Introduction
Tropical protozoan diseases are currently a major public health problem throughout the world. They are infectious diseases caused by unicellular eukaryotic parasites (“protozoans”) of the genera Trypanosome (Chagas disease and sleeping sickness), Leishmania (different forms of Leishmaniasis) and Plasmodium (malaria), Chagas disease, Leishmaniasis, Toxoplasmosis, Amoebiasis, Giardiasis, Cryptosporidiosis and Malaria. The three frst mentioned are classifed as Neglected Tropical Diseases (NTDs) by the World Health Organization, endemic in 149 tropical and subtropical countries, NTDs affect more than 1 billion people annually, including 875 million children in developing economies. Their prevalence is higher in tropical and equatorial countries, where the major number of deaths is due to malaria1. Protozoa are the cause of major number of diseases which spread massive Illness and death, mainly concentrated throughout the tropical world. The lack of commercial markets for potential drugs and the poverty associated with these diseases creates a large burden on the health and economic development of low-income and middle-income countries in Africa, Asia, and the Americas. These diseases are also responsible for over 500,000 deaths per year and are characterized by long-term disability and severe pain. Both the lack Current treatment options are associated with various limitations including widespread drug resistance, severe adverse effects, lengthy treatment duration, unfavorable toxicity profles, and complicated drug administration procedures2-5.
Due to the low of research interest and the high increase of resistance against the existing treatments, as well as increasing ineficiency, toxicity, prolonged treatment schedules and costs, there is an urgent need for cost-effective, safe and easy-to-administer, new lead compounds with novel mechanisms of action4,6-8. Although significant progresses have been made in the past decades, malaria is still considered by the World Health Organization as the frst parasitic disease with 229 000 deaths in 2016 in 91 countries. Chagas disease, African trypanosomiasis and Leishmaniasis are classifed as some of the 17 most important neglected diseases by the WHO. In the absence of effective vaccines, a global antiparasitic strategy relies on a multi-faceted approach based on prevention (i.e. vector control and pharmacological prophylaxis), quick and reliable diagnostic procedures, treatment with effective antiprotozoal drugs and medicinal plants. Natural products remain an interesting source of research for the discovery of new drugs to fght NTDs9. Several studies of crude plant extracts have already identifed potential compounds to treat Chagas’ disease, Leishmaniasis, Toxoplasmosis, Giardiasis, Malaria among other protozoan parasites2-5,8-27. Natural compounds of medicinal plants have shown lower toxicity together with higher specificity, creating an optimistic view of new treatments for diseases, since they contain countless molecules with a great variety of structures and pharmacological activities. Out of 1010 new active substances approved as drugs for medical conditions by regulatory agencies during the past 25 years, 490(48.5%) were from a natural origin.
Natural products derived from plants have played an important role in the control of diseases caused by the protozoal parasites, a classical example is malaria caused by Plasmodium falciparum. Current malaria treatment relies heavily on plantderived products, including the sesquiterpene lactone artemisinin , and the alkaloid quinine24. Malaria and trypanosomiasis, therefore, still constitute a major health problem since the drugs used against these parasites seems to be often unsuccessful and possess high toxicity. Furthermore, another problem with the current antimalarial and antitrypanosomal drugs is occurrence of quick resistance, which increases demand to develop more effective, less toxic, and inexpensive drugs11.
Specific antiprotozoal Phytotherapeutic Products
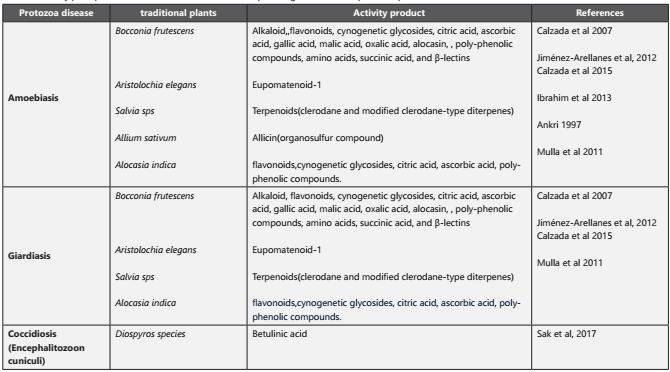
Amoebiasis
Current situation. Amoebiasis is defined as the infection by the protozoan parasite Entamoeba histolytica with or without clinical manifestations. Transmission occurs via the faecal- oral route, either directly by person-to-person contact or indirectly by eating or drinking faecally contaminated food or water. The clinical spectrum ranges from asymptomatic infection, diarrhea and dysentery to fulminant colitis and peritonitis as well as extraintestinal amoebiasis, most commonly the liver where it causes amoebic liver abscess. Although prevalent worldwide, it is more common in areas or countries with poor sanitation, particularly in the tropics28,29.
Anti-amoeba Natural Products. Calzada et al 2007, report that the methanol extract of the aerial parts of Geranium mexicanum exhibited weak activity against Entamoeba histolytica and Giardia intestinalis with IC50 values of 139.9 and 267.7g/ml, respectively. Equally found that methanol extract of the aerial parts of Bocconia frutescens showed moderate activity on Entamoeba histolytica and Giardia intestinalis with IC50 values of 96.4 and 79.3g/ml, respectively. The sanguinarine alkaloid isolated from Bocconia frutescens is known to exhibit toxicity especially for its genotoxicity it could explain the antiprotozoal activity on distinct classes of protozoa such as Trichomonas vaginalis, Entamoeba histolytica and Giardia intestinalis30. Mulla et al, 2011 reported that the aqueous extracts from Alocasia indica Schott (Araceae) exhibited significant in vitro antiprotozoal activity against Entamoeba histolytica and Giardia intestinalis compared to the standard amebicidal with IC50 value 4.78 μg/mL, and 4.12 μg/mL respectively. The ethanol extract exhibited significant in vitro antiprotozoal activity against both protozoa with IC50 value 2.37 μg/mL for Entamoeba histolytica and 4.65 μg/mL in the case of Giardia intestinalis and giardicidal drugs, metronidaze and emetine. This plant contains flavonoids, cynogenetic glycosides, citric acid, ascorbic acid, gallic acid, malic acid, oxalic acid, alocasin, poly-phenolic compounds, amino acids, succinic acid, and β-lectins14. Jiménez Arellanes et al, 2012 looking hexane extract of rhizomes from Aristolochia elegans. The hexane extract has important activity against E. histolytica and G. lamblia, and eupomatenoid-1 is one of the compounds responsible for the antiparasite activity. The antiprotozoal activity of the Hex extract and of pure compounds 1-3was tested against the anaerobic protozoa E. histolytica and G. lamblia . It was observed that the Hex extract was active against these two parasites, exhibiting IC50 = 0.235 and 0.315 μg mL-1, respectively. On the other hand, compound 1 was the most active compound against E. histolytica and G. lamblia, achieving IC50 values of 0.624 and 0.545 μg mL-1, respectively. Compounds 2 and 3 demonstrated moderate antiprotozoal activity with IC50 <275.00 μg mL-1 against both parasites. Because of its important antiprotozoal activity, eupomatenoid-1 was evaluated against T. vaginalis, showing an IC50 = 0.840 μg mL-1.The Hex extract and eupomatenoid-1 were the most active against both E. histolytica and G. lamblia. It should be mentioned that metronidazole was just 1.4 and 4 times more potent than the Hex extract and 2.5 and 10 times more active than eupomatenoid-1, respectively. The antiprotozoal activity of eupomatenoid-1 needs to be supported by a demonstration of its eficacy in animal models as well as by a clear understanding of its action mechanisms. The antiprotozoal activity of neolignans and lignans has scarcely been described in the literature18. Have been identifed Terpenoids from Salvia species to possess biological properties as antiamoebic and antigiardial activities of 14 known clerodane and modifed clerodane-type diterpenes isolated from five Mexican Salvia species against Entamoeba histolytica. Among these, linear lactone was the most active clerodane diterpene against both protozoa with IC50 values of 22.9μM for E. histolytica and of 28.2μM in the case of G. lamblia. These findings give support to the use of Salvia species in the traditional medicine from Mexico for the treatment of diarrhea31. Henciya et al 2017, found that the petroleum-ether extracts of P. juliflora (Fabaceae) leaves were assessed for intestinal, performing antigiardial, and amoebicidal activities and activity against pathogenic protists . In this study was demonstrated high activity against Entamoeba histolytica, with 71.97% mortality after 72-hour exposure to the extract at a concentration of 1000 ppm22. Allicin an organosulfur compound obtained from garlic (Allium sativum) has been investigated the mode of action on E. histolytica, and determined that the preferred target of allicin is mainly the inhibition of cysteine proteinase activity32,33.
Intestinal Coccidiosis
Current situation. Coccidiosis may cause severe intestinal diseases. Coccidiosis is distributed worldwide in poultry and wild birds and has a major economic impact in poultry production due to mortality and morbidity. Epidemiologic studies have shown that the mortality of fowl can range from 5 to 70 %. Economic losses caused by avian coccidiosis, including decreased productivity, usage of coccidiostat, and vaccines, are estimated to amount to $3 billion annually. But the emergence of problems related to drug resistance and drug residues of antibiotics in the chicken meat has stimulated us to seek safer and more eficacious alternative control strategies. Chinese traditional herbal medicines have been utilized for human and animal health for millenniums. Currently, phytotherapies are investigated as alternative methods for controlling coccidian infections34.
Anti-coccidial Natural Products. Zhang et al 2012, showed that D. febrifuga extract was effective against Emeria infection in broiler chickens, a dose of 20 mg extract/kg feed can reduce bloody diarrhea, intestinal lesions, and oocysts excretion and increase body weight gains34. It has been reported that the methanolic extracts from D. sumatrana (Liliidae) and A. fagifolium (Leguminosae - Mimosoideae), was effective for spore elimination to Encephalitozoon cuniculi from immunocompetent BALB/c mice faeces. Notably, the plant extracts reduced spore shedding more effectively than Albendazole; however, the only extract that showed a statistically significant difference was D. sumatrana (p < 0.05). Chemical study of different Diospyros species’ (for example D. glans, D. leucomelas, D. bipindensis) extracts led to the isolation of betulinic acid, its derivatives, as well as other ursane- and lupane-type triterpenoids, having significant antiviral, anti-inflammatory and antimalarian activity35.
Cryptosporidiosis
Current situation. Cryptosporidium is one of the most widespread intestinal parasites and is considered as one of the most common food and waterborne diseases with worldwide spread, acting as a common cause of diarrhea in animals and man. Cryptosporidiosis parvum is a zoonotic protozoan parasite that infects intestinal epithelial cells of man and animals causing major health problem36. In immunocompetent individuals and young children, it is a common cause of severe diarrhea. Cryptosporidiosis is prevalent worldwide, but the prevalence of infection is assumed to be higher in developing countries. Cryptosporidium can be found in surface and groundwater sources susceptible to flooding or faecal contamination and may be present in piped “improved” drinking water systems that use these water sources. Opportunistic infections with Cryptosporidium can cause cryptosporidiosis, a potentially severe and life-threatening illness. Cryptosporidiosis was one of the defining illnesses for acquired immunodeficiency syndrome (AIDS) before the discovery of the human immunodeficiency virus (HIV). Cryptosporidiosis has a higher incidence in developing countries, especially in children, institutionalized patients, malnourished, and immunocompromised individuals (AIDS); In industrialized countries, cryptosporidiosis also occurs in adults due to foodborne or waterborne outbreaks37-40. However, the true global burden of cryptosporidiosis is not known, the number of confrmed cryptosporidiosis cases (N=7 285) reported in EU/EEA countries in 2014 was 5% higher than in 2013. The notification rate in the EU/EEA in 2014 was 2.4 cases per 100 000 population, slightly higher than in 201341,42.
Anti-cryptosporidial Natural Products. The eficacy of three medicinal plants; garlic, ginger and mirazid and a chemical drug metronidazole against Cryptosporidium parvum was evaluated by Abouel-Nour et al 2016, in Male Swiss Albino mice experimentally infected with C. parvum. It was found that garlic successfully eradicated oocysts of infected mice from stool and intestine. Supplementation of ginger to infected mice markedly corrected elevation in the inflammatory risk factors and implied its potential antioxidant, antiinflammatory and immunomodulatory capabilities. Infected mice treated with ginger, mirazid, garlic and metronidazole showed significant symptomatic improvements during treatment. Likewise, Toulahet al, evaluated of garlic plant eficacy in the treatment of cryptosporidiosis in experimentally immumosuppressed rats, found that was significant decrease in the number of excreted oocysts at 5th and 10th day post treatment (P.T.) in treated groups, but no cure rate was detected in treated groups as oocysts still excreted till the end of experiment36,43. Jir et al 2012, determined the protective effect of radix Sophorae flavescentis (RSF) mixture on intestinal mucosa in BALB/c male mice infected with Cryptosporidium parvum. Compared with infection group, the level of oocyst shedding was obviously lower, and the time of the oocyst discharging was significantly shorter in RSF mixture treatment group. The proportion of CD3+, CD4+ T lymphocyte and CD4+/CD8+ T cell ratio in infection group were significantly lower than that of treatment group and control group P < 0.01, and the number of IgA plasmocytes in treatment group was significantly higher than that of control group and infection group P < 0.0144. Another study evaluated the effect of Punica granatum peel on suckling mice infected with experimental C. parvum. At 4 days of age, mice were experimentally-infected by oral administration of 1 × 103 C. parvum oocysts per animal. On day 7 post-inoculation (pi), treated mice received an aqueous suspension of P. granatum peel orally (3 g/kg body weight). these mice did not exhibit any clinical symptoms and no deaths occurred. Oocyst shedding was significantly reduced in the P. granatum-treated mice by day 14 pi (P < 0.05) and was completely eliminated by day 28 pi. The mean weight gain of the P.granatum-treated mice was significantly higher than that of the infected/untreated controls throughout the study (P < 0.01)and improved intestinal histopathology throughout the course of the experiment45.
Giardiasis
Current situation. Giardiasis is one of the most common diseases in many parts of the world including tropical countries. The disease is caused by a protozoan Giardia transmitted via fecal-oral routs in water and food. Swallowing 10 cysts is enough to be contagious with Giardia intestinalis. Annually, 280 million human infections are estimated to occur worldwide, about 200 million people in Asia, Africa, and Latin America have symptomatic infections. Once infected, Giardia causes a generally self-limited clinical illness (i.e., giardiasis) characterized by diarrhea, abdominal cramps, bloating, weight loss, and malabsorption. However, asymptomatic giardiasis occurs frequently, especially in developing countries. Recently, prescribed treatment of giardiasis includes various chemical agents such as metronidazole, tinidazole, ornidazole, mebendazole and albendazole. These drugs have many side effects; while their effectiveness is not always certain, and some have been reported to induce carcinogenic and mutagenic complications in animal models. Also, some of these drugs are contraindicated during pregnancy. Therefore, the use of these medications is controversial because of the reported side effects and drug resistance. To reduce the adverse effects of chemical drugs, researchers are now looking for different ways to find safe alternative remedies including the use of herbs and natural foods as proposed by World Health Organization46,47.
Anti-Giardial Natural Products. Soffar et al 1991, found that of crude Allium sativum extract to be eficient, safe and shortens the duration of treatment in children infected with Hymenolepis nana and G. lambia48. Petroleum-ether extracts of Prosopis juliflora leaves demonstrated high activity against Entamoeba histolytica, with 71.97% mortality after 72-hour exposure to the extract at a concentration of 1000 ppm. Also, showed high antibiotic activity against the protist Giardia intestinalis at 500 ppm6. Likewise, Mulla et al 2011, found that both the aqueous extract and the ethanol extract of Alocasia indica Schott (Araceae) was active against both protozoa Entamoeba histolytica and Giardia intestinalis compared to the standard amebicidal and giardicidal drugs, metronidazole and emetine. The preliminary phytochemical investigation of the extracts of A. indica showed that it contains flavonoids, cynogenetic glycosides, citric acid, ascorbic acid, poly-phenolic compounds14. The activity against E. histolytica and G. lamblia from hexane extract of rhizomes from Aristolochia elegans was determined by Jimenez-Arellanes et al 2012. And they reported that eupomatenoid-1 is one of the compounds responsible for the antiparasite activity, although still the antiprotozoal activity of eupomatenoid-1 needs to be supported by a demonstration of its eficacy in animal models as well as by a clear understanding of its action mechanisms18. Hezarjaribit et al 2015, in a systematic review report the in vitro and in vivo effects of Iranian pharmaceutical plant extracts on Giardia intestinalis, they found in this review that hydroalcoholic extract of asafoetida, Chenopodium botrys, and chloroformic extract of feverfew (Tanacetum parthenium) have the maximum effect (100% eficacy) on in vitro application against Giardia. However, the highest in vivo effect of 100% therapeutic significance was recorded for the extract of Allium sativum at 80 mg/mL concentration. Given the plant species richness of Iran in terms of herbal medicines with fewer side effects, it can be a good alternative to chemical drugs used to treat giardiasis46. Recently, Henciya et al 2017, review the biopharmaceutical potentials of Prosopis spp. and reported that P. juliflora showed high antibiotic activity against the protist Giardia intestinalis at 500 ppm22.
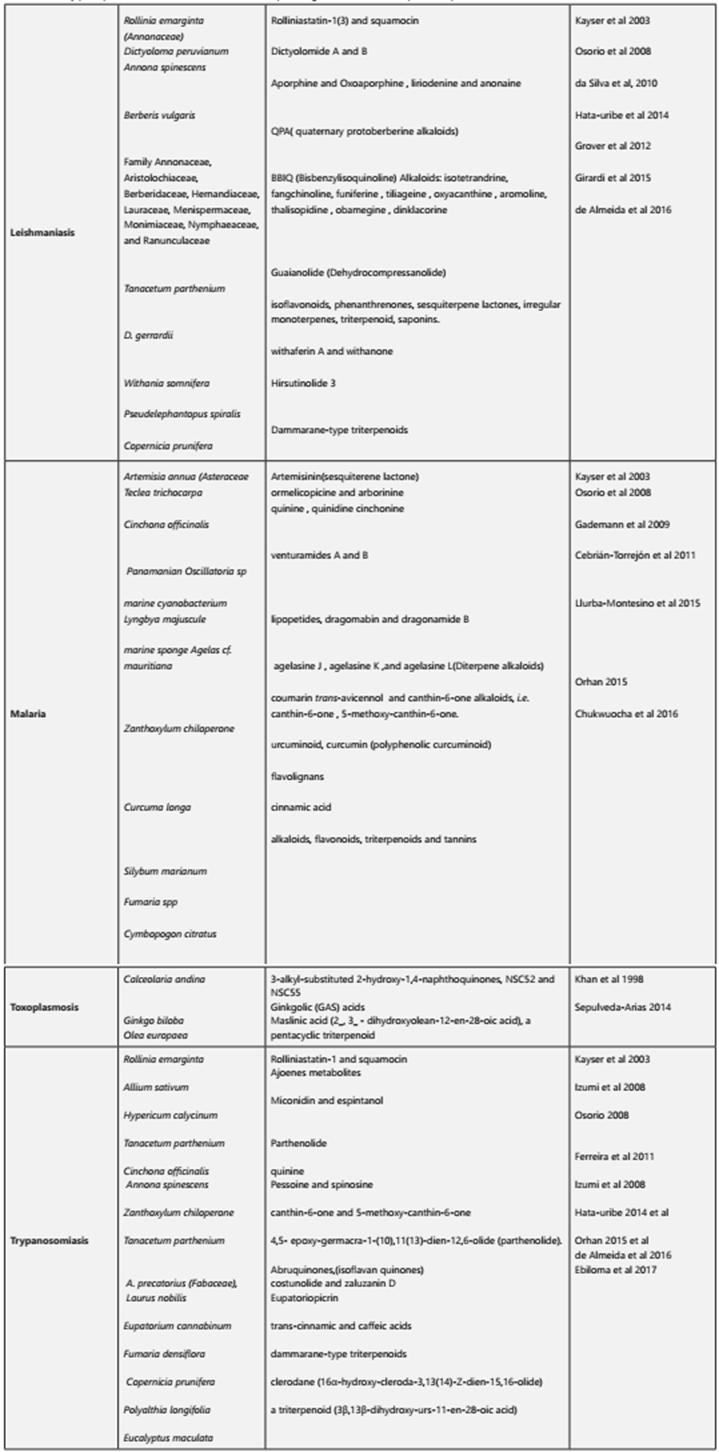
Leishmaniasis
Current situation. Leishmaniasis is one of the most infectious diseases affecting the poorest regions of the world49. Leishmaniasis, caused by several species of flagellated protozoans belonging to the genus Leishmania of the family Trypanosomatidae, manifests in three clinical forms: visceral, cutaneous, and mucocutaneous. Parasites of the genus Leishmania have a dimorphic life cycle, alternating between the motile flagellated promastigote and the intracellular amastigote. Promastigotes are transmitted exclusively to the vertebrate host (including humans and dogs) by the bite of a female phlebotomine sand fly. In its most severe form it is fatal if untreated. Approximately 500,000 new VL cases are appearing every year, with an estimate of 50,000- 70,000 deaths per year worldwide. Other types of the disease, cutaneous Leishmaniasis (CL), mucocutaneous Leishmaniasis (MCL) and post-kalaazar dermal Leishmaniasis (PKDL) are also major problems in many parts of the world. The available drugs for the treatment of Leishmaniasis have been shown to have toxic effects which besides their eficacy reduction urge the development of new therapeutic agents capable of controlling this disease. The basic treatment consists of the administration of pentavalent antimonial sodium stibogluconate (Pentostan® and Glucantime®) and Amphotericin B. All these drugs are limited to some extent by their toxicity and lack of eficacy, the requirement for hospitalization, and cost. Leishmaniasis is becoming the disease of attention and concern because in the last few decades L. donovani has developed drug-resistance and toxicity towards available drugs. While searching for new drugs effective against Leishmaniasis, the disease represents a significant burden especially in the developing countries. Exploration of new less toxic chemotherapeutic agents is being driven by variable effectiveness of the current treatments, their associated side effects and absence of any vaccine50-53.
Anti-leishmania Natural Products. It has been reported that four acetogenins from Rolliniae marginta (Annonaceae) were identifed with antiprotozoal activity by bioassay-guided screening against different Leishmania and T. cruzi strains at 250 μg/mL inhibitory activity up to 89% and 67% was reported for rolliniastatin-1 and squamocin6. Osorio et al 2008, mentioned that bioassay-guided separations led to the isolation and structural elucidation of two 4-quinolinone alkaloids, dictyolomide A and B , from the stem bark of Dictyoloma peruvianum exhibited anti leishmanial activity and this could be attributed to these alkaloids, which induced complete and partial lysis in promastigote forms of L. amazonensis and L. braziliensis, respectively, at 0.35 mM. The alkaloids Aporphine and Oxoaporphine, liriodenine and anonaine , isolated from the roots and trunk bark of Annona spinescens Mart., have shown significant activity against promastigote forms of L. braziliensis, L. amazonensis, and L. donovani. Berberine, a quaternary protoberberine alkaloids (QPA) initially obtained by Buchner and Herberger in 1830 as a yellow extract from Berberis vulgaris L. (Berberidaceae), is probably the most widely distributed alkaloid of all, and is well known for its antiparasitic activity. This metabolite is the main constituent in various folk remedies used to treat cutaneous Leishmaniasis, malaria, and amebiasis, has been used clinically for the treatment of Leishmaniasis for over 50 years, and has been shown to possess significant activity both in vitro and in vivo against several species of Leishmania. Bisbenzylisoquinoline Alkaloids: They are found in the following botanical families: Annonaceae, Aristolochiaceae, Berberidaceae, Hernandiaceae, Lauraceae, Menispermaceae, Monimiaceae, Nymphaeaceae, and Ranunculaceae several biological activities have been reported for BBIQ alkaloids including antiprotozoal activities, in particular against Leishmania sp., Trypanosoma cruzi and Plasmodium sp. The results revealed that 13 BBIQ alkaloids: isotetrandrine, fangchinoline, funiferine, tiliageine, oxyacanthine, aromoline, thalisopidine, obamegine, dinklacorine, was alkaloids with Antiprotozoal Activity, isotrilobine, trilobine, gilletine, and insularine, had IC50 values of less than 5 mM against L.donovani promastigotes, with fangchinoline (IC50 1⁄4 0.39 mM) being as potent as the standard drug pentamidine (IC50 1⁄4 0.4 mM). Of the 15 BBIQ alkaloids tested against L. donovani amastigote forms, phaeanthine showed strong activity (IC50 1⁄4 2.41 mM)17. Equally, a guaianolide purifed from the hydroalcoholic extract of aerial parts of Tanacetum parthenium (L.) Schultz Bip. Dehydrocompressanolide, have shown activity against promastigote forms of L. amazonensis, with 50% inhibition (IC50) of cell growth at a concentration of 2.6 μg/ml. of a guaianolide, and the guaianolide reduced by 10% the survival index of parasites in macrophages when it was used at 20.0 μg/ml49. Dua et al 2011, reported that the chloroform extract of Leucas cephalotes and the petroleum ether extract of Viola canescens exhibited substantial activities against Leishmania donovani with IC50 values of 3.61 g/ml (SI = 8) and 0.40 g/ml (SI = 30), respectively20. Agathosma apiculata extract has been reported active against Leishmania donovani54. Hata-uribe et al 2014, found that two different extracts of D. gerrardii showed antiprotozoal activity, the CH2Cl2/MeOH (1:1) stems extract inhibited L. donovani. A total of 22 secondary metabolites were isolated from fve species and seven new compounds were discovered. These compounds belong to the structural classes of isoflavonoids, phenanthrenones, and terpenes such as, sesquiterpene lactones, irregular monoterpenes and triterpenoid, saponins. Most of them (15 compounds) exhibited in vitro antiprotozoal activity, albeit at differing extent21 By molecular docking the tertiary structure of Leishmanial protein kinase C (LPKC) and two previous experimentally reported natural compounds from Withania somnifera (Solanaceae) using computational methods such as homology modelling and molecular dynamics simulations determined that both withaferin A and with an one were also found to interact with same residues of the active site after the docking process either via H-bonds or hydrophobic interactions, thus validating the accuracy of predicted binding pocket of LPKC and also confrming the inhibitory nature of both natural compounds against the kinase53. Lumpu et al 2013, report that the residual 80% MeOH extract from the leaves, and the total alkaloid extract from stem and root bark were the only subfractions active against Leishmania infantum with IC50 values of 10 mg/ml. None of the samples from the root bark was cytotoxic against MRC-cell lines (CC50464 mg/ml). In general, the aqueous extract (traditional decoction) showed the highest selectivity55. Antileishmanial activity of Azadirachta indica and Emblica oficinalis was evaluated in an animal model with BALB/c mice intracardially infected with promastigotes of L.donovani, observed that the parasite load declined significantly in all infected mice after treatment as compared to the only infected animals. However A. indica in combination with E. oficinalis at the higher dose of 200 mg/kg with each one , significantly reduced the parasite burden in infected animals and maximum reduction was seen in these animals as compared to lower dose of the pure herbs and also when the pure herbs were used alone51. Jain et al 2013, mentioned different papers that evaluated antileishmanial activity of herbal extracts against promastigote and amastigote forms of L. donovani and L. infantum. Finding significant antileishmanial activity in plants species namely Agave americana, Azadirachta indica, Eclipta alba, Piper longum, Nyctanthes arbortristis (Family: Oleaceae), Aesculus hippocastanum, Aloe vera, Coriandrum sativum and Ricinus communis, Agaricus blazei, Plumbago zeylanica, olive tea extract. And this authors concluded that although the use of medicinal plants in treatment of leishmaniasis has been interrupted by unknown mechanism of action, toxicity and low water solubility among others yet scientists are seeking to isolate pure compounds from plant extract as well investigating them in combination with chemotherapeutic agents for antileishmanial activity or immunomodulatory properties or to reduce toxicity 56. Llurba-Montesino et al 2015, tested by a Multiple-TargetScreening (MTS) fifty-eight extracts from 53 herbal medicinal preparations (HMPs) on the German market against parasites of the genera Leishmania, Trypanosoma, and Plasmodium. Sixteen HMPs showed in vitro activity against at least one of the pathogens (IC50 < 10 μg/mL). Six extracts from preparations of Salvia, Valeriana, Hypericum, Silybum, Arnica, and Curcuma exhibited high activity (IC50 < 2.5 μg/mL). Promising results were obtained with Arnica montana and Salvia oficinalis as the most active preparations against the etiologic agent of East African Human Trypanosomiasis (sleeping sickness)4. Hirsutinolide 3, a sesquiterpene lactones, isolated from Pseudelephantopus spiralis (Less.) Cronquist (Asteraceae) displayed strongest activity against the promastigotes and axenic amastigotes of L. infantum with IC50 values of 9.5 and 2.0 mM respectively. Note that the IC50 of 9.5 mM against promastigotes is close to that of the reference miltefosine (8.8 mM)25. De Almeida et al 2016, isolated three dammarane-type triterpenoids from the ‘carnauba’ (Copernicia prunifera) wax, two compounds were effective against the intracellular amastigotes of L. infantum, with IC50 values ranging from 8 to 52μM.16. Ramadani et al 2017, demonstrated that dichloromethane and methanol fractions P. cauliflora (Menispermaceae) radix, have high activity against L. infantum, with IC50 values around 3 μg/ml; their high selectivity index, especially on VERO cells, hypothesises a specific parasiticidal action26. Ahmed et al 2017, evaluated the antileishmanial capability of different extracts of Quercus dilatata Lindl, the evaluation of test extracts was carried out by employing MTT colorimetric assay and found the Ethyl acetate + Acetone and Acetone extracts exhibited remarkable and comparable leishmanicidic potential with IC50 12.91 ± 0.02 μg/ml and 14.40 ± 0.01 respectively and it was found to be absent in the polar extracts. the Chloroform extract showed profound kinase inhibitory activity, cytotoxicity against shrimps and antileishmanial activity52. Li et al 2017, in their review mention that Medda et al found anti-Leishmania activity on Leishmania donovani in the extracts of Swertia chirata and elucidated the inhibitory mechanism. They explained that the activity of DNA topoisomerase I, the enzyme involved in replication, transcription, mitosis and repair of DNA57. Ebiloma et al 2017, isolated compounds active against Leishmania mexicana from Polyalthia longifolia and Eucalyptus maculata23. Vásquez-Ocmín 2018, report the bark extract from Stachytarpheta cayennens is displayed a strong antileishmanial activity on L. donovani intramacrophagic amastigote form (IC50 = 0.95 ± 0.12 μg/mL). And this activity was associated with selective inhibition of the parasite arginase9.
Plasmodium (malaria)
Current situation. Plasmodium infections continue to cause significant morbidity and mortality for residents and travelers to endemic areas. This protozoan parasite is transmitted through the bite of an anopheles mosquito and remains an important public health threat . In 2016, an estimated 216 million cases of malaria occurred worldwide (95% confdence interval [CI]: 196-263 million), compared with 237 million cases in 2010 (95% CI: 218-278 million) and 211 million cases in 2015 (95% CI: 192-257 million). Most malaria cases in 2016 were in the WHO African Region (90%), followed by the WHO South-East Asia Region (7%) and the WHO Eastern Mediterranean Region (2%). The incidence rate of malaria is estimated to have decreased by 18% globally, from 76 to 63 cases per 1000 population at risk, between 2010 and 2016. The WHO South-East Asia Region recorded the largest decline (48%) followed by the WHO Region of the Americas (22%) and the WHO African Region (20%)1.
Anti-plasmodial Natural Products. In one review for plantderived alkaloids, terpenes and phenolics, Kayser et al 2003; mention that Artemisia annua (Asteraceae) is effective against multidrug resistant P. falciparum . Artemisinin (sesquiterene lactone) and its’ derivatives, for example artemether and artesunate, are rapid acting antimalarials6. Osorio et al 2008, mentioned that the family Rutaceae is an important source of antiprotozoal quinoline alkaloids. Seven alkaloids have been isolated from Teclea trichocarpa (Engl.) (a plant used in Kenyan traditional medicine for malaria treatment), including normelicopicine and arborinine, which displayed limited in vitro activities against both chloroquine-sensitive (HB3) and chloroquine-resistant (K1) strains of P. falciparum, with an IC50 of 3.8-14.7 mM. Some DHQ alkaloids obtained from a tunicate species belonging to the genus Didemnum have shown significant and selective antiplasmodial and antitrypanosomal activity. The most pronounced biological activities were found for the alkaloids Lepadin E and F. Lepadin F has an IC50 of 0.47 mM against the P. falciparum. Three alkaloids were identifed by bioassay-guided fractionation, the main alkaloid being crotsparine (1-hydroxy-2-methoxynor- proaporphine), which showed IC50 values of 7.41, 11.29, and 12 mM against the chloroquine-resistant FcB1 and K1, and the chloroquinesensitive Thai strains of P. falciparum, respectively, isoguattouregidine, burasaine, an alkaloid isolated from the roots of several species of the Burasaiagenus, has shown in vitro antiplasmodial activities. The protoberberine group included the alkaloids with the highest antiplasmodial activities. Nitidine (Toddalia asiatica Lam. (Rutaceae), a well-known cytotoxic agent that shows good activity against some chloroquinesensitive and chloroquine-resistant strains of P. falciparum (IC50 around 0.12-0.47 mM). It has been reported that berberine is a potent in vitro inhibitor of both nucleic acid and protein synthesis in P. falciparum. The antiplasmodial and cytotoxic activity of 53 and 24 alkaloids, respectively, isolated from different families. In the frst study, the most active alkaloids against the chloroquine-sensitive clone D-6 of P. falciparum were candicusine and 12-O-methyltricordatine, with IC50 values of 29 and 30 nM, respectively, whereas cepharanthine and cycleapeltine, both with an IC50 of around 67 nM 17. Gademann et al 2009, they mention in their review that many life forms in the marine environment, such as algae, sponges, corals, ascidians, and fungi, have been investigated for their natural product content. The bio activities of compounds against the malaria parasites such as Plasmodium falciparum, including peptides isolated antimalarial venturamides A and B from a Panamanian Oscillatoria sp. Three new diterpene alkaloids, agelasine J, agelasine K, and agelasine L, isolated from the marine sponge Agelas cf. mauritiana from the Solomon Islands. All three compounds displayed moderate activity on P. falciparum. As well as new linear alkynoi clipopetides, dragomabin and dragonamide B, have been isolated from a red Panamanian strain of the marine cyanobacterium Lyngbya majuscula from different sites in the Bocas del Toro. Dragomabin showed good antimalarial activity (IC50 = 6 μM) against chloroquine-resistant Plasmodium falciparum7. Dua et al 2011, reported that extracts of Artemisia roxburghiana, Royleacinerea, Leucas cephalotes, Nepeta hindostana and Viola canescens showed good antiplasmodial activity (IC50 < 5 g/ml). The chloroform extract of Artemisia roxburghiana was the most active (IC50 value of 0.42 g/ml) and the most selective (SI = 78)20. Cebrián-Torrejón et al 2011, determined anti-Plasmodium falciparum resistant and sensitive strains activity the main compounds isolated of Zanthoxylum chiloperone var. angustifolium Engl., Family Rutaceae, such as the coumarin trans-avicennol and canthin-6-one alkaloids, i.e. canthin-6-one, 5-methoxy-canthin-6-one. Showed that Plasmodium falciparum mefloquine resistant Tanzanian strain F32 was the most sensitive strain to avicennol and canthin-6-one 2 (0.5 and 2.0 μg/mL, respectively) considering these data, future improvements in antimalarial chemotherapy based on avicennol and the canthinone alkaloids could generate analogs with increased selectivity and lowered resistance index, and opens new horizons for targeting the increased mortality due to drug-resistant malaria59. Has been reported the isolation and characterization of four new and two known sesquiterpene lactones (STLs) of the germacranolide type from the dichloromethane extract of the aerial parts of Inulamont bretiana DC. (Asteraceae, tribe Inuleae), Interestingly two of news STLs (named: 2a + 2b), showing the lowest activity against the kinetoplastid parasites (Trypanosoma and Leishmania species) and also the lowest cytotoxicity, displayed a somewhat higher activity against P. falciparum with an IC50 of 4 μM and an selectivity indices (SI) of about 3560. Lumpu et al 2013, reported that the aqueous and 80% MeOH extracts and a series of subfractions of each plant part from Alstonia congensis (Apocynaceae) exhibited pronounced antiprotozoal activity against the K1 strain of Plasmodium falciparum with IC50 values ranging from 2 to 5 mg/ml. In general, the aqueous extract (traditional decoction) showed the highest selectivity, especially against Plasmodium falciparum12. DavidOku 2014, found antiplasmodial activity in the methanol leaf extract of Icacina senegalensis(Icacinaceae) a doses of 25.0, 50.0 and 100.0 mg/kg caused 64%, 74% and 80% inhibition of parasitemia respectively. The effect of the extract was significantly (P<0.05) when compared with the control. The antiplasmodial activity of this plant extract might be attributed to the presence of alkaloids, flavonoids and terpenes which have been variously implicated in antiplasmodial activities of many plants61. Memvanga et al 2015, reported that the approximately 120 extracts and fractions obtained from Congolese medicinal plants showed pronounced or good antiplasmodial activity, likewise 37 extract and fractions of plants spices with pronounced antiplasmodial activity. The compost isolated of medicinal plants from the Democratic Republic of Congo with antiplasmodium activity incluyed: AncistrocongolineA-D, Korupensamine A, Ancistrolikokine D,5’-0-Demethylhamatine, Demethyl ancistrobertsonine C,2-Methyl-4-oxo-4 H-1- benzopyrane 5-carboxylic acid ,6-0-demethylancistrobrevine ,A,Yadanziolide A,C, and F, Quindoline, Hydroxycryptolepine, Cryptolepine hydrochloride, Cryptolepine, Neocryptolepine, Biscryptolepine, Cycleanine, Isochondodendrine, Cocsoline, Ursolic acid, Oleanolic acid, Quercitine, Alizarin, Chryzarin, Ellagic acid, Lupeol, Physalin B, 5β,6β-epoxyphysalin B, Strychnogucine A and B, Sungucine, Isosungucine, 18-hydroxyisosungucine, Quinoline and Isoquinoline Alkaloids62. LlurbaMontesino et al 2015, reported that Curcuma longa L. (Zingiberaceae), polyphenol curcuminoid constituents, showed promising antiplasmodial activity with an IC50 value of only 0.59 μg/mL, Specifically, the most abundant urcuminoid, curcumin, exhibited in vitro and in vivo antimalarial activity with IC50 values ranging from 1.84-3.5 μg/mL. The ethanolic extract ID_40 of Hypericum perforatum L. (Hypericaceae) showed high activity againstPfwith an IC50 value of 2.27 μg/mL and with favorable selectivity values. Silybum marianum extract a promising and a useful hit to combat Pf. The isolation of the different flavolignans present in the extract4. The ethanol extracts of Fumaria kralikii (43.45%) and Fumaria rostellata (41.65%) showed moderate activity against Plasmodium falciparum. Among them, Fumaria densiflora extract exerted the highest antiplasmodial (93.80%) and antitrypanasomal effect (55.40%), while the ethanol extracts of Fumaria kralikii (43.45%) and Fumaria rostellata (41.65%) showed moderate activity against Plasmodium falciparum. Therefore, it may be speculated that isoquinoline alkaloids seem to be the most responsible for the antimalarial effect of the Fumaria species. Hand, cinnamic acid derivatives have been stated to possess antiprotozoal activity presumably acting through inhibition of monocarboxylate transport and, therefore, blocking growth of intraerythrocytic Plasmodium falciparum63. Chukwuocha et al 2016, demonstrated the antimalarial activity of the whole Cymbopogon citratus plant, elicited higher anti-malarial activity than the herbal infusion or chloroquine when used as a prophylactic treatment. The antimalarial activity of C. citratus was dose-dependent; a dose of 1600 mg/kg exhibited higher antimalarial activity than that of 3200 mg/kg. This study provides evidence that the whole C. citratus plant has potential for use as an alternative therapy against malaria. This finding may be at least partially explained by the observation that C. citratus produces many compounds that are associated with different pharmacological activities, such as alkaloids, flavonoids, triterpenoids and tannins64. Henciya et al 2017 found high antiplasmodial activity against the malaria-causing protist Plasmodium falciparum the aqueous extract and the methanol extracts of Prosopis juliflora, the isolated sesquiterpene lactones 2 and 3 were the most active compounds with IC50 values of 7.8 mM and 6.9 mM22. Smyrniotopoulos et al 2017, tested for in vitro antiprotozoal activity towards a small panel of parasites (Plasmodium falciparum, Trypanosoma brucei rhodesiense, T. cruzi, and Leishmania donovani) and cytotoxicity against mammalian primary cells the Irish brown alga Bifurcariabifurcata (Sargassaceae family).They observed highest activity was exerted against the malaria parasite P. falciparum (IC50 value 0.65 μg/mL) with low cytotoxicity (IC50 value 56.6 μg/mL)19. Ramadani et al 2017, found A. flava (Menispermaceae) highest antiplasmodial activity (IC50 values less than 3 μg/ml) and the weakest cytotoxicity 26. VasquezOcmin et al 2018, report that the ethanolic extract from Costus curvibracteatus (Costaceae) leaves, Griasneuberthii (Lecythidaceae), Stachytarpheta cayennensis, and Alchornea triplinervia showed strong in vitro activity on P. falciparum (sensitive and resistant strain), suggesting that these activities could be attributed to the main arylpropanoid and iridoid constituents (verbas-coside, isoverbascoside and ipolamiide), of phenolic compounds in the plant leaves9. Llurba-Montesino et al 2018, carried out a review of the critical published information about the antiprotozoal activity of species of the genus Salvia and their chemical constituents Salvia oficinalis L. (Lamiaceae) and found from the 35 species, 45 compounds were isolated and have been tested against the protozoans treated in this review. Diterpenes of the abietane group represent the most active compounds against T. brucei and Plasmodium falciparum, whereas phenolic compounds seem to show higher activity against Leishmania spp. The only flavonoid isolated so far from Salvia species-salvigenin -and tested against protozoa did not show any promising results5.
Toxoplasmosis
Current situation. Toxoplasmosis is an important, widespread, parasitic infection caused by Toxoplasma gondii. Toxoplasmosis is a common parasitic infection and affects onethird of the global population65. Acute infection of toxoplasmosis during pregnancy is detrimental to the developing fetus. In the United States, approximately 1 in 10,000 live births are affected by congenital toxoplasmosis. Although multifactorial in etiology, maternal infection is primarily attributed to the consumption of contaminated meat or water. Infection and transmission to the fetus may result in devastating neurologic impairment66. No effective drug and definitive “gold standard” treatment for T. gondii infection has been available so far, though some medicines have been commonly used in the treatment of T. gondii infection, such as spiramycin, azithromycin, traditional Chinese medicine (TCM), pyrimethamine- sulfadiazine (P-S), trimethoprim-sulfamethoxazole (TMP-SMX), and pyrimethamine-clindamycin (P-C), Most drugs used to treat T. gondii do not significantly affect the tissue cyst stage or the bradyzoites. The tissue cysts produced in the brain are protected by the blood-brain barrier,which makes treatment even more dificult. The eficacy of these same drugs to treat T. gondii infections were examined in this meta-analysis. Generally speaking, spiramycin, azithromycin and TCM were commonly used in the therapy of general populations infected with T. gondii, had a negative conversion rate of 83.4% (95%CI, 72.1%-90.8%), 82.5% (95%CI, 75.9%-87.6%), and 85.5% (95%CI, 71.3%-93.3%),respectively, and no statistical difference was found between these medicines in therapeutic eficacy67.
Anti-toxoplasma Natural Products. The antiprotozoal activity the two 3-alkyl-substituted 2-hydroxy-1,4-naphthoquinones, NSC 113452 (NSC52) and NSC 113455 (NSC55) isolated from the Chilean plant Calceolaria andina L. (Scrophulariaceae), was evaluated against tachyzoites of the RH strain from Toxoplasma gondii in vitro and in murine models of acute toxoplasmosis. In vitro, both NSC52 and NSC55 significantly inhibited intracellular replication of T. gondii. While the results of the in vivo tests showed that oral treatment of mice infected with RH tachyzoites with either NSC52 at 10, 50, or 100 mg/kg/day or NSC55 at 25 or 50 mg/kg/day did not result in significant protection against death. In contrast, when NSC52 and NSC55 were administered i.p., the results revealed significant protection against death68. Kayser et al 2003 reported that quinones have been tested intensively against different parasites including Plasmodium, Leishmania, Toxoplasma gondii6. Ferreira et al 2006, determined the activity against Toxoplasma gondii the compound 2-hydroxy-3-(1’-propen-3-phenyl)-1,4-naphthoquinone (PHNQ6) alone or combined with sulfadiazine and found that treatment with PHNQ6 combined with sulfadiazine protected at least 70 and 90% of mice infected with RH and EGS strains, respectively. The number of brain cysts was lower in mice treated with PHNQ6 alone or combined with sulfadiazine compared to that in control mice. Natural and synthetic naphthoquinones exhibit anti-protozoal activity by generating active oxygen radicals (-OH and -O2- ) that cause lipid peroxidation and alterations in electron transport with inhibition of cell respiration69. Kavitha et al 2012, examined Eurycoma longifolia root extract and fractions as potential sources of new compounds with anti-T. gondii activity of crude extract (TACME) and four fractions (TAF 273, TAF 355, TAF 191 and TAF 401) from E. longifolia, with clindamycin as the positive control and found anti-T. gondii activity in TAF 355 (EC50=0.369 μg/ml) and TAF 401 (EC50=0.882 μg/ml) .this finding signifes that TAF 355 might be potential candidate as an alternative to Clindamycin for the treatment of toxoplasmosis70. Sepulveda et al 2014, performed a systematic literature search, for the 2000-2014 to evaluate the use of natural products as an alternative for the treatment of T. gondii infection, and mentioned that extracts of Lippia multiflora (Verbenaceae), Combretum micranthum (Combretaceae), Vernonia colorata (Asteraceae), Torilis japonica (Apiaceae), Sophora flavescens (Fabaceae), Sinomenium acutum, Pulsatilla koreana, Ulmus macrocarpa, Zingiber oficinale (Zingiberaceae), S. flavescens Antion (Fabaceae), Artemisia annua L (Asteraceae), Azadirachta indica(neem) , Melia azedarach (cinnamon), Eurycoma longifolia Jack (Simaroubaceae), Astragulus membranaceus (Fabaceae) , Scutellaria baicalensis GEORGI (Lamiaceae).The Ginkgolic (GAS) acids from Ginkgo biloba (Ginkgoaceae), extracted the essential oils (EOs) of Thymus broussonetti Boiss (Lamiaceae) and Myristica fragrans Houtt, Myristicaceae, and the maslinic acid (2_, 3_ -dihydroxyolean-12-en-28-oic acid), a pentacyclic triterpenoid obtained from olives (Olea europaea L., Oleaceae) showed variable anti-T. gondii activity71. Yu et al 2015 found that Astragalus membranaceus, Inonotus obliquus , and Licorice sp all have anti-Toxoplasma in vivo and in vitro. However, the polysaccharide of P.poricifolia is weak in vitro10. Wei et al 2015, conducted a meta-analysis to compare the eficacies some medicines have been commonly used in the of treatment of T. gondii infection such as spiramycin, azithromycin, traditional Chinese medicine (TCM), pyrimethamine- sulfadiazine (P-S), trimethoprim-sulfamethoxazole (TMP-SMX), and pyrimethamine-clindamycin (P-C) and reported that the Traditional Chinese medicine (TCM) formulae containing herbs Astragalus membranaceus, Scutellaria baicalensis, Artemisia annua, Ginkgo biloba etc are also used in the treatment of T. gondii infections. however, no statistical difference was found between these medicines in therapeutic eficacy. P-S, TMP-SMX and P-C67. Recently was investigated the potential antitoxoplasma activities of extracts of the endemic plants Centaurea lydia and Phlomis nissolii in a fbroblast cell culture infected with T. gondii trophozoites. Found that C. lydia extract at 55 mug/mL had marked activity against T. gondii trophozoites -the number of trophozoites a 84-fold decrease-, while a 36-fold increase was detected in the P. nissolii extract group, indicating no antitoxoplasma activity72. Ebrahimzadeh et al 2017 reported that methanol extract of F. sellowiana has significant anti-Toxoplasma activity. Further study should be conducted to investigate the potential bioactivity of this extract through bioactivity-guided fractionation27.
Trichomoniasis
Current situation. Trichomoniasis is a sexually transmitted infection (STI) caused by Trichomonas vaginalis, a protozoan which infects the vagina, urethra and paraurethral glands. Is the most common non-viral sexually transmitted disease (STD) in the world. The WHO estimates an incidence of 276 million new cases the Trichomoniasis vaginal (TV) each year and prevalence of 187 million of infected individuals and estimated that over half the new TV infections each year occur in men. By contrast, 89% of prevalent TV cases are found among women. A number of studies have highlighted the fact that at least 80% of TV infections are asymptomatic73-75.
Anti-trichomoniasis natural products. Calzada et al 2007, reported that Carica papaya( Caricaceae) and Cocos nucifera(Arecaceae) showed the best antitrichomonal activity with IC50 values of 5.6 and 5.8g/ml, respectively. And the extracts of Bocconia frutescens, Geranium mexicanum, and Lygodium venustum showed moderate activity with IC50 values ranging from 30.9 to 60.9 g/ml30. Ibrahim et al 2013, carried out a study to evaluate the eficacy of commercially available garlic (Tomex®) Allium sativum (Amaryllidaceae) on T. vaginalis in vitro. The effect of different concentrations of garlic (12.5, 25, 50, and 100 μg/ml) was determined on multiplication and motility of trophozoites at different time points (after 24, 48, 72, and 96 h) in comparison to the same concentrations of metronidazole at the same different time points. The results showed that parasite multiplication inhibition was noticed in proportion of concentration of Tomex® and incubation time. The minimal lethal concentration of Tomex® was 100 μg/ml after 24 h, 50 μg/ml after 48 h, 25 μg/ml after 72 h, and 12.5 μg/ml after 96 h. These results were similar to that of metronidazole. Its antimicrobial activity depends on allicin produced by enzymatic activity of allinase (a cysteine sulfoxide lyase) on allicin after crushing or cutting garlic clove. Allicin and other thiosulfinates are believed to be responsible for the range of therapeutic effects reported for garlic. The main antimicrobial effect of allicin is due to its chemical reaction with thiol groups of various enzymes, e.g., alcohol dehydrogenase, thioredoxin reductase, and RNA polymerase. These can affect the essential metabolism of cysteine proteinase activity involved in virulence of the parasite. Some of the breakdown products of allicin have the ability to cross cell membranes and combine with sulfur-containing molecular groups in amino acids and proteins thus interfering with cell metabolism33.
Trypanosoma (Chagas disease and sleeping sickness)
Current situation: Human African trypanosomiasis (HAT).
Also known as sleeping sickness is a fatal, fly-borne neuroinflammatory disease caused by two agents (protozoa), T. brucei gambiense which is responsible for chronic form of HAT in West and Central Africa, and T. brucei rhodesiense, the etiological factor for the acute form of the disease in East Africa (Fig. 1). The parasites are transmitted by the bite of infected tsetse flies (Glossina spp.). Over 60 million people living in 36 countries are threatened with sleeping sickness and the estimated number of cases is thought to be between 300,000 and 500,000 . In Latin America, infection with T. cruzi is responsible for Chagas’ disease, which is the leading cause of heart disease. Nearly 90 million people in 19 endemic countries are at risk of contracting Chagas’ disease and 16 million people are already infected. Chemotherapy of trypanosomiasis is unsatisfactory for various reasons including unacceptable toxicity, poor eficacy, undesirable route of administration and drug resistance. In this regards, the last few decades have witnessed a surfeit of investigations which have been geared to investigate the effect of common traditionally-used medicinal plants/insect in alleviating the cellular changes produced during trypanosome infection76,77.
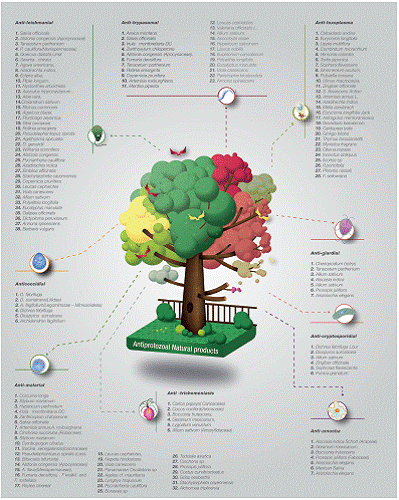
Figure 1 List of plants with proven anti protozoal activity in vitro or in vivo. For each protozoa, the scientific name of the plants with specific antiparasitic activity is showed
Anti-trypanosomal Natural Products. Kayser et al 2003, review, discussed their different features, problems and limitations of the status of validated in vitro and in vivo assays of natural antiprotozoal products. Mentioned that four acetogenins from Rolliniaemarginta (Annonaceae) was identifed with antiprotozoal activity by bioassay-guided screening, against different Leishmania and T. cruzi strains at 250 μg/mL inhibitory activity up to 89% and 67% was reported for rolliniastatin-1 and squamocin respectively. The Ajoenes metabolites, initially isolated from garlic (Allium sativum, Liliaceae), showed significant suppression of Trypanosoma parasitemia in vivo with daily doses of 50 mg/kg over 12 days. This metabolite acting as a covalent inhibitor as well as a substrate of human glutathione reductase (GR) and secondly of the Trypanosoma cruzi trypanothione reductase (TR). Ajoenes also inhibited de novo synthesis of neutral lipids and sterols in T. cruzi epimastigotes, but these effects are not suficient to explain antiproliferative effects of the drug. Other simple phenols include the hydroquinone derivatives miconidin and espintanol, formed from its biosynthesis of a monoterpene to a phenolic, and pholoroglucinol derivatives from Hypericum calycinum (Hypericaceae). Espintanol exhibited an IC90 in the 25-100μg/mL range against twenty different T. cruzi strains.
The mode of action of these semiquinones is unclear. It is attractive to speculate that the sensitivity of Trypanosoma and Plasmodium is due to oxidative stress resulting from the metabolic oxidation of semiquinone radicals or benzoquinones6. Izumi et al 2008, isolated and identifed from the hexane and dichloro-methane fractions of Tanacetum parthenium 4,5-epoxy-germacra-1-(10),11(13)-dien-12,6-olide (parthenolide). The study results indicated that parthenolide is effective against proliferative epimastigote and amastigote forms of T. cruzi Y strain. A progressive increase in the antitrypanosomal effect was observed during the purification process of the compound, the sesquiterpene lactone, parthenolide (lactone of the germacranolide class, has been studied for its ability to inhibit IjB kinase, which is important in cytokine-mediated signaling), was almost four times more effective against epimastigote forms than the reference drug, benznidazole). The pure compound showed IC50/96h and IC90/96h of 0.5 μg/ml and 1.25 μg/ml, respectively. The cytotoxic effect of parthenolide in LLMCK2cells was 3.2 μg/ml (CC50/96h) and the selectivity index was 6.4. The internalization index of T. cruzi in LLMCK2cells was reduced almost 51% at the concentration of 2 μg/ml of parthenolide, and 96.6% at 4 μg/ml15. Osorio et al 2008, mentioned that the family Rutaceae is an important source of antiprotozoal quinoline alkaloids. The treatment of trypanosomiasis, has afforded active compounds identifed as 2-substituted quinoline alkaloids, especially chimanine B and chimanine D with an IC90 of around 0.14 mM, and 2-npropylquinoline, with an IC50 of around 0.29 mM, against the epimastigote forms of T. cruzi. Quinine, quinidine, cinchonine, and cinchonidine have significant trypanocidal activity with IC50 values of 4.9, 0.8, 1.2, and 7.1 mM, respectively, in T.b. brucei. On the other hand, isoguattouregidine, argentinine (an aminoethyl phenanthrene), and 3-hydroxynornuciferine, evaluated at around 0.8 mM, presented significant trypanocidal activity against the bloodstream form (trypomastigote) of T. cruzi with partial lysis of 92, 81, and 68%, respectively.
Among the isoquinoline alkaloids with trypanocidal effect are the aporphine alkaloids predicentrine, glaucine, and boldine, which showed inhibition of the in vitro growth of T. cruzi epimastigotes with IC50 values of 0.08, 0.09, and 0.11 mM, respectively. Thebenzyl isoquinolinic alkaloids are widely distributed in Nature and they have been isolated from different plants commonly used in traditional medicine for the treatment of parasitic diseases. Among the active constituents are the phenolic benzyl isoquinolin escoclaurine and norarmepavine, which have been shown to inhibit the growth of T. cruzi epimastigotes in vitro with an IC50 of around 0.30 mM. Pessoine and spinosine, members of the small group of catechol icisoquinoline alkaloids with a protoberberine skeleton, were isolated from the trunk bark of Annona spinescens (Annonaceae). Pessoine was evaluated for its trypanocidal activity in vitro and induced a partial lysis (55%) in the trypomastigote forms of T. cruzi at 0.79 mM. Sanguinarine and berberine have been reported as trypanocidal agents against T. b. brucei with IC50 values of 1.9 and 0.5 mM, respectively.
Bisbenzyl isoquinoline(BBIQ) Alkaloids: They are found in the following botanical families: Annonaceae, Aristolochiaceae, Berberidaceae, Hernandiaceae, Lauraceae, Menispermaceae, Monimiaceae, Nymphaeaceae, and Ranunculaceae. A number of biological activities have been reported for BBIQ alkaloids including antiprotozoal activities, in particular against Leishmania sp., T. cruzi and Plasmodium sp. Of the 12 BBIQ alkaloids tested for antitrypanosomal activity, eight: isotetrandrine, aromoline, thalisopidine, isotrilobine, trilobine, phaeanthine, daphnoline, and berbamine, had IC50 values of 1-2 mM against T. b. brucei bloodstream trypomastigote forms. Thalisopidine displayed the strongest trypanocidal activity (IC50 1⁄4 1.14 mM). The alkaloids daphnandrine, gyrocarpine , phaeanthine, cocsoline, daphnoline, and isochondodendrine completely lysed the trypomastigote forms of T. cruzi at a concentration of 250 mg/mL (0.40-0.45 mM). Five alkaloids, limacine , isotetrandrine, phaeanthine, curine , and cycleanine, were tested orally for their trypanocidal activity in T. cruzi-infected BALB/c mice in the acute phase; curine and cycleanine showed high eficacy and negative parasitemias 5-7 weeks after inoculation at 10 mg/kg (0.002 mM/kg) daily for 10 days of treatment17. Ferreira et al 2011, evaluate the chemical composition of each organ of Zanthoxylum chiloperone and the eficacy in reducing Trypanosoma cruzi parasitaemia in vivo animal model with Balb/c mice infected acutely with Trypanosoma cruzi (CL or Y strain) for 2 weeks assays of oral or subcutaneous treatments at 10 mg/kg daily for 2 weeks with the ethanolic extract of leaves of Zanthoxylum chiloperone were examined and compared with benznidazole at 50 mg/kg and found that canthin-6-one was the main compound of stem and root bark and 5-methoxy-canthin-6-one in leaves and fruits. In the treated mice, the absence of parasites in blood was attested by control of parasitaemia 33 days after infection, and a significant decreased (P < 0.05) of ELISA serology 30 days post-infection was observed, no significant differences were noted when serology was evaluated at 60 days post infection78. Dua et al 2011, observed that the petroleum ether extract of Viola canescens exhibited activity against Trypanosoma cruzi with an IC50 value of 1.86 g/ml (SI = 7). Also the extracts of Artemisia roxburghiana (leaves), Mentha piperita (leaves) showed activity against Trypanosoma brucei rhodesiense and Leucas cephalotes (whole plant) against Trypanosoma brucei rhodesiense and Leishmania donovani, respectively without any cytotoxicity20. Gökbulut 2012, reported that the crude dichloromethane extract of the flowering aerial parts of Inulamont bretiana DC. (Asteraceae, tribe Inuleae) exhibited activity against T. brucei (IC50 = 3.38 μg/mL)13. Lumpu et al 2013, reported that the aqueous and 80% MeOH extracts and a series of subfractions of each plant part from Alstonia congensis (Apocynaceae) exhibited, good activity against Trypanosoma brucei brucei and Trypanosoma cruzi with IC50 values ranging between 5 and 10 mg/ml55. Lumpu et al 2013, reported that the aqueous and 80% MeOH extracts and a series of subfractions of each plant part from Alstonia congensis (Apocynaceae) exhibited, good activity against Trypanosoma brucei brucei and Trypanosoma cruzi with IC50 values ranging between 5 and 10 mg/ml12. Abimbola et al 2013, established the in vitro and in vivo antitrypanosomal activity of Peristrophe bicalyculata, and its potential as a possible drug candidate in the management of trypanosomiasis. Although the exact mechanism of action is unknown, previous reports have attributed the trypanocidal activity of a number of tropical plants to their flavonoids, alkaloids and other phytochemicals constituents. Also, it has been suggested that many natural products exhibit their trypanocidal effect by interfering with the redox balance of the parasites acting either on the respiratory chain or on the cellular defenses against oxidative stress. Thus, the antitrypanosomal activity of P. bicalyculata may be attributed to its high antioxidant activity and its phenol, flavonoids and alkaloid contents77. Hata-uribe 2014 et al, identifed abruquinones, a series of isoflavan quinones from A. precatorius (Fabaceae), as responsible for strong and selective in vitro inhibition of T. b. rhodesiense on the crude extract. Also isolated several sesquiterpene lactones that showing activity against T. b. rhodesiense from Laurus nobilis L. (Lauraceae), costunolide and zaluzanin D and eupatoriopicrin from Eupatorium cannabinum L. (Asteraceae). Germacrolides, i.e.costunolide and eupatoriopicrin (sesquiterpene lactones), showed a higher inhibition (IC50 of 1.3 ± 0.4 μM and 1.2 ± 0.2 μM, respectively) on the protozoon, than the guaianolide zaluzanin D (IC50 of 10.8 μM). However, none of the sesquiterpene lactones showed suficient activity and selectivity to warrant in vivo testing21. Montesino et al 2015, investigated the potential of legally approved and marketed herbal medicinal products (HMPs) on the German market as antiprotozoal agents. The extract of Arnica montana L. (Asteraceae) (IC50 = 1.12 μg/mL and SI = 10.86) was included in the study as an herbal positive-control, since the anti-trypansosomal activity of its main sesquiterpene lactone constituents. The tincture under study was analyzed by UHPLC/ESI-QqTOF-MSMS and found to contain the main sesquiterpene lactones of the helenalin type known from this plant. With an IC50 value of 1.86 μg/mL against T.brucei, the extract of Salvia oficinalis L. (Lamiaceae) appears to be a promising hit for further evaluation. The antitrypanosomal and antimalarial activity of other species of the genus Salvia have been previously reported, however, many hitherto isolated and active compounds also showed non selective toxicity. The tested extract presented a favorable value of SI = 17.3. The only preparation that showed activity against T. cruzi (IC50 = 5.86 μg/mL and SI =7.9) was the ethanolic extract ID_26 of Valeriana oficinalis L(4). Orhan 2015 et al, found highest antitrypanasomal activity with the ethanol extract of Fumaria densiflora (55.40%), trans-cinnamic (4.32 mg/g) and caffeic (3.71 mg/g) acids were found to be the dominant phenolic acids in Fumaria densiflora63. Bashir et al, in 2015 done a literature review and finding a total of 215 plants species from 82 families. While two herbal formulation and three insect, were reviewed for in vivo anti-trypanosomal activity and additionally, some of the plants were investigated for possible ameliorative effects on the trypanosome-induced pathological changes. Phytochemistry studies of the anti-trypanosomal plants led to the isolation of 96 specific bioactive anti-trypanosomal compounds from different parts of the plants76. De Almeida et al 2016, isolated three dammarane-type triterpenoids from the ‘carnauba’ (Copernicia prunifera) wax, two compounds displayed activity against trypomastigote forms of T. cruzi with IC50 values of 15 and 35μM, respectively. It was possible to conclude that the detected antiprotozoal bioactivity of ‘carnauba’ (C. prunifera) wax extracts could be related to the presence of the natural dammarane triterpenoid derivatives16. Ebiloma et al 2017, isolated active compounds from Polyalthialongifolia (Annonaceae) and Eucalyptus maculata (Myrtaceae), in particular, display promising activity against drug-sensitive and multi-drug resistant Trypanosoma bruceias well as against T. congolense. The EC50 value of a clerodane (16α-hydroxy-cleroda-3,13(14)-Z-dien-15,16-olide) isolated from Polyalthia longifolia was as low as 0.38 μg/mL, while a triterpenoid (3β,13β-dihydroxy-urs-11-en-28-oic acid) isolated from Eucalyptus maculata displayed an EC50 of 1.58 μg/mL. None of the isolated compounds displayed toxicity towards Human Embryonic Kidney cells at concentrations up to 400 μg/mL23.
Future perspectives and upcoming directions
As can be analyzed, the perspectives in the research of antiparasitic products from natural products are vast, given the great biodiversity present in countries where tropical diseases produced by intestinal or blood parasites are prevalent, that is why the different groups of research we must continue deepening the nature of the chemical compounds present in the natural products of our countries, in order to identify new candidate molecules to be used as medicines for the treatment of these diseases. In addition, as can be seen, there are multiple secondary metabolites identifed as active compounds in plants of the tropical and subtropical regions of the world, so it is urgent to carry out more bioinformatic analysis of enzymatic targets of the metabolic and / or replicative pathways of these parasites that can be inhibited by the native compounds already identifed, as well as studies in animal models, as well as scaling up to clinical trials in order to offer alternatives therapeutics for these parasites diseases ( many of which are neglected tropical diseases), to the inhabitants of the developing countries. Finally the reader can see in Figure 1 (Phototherapy tree against protozoa) and table 1 and 2 (Activity principles from some antiprotozoal traditional medicine plants) a summary of the information about phytoterapy products reports against protozoa parasites.
Table 1 Activity principle found in some traditional medicine plants against intestinal protozoa parasites.