Introduction
The development of new therapies for oncological patients has generated an increase in life expectancy, but in the same way, this has led to a higher risk of infection. The high rates of infections are a result of several conditions like cancer chemotherapy, immunosuppressants, neutropenia, surgeries, malnutrition, chronic inflammatory diseases, organ transplantation and a greater exposure to invasive techniques such as catheters, dialysis and respiratory support, due to the increase in the length of their hospital stays1,2.
Antibiotic resistance is one of the most important public health problems in the world. Its origin is multifactorial. The main associated aspects described are an inappropriate use, the lack of effective surveillance systems, the absence of legislation to regulate the market, and its widespread use in animals3-5. Nowadays, antimicrobial resistance tests are crucial to determine empirical and non-empirical therapy schemes that should be followed by health professionals. Worst case scenario, a pathogen develops resistance against three or more classes of antibiotics, making it multi-resistant, an increasing daunting situation, without many pharmacological alternatives available3,4,6-10.
The pattern of infectious disease in cancer patients has changed over time. Back in the early 1.980s, Gram-negative microorganisms were responsible for nearly two-thirds of infections; however, at the end of that decade, the pattern changed, being overtaken by Gram-positive ones. In this case, there is a reappearance of multi-resistant Gram-negative bacteria5,11. Otherwise, Gram-positive infections mortality ranges from 5% to 20%12-14, compared with 18 to 40% for Gram-negative ones3,15.
In Colombia, 80.000 new cases of cancer were diagnosed in 2.015; a trend expected to increase by 2.035 in 155.000 new cases per year16. Furthermore, it is also expected an increase in infections prevalence, antibiotics use, antibiotic therapy resistance and associated mortality. As a disadvantage, the country has a small number of reference centers that publish epidemiological, clinical and microbiological behaviors about this population, and when compared, there are discrepancies with international reports17-20.
In our region, cancer has been establishing as a serious and growing public health problem, occupying the first causes of morbidity and mortality in the Colombian population. Oncologos de Occidente, an institution that covers between 85% and 95% of patients with cancer of the Colombian coffee region area, facilitates the study of their cancer clinical records, in order to design screening interventions and reduce the rates of associated morbidity and mortality21.
For this reason, a cross-sectional study was carried out analyzing the microbiological characteristics of infections in patients from an oncological reference center in the Colombian coffee region area during 2014-2016.
Patients and methods
A cross-sectional, descriptive study on microbiological isolates from cancer patients during a 3-year period in Colombia was realized. Our study group was established by patients over 18 years old with histopathologically cancer diagnosis (include hematological malignancies); treated in “Oncólogos del Occidente S.A.” located in Armenia, Manizales, and Pereira (Colombia) from January 2014 to December 2016; who had suffered an infectious process defined as patients with fever, neutrophil count below 1.500/mm31 or any other cause of clinical suspicion of infection during their hospital stays; and that, were systematically cultivated with antibiogram to confirm an infectious focus.
Laboratory procedures
Samples were processed at the Oncólogos del Occidente S.A. Microbiology tests were performed using automatic BACTEC systems. If there was a high suspicion of colonization or bacterial contamination, the process was restarted from the sowing in the respective agars to the identification of the agent, with the subsequent clinical and paraclinical correlation that allowed verifying the infection. All the processes performed by the laboratory were standardized.
Source of information
The “Sistema de Administración de Historias Clínicas Oncológicas” (SAHICO) by its initials in Spanish, is the registration system used by Oncólogos del Occidente S.A. to store their medical records. The laboratory, based on the Performance Standards for Antimicrobial Susceptibility Testing (CLSI 2015 guideline) (22, reported through the software WHONET 5,6: date of the sample collection, source of the positive sample, etiological agent, minimum inhibitory concentration, antimicrobial in-vitro resistance development, and bacterial resistance.
Statistical análisis
With the identification of patients, the databases of SAHICO and WHONET were mixed. Medians and interquartile range (IQR) of continuous and discrete variables were examined. Nominal variables were analyzed using absolute and relative frequencies. All analyzes were performed using STATA 14,2 official version.
Ethics approval
Approved by the Bioethics Committee of the Universidad Tecnológica de Pereira23 and the Infections Committee of Oncólogos del Occidente S.A. The authors declare that there is no conflict of interest.
Results
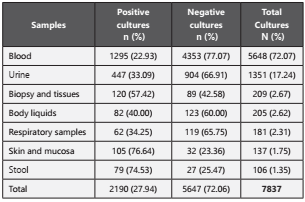
From January 1, 2014, to December 31, 2016, we collected data on over 1.216 patients with cancer diagnosis from a reference oncological institution in three cities from Colombia. The 51,97% (n=632) were females compared to 48,03% (n=584) males. The mean age was 58,79 ± 17,83 years (range: 18-95 years) with the following distribution: < 20 years (n=24, 1,97%); 20-44 years (n=228, 18,75%); 45-65 years (n=463, 38,08%); and ≥65 years (n=501, 41,20%). A total of 7.837 cultures made during the study period. The service with the largest number of cultures requested was hospitalization (66,88%; n=5.241), followed by the Intensive Care Unit (30,48%; n=2.389), operating rooms (2,07; n=162) and external consultation (0,57%; n=45). The culture positive rate was 27,94% (n=2.190) and 72,06% (n=5.647) were negative; of those, 6.938 samples (88,53%) were obtained before starting antibiotic therapy. The table 1 shows the culture positivity according to sample type.
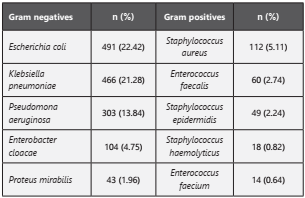
Of all infections, 79,54% (n=1.742) were a Gram-negative bacterium, 15,71% (n=344) were a Gram-positive bacterium and 4,74% (n=104) had a yeast like fungi. Main one’s isolated bacteria were described in Table 2.
According to sample type, we found that for E. coli: 41,14% (n=202) corresponded to a blood culture; 31,57% (n=155) to urine culture and 10,39% (n=51) to skin and mucous membranes samples. For K. pneumoniae we reported: 63,73% (n=297) corresponded to blood culture, 17,17% (n=80) to urine culture and 6,65% (n=31) to respiratory tract. For P. aeruginosa: 49,50% (n=150) to blood cultures, 23,43% (n=71) to urine culture and 9,90% (n=30) to respiratory tract nálisi. For S. aureus: 81,25% (n=91) to blood culture, while 11,60% (n=13) corresponded to biopsy and tissue samples.
We performed a blood culture nálisis to identify the most prevalent pathogens, recognizing Klebsiella pneumoniae (22,93%, n=297), Escherichia coli (15,60%; n=202), Pseudomonas aeruginosa (11,58%; n=150) and Staphylococcus aureus (7,03%; n=91).
The Table 3 shows the most prevalent profiles of antibiotic resistance founded in Gram-negative bacteria. The main mechanisms of antibiotic resistance reported for Gram-negative are extended-spectrum β-lactamases (ESBLs) (45,45%; n=235) and AmpC-type 7β-lactamases (AmpC) (37,71%; n=195); Otherwise, for Gram-positive the 47,20% (n=116) were methicillin-resistant Staphylococcus aureus (MRSA).
Table 3 Resistance profile of most frequently isolated Gram-negative bacteria (Escherichia coli, Klebsiella pneumoniae and Pseudomona aeruginosa). TPM/SMX: Trimethoprim Sulfamethoxazole
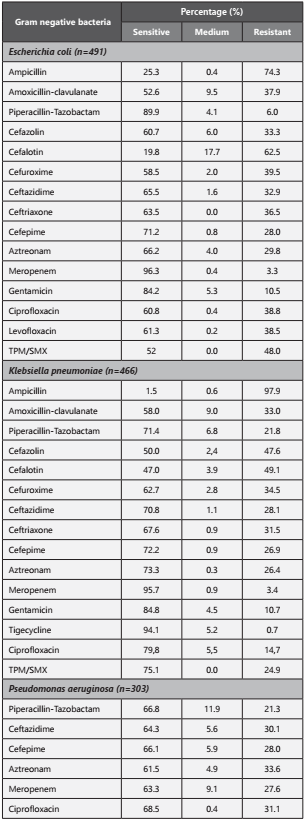
According to the evidence the high negative blood culture in our study can be explained by patient´s self-medication, prophylactic therapy or antibiotic therapy on admission, reflected with a culture positivity of 27,94%, which is similar to reported studies28,29; with an average of 1.8 positive cultures per patient, greater than 0,04 -1,64 documented6,11,27-28. Nevertheless our findings on bacterial isolation (Gram-negative 79,54%, Gram-positive in 15,71% and fungus in 4,74%), contrasted with other studies with distributions as Gram-positive (26,70% - 76,10%), Gram-negative (23,90% - 60,23%) and fungi (11,90% - 14%)8,11,28-30. These data supports the shift from gram-positive to gram-negative agents in our region, which may suggests an apparently adequate management of central venous accesses, a failure in biosafety and sanitation protocols, a lack of antibiotic prophylaxis with fluoroquinolones in neutropenic patients or even worse, a significantly increase in resistance for them, as seen in our findings with a ciprofloxacin resistance of 38,8% for E. coli and 14,7% for K. pneumoniae. This resistance pattern is commensurable with the reported in an investigation made in Taiwan (2010), where the Gram-negative prevalence was 60%29.
We could not correlate the clinical information and the microbiological characteristics due to the absence of the former in our database, main limitation of our retrospective study, this is a possibility that remains to be execute in cohorts that allow rigorous long term follow up of patients to assess variables that we missed: type of cancer, venous access (central or peripheral line), hospital stay, pharmacotherapies, related costs and survival rates, between others.
The present study defined the pathogens’ characteristics of infections in a population with oncological diagnosis which allow us to do the next conclusions. According to our findings, we recommend prior taking of cultures, the beginning of empirical treatment with coverage for Gram negative such as Escherichia coli, Klebsiella pneumoniae and Pseudomona aeruginosa; establishing antibiotic dispensing policies (plus therapeutic de-escalation) thus achieving a microbiological risk control and improve the epidemiological surveillance. Empirical use of beta-lactams with extended spectrum or cephalosporins of 1 to 3 generation is not recommended due to the high resistance found. Alternatively, if resistance to an antibiotic group already exists, we would suggest to start a cyclic replacement, which is performed with antibiotics of different mode of action until the resistance to the former relapses5,7,10,26,30,31.