Introduction
Since the early twentieth century, it was suspected that cell transformation could involve external agents called “filterable agents free of cells”, capable of reproducing neoplastic manifestations in experimental animals1,2. Later on, these “agents” were classified as viruses, giving rise to the concept of oncogenic viruses in 19703.
Viruses are etiological agents of a vast number of human pathologies, from acute self-limiting infections to potentially fatal conditions, among of them cancer which is consider a worldwide public health problem. The rate of incidence of cancer includes 14.1 million new cases and 8.2 million deaths per year; of those, 57% (8 million) of the new cases and 65% (5.3 million) of cancer deaths occur in developing countries3.
The different types of cancer of viral origin are multifactorial and, like non-infectious ones, seem to be a biological anomaly since the tumors do not increase the transmissibility of the virus, nor do they offer any specific advantage. In short, cancer seems to be the final event of the infection. Due to this, most people exposed to these viruses do not develop malignancy, confirming the need for other factors to trigger oncogenesis4.
The cause of these malignancies is based on postulates as old as the one attributed to Galileo Galilei: “An agent causes disease when it is necessary and sufficient for the disease to occur”. This postulate has evolved through scientist such as Henle, Koch and Hill, that established the criteria needed to declare microorganisms as the causal agents of disease. These criteria include the biological characteristics of the microorganism, its effects on the host, the reproducibility of these by different researchers and the epidemiological relationships5. Thanks to these scientific methods, the infectious agents that cause cancer have been divided into two categories: direct carcinogens, which express oncoproteins that contribute to the transformation and indirect carcinogens that through the necessary process of infection and inflammation, eventually cause carcinogenic mutations4.
It is estimated that 15% of cancer cases are related to viral agents, which is why, in this review of literature, readers are offered an update on the subject, which considers the different oncogenic viruses, their pathogenic mechanisms and the interaction with the host cell, which culminates in tumorigenesis.
Materials and methods
Search Strategies
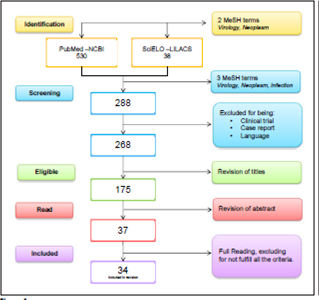
The PRISMA statement (http://www.prisma-statement.org/) was incorporated, a systematic search was made in the electronic databases: NCBI (PuMed), MEDLINE (OvidSP), Scopus (ScienceDirect), combining the terms Search (MesH): “Neoplasm”, “Virology”, “Related” and “Etiopathogenic”, the DOI of the articles was verified at http://www.doi.org/.
Inclusion and exclusion criteria
We included articles that offered a review of the topic in humans in the last 5 years, with the indicated search strategy; The selection of articles is detailed in figure 1.
Types of studies excluded: (I) Clinical trials of drugs and vaccines; (II) Comparison of treatments or diets in patients; case reports; (IV) Syndromes (V) comments and editorials. Data on geographical location, study population, study design and results were extracted, according to the acronym PICO, see table 1.
Table 1 Inclusion criteria according to de acronym PICO *.
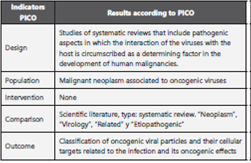
*The PICO process (acronyms defining (P) population, (I) intervention, (C)comparison and (O) outcome).
The full text documents were independently evaluated by two reviewers, the disagreements were resolved by consensus, with the participation of a third party when it was necessary (Figure 1).
Evaluation of quality
The evaluation of the systematic reviews included in the comparative analysis was done through the: "Critical Appraisal Skills Program" (CASP), http://www.casp-uk.net/#!casp-tools-checklists/c18f8 a minimum inclusion score of 6/10 was established, determined by two authors and based on the analysis of the text of the published version.
The methodological quality of the 45.95% of the studies is between 8 and 10 points. The average score is 7.37. No publication showed a methodological quality of less than six.
Results
A total of 34 articles were selected for the comparative analysis, of which 33 (97%), relate the cancer with molecular details of viruses considered oncogenic. The majority of these (29.4%) correspond to journals in the United Kingdom, with an average H index (IHP) of 133, followed by publications from the United States and China. Of the 34 articles, 10 of them present a review about the Epstein Barr virus (29.4%); 8 of Kaposi’s sarcoma virus (23.5%); 6 of human papilloma virus (17.6%); 5 of hepatitis C (14.7%); 4 of hepatitis B (11.7%); 3 of HTLV-1 (8.8%); 2 of Merkel virus (5.8%) and 1 of Cytomegalovirus (2.9%).
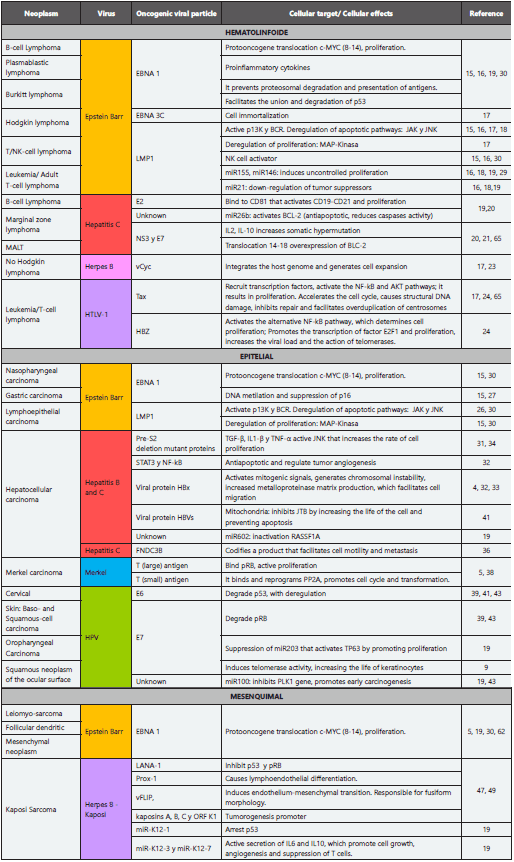
Viruses have been identified as etiological agents of cancer since 1909 when Peyton Rous started his research on the transmissibility of sarcoma in hens6, followed by the description of oncogenic viruses in mammals in the thirties, with accelerated development in tumor virology in the subsequent decades7. The twentieth century culminated with Harld zur Hausen’s declaration of the human papillomavirus as a highoncogenic risk and describing it as a “stage setter” for cervical cancer based in long term researchs2,8; this announcement was recognized with the Nobel Prize in 20089. With the advances in genomics and the description of the pathogenicity of the different viral families, viral molecules closely related to the development of cancer have been identified4. The families of viruses related to cancer in the different publications are: Herpesviridae - double-strand linear DNA, Hepadnaviridae - double-circular DNA chain, Flaviviridae - singlechain RNA, Retroviridae - single-chain RNA, Polymaviridae - double-stranded circular DNA, Papilomaviridae - Double linear DNA4. These families have viruses of different genomic and transmissibility characteristics (sexual, oral fecal, fomites, aerial droplets and even blood products transfusions); cellular tropism (immune system, mucous membranes or parenchymal tissues). In addition, they can have diverse pathogenic mechanisms (Table 2) including: infection, inflammation - TNF, IL6, IL8 -, viral persistence and oncogenic mechanisms (Table 3) such as transformation, key in the multiple steps necessary for viral oncogenesis10,11. Recently, Lin-Tao, et. al., postulated that after the transformation, there is an uncontrolled proliferation and metabolic reprogramming that facilitate the production of energy and molecular synthesis12. In the last four decades, the international agency for cancer research IARC reports to all these families as human carcinogens with different degrees of risk, the highest degree is # 1 in which there is enough evidence linking a certain virus with a malignancy, such as the Epstein Barr virus with lymphomas and epithelial tumors, as detailed in Table 3,14.
Table 2 Viral families, oncogenic viruses, genomics characteristics, transmissibility and pathogenesis.
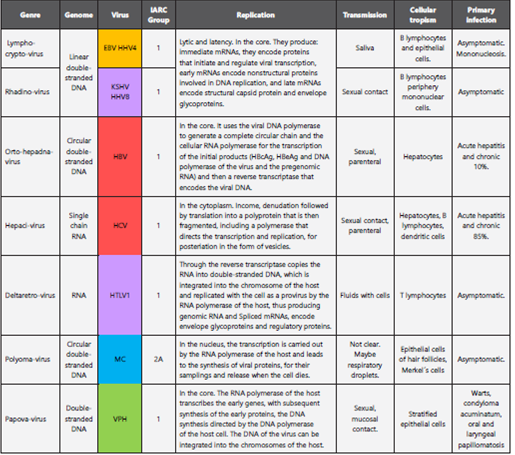
Adapted from Sherris Medical Microbiology, 5th edition, Kenneth Ryan-George Ray, Mc Graw Hill editorial. Medical microbiology: Jawetz, Melnick and Adelberg, 25th edition, Mc Graw Hill publishing house. IARC: International Agency for Research on Cancer.
Virus related to tumors of hemolymphoid origin
Epstein Barr: This virus infects B cells and is expressed in different ways, depending on the oncological pathology it generates: 1. Type I latency 15, characterized by the nuclear antigen EBNA-1. It promotes cell proliferation by activating the c-MYC complex and it prevents the presentation of antigens avoiding the proteosomal degradation characteristic of Burkitt’s lymphoma 16; 2. Type II latency, EBNA in conjunction with membrane proteins LMP-1 and 215; characteristic of Hodgkin’s lymphoma, in which LMP-1 deregulates the apoptotic pathways by activating p13K and the B cell receptor (BCR), which, in turn, promotes genetic damage by translocation and negative regulation of tumor suppressors through miR21; the immortalization of these cells is attributed to EBNA-3C by interaction with cell cycle control points and apoptotic block17,18; 3. Type III latency associated with post-transplant lymphoproliferative disorders is associated with elevation of cellular miR155 that activates BCR, promoting proliferation15,19.
Hepatitis C (HCV): This virus establishes its oncogenic action in marginal zone lymphoma and others with greater malignancy such as diffuse large B-cell lymphoma by means of the structural protein E2, which, by binding to CD81, activates proliferation upon stimulation CD19, CD21 and BCR20; Through NS3 and E7, pro-inflammatory interleukins are activated that increase somatic hypermutation and genetic translocation21, with the consequent overexpression of BLC-2 and the reduction of caspase action, causing apoptotic dysfunction. MirR122 is a miRNA specific to the liver18,22, whose mechanism of action is still uncertain. It was described by Lin and Flemington19 as a positive regulator of virus replication, which causes an increase in the number of copies.
Herpes type 8: It is associated with lymphomas in the serous cavities (PEL). The production of IL-6 induces the production of its human analog, resulting in cell proliferation. In addition, this viral cyclin gene (vCyc), is integrated into the genome of the B lymphocytes, generating the right environment for cell expansion and facilitating transformation23.
Human lymphotrophic type 1 (HTLV1): HTLV1 is the causal agent of adult T cell leukemia. It infects dendritic cells and T lymphocytes, where the viral protein Tax recruits transcription factors, resulting in accelerated mitosis. The multiple integration of multiple viral copies in the host genome promotes the structural damage of DNA24. The HBZ protein activates the alternative pathway NF-kB, promoting proliferation and activation of the transcription of E2F1 that increases the viral load and the action of telomerases25.
Viruses Related to Tumors of Epithelial Origin
Nasopharyngeal and Oropharyngeal carcinoma: it is a distinctive histological subtype of head and neck tumors, with variants such as keratinized and non-keratinizing carcinoma26. This in turn, has a subtype with abundant lymphoid infiltrate, associated with virus infection. EBV infects epithelial cells by transforming beta 1 growth factor, to then activate type II latency and express EBNA1 and LMP1 and LMP2A. This causes dysregulation of proliferation through MAP-kinase, c-MYC and suppression of p13K and p16. EBV expresses BARTs, a family of multi-spliced transcriptional products of the viral genome, with high expression in infected epithelial cells. Its main function seems to be to attack non-coding cellular RNAs responsible for regulating genetic expression. BARTs produce multiple microRNAs that have apoptotic and anti-proliferative mechanisms26.
Gastric carcinoma: It has been associated with bacterial infection by Helicobacter pylori27, as well as with infection by EBV. Positive tumors for this virus occur in two histological types: conventional adenocarcinomas (16%) and gastric carcinomas with lymphoid and epithelial phenotype (89%)26. Tumors positive for EBV are characterized by infecting almost all tumor cells; they are detected by the presence of antigens EBE1 and 2, small non-coding RNAs, with oncogenic properties, such as efficient proliferation of transformed B cells and Activation of insulin-1-like growth factor as an autocrine factor for proliferation27,28. The infection is established in its type I or II latency form15, with expression of EBNA1, BARTs, and LMP2A, which play an essential role in epigenetic abnormalities by promoting the methylation of PTEN (enzymesuppressor tumor).
Lymphoepithelial carcinoma: It is a poorly differentiated carcinoma with dense lymphocytic infiltrate26. It has been described in locations such as stomach, esophagus, tonsils, salivary glands, parotid29, thymus, lungs and intrahepatic biliary epithelium30. The poor differentiation of these epithelial cells and an inflammatory environment are the oncogenic characteristics of the Epstein Barr virus, characterized by type II latency that interrupts multiple cellular processes and signaling pathways mentioned above.
Hepatocellular carcinoma (HCC): This carcinoma has low development associated with fibrosis. Epidemiologically it has been related in 80% of the cases with the chronic infection by the Hepatitis B (HBV) and the Hepatitis C virus (HCV), turning the infection into one of the most important risk factors. In the natural process of viral hepatitis, cirrhosis is not reached until 20 years after infection and oncogenesis takes at least 10 more years31. These viruses are widely distributed throughout the world and have a range of oncogenic mechanisms32 as follows:
The inflammatory microenvironment that promotes the activation of the transforming growth factor beta (TGF-β) stimulates mitogenic factors such as JNK (of the MAPK family), producing rapid cell division with decreased genetic repair. Other endogenous inflammatory products such as IL- 1β, TNF-α and IL-6 (which generate resistance to apoptosis), are capable of activating these pathways. TGF-β maintains a relationship with pSmad3C under normal conditions, but when the latter is persistently phosphorylated, a mitogenic effect occurs and inhibits the apoptosis of transformed cells, a critical step for the progression of malignancy31.
The induction of oxidative damage in lipids, mitogenic proteins such as MAPL, P13K, p53 and β-catenin, and in DNA. These reactive oxygen species induce intracellular calcium signaling, which increases their mitochondrial concentrations by activating STAT3 as antiapoptótico32.
The insertion of viral DNA into the genome of the host in early stages of infection, which in some cases results in major genetic alterations, such as genomic instability, deletions, amplifications and chromosomal translocations. Multiple studies have described common integration sites, generally close to segments that control proliferation, survival, differentiation and immortalization, with advantages for tumorigenesis such as production of mutated HBx and mutated PreS232.
The products of the virus like Hbx and PreS / S. Hbx is a viral protein that activates aforementioned mitogenic factors. It also binds directly to p53 inhibiting apoptosis; it has been identified as a paracrine activator of stellate cell activation, promotes cell migration by increasing intracellular metalloproteinase and angiogenesis when activating HIF1 that activates VEGF and ANG232. PreS / S is a viral antigen retained in the endoplasmic reticulum of the hepatocyte that generates oxidative stress, this retention activates cyclin A and therefore proliferation, as well as the over-duplication of centrosomes. It also activates hTERT that increases the activity of telomerase, generating telomere instability and even polyploidy and inhibits JTB by increasing cell motility, facilitating metastasis32,33. PreS2 also interacts with c-Jun and hyperphosphorylates pRB34.
The aberrant methylation of promoter areas (CpG islands), which inactivate tumor suppressors in early stages of the disease. The enzyme DNMT is in charge of maintaining methylation patterns of the cell, with an up-regulation of this enzyme by Hbx. It has a preference for methylation near tumor suppressors such as RASSF1A, p16, p21, as well as adhesion controllers cellular such as E-cadherin, which, when inhibited, facilitates tumor metastasis32.
The control of the microRNAs: Multiple interaction between the virus and these non-coding particles22 have been established. The following are highlighted: miR602 attacks RASSF1A; miR143 usually considered a tumor suppressor, which contributes to metastasis by over expression, inhibiting the expression of FNDC3b of the fibronectin 3 gene, whose product regulates cell motility19. HBV and HCV share multiple mechanisms of transformation and tumorigenesis, with small differences; one of them is the inhibition of the tumor suppressor of promyelocytic leukemia (PML)35,36. This possesses nuclear domains (PML-NBs), involved in the regulation of apoptosis, cellular senescence and antiviral response. It has been shown that HCV nuclear proteins interfere with multiple regulatory host particles including p53 by attacking their coactivator PML.
Merkel’s carcinoma: This is one of the most aggressive and lethal types of skin cancer in up to 30% of patients. It occurs mostly in immunocompromised patients37. In 2008 its relationship with the virus of the polyomaviridae family was found. The Merkel cell carcinoma virus (MCV) has two oncoproteins; the long T (LT), which binds to pRB stimulating proliferation and the small T protein (Ts) which binds and reprograms the PP2A protein, to induce cell cycle progression and transformation. It also activates cell translocation factors such as 4E-BP1 that favor integration and genetic instability37,38.
Oropharyngeal squamous cell carcinoma: This is a unique clinical entity, characterized by the anatomical location in the junction of soft and hard palate to the hyoid bone, including the base of the tongue It is associated with the human papilloma virus (HPV). It affects a heterogeneous population with determined risk factors and it is related to high-risk serotypes (HPV 16 and 18)39 such as cervical carcinoma40. The virus has a common pathogenic pathway characterized by an advance in malignancy in which each time there is amount of virus present. Dysplastic changes are found in early stages, followed by carcinomas in situ, to culminate in infiltration of squamous cell carcinoma. It is noteworthy that oropharyngeal cancers are reported with viral changes for HPV 6 and 11, usually considered low risk. It is postulated that their behavior is not associated with benignity in this location39.
The HPV has certain viral proteins with different functions during the infection and progression of the disease. E1 is essential for cellular regulation and thus, it supports viral replication. E2 is inactivated to increase the number of viral and epithelial cells with the integrated viral genome. The viral oncoproteins E6 and E7 cause genomic instability by coupling to the host genome and inactivate and/or reduce the half-life of p53 and pRB respectively. This translates into a lack of control during the cell cycle and the activation of cell signaling that causes uncontrolled cellular differentiation and proliferation by increasing the activity of p1641,42. The oncoproteins in turn have effects on the miRNA. E7 suppresses the activity of miR203 in epithelial cells, promoting proliferation and it is believed to facilitate viral replication, forcing the cell into the S phase, where the viral DNA is easily amplified39. E6 inhibits miR219 which overstimulates the LAMB3 gene involved in cell migration and tumorigenesis17. Pedroza et. al, relates other microRNAs related to early carcinogenesis, such as miR100, which promotes the activation of the PLK1 gene that promotes early carcinogenesis, as well as miR10a, miR196a and miR132 that are upregulated and contribute to cell transformation through the HOX and miR886-5p overexpression. This inhibits the process of apoptosis through the BAX gene43.
The HPV has also been associated with squamous neoplasms of the ocular surface (pterygium, papilloma, carcinoma in situ, conjunctival intraepithelial neoplasia), which comprise the same phases of pre-malignancy and malignancy44 and an oncogenic pathway similar to that previously described. However, the clinical evidence reported is scarce and is basically explained by two mechanisms: suboptimal study designs and techniques to identify HPV in biological specimens45.
The role of HPV in keratinocytic carcinomas such as basal cell carcinoma and squamous cell carcinoma of the skin is not fully understood. Multiple clinical, epidemiological and experimental studies suggest a carcinogenic role of type B HPV in the development of these malignancies. This includes the viral characteristics (presence of E6 and E7 that degrade tumor suppressors and activate telomerase) and other factors such as exposure to UV radiation and the response of immunocompromised patients. That is, a patient with a deficient immune system46 has more viral copies that cause damage to the DNA and prevent its repair, blocking in turn apoptotic pathways, these added factors culminate in the development of cancer9.
Tumors of Mesenchymal Origin
There are multiple presentations of this type of malignancy, such as leiomyosarcoma, follicular dendritic cell sarcoma and myopericytoma30 that have been related to the Epstein Barr virus and the nuclear antigen EBNA-1, which mediates oncogenesis, mainly by the translocation of the c-MYC protooncogene and the subsequent proliferation.
Herpes virus 8 or Kaposi’s sarcoma virus induces transformation of mesenchymal cells, through different routes. LANA-1 binds to tumor suppressor p53 and inhibits the ability to induce apoptosis, as well as the pRB controller to modulate the cell cycle transition from G1 to S, where it allows replication and viral latency47,48. Prox-1 and vFLIP induce the lymphoendothelial and endothelial-mesenchymal transition resulting in the fusiform morphology of the cells. Kaposins A, B and C, and the K1 ORF are tumor promoters49. Lin and Flemington, describe the viral microRNA K12-1, which allows the increase of the cell cycle speed when arresting p53, K12-3 and K12-7. This selectively activate the secretion of IL6 and IL10 promoting cell growth, the angiogenesis and the suppression of T cells19.
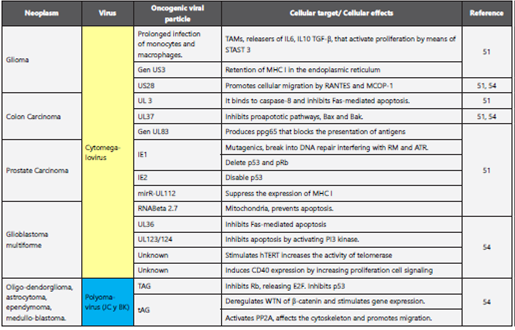
In this review, seven viruses are reported as causal agents of cancer in humans, but the literature proposes some others that have viral characteristics compatible with the oncogenic mechanisms described above, along with epidemiological and experimental associations that sustain them5,50,51. Among them, the Cytomegalovirus of the herpesviridae family are found as potential causal agents of CNS malignancies (gliomas and multiform glioblastoma)52, colon and prostate carcinoma53,54. JC and BK viruses of the polyomaviridae family are causal agents of tumors of the central nervous system1,54 and colorectal carcinoma50. The postulated mechanisms for tumorigenesis are explained in Table 4. There has been descri be another virus linked to the development of breast cancer like bovine leukemia virus (BLV), mouse mammary tumor-like virus (MMTV) both retroviruses55 their oncogenic mechanism still no clear, there is substantial information, but cannot be regarded as conclusive56 more over in recent studies has been demonstrated the interaction of different types of virus, Drop et. al.,57 describes confection between 22-34%, involucre tow or more of the following viruses: BKV, HPV, CMV, HSV and EBV, this combinations can induce transformation or ether exhibit a more rapid developing; so that associated with esophagus, prostate, bladder, breast, lung, colon and even central nervous system58, not even this association are no documented properly.
Discusion
Pathologies of viral origin begin at the time of infection, although this is only one of the steps for the development of malignancy. The process of cellular transformation begins with the deregulation of cell signaling induced by the virus, stimulating oncoproteins that tend to have self-sufficiency in replication and adaptation, insensitivity to inhibitory and apoptotic signals, as well as to angiogenesis, tissue invasion and metastasis59,60. These provide proliferative advantages even under conditions of nutrients and poor oxygen61.
The Epstein Barr virus is the oncogenic prototype, one of the most ubiquitous and successful known viruses. It has developed strategies to infect multiple cell types, evade the immune system, develop viral latency and contribute to the development of malignancies in the three types of tissue mentioned in this review (lymphoid, epithelial and mesenchymal), to the point that 1% of human cancer types are related to these agents7,30,62.
Viral infections are one of the risk factors for the development of cancer. Research and understanding of the pathogenic mechanisms will allow the development of strategies for the prevention of these malignancies. The development of effective vaccination strategies, as reported by Stanley in a recent review about hepatitis B virus infection, indicating that there are approximately 250 million people infected and about 887,000 deaths in 2015. In addition, it appears that up to 50% of children infected at an early age develop cirrhosis or carcinoma hepatocellular. The implementation of the universal vaccination program in 1992, resulted in a decrease in the rate of carriers of the virus. Regarding the human papillomavirus, approximately 290 million women are estimated to be infected. In an optimal scenario, in countries with more than 50% vaccine coverage, the v2VPH v4 HPV vaccine could reduce the incidence of cervical cancer by 70%, and the v9VPH could reduce it by 90%. The infections by HPV16 and 18 were decreased by 68%, with a reduction of cross infection by strains 31, 45 and 3363. In the near future, specific treatments should be implemented, perhaps through vectors of genetic therapies or by the use of oncolytic therapy agents64,65.