Introduction
The congenital transmission of Chagas disease poses a challenge in the control of this parasitic disease, since it not only does it affect endemic countries in Latin America but also non-endemic countries in other continents where, because of migration, there are women of childbearing age carriers of Trypanosoma cruzi1 infection.
Comprehensive care for this disease includes not only early detection and treatment of the newborn or infant, but also new challenges in care and medical guidance for infected women. In Colombia, there is little academic production on this matter. For this reason, the country is an adequate scenario to outline a review of the strategies that have shown efficiency in countries and similar contexts and to discuss the suitability of each of these strategies in the national context. Therefore, this first consensus seeks to integrate experts on issues related to pediatrics, prenatal care and health care system stakeholder’s, daily decisions makers for this this group of women and children, along with experts in Chagas disease, so as to issue recommendations for the care of these populations in Colombia.
Objective of the consensus
To generate a series of recommendations on diagnosis, follow-up and treatment of congenital Chagas and to guide fertile women with Chagas in Colombia.
Clinical Questions answered in the consensus
- What is the prevalence of T. cruzi infection in pregnant women in Colombia?
- What is the risk of congenital transmission of T.cruzi?
- What are the most appropriate tests and procedures for diagnosis of T. cruzi infection in pregnant women and newborns?
- What are the recommended drugs for the etiological treatment of Chagas’ disease in newborns and infants?
- What are the basic recommendations for the etiological treatment of Chagas disease for fertile women with con- firmed diagnosis?
- What are the complementary public health measures for the management of congenital Chagas?
Patients to whom the consensus is addressed
Pregnant women in Colombia, newborns and women of childbearing age.
Consensus Users
Medical services, health care institutions, public health groups at municipal, departmental and national level in Colombia.
Methodology
Review of the bibliography and evaluation of the evidence
Literature search related to epidemiology, transmission, diagnosis, treatment and public health strategies for the vertical transmission of T. cruzi and congenital Chagas from 1980 to April 2013 was performed. The following Algorithms were designed within the Medline information base: («Chagas Disease/congenital»{Mesh}); («Chagas disease/ congenital»Mesh) AND «prevention and control»(Subheading); Chagas disease/congenital»Mesh) AND «Diagnosis» (Mesh) OR («Chagas disease/congenital»{Mesh} AND «Chagas disease/ diagnosis»Mesh); («Chagas disease/ congenital»Mesh) AND «Therapeutics»MeshOR («Chagas disease/ congenital»{Mesh} AND «Chagas disease/ therapy»Mesh) and («Pregnancy»Mesh) AND «Chagas disease»Mesh NOT («Chagas disease/congenital»{Mesh})
It also included the literature submitted by experts and derived from secondary research from other consensus published on the same subject. The bibliography was selected by summaries, after which the full text of the publication was evaluated. Publications with duplicate information were removed from the record for analysis. When the information of the same article was relevant for more than one topic, it was included in several searches. The data extraction from the articles was done by evidence graphs, verifying the data and the ratios reported and, in some cases, the ratios were calculated from the data provided in the text of the articles. The evaluation of each publication is carried out) through the methodology of reporting of observational studies for the evaluation of the evidence (STrengthening the Reporting of Observational studies in Epidemiology STROBE).
Development of expert consensus based on Delphi methodology
The inclusion of experts was based on the recommendations described by McGinis et al.3 Panels of 3 experts per theme were organized for each of the aspects related to decision making for the identification and management of congenital Chagas disease in the areas of: gynecology, pediatrics, infectology or public health, health promotion entities, as well as representatives of the Ministry of Health and departmental health secretariats. In addition, 2 international experts on the specific subject of congenital Chagas participated in the consensus.
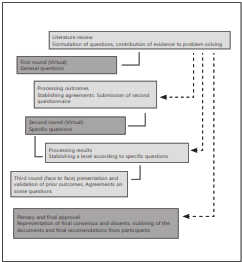
The participation of the consultants was administered in 3 rounds, being the first 2 virtual and the last one, face-to- face. These were carried out from December 2012 to May 2013. The questionnaires were sent by electronic mail with hidden carbon copy and reminders for completion. During all the rounds participants were submitted a text with the best available evidence for each topic, accompanied by questions leading to decision making with the patient. During the first round, she asked about general aspects related to epidemiology, diagnosis and clinical guidance for children, pregnant women and women of childbearing age with Chagas disease through 15 questions questionnaire. During the second round, the results of the first round were presented and evidence was included for questions in which there was no consensus in the first round; In addition, 10 more specific questions were included. During the third round, in person, consolidated results of the previous rounds were presented, disagreements were resolved based on the points of view of the experts from each area and the final version of the document was approved. Throughout the process participants could suggest adding other studies to the searches (Figure 1)
Consensus data analysis
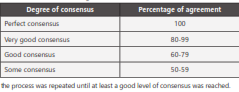
Since the questionnaires were in some cases based on open- ended questions, the responses were analyzed at a qualitative level and similar response groups were organized over the total to establish the percentage agreement. It was considered as a consensus based on a 60% agreement percentage plus the validation of the responses during the third round. In cases where the agreement was below 60%, a new question was drafted related to the same topic in the following round and, if no consensus was reached, it was recorded as a lack agreement (Graph 1).
Classification of evidence and formulation of recommendations
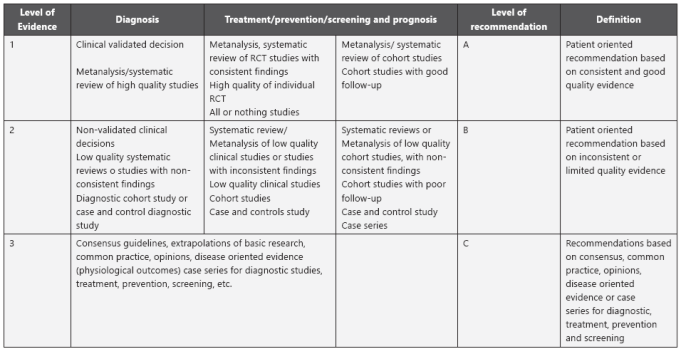
The level of evidence and the degrees of recommendation were assessed and cataloged per the Strength of Recommendation Taxonomy (SORT).
Thus, the quality of evidence was classified into three levels: good, limited, and other. Similarly, the recommendations were classified into 3 categories per the quality of the evidence and the degree of consensus (Graph 2).
Results
In total, 545 titles under the algorithms designed were found, the information provided by the researchers and the review of the gray literature. With the reading of the summaries, 172 texts were chosen for full text reading, of which, finally, 81 texts were included to contribute the evidence presented in the 3 rounds questionnaires. The relevant results of each aspect are shown below:
Section 1. Epidemiology
Prevalence of Chagas in women of childbearing age in Colombia
According to estimates by the Pan American Health Organization, the prevalence of Chagas’ disease in Colombia is 437,960 infected and approximately 107,800 women of fertile age (15 to 44 years) suffer from disease4.There is a lack of precise data on the prevalence of infection in women, both in the general population and in areas where Chagas disease is considered endemic. One of the possible reasons for this lack of knowledge is that screening for diseases such as HIV, toxoplasmosis, syphilis and hepatitis B, but not for Chagas disease, is included during mandatory prenatal care 5,6. It is estimated that in Colombia there were 665,499 births in 20117, and, per estimates of the population at risk of transmission (10.5%), it can be reasoned that about 69,877 of these pregnant women lived in endernic areas.
Since there is no information on the prevalence of Chagas disease among pregnant women in the general population, it was decided to consider the prevalence of seropositive individuals in blood bank donors as a reference data to obtain an estimate of the magnitude. However, it is also known that prevalence is usually lower in blood banks8. This would be explained by the selection of healthy patients and the repetitive stimulation of the donation by suitable donors9. There- fore, considering that estimates of reactivity prevalence for IgG. anti-T Cruzi in blood banks vary amongst departments between 0.3 and 0.8% 4,10, it could be gathered that the prevalence in pregnant women is higher than these figures and could be close to 1%.
Prevalence of Chagas’ disease in pregnant women in en- demic populations
A recent study determined that the overall prevalence of T. cruzi infection in pregnant women from 5 endernic departments was 2.7%, as follows for each department: Casanare: 4.0%; Santander: 3,3%; Boyacá: 3.2%; Arauca: 2.1% and Meta 0.2%, while the range among the 63 participating municipalities oscillated between 0.0 and 20.2%11. In addition, a study carried out in 2 municipalities of Boyacá during 2006 and 2007 indicated that the prevalence for Miraflores 4.0% and for Moniquira 2.8%. In conclusion, considering the few local data, it could be estimated that the prevalence of T. cruzi infection in pregnant women in endemic Colombian departments ranges from 2% to 4%, while in some municipalities it may reach 20%.
Magnitude of Congenital Chagas Disease
There is confirmed evidence of the congenital transmission of T. cruzi in at least 9 cases in Colombia. Six of these were diagnosed by blood culture a study about the endemic area of Boyacá 12,13; Another 2 cases were diagnosed in the department of Santander; and a case of a premature newborn (28 weeks) in the department of Caldas, who was diagnosed by thick blood smear and 8 days after birth and died due to the infection before receiving treatment. On the other hand, in acute Chagas outbreaks, 2 cases of pregnant women have been reported: one occurred before 20 weeks’ pregnancy and led to an abortion and the other in Villanueva Bolivar in a 32 weaker, causing the death of the mother and the child.
In terms of population, the Pan American Health Organization estimates that the annual incidence of congenital Chagas congenital in Colombia is 0.104 and causes an average of 1,000 cases per year4. However, there are only two studies in the literature aimed at estimating the magnitude of the problem. The first of one was developed in an endemic area of the department of Boyacá, with a frequency of transmission of 27.3% (6/22) by blood culture, which has been one of the highest frequencies reported in the literature15. In contrast, the second study, per preliminary results, has followed done a follow up of 114 children of seropositive mothers in endemic areas from birth and every 3 months up to 12 months of age, and so far, has not shown the presence of cases with the use of parasitological or serological methods11. Thus, although in Colombia there is evidence of the frequency of transmission of congenital T. cruzi, it is considered that more studies are required and research in grey literature to establish the magnitude of the problem in the country. For now, in the only 2 studies available in Colombia, the frequency of congenital T. cruzi transmission ranges from 0 to 27.3%. Graph 3 summarizes the estimates of transmission rates of congenital Chagas based on data from Colombia and other countries.
Graph 3

ELISA: Enzyme linked immunosorbent assays; FC: complement fixation test; HAI: Indirect Hemagglutination; HC: blood culture; IgM: immunoglobulin M; IIF (IFI) indirect immunofluorescence; MH: microhematocrit; NA: not applicable; NI: no information; PCR polymerase reaction chain; Stat-Pack immunochromatographic rapid test; XD xeno diagnosis. a: as there were twins. b: historical data from local hospital from 2005-2006
Cost effectiveness of strategies
Some studies of cost-effectiveness of screening in endemic countries such as Chile, 16 and Bolivia17, and in non-endemic countries such as Spain2, indicate a good cost-effectiveness ratio for screening and subsequent follow-up of newborns born to infected mothers and to the women themselves to reduce the burden of disease in both endemic and non-endemic areas compared to not performing the diagnosis. In addition, in other countries, strategies for the active search of cases have been suggested for cases in door to door pregnant women in rural areas difficult to access to improve the coverage of the diagnosis18. Preliminary results from a study in Colombia show that the inclusion of a congenital Chagas’ surveillance program is a cost-effective strategy, being more cost-effective in endemic areas19.
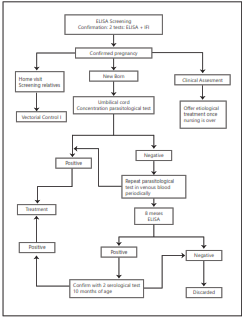
Recommendation 1: Although there is no precise data on the magnitude of Chagas disease in women in pregnant women and newborns in Colombia, estimates suggest that the magnitude is potentially sufficient to consider the inclusion of screening for T. cruzi infection in women during pregnancy as part of the test suite during prenatal control. This strategy would potentially be more cost-effective in areas where the disease is considered endemic. Screening is recommended for pregnant women who have lived in recognized endemic areas or have been in contact with risk factors at any point in life. The most widely recognized risk factors are: knowledge of the “pito” vector, ground floor dwelling, palm roof or bahareque wall located less than 2,000 m above sea level, and having relatives diagnosed Chagas. (perfect Consensus). Level of evidence 3. (Degree of recommendation B).
Section 2. Diagnosis in pregnant women
As for the diagnostic tests for Chagas’ disease in chronic phase, the most appropriate are the serological tests such as the immunoenzymatic test (ELISA) and indirect immunofluorescence (IFI). The reported sensitivity for the ELISA test is 97 to 100%, while its specificity is 96.3 to 100% 20-22. Regarding the IFI test, it is estimated that it has sensitivity from 93.3 to 100% and specificity from 99 to 100% 21,23. These results are like those reported in Colombia from both serum and filter paper samples24. Furthermore, a recent study in Colombia compared the intra-reproducibility of serological tests for identifi- cation of T. cruzi infection in pregnant women (ELISA, IFI and indirect hemagglutination-HAI in serum and ELISA on filter paper). This study concluded that the ELISA reproducibility in serum was 0.98, higher than the reproducibility of the other serological tests and well above the ELISA reproducibility on filter paper, which was 0.5525.
Recommendation 2: ELISA is recommended for serological screening in pregnant women. In a reactive case, confirmation must be made through another conventional serological test with a different principle or with a different antigen, such as IFI or HAI, which has been previously validated in Colombia. (Good consensus). Level of evidence 1. (Level of recom- mendation A).
On the other hand, some researchers have evaluated the usefulness of rapid immunochromatographic tests for chronic Chagas’ screening with good results in both sensitivity and specificity in the general population, with values above 95% 26-30. In Colombia, there is only one evaluation report of the Stat-Pack test in population from 5 to 18 years of age in the department of Casanare, with a sensitivity of 85.4% and specificity of 98% when compared with conventional serological tests and 46.7% when compared to ELISA on filter paper, showing a lower sensitivity in relation to the studies31. A population study of pregnant women from Argentina, Bolivia, Honduras, and Mexico, using umbilical cord blood samples, found that the rapid test on average sensitivity and specificity of 94.6 and 99,0%, respectively, suggesting the use of this test as alternative screening during childbirth32. Finally, some studies show that the sensitivity of rapid tests could be affected using whole blood samples, since sensitivity values increase when serum samples are analyzed33,34. Although there are limitations are identified in terms of reproducibility, in certain regions, rapid tests could be useful in special situations where there is no alternative, by allowing quick decisions.
Recommendation 3. In cases where the pregnant woman is in labor and there is no previous standard serology for T. Cruzi antibodies, an alternative would be the use of rapid test for Chagas on admission to labor. Since the rapid test could have a low sensitivity, in case of a negative result and there are risk factors, the serum test should be administered. A positive test result would lead to the immediate assessment of the newborn, but it will also have to perform confirmatory serological tests on the mother. The use of rapid tests would facilitate the screening strategies; thus, it is necessary to en- courage validation studies in the national and especially rural context. (Good consensus). Level of evidence 2. (Degree of re- commendation B).
Section 3. Diagnosis in the newborn
Microhematocrit in the newborn. This parasitological method of blood concentration from capillaries allows the microscopic identification of T. cruzi in movement, by reading the leukocyte phase in the slide, a procedure that includes the rupture of the capillary. Its main advantage is that it can be performed on the first level settings and requires a low volume of blood, which is ideal for newborns35. In classical studies, it is estimated that the sensitivity of this test varies between 80 and 90%36. Later in Bolivia the so-called modified microhematocrit was introduced, which consists on the direct visualization of the capillary without requiring its rupture, generating better biosafety conditions in the procedure and additionally allowing the estimation of the parasitic load. The quantification of the load has allowed to determine a direct relation of the level of parasitemia, detected by modified microhematocrit, with the severity and lethality of congenital Chagas disease. There is a recent modification to this method called “Eppendorf” micro method, which consists of taking the sample in conical plastic vials (“Eppendorf”) of 1.5 ml, in place of capillaries. This test has shown excellent operational characteristics compared to xenodiagnostics in Argentina. These 3 concentration techniques (microhematocrit, modified microhematocrit modified and “Eppendorf ” micro method) have been widely recommended at international level, as a routine test for the diagnosis of congenital Chagas during the first months of life39. These tests can be performed from cord blood or peripheral blood, having a better performance during the first months of the child’s life and increasing the possibility of detection with the repetition of the test and with an optimal training of the reader40. In Colombia, this method has been used for the diagnosis of acute Chagas in endemic areas and for research of congenital Chagas, in reference centers.
Recommendation 4. It is recommended to carry out concentration parasitological tests (microhematocrit and modi- fied microhematocrit and “Eppendorf ” micro method for the diagnosis of congenital T cruzi infection in the newborn and during the first months of life. (Excellent consensus). Level of evidence 2. (Degree of recommendation B).
Recommendation 5. Given that the performance of the con- centration tests depend to a large extent on the skill of the operator, it is recommended to train health service personnel so they are administered properly, especially in institutions where deliveries take place. (Very good consensus). Level of evidence 2. (Degree of recommendation B).
Serological tests in infants. It is known that serological tests for the detection of anti-T cruzi IgG are useful for the diag- nosis of congenital Chagas only after enough time has elapsed that allows the removal of passive IgG antibodies from the mother, so that the detected antibodies do correspond to the the baby39. According to different investigations, the time for the removal of maternal IgG antibodies varies. While a study in Santa Fe, Argentina, evidenced the elimination of maternal antibodies to at 6 months,41 other investigators have proposed a threshold of 839.,42 or 9 months, 43,44 for the performance of these tests. Finally, there are some proposals to perform the serological tests at month 12 as it coincides with the date of application of vaccines and other public health interventions45. Alternatively, the Shed Acute Phase Antigen (SAPA) antigen, expressed mainly in the acute phase of the disease, has been proposed for the diagnosis in infants as of 3 months of age. This is because anti-SAPA IgG class specific antibodies transferred from the mother to the baby transplacentally are eliminated from the baby between days 30 and 90. Different studies performed using SAPA antigen in an ELISA test showed their potential for the diagnosis of congenital infection. A limitation for the use of this test is the low availability of the antigen.
Recommendation 6. It is recommended that in situations where the initial parasitological tests are negative, the determination of congenital infection by means of conventional T-cruzi anti- IgG serological tests is administered at 8 months of age. Any negative test at that time discards congenital transmission. Any positive test at 8 months should be con- firmed after 10 months by 2 serological tests with different principles. (Very good consensus). Level of evidence 2. (Degree of recommendation B).
PCR: In a Colombian study, the polymerase chain reaction (PCR) displayed a sensitivity of 88.8% and specificity of 92.5% for chronic Chagas’ disease in an adult population50. In addition, other studies in Colombia have shown 80% sensitivity and 100% specificity in adult patients51, it is recommended to take serial samples at different times to increase the likelihood of detecting parasites in the sample. However, there are no local studies that allow to have these estimates about the use of this test for diagnosis of congenital Chagas in Colombia. On the other hand, international publications show that PCR could have a great diagnostic value of the congenital infection and especially the real-time PCR (qPCR) given its: sensitivity higher than that of parasitological tests, early detection of congenital infection, quantitative result (parasite load) and information of epidemiological interest as the determination of lineages of the parasite53-56. However, in the latter years it has been reported that T cruzi DNA can be transferred transplacentally, being able to circulate for a time in the baby without infection and suggesting that the use of the PCR test is should be after 3 months old to avoid false positives57. Despite the potential benefits of the test, it must be considered that it requires a specialized infrastructure and that it is still in the process of standardization and validation in the different laboratories of the endemically countries68,59. Therefore, PCR is not recommended as a routine method for diagnosis33,39.
Recommendation 7. Polymerase chain reaction (PCR) test has shown its potential for the detection of T. cruzi congenital infection. However, this test requires more laboratory infras- tructure than conventional tests and additionally it needs to be validated specifically for the newborn population. On this basis, it is not recommended to incorporate this test into the routine diagnosis until the above criteria are met. (Good consensus). Level of Evidence 2. (Degree of recommendation B).
Section 4. Clinical Manifestations of Congenital Chagas Disease
Between 60 and 90% of cases with congenital Chagas disease may be asymptomatic. Symptoms may appear at an early stage if be it appears during the first 30 days of life, or later, if it appears after this period36. There are no pathognomonic symptoms, but some authors have reported that clinically some of the most severe forms of the disease do not differ greatly from infections of the TORCH group (to- xoplasmosis, rubella, cytomegalovirus, herpes, and others) Such as low birth weight, prematurity, generalized edema, hepatosplenomegaly, respiratory distress and some more severe cases of hydrops fetalis and death 37,60. With regards to the association of prematurity with congenital infection evidence is disparate, as some authors have reported a de- crease in the gestational age of infected infants compared to healthy infants61, there are other investigators who do not find any difference regarding the age at birth between the children of infected and non-infected mothers 48. -hepatomegaly and splenomegaly are among the most commonly reported signs in congenital cases48,60,62 and there are also low frequency symptoms, such as anasarca, petechia, myocardi tis and meningoencephalitis60,62,63. Although the presence of symptoms and signs does not usually reach high percentages, the presence of them should warn the treating physician to the possibility of congenital Chagas57.
Recommendation 8. It is recommended that newborns with clinical signs such as low birth weight, prematurity, respiratory distress, hepatomegaly, splenomegaly, neurological anomalies, cardiomegaly, hydrops fetalis, fetal death and with an epidemiological history associated with T. cruzi infection are discarded the diagnosis of congenital Chagas, using the appropriate technique given the case. (Excellent consensus). Level of evidence 2. (Degree of recommendation A).
Section 5. Etiological treatment of newborns
The effectiveness of the etiological treatment in children with congenital infection during the first year of life is considered to be close to 100%, with disappearance of parasitemia and seronegativization after a few months’ posttreatment. These findings have been consistently published in studies conducted in Paraguay, Argentina53 ’64-66 and Botivia40, 42,45 among others. Thus, the goal of etiological treatment in the baby is clearly the cure, as evidenced by all methods.
Recommendation 9. The efficacy of trypanocides treatment for children with congenital Chagas’ disease is close to 100% before the first year of life and, therefore, etiological treatment at an early stage is recommended for all cases. The criteria for healing is the negative results in parasitological and serological tests. (Very good consensus). Level of evidence
1. (Degree of recognition A).
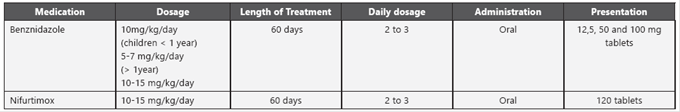
Per international recommendations and although there are no comparative clinical trials between the two available drugs (benznidazole and nifurtimox), both are used to treat congenital cases39. Recommended dosage is specified in Table 4.
Section 6. Etiological treatment of women in child- bearing age
The objectives of etiological treatment are to eliminate infection, reduce parasitic burden and reduce the likelihood of complications and death 67,68. In observational studies there is evidence of the positive effect of etiological treatment on the elimination or reduction of parasitaemia69 and there is some evidence of its effect on the reduction of morbidity and mortality in adult patients treated with benznidazole70-72. To reach the highest levels of evidence, clinical trials such as: Adult Treatment (TRAENA) in Argentina and the Benznidazole Evaluation for Interrupting Trypanosomiasis (BENEFIT) clinical trial are being developed in a multi-centered way in 5 endernic countries74. On its side, the Colombian guideline for the integral treatment of Chagas disease recommends that etiological treatment should be offered to all patients under 18 and, per medical criteria, for adults in the chronic phase6. Therefore, etiological treatment for fertile women is indicated with different levels of evidence and strength of recommendation, being a population that could benefit for it as they are mainly young and asymptomatic.
Moreover, there is increasing evidence of the benefit of etiological treatment in women for prevention of congenital transmission in future pregnancies75. Etiological treatment is clearly contraindicated during pregnancy. During the chronic phase of the disease, it is recommended to postpone the initiation of etiological treatment during nursing, in order not to alter the mother-child bond in the event of adverse effects of the medication on the mother. When prescribed in fertile women, contraception should be provided because trypanocides drugs have teratogenic potential.
Recommendation 10. It is recommended to provide etiological treatment with benznidazole or nifurtimox to fertile women. It is contraindicated during pregnancy and it is recommended to delay the start of treatment until the end of nursing. In all cases, contraception is recommended for fertile women during the administration of etiological treatment for Chagas. (Very good consensus). Level of Evidence 2. (Degree of recommendation B).
Adverse effects of etiological treatment
Etiological treatment with available trypanocides (benznidazole or nifurtimox) may generate some adverse effects; However, these drugs are always better tolerated in children than in adults76. Adverse effects can be well controlled through appropriate symptomatic management and the possibility of rigorous medical follow-up allows the completion of treatment in more than 85% of patients.
Recommendation 11. It is recommended that the patient be informed in advance about the possibility of adverse effects of the etiological treatment of the Chagas disease. It is also recommended that patients receive constant medical follow- up during the treatment period, to promote good adherence to treatment, as well as the end of treatment. (Very good consensus). Level of evidence 2. (Degree of recommendation B).
Section 7. Considerations about activities in the family of the pregnant woman
The recommendation to study the relatives of patients with congenital Chagas ‘disease and pregnant women with Chagas’ disease is widely disseminated39,45,67. Since congenital infection can occur in one or more children of seropositive pregnant women, it is possible that there are children older than the neonate with an undiagnosed congenital infection77. In addition, in a local study in Casanare, Colombia, the prevalence of Chagas disease in relatives of positive pregnant women was 9.8%. Other studies in Argentina have found that being a sibling of a child infected with T. cruzi is a risk factor for acquiring the infection, which indicates that family members should be targeted by screening programs33.
Recommendation 12. Diagnostic activities for the family group of HIV-positive mothers (with emphasis on children) are recommended to confirm other possible cases and en- sure access to treatment and follow-up. (Excellent consensus). (Level of Evidence 1). (Grade of recommendation A).
Recommendation 13. Comprehensive counseling is recommended at the time of diagnosis to both the pregnant woman and her family, with a clear orientation regarding comprehensive treatment and mental health support to minimize the impact of the diagnosis. (Excellent consensus). (Level of Evidence 3). (Degree of recommendation C).
Section 8. Considerations on vector control and other public health measures
Vector control, not only has an impact on the most important transmission route (vectorially), but it is also an indirect control tool for congenital Chagas79 because it reduces the likelihood of new infected mothers or seropositive mothers who have been reinfected, avoiding parasitemias that make mother-child transmission more likely80. Some studies in Bolivia have shown that the children of mothers from areas with higher exposure to reinfection, caused by a higher vector presence, are more likely to have worse outcomes at birth, such as lower birth weight or postpartum complications81. Similar findings in Bolivia confirm that there is less severity of congenital disease in the absence of reinfection of the mother during pregnancy.
Recommendation 14. Home visits for vector control is recommended and sustained surveillance activities in areas where of HIV-positive pregnant women and persons undergoing etiological treatment for Chagas disease are at risk Vector transmission live. (Excellent consensus). Level of evidence 1. (Degree of recommendation A).
Recommendation 15. Given the logistical difficulties of monitoring the mother-child binomial as well as the family back- ground, it is recommended that vector-borne disease control programs integrate efforts and platforms with strategies al- ready in place, such as sexual and reproductive health, indi- genous communities and programs for the care of children. (Excellent consensus). Level of evidence 3. (Degree of recommendation C).
The consensus recommendations are summarized in Figure 2.
As indicators of the implementation of these recommendations, the following are proposed:
% of pregnant women with screening tests for T. cruzi infection
% of pregnant women with confirmatory tests for T. cruzi infection
% of children of HIV-positive mothers with parasitological tests the first month of life
% of children of seropositive mothers with serological tests between 9 and 12 months
% of children with confirmed diagnosis receiving etiological treatment
Potential barriers to implementation of recommendations
There are potential barriers, first, the acceptance of the recommendations by health professionals. Second, the dissemination of recommendations in the national territory. Thirdly, the reforms to the health system that eventually modify the roles of the stakeholders or that limit the opportunity of the processes.
Limitations
In Colombia, there is little information about this topic, since most of the evidence presented comes from studies performed in other Latin American countries. Although the consensus was reached with representatives from all sectors of in health care it did not include patients. In addition, the possible limitations due to health reforms generated or implemented after the consensus was drafted, that modify the itinerary of care and responsibilities of the different stakeholder of the system, are not considered.
Conclusion
The methodology for the elaboration of this consensus is based on the compilation and organization of the abundant international experience, in comparison to domestic experience, to establish common points and potential applications to the Colombian context. The recommendations are based on a collective construction from stakeholders from the scientific, clinical and public health care fields of various levels of care related to decision making in this population group. This consensus constitutes the first effort to generate a tool to base the application of practices in the health system that optimize the opportunity of diagnosis and treatment of people infected with T cruzi, with emphasis on women and children. Additional efforts on the part of the authorities should be carried out to guarantee the dissemination and application of the recommendations in the national territory. Finally, the consensus update should be given once the evidence on new or better procedures is identified.