Introduction
Mediastinal lymphadenopathies are a diagnostic challenge. In breast cancer, metastases can be found in lymph nodes of the mediastinum during the progression phase. These lymph nodes must be sampled to reach a diagnostic conclusion1. In the United States, most patients do not present metastases at the time of diagnosis, but 30% of patients who initially had a limited disease and initiate treatment may have distal metastases in the bones, lung, liver, and lymph nodes2.
Patients with breast cancer undergo alterations in the expression of estrogen receptors (ER), progesterone receptors (PR), and in the amplification of human epidermal growth factor receptor 2 (HER2) during tumor progression, possibly influenced by adjuvant therapies, which significantly impacts on the survival. Therefore, the molecular re-characterization in relapse improves the patient's management and survival3.
There are several techniques available for sampling lymph nodes, such as conventional transbronchial needle aspiration (c-TBNA), computed tomography-guided transthoracic needle aspiration (CT-guided TNAB), ENB (Electromagnetic Navigation Bronchoscopy), transesophageal ultrasound-guided fine-needle aspiration (EUS-FNA), and mediastinoscopy1-5. These techniques have certain disadvantages, such as the low possibility of obtaining a sample in lesions smaller than 2 cm with bind puncture, problems with gaining access to central mediastinal locations by a percutaneous approach, the non-availability of techniques like ENB in our environment, the limited access to certain lymph stations by EUS-FNA and mediastinoscopy; in addition, some complications such as pneumothorax, bleeding, air embolization, dysphonia, among others, may occur5.
Endobronchial ultrasound-guided TBNA (EBUS-TBNA) is the most recent advance in the field of pulmonary medicine. EBUS-TBNA has revolutionized the scene for the pathologist and the interventional pulmonologist. It is the most common method used today for the sampling of mediastinal lymph nodes in patients in which oncological pathology is suspected. It has replaced the mediastinoscopy as the first option for the evaluation of lung cancer, in both early and advanced stages, as it can provide the pathological diagnosis, mediastinal staging and the necessary tissue for molecular analysi6. Although its diagnostic accuracy is high (96-98%) in malignancies, it is generally not as good for benign conditions (55-67%)7.
The technique is performed in real-time under direct visualization of the needle, using simultaneous ultrasound and optical images4. Doppler mode is used to visualize the vascular structures and achieve a precise localization of the lymph node stations. This technique uses a needle of 19G to 22G caliber and allows material acquisition for cytology and cellblock construction. ROSE evaluation is carried out at the site of cytological aspiration, which improves diagnostic performance and helps with clinical decision making8. The detection rate of mediastinal lymphadenopathies by EBUS-TBNA, based on the systematic review by Deng et al5, is 86.06+/-9.7%, the diagnostic sensitivity, specificity, accuracy, positive predictive value y negative predictive value for the staging of lung cancer is 85.48%+/-12.89%, 99.09%+/-3.15%, 92.88%+/-4.99%, 98.70%+/-3.03%, 83.03%+/-15.45%, respectively. The most common complications are hemorrhage (0.61%), pneumothorax (0.37%) and infection (0.30%), with a very low mortality of 0.04%. EBUS-TBNA is currently considered a safe procedure to be performed in both outpatients and hospitalized patients9.
In patients with a previous malignity diagnosis, with or without treatment, a new enlarged mediastinal lymph node can indicate a relapse, a second malignity, or a granulomatous condition. The EBUS-TBNA has become an efficient and minimally invasive alternative for the evaluation of these patients10,11. This technical note discusses its usefulness for the diagnosis and molecular classification of progressive breast cancer12.
EBUS-TBNA and ROSE
Linear EBUS is a device that has a larger diameter than conventional bronchoscopes, approximately 7 mm. It is introduced through the mouth, under deep sedation or general anesthesia, using a laryngeal mask, orotracheal tube or trans-oral laryngoscopy. It has a 2 mm test channel, through which a 19G or a 22G needle is advanced. Its field of vision is oblique, between 35° to 45°, so usually, before the procedure, a diagnostic bronchoscopy has been performed, in order to evaluate in detail, the airway. It has a specially designed convex transducer incorporated at the tip, which emits an ultrasound beam parallel to the major axis of the endoscope, from 50° to 90°, with frequencies between 5 and 12 MHz. With the high frequencies, the shallow layers are very well evaluated. It is ideal for detecting mediastinal lesions. EBUS is equipped with color and pulsed Doppler to detect blood flow. It has a processor that gives the image of video bronchoscopy and at the same time, the ultrasound image and pulsed Doppler, which guarantees the accuracy of the puncture. To improve the image, EBUS is equipped with a distal balloon that inflates when the lymph node stations or the lesions to be studied have been identified13,14.
The advantage lies in being able to perform the directed puncture in real time through the bronchial wall, not through the chest wall, as it is the case with the CT guided TNAB, so the lymph nodes and their relation to the great vessels can be identified, and the needle's puncture can be controlled in an efficient and secure manner. The detection of adenopathies by EBUS is possible even with lymph node sizes of 5-10 mm. The lymph node stations that can be accessed are 1, 2, 3, 4, 7, 10, 11, and 1215. However, it is necessary to do preliminary planning of the procedure, taking into account the images, either one or both, of the CT scan and the positron emission tomography (PET scan). Since the puncture of the stations must be sequential, a route should be previously designed; it starts with the highest ones, in order to perform a correct tumor staging in lung cancer13,14 or progressive breast cancer, to get to the affected station. ROSE has allowed the improvement of the quantity and quality of the samples, specifically to identify the mediastinal compromise, to diminish the interventions, and to carry out complementary studies in the cellblocks16,17.
Study of mediastinal lymph nodes with EBUS-TBNA and ROSE in progressive breast cancer
Interventional pulmonology and pathology have a standardized protocol of EBUS-TBNA, ROSE, and cytopathology. The procedure was ambulatory in most of the cases, and it was performed under general intravenous anesthesia, with access through a laryngeal mask or orotracheal tube. All cases were punctured with a 21G needle (Figure 1. A, B, and C); cytology was performed on a liquid basis (Figure 1. D); it was obtained enough material for the cellblock; in addition, there were performed immunohistochemistry, digital analysis, and fluorescent in situ hybridization (FISH).

Figure 1 A. Endobronchial ultrasound equipment. B. Introduction of the needle inside the instrument. C. Real-time puncture, where the needle is visualized inside the lesion*. D. Material recovered during puncture.
The cases were carried out for three and a half years, from January 2015 to June 2018. The EBUS-TBNA and ROSE were performed in 7 patients with a history of breast cancer and clinical suspicion of progression (Figure 2). The average age was 63.5 ± 12.98 years; the most frequently compromised station was 7 (5 patients), followed by 11L (1 patient), and 11R (1 patient). We performed one pass (5 cases) and two passes (2 cases), which were colored with Diff-Quick and evaluated on-site. This allowed us to confirm the metastatic compromise in real time during the procedure.
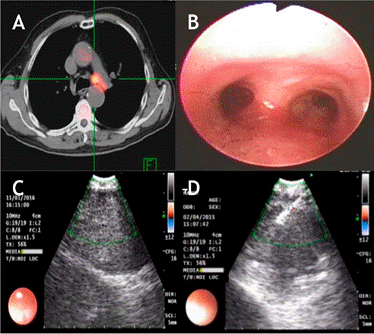
Figure 2 A. PET-CT with a hypermetabolic mediastinal lesion. B. Endoscopic appearance without endobronchial lesions. C. Lesion identified by ultrasound. D. Needle puncture in real time for sampling.
Afterward, the sample was sent to the cytopathology laboratory, where liquid-based cytology was performed with Papanicolau staining (PAP) and cellblock with Hematoxylin and Eosin staining (H & E) (Figure 3. A and B). In all cases, the immunohistochemical technique was performed with the antibodies GATA-3 (L50-823), TTF-1 (SP-141) and CK-19 (A53-B / A2.26), in order to confirm the primary origin in the mammary gland; subsequently, there were measured the biomarkers RE (SP-1), RP (1E2), HER2 (4B5), and Ki-67 (30-9) (Table 1).
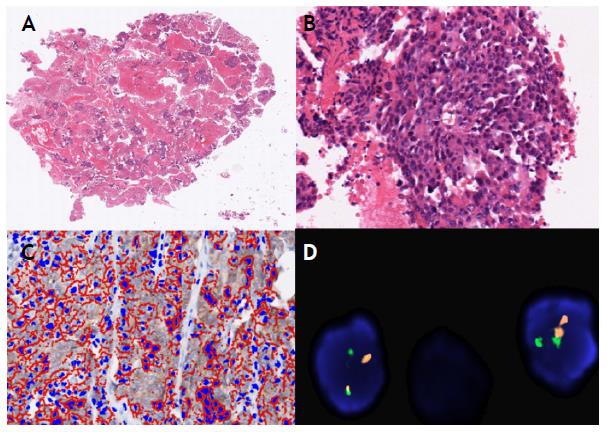
Figure 3 A and B. Cellblock, quality and quantity of tumor cells that allows the diagnosis and molecular characterization, H & E staining (4x and 20x). C. Digital analysis HER2 (Score 2+), equivocal (20x). Immunohistochemistry technique and analysis with software approved by the FDA. D. Study of the amplification of the HER2 oncogene: Not amplified. FISH technique (60x).
Table 1 Molecular classification of breast cancer; it was performed a digital analysis with FDA approved software
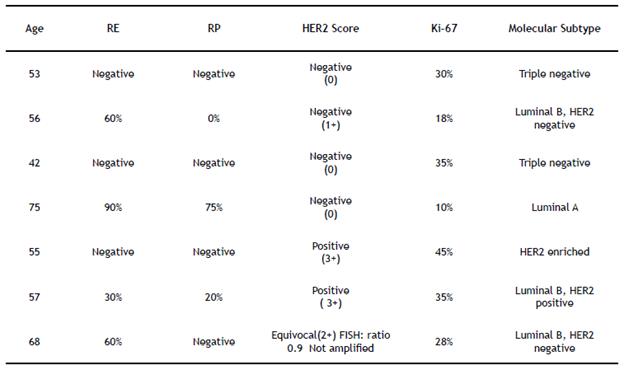
The digital analysis of breast biomarkers was carried out with software approved by the FDA (Food and Drug Administration), which allows characterizing, according to the expression of biomarkers, the molecular subtype of breast cancer. All cases were accepted for digitization by the scanner (Figure 3. C). One case required FISH to evaluate the amplification of the HER2 oncogene, which gave an unamplified result in the final diagnosis (Figure 3. D). None of the patients required mediastinoscopy or additional procedures.
Conclusion
EBUS-TBNA is an efficient ambulatory procedure for the study of mediastinal lesions, as well as paratracheal or peribronchial adenopathies in suspected cases of breast cancer progression. This study should be well planned, sequenced, and standardized, with the accompaniment of a pathologist.
The immediate on-site feedback to the interventional pulmonologist and the early evaluation of the samples allows having enough material for in situ decision making, thus avoiding additional procedures.














