Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Infectio
Print version ISSN 0123-9392
Infect. vol.15 no.3 Bogotá July/Sept. 2011
1Grupo de Investigación Dermatológica, Facultad de Medicina, Universidad de Antioquia, Medellín, Colombia
2Centro de Investigación en Biomoléculas, Universidad Industrial de Santander, Bucaramanga, Colombia
3Centro de Investigación de Excelencia, CENIVAM, Bucaramanga, Colombia
Recibido: 08/10/2010; Aceptado: 03/08/2011
Abstract
Objective: To determine anti-Candida albicans activity, cytotoxicity and drug interaction of essential oils and extracts from plants collected in Colombia.
Materials and methods: The antifungal activity was evaluated following the AFST-EUCAST protocol. With most active samples, the inhibition of the formation of germ tubes and budding, the in vitro pharmacodynamics, using time-kill assays, and the interaction with itraconazole and amphotericin B following the chequerboard technique were evaluated. The cytotoxicity assay for all samples was done using MTT.
Results: Strong activity in 17.57% of the samples was found. The lowest MIC values were obtained with Piper bredemeyeri Jacq and Lippia origanoides Kunth (B) oils and Morinda royoc L extract. The three samples inhibited the formation of germ tubes and budding. P. bredemeyeri Jacq oil and M. royoc L extract samples showed fungicidal activity at 2xMIC. A synergistic effect was obtained with the combination of P. bredemeyeri Jacq oil and itraconazole, but not for the combination with amphotericin B. Active samples against C. albicans were not cytotoxic on Vero cells ATCC CCL-81, excluding P. bredemeyeri Jacq oil.
Conclusions: The results of this study suggest that Colombian medicinal and aromatic plants represent an untapped source of compounds with anti-C. albicans activity that could be a resource in the development of new therapeutic natural products.
Key words: Essential oils, extracts, Candida albicans, antifungal activity, cytotoxicity, synergism
Resumen
Objetivo. Determinar la actividad anti-Candida albicans, la citotoxicidad y la interacción con antifúngicos de aceites y extractos de plantas recolectadas en Colombia.
Materiales y métodos. La actividad antifúngica fue evaluada siguiendo el protocolo Antifungal Susceptibility Testing Subcommittee of the European Committee on Antimicrobial Susceptibility Testing (AFST-EUCAST). Con las muestras más activas se evaluó la inhibición de la formación de tubo germinal y la gemación, la farmacodinamia mediante curvas de tiempo muerte y la interacción con itraconazol y anfotericina B. Se determinó la citotoxicidad mediante la técnica MTT.
Resultados. Se encontró actividad en 17,57 % de las muestras. La mayor actividad se obtuvo con los aceites de Piper bredemeyeri Jacq y Lippia origanoides Kunth (B) y el extracto de Morinda royoc L. Las tres muestras inhibieron la formación de tubo germinal y la gemación. El aceite de P. bredemeyeri Jacq y el extracto de M. royoc L mostraron actividad fungicida con dos veces la concentración inhibitoria mínima. Se encontró un efecto sinérgico por la combinación del aceite de P. bredemeyeri Jacq e itraconazol, pero no con anfotericina B. Las muestras activas no fueron citotóxicas, excepto el aceite de P. bredemeyeri Jacq.
Conclusión. Los resultados de este estudio sugieren que las plantas de Colombia son una fuente no explorada de compuestos con actividad anti-C. albicans, útiles para el desarrollo de nuevos productos terapéuticos.
Palabras clave: aceites esenciales, extractos, Candida albicans, actividad antifúngica, citotoxicidad, sinergismo.
Introduction
Many researchers, particularly the ones from countries with a rich biodiversity, have contributed to the detection of new antifungal compounds in medicinal plants. Screening by using in vitro evaluation is a useful tool for the discovery of new potential antifungal agents from natural products such as essential oils and extracts derived from plants (1). Colombia is the second richest country in the world in biodiversity, and its floral diversity is estimated at 40,000 species of vascular plants (2). Although Colombia possesses a rich tradition in the use of medicinal plants, the antifungal activity of medicinal plantsâ derivates has not been deeply studied.
Candidiasis is a common infection of the skin, nails, oral cavity, esophagus, and vagina, caused by yeast of the Candida genus. Systemic yeast infections are a common consequence of immunosuppression, long-term indwelling catheters, and endocrinopathies. Candida albicans is the most common pathogen causing that fungal opportunistic infection. Additionally, it has the ability to adhere to host surfaces or to prosthesis leading to the formation of bioï¬lms which further facilitate adhesion, infection and resistance to the antifungals (3).
In Colombia, different studies have shown the importance of C. albicans as the principal agent causing bloodstream fungal infection (44.7%) in tertiary care level hospitals (4), and as cause of invasive infection (43.6% and 54.5%) (5,6).
The number of clinical infections worldwide by C. albicans has risen considerably in recent years, and the incidence of resistance to traditional antifungal therapies is also increasing (7). In addition, drug-related toxicity, significant drug interactions and insufficient bioavailability of the conventional antifungals, have encouraged the search for new alternatives among natural products (8).
The aim of this study was to evaluate anti-C. albicans activity and cytotoxicity of essential oils and plant extracts obtained from aromatic and medicinal plants of different Colombian regions. Furthermore, the combined effects of itraconazole and amphotericin B with the most actives samples, and their pharmacodynamics were evaluated in vitro by the chequerboard method and the time-kill curves, respectively.
Materials and methods
Plant material
Stems and leaves of 74 plants were collected in different regions of Colombia, from 2005 to 2008, as part of a survey conducted by CENIVAM, a research center devoted to the study of aromatic plants and essential oils in Colombia. The taxonomic identification of the botanical samples was performed by José Luis Fernandez at the Herbario Nacional de Colombia, Instituto de Ciencias Naturales, Facultad de Ciencias, Universidad Nacional de Colombia at Bogotá, where voucher specimens were deposited.
The selected plants belong to the following genera: Lippia (Verbenaceae) (26); Salvia (Lamiaceae) (7); Lepechinia (Lamiaceae) (3); Piper (Piperaceae) (3); Baccharis (Asteraceae) (2); Hedyosmum (Chloranthaceae) (3); Illicium (Schisandraceae) (2); Cymbopogon (Poaceae) (3); Minthostachiys mollis (Lamiaceae) (2); Thymus vulgaris (Lamiaceae) (2); and Rosmarinus officinalis (Lamiaceae) (2). Additionally, the following plants were also selected: Cananga odorata (Annonaceae), Tagetes lucida (Asteraceae), Eucalyptus citriodora (Myrtaceae), Turnera aff. diffusa Willd. ex Schult (Turneraceae), Nectandra tomentella (Lauraceae), Sigesbeckia agrestis (Asteraceae), Lantana fucata Lindl (Verbenaceae), Hyptis perbullata (Lamiaceae), Origanum vulgare (Lamiaceae), Zingiber officinale (Zingiberaceae), Cascarilla saravena (Euphorbiaceae), Achyrocline alata (Kunth) DC (Asteraceae), Pimenta racemosa (Myrtaceae), Elettaria cardamomum L (Zingiberaceae), Aloysia triphylla (Verbenaceae), Curcuma monteria (Zingiberaceae), Chenopodium ambrosioides L (Amaranthaceae), Bursera graveolens (Kunth) Triana & Planch (Burseraceae), and Morinda royoc L (Rubiaceae).
Extracts and essential oils extraction
Essential oils (59) and extracts (15) were evaluated. The essential oils were extracted from dried stems and leaves (300 g) by microwave-assisted hydrodistillation as described (9). The extracts were obtained from 40 g of dried leaves from each plant, macerated with 200 ml ethanol and left in suspension for 7 days at room temperature (28 °C). The mixture was filtered and concentrated using a Buchi rotavapor. Stock solutions of both oils and extracts, were prepared in DMSO (≤1% v/v), for subsequent bioassays.
Antifungal susceptibility testing
The Minimum Inhibitory Concentrations (MIC) of all samples were determined by the Antifungal Susceptibility Testing Subcommittee of the European Committee on Antibiotic Susceptibility Testing (AFST-EUCAST) protocol (10). Candida albicans ATCC 10231, C. albicans ATCC 90028 and a clinical blood isolate randomly selected from a group of clinical isolates of the Candida species collected in our laboratory were used at inoculum size of 1-5 x 105 CFU/ml. The oils and extracts were evaluated at five concentrations of 31.25-500 µg/ml. Oils and extracts were considered active when they exhibited MIC values equal or greater than 500 µg/ml. Itraconazole (Sigma-Aldrich, Co., MO, USA) was used as a positive control at a range of 0.031-16 µg/ml with the strains C. krusei ATCC 6258 and C. parapsilosis ATCC 22019. A negative control (inoculum without treatment) was also included. MIC values were expressed as a geometric mean (GMMIC) of tests performed in duplicate in three different assays against each Candida strain and the clinical isolate.
Effect on germ tube formation and budding
The effect on germ tube formation and budding was evaluated according to Ishida, et al. .(11). Different concentrations (31.25-500 µg/ml) of P. bredemeyeri Jacq, L. origanoides Kunth (B) oils and M. royoc L extract were tested with the three yeasts. A suspension of 1-5 x 105 CFU/ml in RPMI of each yeast was prepared, and 100 µl were added to the same volume of each sample concentration. Positive and negative controls with nystatin at 6.25 µg/ml (La Santé, Bogotá, Colombia) and cells without treatment, respectively, were included.
For germ tube assays, after two hours of incubation at 35 °C, the absence or presence of germ tubes was determined using a light microscopy (Nikon, Eclipse E200, Japan). Each sample was tested in duplicate using two different assays. To evaluate the effect on budding, after three hours of incubation at 35 °C, the budding cells/ml were counted using a hemacytometer.
The statistical analysis of the effect on budding, was performed by R version 2.9.1 (The R Foundation for Statistical Computing, ISBN 3-900051- 07-0) using ANOVA after transforming the response variable with square root. The Tukey multiple comparison test was used to compare strains with and without treatment. A 0.05 significance level was employed.
Time-kill assay
The in vitro pharmacodynamics of P. bredemeyeri Jacq oil and M. royoc L extract against C. albicans ATCC 90028 were performed by the method described by Klepser, et al.(12). An initial inoculum ranging from 1-5 x 105 CFU/ml was seeded on each sample, at concentrations of 0.25, 0.5, 1 and 2 times the MIC. The samples were incubated at 35 °C with agitation. At 0, 4, 8, 12, and 24 hours, volumes of 10 µl were then spread onto potato dextrose agar (Oxoid, Basingstoke, Hampshire, UK) and incubated at 37 °C for 24 hours to determine the number of CFU/ml. The limit of detection was 100 CFU/ml. Time-kill curves with itraconazole and amphotericin B (Sigma-Adrich, Co, MO, USA) were used as fungistatic and fungicide controls, respectively. Experiments were carried out in duplicate in two separate experiments. Time-kill curves were constructed by plotting of mean ± standard deviation (SD) of colony count (log10 CFU/ ml) as a function of time (hours) with the Prisma ™ (GraphPad Software, Inc., USA, 2007) statistical package. Fungicidal activity was defined as ≥ 3 - log10 (≥ 99.9%) reduction in CFU/ml from the starting inoculum (0.5 – 2.5 x 105 CFU/ml).
Interaction of essential oils and drugs
Assays were performed using the chequerboard method (13). Candida. albicans ATCC 10231 was used at a final concentration of 0.5-2.5 x 105 CFU/ ml. The final concentrations of the itraconazole and amphotericin B ranged from 0.004 µg/ml to 2 µg/ml and the essential oil of P. bredemeyeri Jacq from 7.813 to 500 to µg/ml. The fractional inhibitory index (FICI) was calculated, and the interpretation was determined as follows: ≤0.5, synergistic effect; >0.5 but <4 no interaction; and ≥4 antagonistic effect (13).
Citotoxicity assay
Cercopithecus aethiops African green monkey kidney cells (Vero cell line ATCC CCL-81) were used. The cells were grown in Eagleâs minimum essential medium (MEM) supplemented. The cytotoxicity of the essential oils and their components was examined in vitro using an MTT (dimethylthiazol-2-yl]-2,5-diphenyl tetrazolium bromide) (Sigma, New Jersey, USA) assay as described by Betancur-Galvis, et al..(14). The minimal dilution of the essential oils that induced 50% growth inhibition of the cells was expressed as inhibitory concentration 50% (IC50). The IC50 values for each compound were obtained by linear regression analysis of the dose-response curves generated from the absorbance data with the R (Development Core Team, Vienna, Austria, 2008) statistical package. IC50 values were expressed as the mean ± standard deviation (M ± SD) of two independent experiments done in quadruplicate.
Results
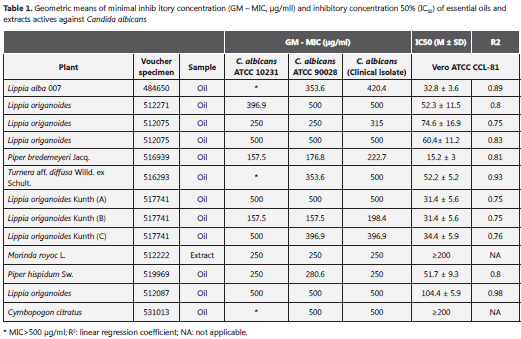
The MICs of 13 active samples (GM-MIC range 157.5-500 µg/ml) tested against C. albicans and the respective IC50 values, are presented in Table 1. The most active samples were the oils from P. bredemeyeri Jacq (GM-MIC of 157.5, 176.8 and 222.7 µg/ml for C. albicans ATCC 10231, C. albicans ATCC 90028 and the clinical isolate, respectively) and L. origanoides Kunth (B) (GM-MIC of 157.5, 157.5, 198.4 µg/ml for C. albicans ATCC 10231, C. albicans ATCC 90028, and the clinical isolate, respectively). Also M. royoc L extract was active (GM-MIC = 250 µg/ml) with the three evaluated yeasts (Table 1).The MIC values of the positive control itraconazole with C. parapsilosis ATCC 22019 and C. krusei ATCC 6258 (0.169 and 0.999, respectively), were in the acceptable range according to the standard protocol (0.3125- 0.250 and 0.0625-0.250 µg/ml, respectively).
Piper bredemeyeri Jacq and L. origanoides Kunth (B) oils inhibited the formation of germ tubes in the two strains and in the clinical isolate at a ≥31.25 µg/ml concentration. Also, M. royoc L extract inhibited the formation of germ tube in both: C. albicans ATCC 90028 strain and in the clinical isolate, at the same concentrations. The inhibition in C. albicans ATCC 10231 was at a ≥125 µg/ml concentration. Germ tubes were observed in the negative control, but not in the positive control. In addition, the treatment of the strains and the clinical isolate with P. bredemeyeri Jacq and L. origanoides Kunth (B) oils, and M. royoc extract, inhibited the process of budding, at 250 µg/ml. The decrease of budding in treated blastoconidia, compared with the negative control was significantly different (p<0.05). The positive control inhibited the budding, and in the negative control there was a normal rate of budding.
The killing activity of P. bredemeyeri Jacq oil and M. royoc L extract against C. albicans ATCC 90028 as well as itraconazole and amphotericin B is represented in Figure 1. At 2 times the MIC, P. bredemeyeri Jacq oil, after 4 hours of incubation, showed fungicidal activity similar to amphotericin B (Figure 1a and Figure 1c, respectively). It was found that at 0.5 times and 1 time the MIC concentration there was a reduction in growth until approximately 8 hours, when colony counts continued to grow near the initial starting inoculum. M. royoc L extract showed fungicidal activity at 2 times the MIC after 8 hours of incubation (Figure 1b). At lower concentrations (0.25, 0.5 and 1 times the MIC), we did not find significant reduction in CFU/ml (<3 - log10) compared with the starting inoculum.
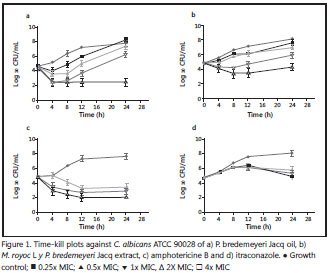
A synergistic effect was obtained for the combination of itraconazole and P. bredemeyeri Jacq (FICI range 0.09-0.13), but no interaction was detected for the combination of P. bredemeyeri Jacq and amphotericin B (FICI=1.06).
According to the American National Cancer Institute (USA) criteria samples with anti-C. albicans activity were not cytotoxic on Vero cells excluding P. bredemeyeri Jacq oil (IC50=15.2 ± 3) (Table 1).
Discussion
Plants, marine organisms, and microbes usually produce biologically active compounds as a defense against predators and competition with neighbors. Thus, it seems logical that most of the drugs derived from natural sources have anticancer or anti-infective properties. Important antifungal agents such as polyenes, echinocandins/ pneumocandins, aureobasidins and sordarins had their origin in natural products (15).
In this study, oils and extracts from medicinal and aromatic plants were tested against two C. albicans strains and a clinical isolate. Currently, there is no agreement on the acceptable level of activity for natural products when they are compared to standard drugs. Aligiannis, et al.(16), suggested a classification of antifungal activity in plant derivates (antifungal activity based on MIC results) as follows: strong inhibitors, MIC up to 0.5 mg/ml; moderate inhibitors, MIC between 0.6 and 1.5 mg/ml; weak inhibitors, MIC above 1.6 mg/ml. According to these criteria, we found strong anti-C. albicans activity in 17.57% of the evaluated samples (Table 1).
Previous studies have shown the antifungal activity in extracts and oils of plants belonging to Piper, Morinda, and Lippia genus (17-19). López, et al.(19), determined a MIC value of 100.000 µg/ ml against C. albicans of the methanolic extract from the plant Piper lanceafolum, using an agar diffusion method. In contrast, in this study, the microdilution standard method AFST-EUCAST was used, and lower MICs with P. bredemeyeri Jacq were found (GM-MIC of 157.5, 176.8, and 222.7 µg/ml, with the three evaluated yeasts) (Table 1).
This is one of the few studies focused on evaluating the anti-C. albicans activity of L. origanoides oils using a standard method for determination of MIC by broth dilution of fermentative yeast. Oliveira, et al .(20), also found important activity against C. albicans in an oil of L. origanoides using an agar diffusion method; however, we cannot compare our results with theirs, because they used a technique that determined inhibition zone (mm) but not MIC (µg/ml).
Candida albicans is a dimorphic fungus, able to grow as yeast or filamentous form. The yeast to hyphal transition begins with the formation of a germ tube and it is the initial stage of hyphal formation (21). The oils from P. bredemeyeri Jacq and L. origanoides Kunth (B) and the M. royoc L extract inhibited the germ tube formation. It is possible that those oils and extracts are acting on an important process in the morpho-transformation of C. albicans, as wall or membrane integrity or reorganization of the cytoskeleton (22, 23). This characteristic could make a new antifungal more selective towards the infecting germ than towards the host cells.
The concentration at which P. bredemeyeri Jacq and L. origanoides Kunth (B) oils were able to inhibit the germ tube formation of C. albicans (≥31.25 µg/ml or ≥125 µg/ml) are within the concentration ranges published for this activity with Melaleuca alternifolia oil (tea tree oil) (0.25 and 12.5% (v/v, approximately 2.25 and 112.5 µg/ml, respectively) (24). Those results make our findings promising if we consider that tea tree oil is used as commercial product for treatments of fungal infections as oropharingeal and vaginal infections by C. albicans (24-26).
Candida albicans has isotropic growth by budding and several antifungal drugs and plant extracts can inhibit budding of yeast cells (21, 27). In this study the treatment of the strains and the clinical isolate with P. bredemeyeri Jacq and L. origanoides Kunth (B) oils, and M. royoc extract, decreased blastoconidia budding at 250 µg/ml compared with the negative control (p<0.05). Previous studies have suggested that the deleterious effect of the essential oil on the cell wall of the fungus could be the main reason for the decrease in the rate of yeast budding, because the cell wall is necessary for cell division (27).
The time-kill studies are useful for the evaluation of the pharmacodynamicsâ characteristics of new antimicrobial agents (28). The oil of P. bredemeyeri Jacq and the extract of M. royoc L showed a fungicidal activity on C. albicans at 2 x MIC after 4 and 8 hours, respectively. It is clinically more important to find fungicidal compounds than fungistatic, particularly in HIV patients, because the prophylactic use of fungistatic drugs has been associated with an increased frequency of innate or acquired drug resistance in clinical isolates (29).
Chemical analyses of most active essential oils against C. albicans have been carried out in our laboratory. The main compounds of P. bredemeyeri Jacq oil were terpenes alpha-pinene (20.3%) and beta-pinene (32.3%) (unpublished data). Other studies have demonstrated that those terpenes act on cellular integrity, inhibition of the respiration and ion transport processes, and increase membrane permeability in C. albicans (30, 31). Hence, it is possible that the antifungal activity of P. bredemeyeri Jacq oil could be explained by a higher concentration of those monoterpene hydrocarbons. On the other hand, chemical analyses of oil from L. origanoides Kunth (B) have also been carried out in our laboratory. The main components were the oxygenated monoterpenes thymol (43.8%) and carvacrol (17.3%). The antimicrobial activities of those terpenes have also been demonstrated, and their mechanism of action has been associated with membrane permeability (20).
To the best of our knowledge, this is the first time that antifungal activity of M. royoc L extract is described. Morinda genus contains substantial amounts of anthraquinones, especially in the roots. The antifungal activity of those quinones has been demonstrated (32), so it is possible that anti-C. albicans activity of M. royoc L extract may be produced by the presence of such molecules.
In our laboratory, the antifungal activity of L. origanoides Kunth (B) and M. royoc L extract against Aspergillus fumigatus, A. flavus, C. krusei and C. parapsilosis has been demonstrated. Unlike P. bredemeyeri Jacq oil only showed activity against C. krusei (unpublished data).
Potential synergy of essential oils with antibiotics has been previously considered with the aims of increasing the rate of fungal killing, shortening the duration of therapy, avoiding the emergence of drug resistance, expanding the spectrum of activity, and decreasing drug-related toxicity by allowing lower doses of antifungal agents to be administered (33). In this study, a synergistic effect was obtained when itraconazole and the oil from P. bredemeyeri Jacq were combined (FICI range, 0.09-0.13). No interaction was detected with the combination of the oil of P. bredemeyeri Jacq and amphotericin B (FICI=1.06).
The criteria of cytotoxic activity for the crude extracts as established by the American National Cancer Institute (USA), is an IC50 of less than 30 µg/ml (34). According to these criteria, we consider that oils with anti-C. albicans activity and M. royoc L extract were not cytotoxic on Vero cells (IC50≥200 µg/ml) excluding the oil of P. bredemeyeri Jacq (IC50 =15.2 ± 3 µg/ml) (Table 1). González, et al..(35), orally administered an extract of M. royoc in rats and they did not find toxic effects. Both our results of in vitro citotoxicity, as well as the toxicity in vivo demonstrated by González, et al..(35), suggest that M. royoc L extract may be a candidate for developing an antifungal of natural origin against C. albicans. The discovery of a novel natural product for therapeutic use is a slow process. For example, Taxol, an anticancer agent developed from the plant Taxus brevifolia, was discovered after a random screening of 35,000 plant samples that took more than 25 years (36, 37).
In conclusion, the results of this study suggest that Colombian medicinal and aromatic plants represent an untapped source of compounds with anti- C. albicans activity that could be a resource in the development of therapeutically natural products.
Acknowledgements
Grant RC-245-2011 (Fondo Nacional de Financiamiento para la Ciencia, la Tecnología y la Innovación, Francisco José de Caldas).
Correspondencia: Ana Cecilia Mesa-Arango: Carrera 51D N° 62-29, Departamento de Microbiología y Parasitología, Facultad de Medicina, Universidad de Antioquia, Medellín, Colombia. Phone number: (574) 219-6050; Fax: (574) 219-6055. e-mail: amesa@medicina.udea.edu.co.
References
1. Rai M, Mares D. Plant-derived antimycotics: current trends and future prospects. New York: Food Products Press; 2003. [ Links ]
2. Fonnegra R, Jiménez S. Plantas medicinales aprobadas en Colombia. Medellín: Editorial Universidad de Antioquia; 2007. [ Links ]
3. Parahitiyawa NB, Samaranayake YH, Samaranayake LP, Ye J, Tsang PW, Cheung BP, et al. Interspecies variation in Candida biofilm formation studied using the Calgary biofilm device. APMIS. 2006;114:298-306. [ Links ]
4. Cortés JA, Reyes P, Gómez C, Buitrago G, Leal AL. Fungal bloodstream infections in tertiary care hospitals in Colombia. Rev Iberoam Micol. 2011;28:74-8. [ Links ]
5. Zuluaga A, de Bedout C, Restrepo CA, Parra HH, Arteaga MA, Restrepo A, et al. Susceptibility to fluconazole and voriconazole of Candida species isolated from intensive care units patients in Medellin, Colombia (2001-2007). Rev Iberoam Micol. 2010;27:125-9. [ Links ]
6. Torres NA, Álvarez CA, Rondón MA. Evaluación mediante tres técnicas de susceptibilidad a fluconazol en especies de Candida aisladas en pacientes con infecciones invasoras: Bogotá, Colombia. Rev Chil Infectol. 2009; 26:135-43. [ Links ]
7. Lai CC, Tan CK, Huang YT, Shao PL, Hsueh PR. Current challenges in the management of invasive fungal infections. J Infect Chemother. 2008;14:77-85. [ Links ]
8. Cavaleiro C, Pinto E, Gonçalves MJ, Salgueiro L. Antifungal activity of Juniperus essential oils against dermatophyte, Aspergillus and Candida strains. J Appl Microbiol. 2006;100:1333-8. [ Links ]
9. Stashenko EE, Jaramillo BE, Martínez JR. Comparison of different extraction methods for the analysis of volatile secondary metabolites of Lippia alba (Mill.) N.E. Brown, grown in Colombia, and evaluation of its in vitro antioxidant activity. J Chromatogr A. 2004;1025:93-103. [ Links ]
10. Cuenca-Estrella M, Moore CB, Barchiesi F, Bille J, Chryssanthou E, Denning DW, et al. Multicenter evaluation of the reproducibility of the proposed antifungal susceptibility testing method for fermentative yeasts of the Antifungal Susceptibility Testing Subcommittee of the European Committee on Antimicrobial Susceptibility Testing (AFST-EUCAST). Clin Microbiol Infect. 2003;9:467-74. [ Links ]
11. Ishida K, de Mello JC, Cortez DA, Filho BP, Ueda-Nakamura T, Nakamura CV. Influence of tannins from Stryphnodendron adstringens on growth and virulence factors of Candida albicans. J Antimicrob Chemother. 2006;58:942-9. [ Links ]
12. Klepser ME, Ernst EJ, Lewis RE, Ernst ME, Pfaller MA. Influence of test conditions on antifungal time-kill curve results: Proposal for standardized methods. Antimicrob Agents Chemother. 1998;42:1207- 12. [ Links ]
13. Vitale RG, Afeltra J, Dannaoui E. Antifungal combinations. In: Ernst EJ, Rogers PD, editors. Antifungal agents: Methods and protocols. Totowa, N.J.: Humana Press; 2005. [ Links ]
14. Betancur-Galvis LA, Forero JE, Morales GE, Forero JE, Roldán J. Cytotoxic and antiviral activities of Colombian medicinal plant extracts of the Euphorbia genus. Mem Inst Oswaldo Cruz. 2002;97:541-6. [ Links ]
15. Jacob, MR, Walker IA. Natural products and antifungal drug discovery. In: Ernst EJ, Rogers PD, editors. Antifungal agents: Methods and protocols. Totowa, N.J.: Humana Press; 2005. [ Links ]
16. Aligiannis N, Kalpoutzakis E, Mitaku S, Chinou IB. Composition and antimicrobial activity of the essential oils of two Origanum species. J Agric Food Chem. 2001;49: 4168-70. [ Links ]
17. Duarte MC, Figueira GM, Sartoratto A, Rehder VL, Delarmelina C. Anti-Candida activity of Brazilian medicinal plants. J Ethnopharmacol. 2005;97:305-11. [ Links ]
18. Holetz FB, Pessini GL, Sanches NR, Cortez DA, Nakamura CV, Filho BP. Screening of some plants used in the Brazilian folk medicine for the treatment of infectious diseases. Mem Inst Oswaldo Cruz. 2002;97:1027-31. [ Links ]
19. López A, Hudson JB, Towers GH. Antiviral and antimicrobial activities of Colombian medicinal plants. J Ethnopharmacol. 2001;77:189-96. [ Links ]
20. Oliveira DR, Leitao GG, Bizzo HR, Lopes D, Alviano DS, Alviano CS et al. Chemical and antimicrobial analyses of essential oil of Lippia origanoides H.B.K. Food Chemistry. 2007;101:236-40. [ Links ]
21. Calderone RA, Fonzi WA. Virulence factors of Candida albicans. Trends Microbiol. 2001;9:327-35. [ Links ]
22. Toenjes KA, Munsee SM, Ibrahim AS, Jeffrey R, Edwards JE Jr, Johnson DI. Small-molecule inhibitors of the budded-to-hyphal-form transition in the pathogenic yeast Candida albicans. Antimicrob Agents Chemother. 2005;49:963-72. [ Links ]
23. Hawser S, Islam K. Comparisons of the effects of fungicidal and fungistatic antifungal agents on the morphogenetic transformation of Candida albicans. J Antimicrob Chemother. 1999;43:411-3. [ Links ]
24. Carson CF, Hammer KA, Riley TV. Melaleuca alternifolia (tea tree) oil: A review of antimicrobial and other medicinal properties. Clin Microbiol Rev. 2006;19:50-62. [ Links ]
25. Jandourek A, Vaishampayan JK, Vazquez JA. Efficacy of Melaleuca oral solution for the treatment of fluconazole refractory oral candidiasis in AIDS patients. AIDS. 1998;12:1033-7. [ Links ]
26. Hammer KA, Carson CF, Riley TV. In vitro activity of essential oils, in particular Melaleuca alternifolia (tea tree) oil and tea tree oil products, against Candida spp. J Antimicrob Chemother. 1998;42:591-5. [ Links ]
27. Nakamura CV, Ishida K, Faccin LC, Filho BP, Cortez DA, Rozental S, et al. In vitro activity of essential oil from Ocimum gratissimum L. against four Candida species. Res Microbiol. 2004;155:579-86. [ Links ]
28. Pfaller MA, Sheehan DJ, Rex JH. Determination of fungicidal activities against yeasts and molds: Lessons learned from bactericidal testing and the need for standardization. Clin Microbiol Rev. 2004;17:268-80. [ Links ]
29. Monk BC, Goffeau A. Outwitting multidrug resistance to antifungals. Science. 2008;321:367-9. [ Links ]
30. Lima IO, Oliveira RG, Lima EO, de Souza EL, Farias NP, Navarro DF. Inhibitory effect of some phytochemicals in the growth of yeasts potentially causing opportunistic infections. Braz J Pharm Sci. 2005;41:199-203. [ Links ]
31. Cox SD, Mann CM, Markham JL, Bell HC, Gustafson JE, Warmington JR, et al. The mode of antimicrobial action of the essential oil of Melaleuca alternifolia (tea tree oil). J Appl Microbiol. 2000;88:170-5. [ Links ]
32. Xiang W, Song QS, Zhang HJ, Guo SP. Antimicrobial anthraquinones from Morinda angustifolia. Fitoterapia. 2008;79:501-4. [ Links ]
33. Shin S, Lim S. Antifungal effects of herbal essential oils alone and in combination with ketoconazole against Trichophyton spp. J Appl Microbiol. 2004;97:1289-96. [ Links ]
34. Suffness M, Pezzuto JM. Assays related to cancer drug discovery. In: Hostettmann K, editor. Methods in plant biochemistry: Assays for bioactivity. London: Academic Press; 1990. [ Links ]
35. González Y, Scull I, Bada AM, González B, Fuentes D, Arteaga ME, et al. Ensayo de toxicidad a dosis repetidas del extracto acuoso de Morinda royoc L. en ratas Cenp: SPRD. Rev Cubana Plant Med. online. 2003, vol.8, n.2, pp. 0-0. [ Links ]
36. Chivian E. Biodiversity: Its importance to human health. A Project of the Center for Health and the Global Environment. Cambidge: Harvard Medical School;2002. [ Links ]
37. Wall ME, Wani MC. Camptothecin and taxol: From discovery to clinic. J Ethnopharmacol. 1996;51:239-54. [ Links ]














