type of waste in discontinuous systems with the addition of neutralizing agents generates a rapid hydrolysis of the raw material, and its main product is lactic acid (80%), as well as other by-products in lesser proportions (Gu, Liu, & Wong, 2018; Omar, Aini, Rahman, Hafid, & Yee, 2009). However, modifications of the culture system can affect the production of organic acids. Gu et al. (2018), determined that a semicontinu-ous culture, adding daily between 50 g/d and 150 g/d of organic load, drastically reduces the production of lactic acid at percentages lower than 10% and favors the production of butanoic acid. RedCorn (2016) determined that the optical conditions of temperature, pH, fermentation time, and initial concentration of food residues, us-ing natural microbiota as a catalyst, are 41 °C, pH 5.5, 20 h, and 150 g VS/L, respectively. Sakai et al. (2011), emphasized that food waste containing a high amount of total sugar close to 129 g/kg of waste can be transformed via enzymatic hydrolysis to glucose, reaching a concentration of 80 to 83 g/kg of waste. Kim et al. (2003), found a high LA yield of 0.91 g/g from coffee residue. From this residue, 80 g/L of LA were obtained in only 48 hours. Ohkouchi and Inoue (2006) also obtained a high yield of 1.11 g LA/g food waste, where the waste used showed a starch content of over 60% of total carbohydrates. The balance of nutrients and the amount of fermentable sugars in the culture medium is difficult to control when domestic wastes are used, since the chemical composition of these materials may vary from day to day. For that rea-son, Ohkouchi et al. (2007), determined that a 10:1 ratio of sugar/nitrogen must be supplied in order to generate high concentrations of LA. The study presented by Pleissener et al. (2017), on the manufacture of LA in a technical scale of 50 L under real process conditions, without sterilization of the medium, demonstrated the feasibility of the use of food waste for the industrial production of LA, with a starch product yield of 0.75 g/g and with no considerable production of other organic acids.
Dairy industry. Approximately 85% to 90% of all milk used for cheese production is discarded as whey, which is mainly composed of lactose, proteins, and minerals. In Colombia, in 2009, for example, 1084 million liters of milk were used for cheese production, and 921.7 million liters were discarded as whey (Parra, 2009). There are two types of whey: sweet and acid. Both are successfully used in the production of LA, supplemented with yeast extract, glucose, peptone, soybean meal, or corn steep liquor. This sub-product is the most common substrate for the fermentative production of LA using LAB, and it has already been employed on an industrial scale by Shefield By-Products (Hofvendahl & Hagerdal, 2000; Solá-Villatoro, 2006). L. delbrueckii spp. bulgaricus, L. helveticus, and L. casei are among the strains that have been used for the production of LA from whey. However, L. helveticus is preferred, because of its high rate of production (Panesar et al., 2007). Urribarrí et al. (2004), determined that in a continuous culture of L. helveticus using deproteinized whey, the optimal conditions for the growth of this microorganisms are 40 °C, with pH 5,9 and a dilution rate of 2 h-1. The maximum specific growth rate was 0.47 h-1, and the specific rate of substrate consumption was 0.06 kg/m3. Volumetric productivity for the conversion of lactose to LA in continuous bioreactors has been reported to be about 2 to 4 g/Lh. Roukas and Kotzekidou (1998) found an increase of LA using fed batch fermentation and immobilized cells of L. casei and Lactococcus lactis with deproteinized whey as a substrate. Moreover, in the dairy industry, 1% of the total production of yogurt it is considered to be waste, with a high content of sucrose, glucose, and galactose, and it is more polluting than whey. Yogurt serum was used by Alonso et al. (2010), for the production of LA in a batchtype process, obtaining a yield of 90% and a maximum LA production of 25.90 g/L. The chemical oxygen demand (COD) was measured for yogurt serum before fermentation, showing a concentration of 90 g/L. After fermentation, it was reduced to 72.50 g/L. This demonstrates that the use of waste in fermentation pro-cesses helps to reduce the environmental impact. Table 3 shows a review of some economical raw materials that have been explored for LA fermentation.
Considering that some wastes are richer in nitrogen (e.g. wheat bran, corn cobs) and others as a source of carbon (e.g. starchy materials), depending on the availability of these raw materials in a region, the process could be optimized with reduced costs through a formulation that combines these wastes, reducing the purchase of commercial inputs. Less-developed regions such as Latin America and Africa produce millions of tons of the above waste annually, discarding it in most cases, without an integrated environmental plan. In spite of the commercial challenges that the generation of a new industry implies, the use of these raw materials for the production of LA in these regions of the planet is considered to present an opportunity for environmental and economic development.
SIMULTANEOUS SACCHARIFICATION AND FER-MENTATION OF LA
The bioconversion of materials to LA can be done in one step by coupling enzymatic hydrolysis and microbial fermentation. This process is known as simultaneous saccharification and fermentation (SSF), (Marques et al., 2008; Watanabe et al., 2012). A benefit of SSF is the decrease in the inhibition by the substrate caused by the accumulation of sugars generated when enzymatic hydrolysis is performed separately, which allows an increase in the rate of saccharification and productivity. However, when cellulosic materials are used, inhibition by cellobiose remains even when SSF is implemented.
The addition of β-glucosidase at the beginning of SSF could help to avoid this problem (M. Adsul, Khire, Bastawde, & Gokhale, 2007; John et al., 2007; Ping et al., 2005). SSF can be successfully carried out when the microorganisms and enzyme systems have similar culture conditions, because if there is a marked difference between them, the production of LA is drastically affected. For that reason, identification of the microbial and biochemical kinetics is of great importance for determining the optimum conditions for improving the performance of SSF (John et al., 2006; Venkatesh, 1997). Marques et al. (2008), conducted a comparison of separate and SSF using paper industry waste. They obtained better results with fermentation in a single step, and the production of LA increased from 51.90 g/L to 72.90 g/L. As mentioned above, the use of commercial enzymes for the hydrolysis process generates a considerable expense for the manufacture of LA. Due to this, other types of cultures have been explored. For example, microorganisms produce LA capable of excreting enzymes to carry out the hydrolysis, and the mixture of different species of microorganisms (co-cultures) for the development of SSF increases the economic feasibility of the whole process (Pleissner et al., 2017; Watanabe et al., 2012). Miura et al. (2004), developed a mixed culture for the production of LA using corn cob as a substrate; A. thermophilus (producer of cellulase) was in charge of enzymatic hydrolysis, while Rhizopus sp. was used for the production of LA. The same co-culture strategy was used by John et al., (2007). They mixed two strains of Lactobacillus in order to degrade cassava bagasse: L. casei and L delbrueckii. Aerobic microorganisms such as Aspergillus sp. are used in the industry to produce amylase. Kurosawa et al. (1988), performed an immobilization of a mixture of Aspergillus awamori for saccharification and Streptococcus lactis (aerotolerant anaerobic) for fermentation with starchy substrates. They generated 25 g/L LA from 50 g/L of substrate.
GENETIC STRATEGIES FOR INCREASING THE PRO-DUCTION OF LA
The metabolism of LA microorganisms has been modified using genetic and metabolic engineering tools in order to increase their production of LA or to introduce exogenous genes in order to convert the microorganisms into producers of this metabolite. Rhizopus strains have two lactate dehydrogenase (LDH) genes, ldhA and ldhB. LdhA is believed to be primarily responsible for the production of LA (John et al., 2009; Christopher D. Skory, Mertens, & Rich, 2009). That is why Skory (2004) tried to increase LDH activity by determining the length of the ldhA gene and increasing the number of copies of this gene in R oryzae. This resulted in an increase of specific LDH activity and LA production. Furthermore, there was a decrease in by-products when compared with the control case. In other studies, Rhizopus sp. was transformed in order to decrease the activity of the alcohol dehydrogenase enzyme, obtaining a mutant strain capable of generating 35% less ethanol and about ten times more LA than the parent strain (Christopher D Skory, Freer, & Bothast, 1998). Moreover, L. delbruekii UC-3 is a strain mutated by exposure to ultraviolet light. It has the ability to tolerate high sugar concentrations, making it an attractive microorganism for LA fermentation with molasses waste and sugarcane bagasse. This microorganism also has enzymes that can degrade cellobiose and could be used with this type of substrate (M. Adsul et al., 2007; Dumbrepatil et al., 2008). Zhou et al. (2011), obtained a recombinant strain with multiple delections (E. coli CICIM B013-070) able to produce 125 g LA/L from glucose. Its low requirements for minerals and its microaerobic conditions make this strain suitable for large-scale use. Yeast does not have the Ldh enzyme, and therefore it lacks the ability to produce LA. Ishida et al. (2005), reported the successful transformation of Saccharomyces cerevisiae by the insertion of two copies of the LDH gene on the genome, which express bovine LDH under the control of native promoter pyruvate de-carboxylase1 (PDC1). This strain proved to be an efficient and pH-tolerant microorganism, which produced approximately 50 g/L LA(+) with high optical purity with-out the need for neutralization (M.-T. Gao, Shimamura, Ishida, & Takahashi, 2009). The problem presented by S. cerevisiae is the large amount of ethanol produced. Kluyveromyces lactis has only one gene that codifies for pyruvate decarboxylase, while S. cerevisiae has three genes for this enzyme. Sinhg et al. (2006), suppressed this gene in K. lactis and avoided the production of ethanol. Thus only LA was generated, with a high concentration of 109 g/L. In more recent studies, Lee et al. (2017), obtained K. marxianus yeast able to co-express two foreign Ldh enzymes, each with a different optimum pH (basic or acid), and the production of LA improved. These results show the importance of a pH drop in the culture and its effect on the production of lactic acid.
CONCLUSIONS
The high content of cellulose, hemicellulose, starch, and lactose in agroindustrial and household wastes make them a replacement for expensive pure sugars as carbon sources. In some cases, they can also be utilized as a nutrient source for LA production by fermentation. However, the use of these materials in LA production has some drawbacks, due to the pretreatment required. SSF and the use of recombinant microorganisms have made a great contribution to the increase of LA production, overcoming some problems, such as high content of undesirable by-products and inhibition by sugars in the media. In the pretreatment of the wastes, enzymatic hydrolysis is considered to be more beneficial for the performance of the microorganism, this in spite of the higher cost of this treatment. Although both fungi and LAB are reported to give very good yields in the production of LA using these residues, LAB are considered more promising, due to their operational advantages and the greater facility of genetic transformation, which increases production. In spite of the commercial challenges that the generation of a new industry implies, the use of these raw materials for the production of LA in regions as Latin America and Africa is considered to be an opportunity for environmental and economic development.