INTRODUCTION
Quinoa (Chenopodium quinoa Willd.) is a plant of the Amaranthaceae family that grows mainly in the Andean region, which is the center of origin of the species (Apaza et al. 2013). Quinoa grains have a high nutritional value due to its ideal balance of essentials amino acids, fatty acids, content of vitamins and minerals such as iron and calcium (Muñoz & Acevedo, 2002). Quinoa is qualified as a functional food (Filho et al. 2017; Abugoch, 2009) because its nutritional quality is superior to other grains like rice and wheat (Vilcacundo & Hernández, 2017; Navruz-Varli & Sanlier, 2016). According to the United Nations Food and Agriculture Organization FAO (2010), Quinoa is a promissory crop by reason of its multiples uses, high adaptability to a wide range of climatic conditions, tolerance to abiotic factors and high genetic diversity (Montoya et al. 2005; Mújica & Jacobsen, 2006; Vargas et al. 2015). However, Quinoa seed quality is influenced by the low germination rate and reduced vigor; seeds lose viability more rapidly than cereals because of the porosity in the integument, which allows a seed to easily gain or lose moisture and may initiate germination in the panicle (Spehar & Santos, 2002).
Quinoa seeds have an orthodox behavior, because they naturally dehydrate up to a water content in equilibrium with the environment that implies a subsequent artificial drought tolerance up to about 5% water content without losing viability (Ellis et al. 1990). In this type of plant, seed cells get dehydrated due to losses of vacuolar water, supplied from their mother plant to seed during the maturity process, which helps keep viability and storage potential (Magnitskiy & Plaza, 2007). Conservation techniques of orthodox seed require a minimum physiological activity (Calle et al. 2010). However, some non enzymatic events occur in low water contents, this reaction leads to seed aging, producing an alteration on functional proteins that weakens the metabolic system and limits the capacity to defend itself from free radical damage and to repair damages throughout the germination process (Castellión et al. 2010).
The moisture content is a relevant factor that can affect the rate of damaging and aging reactions during seed storage period (Castellión et al. 2010). One of the conditions that need to be met in Quinoa seeds storing is having about 10% of moisture to increase longevity (Ellis et al. 1988). Under unsuitable conditions of relative humidity 75 - 80% and temperatures of 10 - 20ºC respectively, seeds lose viability in a short period of time (Ellis et al. 1993). Storage conditions can influence the dormancy pattern output and seed longevity along with seed variety from contrasting environments (Strenske et al. 2017; Castellión et al. 2010).
The first long term seed conservation was carried out in the Bolivian C. quinoa germplasm bank, with the conservation of 247 accessions of quinoa (Rojas et al. 2003). According to the FAO (2010), ex situ conservation of 16,263 accessions of quinoa and wild relatives was carried out in 59 seed germplasm banks in 30 countries around the world (Rojas et al. 2013). Storage and conservation of germplasm as well as commercial seed production, require adequate viability prediction and monitoring (Ceccato et al. 2013).
As a reproductive organ of plants, seed has a vital role to play in the propagation and perpetuation of the species; likewise as a source of food for animals and humankind. In agricultural production, the seed quality is essential for crop success. This quality depends on, among other features, its genetic, physiological, health and physical characteristics (Doria, 2010).
According to Antuna et al. (2003), physiological quality includes structures and processes that allow the seed to maintain high rates of viability; this quality component must be checked before sowing or storage. Seed storage allows distribution in time and space. Conditions associated with seed quality like germination and viability must be preserved. Water content, temperature and oxygen are fundamental factors in the control of seed longevity (Roberts & Ellis, 1989).
Considering the limited information on quinoa seed viability stored under different temperatures, the objective of this research was to determine the effect of cold storage in the viability of four accessions of Ch. Quinoa from different origin.
MATERIAL AND METHODS
The evaluation was developed during the years 2014 - 2015 at the Universidad de Ciencias Aplicadas y Ambientales U.D.C.A, in the agrarian unit ‘El Remanso’ (4° 47’ 57.98” N 74° 2’ 47.17” W, altitude 2.560 m.a.s.l) located in the savannah north of Bogotá.
Seed material was obtained from four accessions of Chenopodium quinoa. Three of them were from production zones of Colombia: Nariño, Boyacá and Bogotá (Usme); the fourth one was a Bolivian accession donated by the Jardín Botánico de Bogotá in the meeting “Traveling Exhibition of Quinoa - Colombia 2014”. The time after seeds are stored is referred to as post-harvest period, which corresponded to one month for Nariño and three months for the other accessions.
A completely random design was used in a 3x4x2 factorial arrangement (temperature, accession and substrate, respectively) with three repetitions and an experimental unit consisting of one hundred seeds. Germination assessment was carried out in greenhouse, using as commercial peat soil substrate and soil enriched with organic matter, in trays of 128 alveoli.
To identify the initial state of the seeds, physiological tests of moisture content, imbibition and viability were performed. The initial moisture (fresh weight percentage FW%) of the seeds was determined by its constant dry weight after drying. For this purpose, the samples were dried in an oven at a temperature of 103ºC, during 17h as recommended by the ISTA (International Seed Testing Association).
The imbibition curve was estimated from 10g of seed, with measures of weight every hour for six hours. The viability test was performed using the tetrazolium test technique in Petri dishes with tetrazolium chloride (2,3,5-triphenyl-2H-tetrazolium chloride) at 1% and temperature of 30°C in a period of two hours. The seeds were observed by stereoscope and the one that presented homogeneous staining of the embryo (in line with the cotyledons and the radicle that surrounds the perisperm in the form of a ring) was defined as viable seed; according to the first two seeds of figure 1.
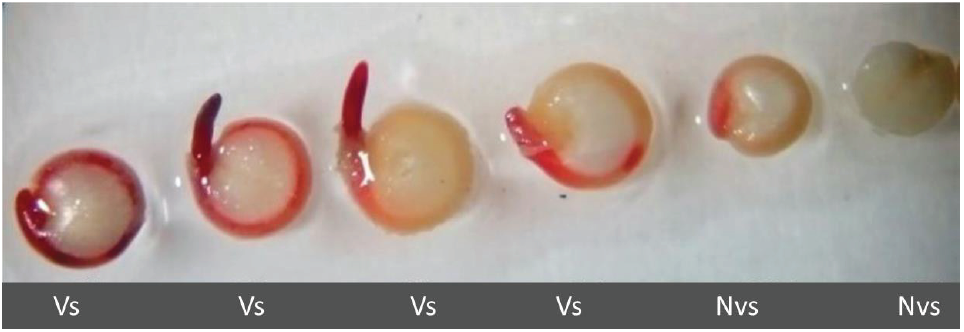
Figure 1 Reddish stain of the embryo in the Nariño accession. Viability test by tetrazolium (2,3,5-triphenyl-2H-tetrazolium chloride) 1%, in seeds of Chenopodium quinoa Willd. Vs: Viable seed; Nvs: non-viable seed.
The different accessions were stored for 10 months, at temperatures of 4, -20 and -80°C, using hermetic bags, covered with aluminum foil to ensure a minimum gas exchange and absence of light, temperatures of seeds were maintained in a cooler and a freezer. Germination tests were carried out at 2, 4, 6, 8 and 10 months of conservation. Germination tests were carried out prior to storage (ambient conditions) at the beginning of the assay and after ten months under the same conditions (control).
Data were subjected to analysis of variance (ANOVA). Post hoc tests of means comparison were performed using Tukey’s honestly significant difference (HSD). The analyses were performed with the R Core Team statistical package, 2015.
RESULTS AND DISCUSSION
The initial moisture content corresponded to 10.66, 11.59, 11.80 and 12.47% for the Nariño, Boyacá, Bogotá (Usme) and Bolivia accessions, respectively. The seed water content is a relevant factor that can affect the rate of damaging and aging during long time storage (Justice & Bass, 1978). An alternative to reduce deterioration and enhance longevity of orthodox seeds is decreasing water content and storage temperature.
The imbibition curve of the four accessions of quinoa (Figure 2 and the differentiation between germination phases I and II were observed. Phase I corresponds to the rapid increase in water absorption as shown from 0 to 5 hours with an exponential behavior of the curve, evidencing weight gain in the four accessions. The highest rate occurred after one hour of imbibing the seed. After five hours, the absorption of water decreases; thereafter, the weight begins to stabilize in the transition to phase II. In the second phase, Hartmann et al. (2011) showed that metabolism could be activated because of protein synthesis, carbohydrates and lipids; perisperm is the unique carbohydrate source, after emergence cotyledons develop chlorophyll and are efficient photosynthetic organs.
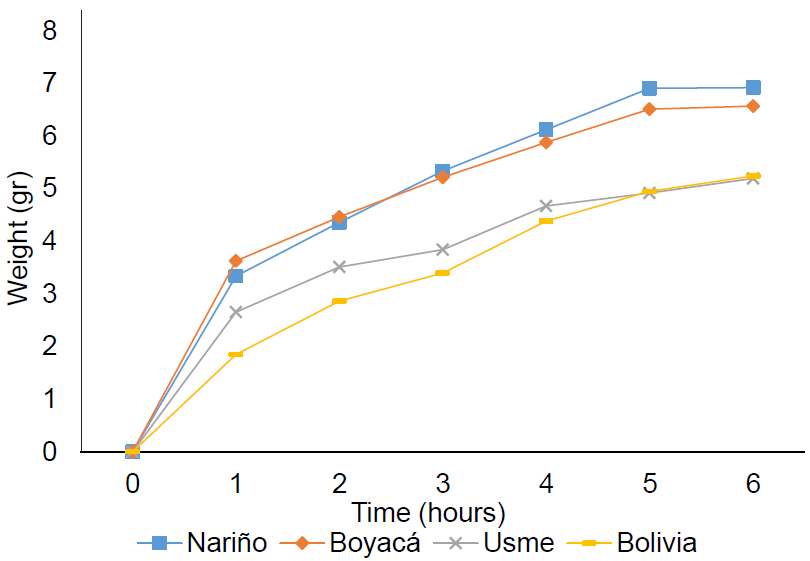
Figure 2 Imbibition curve of Chenopodium quinoa Willd. in accessions from Nariño, Boyacá, Bogotá (Usme), Bolivia.herefore, the nutrition is available for the embryo and it starts growing up (Rosa et al. 2009).
Quinoa seeds have a high and rapid permeability, under optimal physiological and environmental conditions, it has a high germinative potential. Depending on the environmental conditions, this aspect represents a risk, because the seed can germinate in pre-harvest period (Gubler et al. 2005; Kermode, 2005).
Regarding the viability quantification by the tetrazolium method, the accessions have shown percentages lower than 80%. It is noteworthy that genetic and physiological quality of quinoa seed had a viability and germination rates close to 100% in the harvest. However, its loss of life and viability is faster compared to cereals due to the porosity in the integument, which allows it to gain or lose moisture easily (Spehar & Santos, 2002).
In seed viability, the measures indicated that the accessions from Boyacá and Nariño reached 71 and 70%; while the accessions from Bolivia and Usme (Bogotá) registered 55 and 10%, respectively. The lower viability in the Bolivia and Bogotá (Usme) accessions can be attributed to post-harvest management conditions, genotype, seed provenance and prevailing conditions during seed development (Gómez-Tejero et al. 2006). Castellión et al. (2010) obtained viabilities close to 100% under storage conditions at -20°C and seed humidity content of 6%. Nevertheless, in the present research seeds were studied in a post-harvest period of 1 to 3 months without specific storage conditions, seeds were acquired from growers in different zones, fresh seeds were not available, additionally, viability technique with tetrazolium sales, is subjective and not discriminate seed in dormancy.
The germinative potential percentage of germination for each accession of quinoa assessed at different storage temperatures, presented no significant differences within the assessment times (Table 1) during the period. Nevertheless, according to data in table 1, the highest germination percentages occurred under storage temperatures of -20 and 80°C (Figure 3). Walters et al. (2004) mention the different degrees of low temperatures that contribute to prolong seed´s lifespan; this aspect depends on intrinsic properties, handling and harvest of seed. Moreover, pre-storage under higher temperatures had dramatic effects on longevity.
Table 1 Germination percentage of four accessions of quinoa, under different storage temperatures and two substrates. Glass house, U.D.C.A.
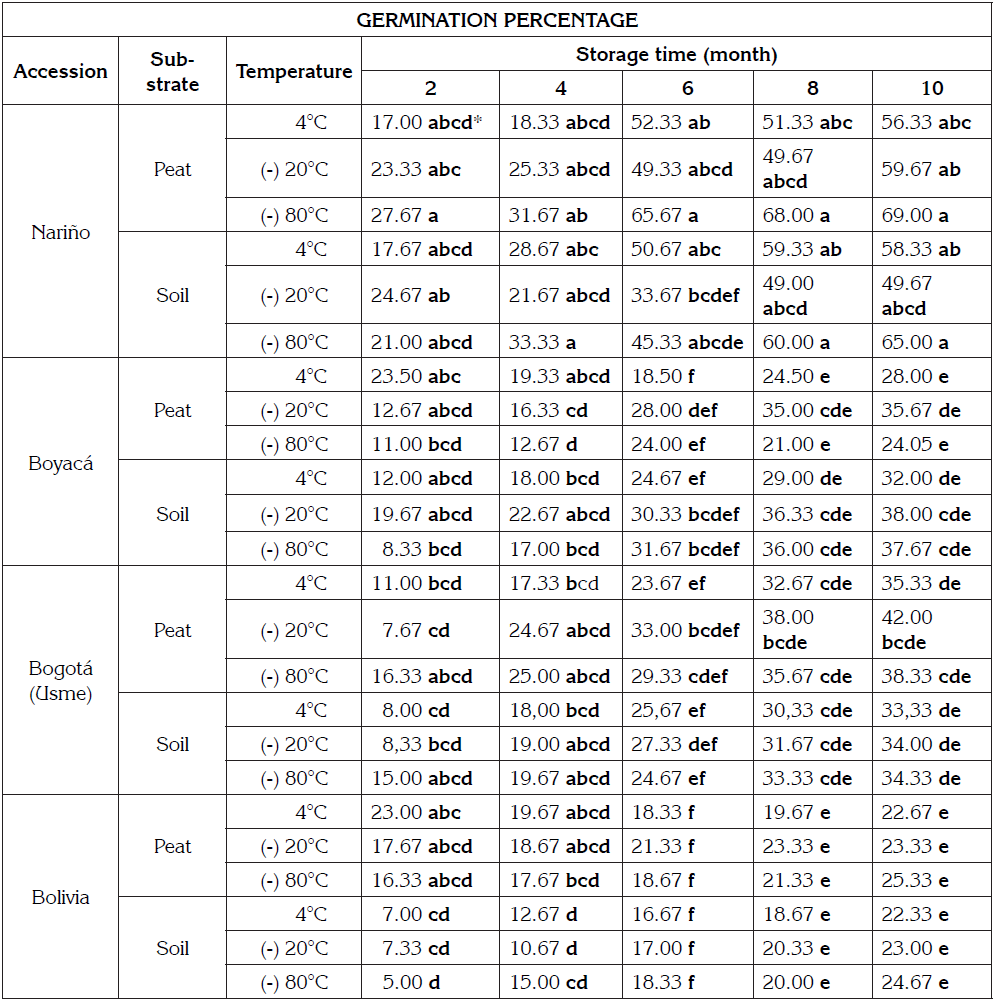
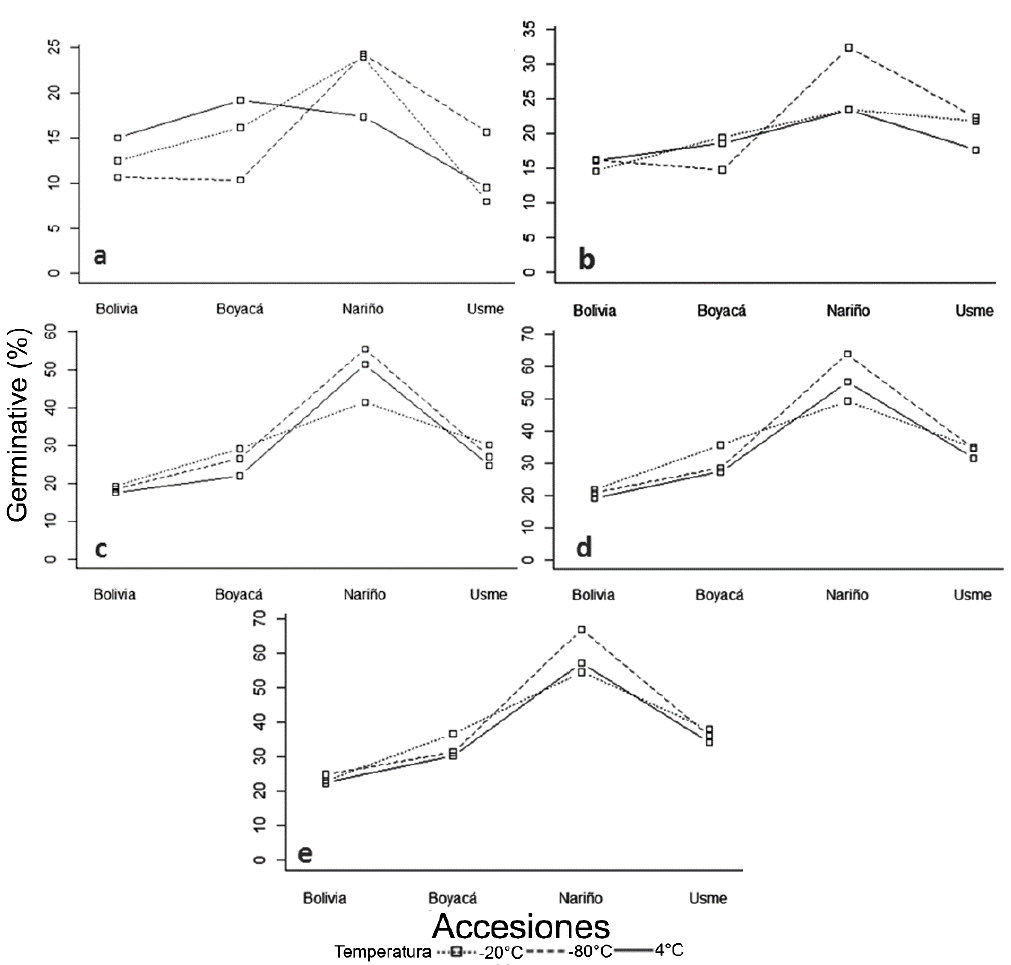
Figure 3 Germination percentage in four accessions of quinoa (Boyacá, Bolivia, Nariño, Bogotá-Usme) with respect to the temperature treatment in five storage times (months 2, 4, 6, 8 and 10), respectively figures from a to d
Furthermore, the effect of substrate had no significant differences, in contrast, it showed greater germinative power in commercial peat treatment. As regards interaction between substrates and accessions, there were significant differences registered in the second month of storage (figure 4a; p-value: 4.8E-03) for cultivars of Bolivia and Boyacá, while for Nariño they happened in the sixth month (Figure 4b).
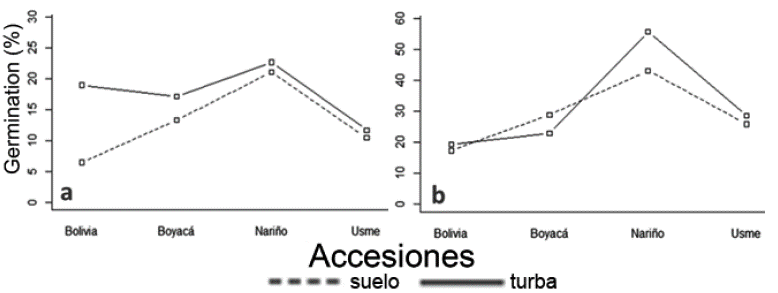
Figure 4 Accession and substrate interaction on percentage of quinoa germination. a) month two of storage; b) month six of storage.
Percentage of germination showed notable differences between accessions over the months of evaluation. The Nariño accession obtained the greatest response. This high percentage of germination can be attributed to the short time of harvest, which indicates its high seed viability, according to tetrazolium test. In addition, the Nariño accession registered the lowest seed moisture content (10.66%). Moisture content close to 10% was reported as an optimum condition for storing orthodox seeds.
The accessions of quinoa were measured twice. First, before starting the assay, and second, ten months later without storage (control) at room temperature under laboratory conditions (Figure 5). The losses of germinative potential were evidenced for all accessions. A similar behavior was found on the two substrates evaluated. Results were concordant with those presented by Castellión et al. (2010) , who indicated losses in viability and germination in quinoa seeds stored under ambient conditions, evidencing different responses between cultivars. The foregoing was associated with the origin of the cultivar, genotype, and physiological harvest conditions.
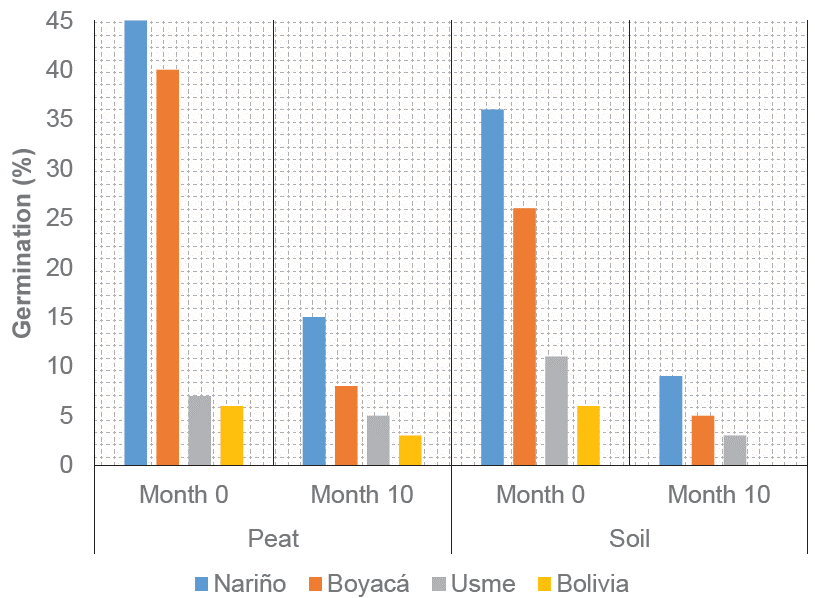
Figure 5 Behavior of quinoa seeds under ambient temperature conditions at the beginning of the evaluation and after ten months (Month 0 and 10, respectively).
Accessions showed a progressive increase in the percentage of germination over the months of cold storage. This indicates that temperature changes contribute to breaking the dormancy in quinoa seeds. According to Ceccato et al. (2011), the lack of dormancy in quinoa cultivars highlights the importance of determining the effect of environmental and physiological factors over changes in the dormancy levels of quinoa seeds during its development, maturation and post-harvest storage. Similar works in sesame (Almeida et al. 1999), cucumber (Torres et al. 2002) and passion fruit (Catunda Aragão et al. 2003), registered an increase in germination after some months of storage.
Quinoa is a seed featuring orthodox behavior, which enables its storage under physiological conditions (maturation, water content) and an appropriate environment. According to the results of this study, it is recommended to keep under cool conditions seeds intended for establishing new crops. Viability is progressively affected by the post-harvest period or the time that passes without adequate storage conditions. Furthermore, seeds destined to germplasm banks are recommended to be conserved at temperatures equal to or lower than -20°C. Results for 4°C were not significant, however there were better responses to lower temperatures.














