Introduction
Currently, infective endocarditis (IE) is a rare disease with a yearly incidence estimated in 3-10 per 100,000 people,1 remaining a major cause of morbidity and mortality, apart from improvements both in diagnostic and therapeutic management.2 Since cardiac valves can be destroyed lead-ing to heart failure and vegetations can embolize, mainly to the brain but also to other organs, if left untreated IE can assume 100% mortality3. The epidemiology varies worldwide, and concerning the etiology there have been some changes in the last decades, not only in regard to the microbiological agents, but also to the predisposing factors. While rheumatic heart disease represents the main risk factor and the leading cause of IE in lowincoming countries, in high-incoming ones, virulent staphylococci have over-whelmed penicillin-sensitive oral streptococci, especially in relation to health care infections and invasive procedures1 .The major traditional risk factors are native valve disease, diabetes, immunosuppression and intravenous drug use. Apart from that, IE is increasingly associated with prosthetic valve and intracardiac devices4. Diagnosis relies on clinic aspects, microbiology and echocardiography, either transthoracic or transesophageal echocardiogram, which remains the technique of choice for the diagnosis of IE. Prognosis is determined by patient characteristics, the presence or absence of cardiac and non-cardiac complications, the infecting organism and the echocardiographic findings2.
Clinical case
We present the case of a 50-year-old homeless man, with unknown past medical history, who was admitted at the emergency room after being found lying on the street, complaining of lumbar pain. On examination, he was cachectic with evident poor hygiene. Initially conscious, he became confused, showing signs of shortness of breath (respiratory rate 30 breaths/min), hypotensive (blood pressure of 79/33 mmHg) and tachycardic (pulse rate of 130 bpm). His cardiac sounds were arrhythmic, presenting grade II diastolic murmur and pulmonary examination was significant for crackles on the posterior basal region, bilaterally. He had no visible puncture wounds and the remaining observation was unremarkable. Assessment of arterial gases was compatible with severe metabolic acidemia (pH: 7.096; pCO2: 12.4 mmHg; HCO3: 3.7 mEq/L) with raised lactates (19.1 mg/dL) and laboratory tests revealed leucocytosis (white blood count of 49.2 × 109/L with 96.3% neutrophils), thrombocytopenia (platelets of 14 × 109/L), azotaemia (urea nitrogen: 231 mg/dL and cre-atinine 2 mg/dL), elevated myocardial necrosis markers (myoglobin: 1314.8 ng/mL; creatine kinase: 732 UI/L and troponin T hs: 672 ng/L with reference normal less than 13 ng/L) as well as C-reactive protein (10.2 mg/dL). The electrocardiogram showed atrial fibrillation with rapid ventricular response, ST-segment depression on inferior derivations and Q waves on the precordial ones. According to this, initially, severe sepsis and type 2 acute myocardial infarction were assumed. However, transthoracic echocardiogram showed diffuse left ventricle hypokinesia with depressed ventricular ejection and massive aortic valve vegetation with perivalvular abscess leading to severe aortic regurgitation (Fig. 1). The patient died 12 h after admission with the final diagnosis of native valve infective endocarditis. Meanwhile, the 3 blood cultures collected on admission were incubated in BACTEC(r) (Becton and Dickinson) automated blood culture monitoring system, and since growth index of blood was positive in all, samples were cultured on blood agar plate and incubated at 37 ◦ C in 5% CO2. After 48 h, colonies were finally identified as Aggregatibacter aphrophilus, using matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS) sequencing method.
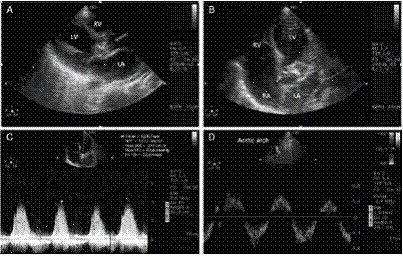
Figure 1 Transthoracic echocardiogram: parasternal long-axis view in panel A showing a voluminous mass attached to aortic valve (red arrow) compatible with a vegetation obstructing the left ventricle outflow tract (LVOT); apical five chambers view in panel B showing an echo lucent area in the aortic root area suggesting the presence of a paravlvular abscess (red arrow); continuous Wave Doppler through the LVOT showing a dense aortic regurgitant flow with a very short pressure-half time (panel C); panel D - Pulsed Wave Doppler on descending part of aortic arch showing holodiastolic flow reversal in favor of severe aortic regurgitation. LA: left atrium; RA: right atrium; LV: left ventricle; RV: right ventricle.
Discussion
Aggregatibacter aphrophilus, which includes both species Haemophilus aphrophilus and H. paraphrophilus, is a Gramnegative bacteria member of the normal flora of the human oral cavity and pharynx 5. It was described, in 1940, as an atypical cause of IE and currently is an agent of the HACEK group (Haemophilus parainfluenzae, Aggregatibac-ter actinomycetemcomitans, Aggregatibacter aphrophilus, Aggregatibacter paraphrophilus, Cardiobacterium spp., Eikenella corrodens and Kingella spp.), responsible for 1.4-3% of cases of IE6. A. aphrophilus may also cause brain abscess and has been isolated in different sites, including peritoneum, pleura, wound and bone5. These organisms are fastidious and antimicrobial susceptibility testing is difficult and impractical in many laboratories, which can be problematic especially in those without automated identification methods. Since ampicillin- resistant strains were recognized, the recommended empirical treatment of IE due to these organisms is therefore a broad-spectrum cephalosporin or a fluoroquinolone7. Although outcomes are generally successful, the IE HACEK infections can be extremely serious, especially if the organism is not early identified8.
This case presents special relevance, not only for its presentation with sepsis and severe aortic regurgitation, but also highlights the serious complications that infections related to fastidious growth bacteria can assume, demanding a prompt diagnosis and treatment. Moreover, since A. aphrophilus is a member of the normal oral human flora, dental programs campaigns and health community surveillance are needed in order to prevent these kinds of infections.
Ethical disclosures
Protection data of people and animals. The authors state that for this investigation have not been performed experiments on humans or animals.
Confidentiality of data. The authors declare that this article does not appear names or data of the patient.
Right to privacy and informed consent. The authors declare that this article does not appear patient data.














