Introduction
The bacteria belonging to the genus Ralstonia spp. are gramnegative, non-fermenting, environmental bacilli and are found mainly in humid environments1. They are resistant to most disinfectants and have the capacity to form biofilms2. The main species within the genus are R. pickettii, R. solanacearum, R. insidiosa and R. mannitolilytica3. They are not usually pathogenic bacteria but have occasionally been found to cause infections associated with health care as an opportunistic pathogen. Most of these infections were associated with the use of contaminated solutions or products and all types of infections were reported4. They can produce outbreaks in selected populations, although usually with few affected cases because of the low virulence of the bacteria. However, large outbreaks may occur because it is not easy to find the source of the infection and control it adequately. Patients with chronic kidney disease undergoing dialysis are immunosuppressed due this condition, which make them more susceptible and vulnerable to be infected by opportunistic pathogen like Ralstonia spp5,6. The aim of the present study is to describe an experience of identification and control of an outbreak of Ralstonia spp. in a renal unit in the city of Cali, Colombia.
Material and method
On December 20, 2017, the Departmental Health Secretariat received notification of an outbreak in a dialysis unit. During the previous 2 weeks, 11 patients had presented mild symptoms of fever and/or chills during or after the dialysis session. Accordingly, blood cultures were taken and antibiotic therapy was begun. The cultures of all the patients tested positive for Ralstonia pickettii/mannitolilytica. All patients belonged to a renal unit and underwent hemodialysis for acute or chronic kidney disease.
The first visit was made on December 22, 2017, and, on reviewing the clinical histories and laboratory results of the reported patients, the outbreak was confirmed. It was defined like an outbreak because the number of positive blood culture exceeded what it was expected for this renal unit and identification tests show a unusual bacteria7.
The following confirmed case definition were made: Patient in renal unit who had a temperature of ≥38°C and/or chills, during or after dialysis therapy and had a positive blood culture for Ralstonia spp.
A review was made of the medical records of all the patients who had presented some type of reaction during or after the dialysis session. Active daily surveillance was maintained until February 12, 2018.
Traceability of drugs and devices
As part of the fieldwork, a review was made of the traceability of the batches of drugs and devices in use for each patient infected in the renal unit from 15 days before the onset of symptoms until the diagnosis of bacteremia.
Environmental and drug cultures
Environmental cultures were carried out on surfaces, walls, air, equipment, air conditioning systems, tap water and wáter from the treatment plant: rings, osmosis and permeated wáter from the dialysis machines. Repeated cultures were also performed on supplies, disinfectants, devices and medications.
Drug Cultures were performed following the method of the current United States Pharmacopeia (USP) which concurs with the International Pharmacopoeia of the World Health Organization8 and is accepted in Colombia9. Additionally, prefilled heparin was tested using a method recommended by international expert and previously documented by the Spanish Society of Infectious Diseases and Clinical Microbiology10.
Surveillance of the event was maintained as well as enforcing and monitoring of control and prevention measures including standard precautions, hand hygiene, cleaning and disinfection of equipment and environments, and contact precautions.
Results
Description of the cases
769 patients were treated for hemodialysis, 295 (38.4%) had a temporary central catheter and 474 (61.6%) had an arteriovenous fistula/graft. 362 were treated in the southern unit and 347 in the northern unit. Additionally, 60 patients were treated extramurally during the period of the outbreak.
124 patients were identified with bacteremia due to Ralstonia spp., 122 (98.4%) with catheter access and 2 (1.6%) with fistula access. There were 67 (54.0%) affected patients in the southern unit, 54 (43.6%) in the northern unit and 3 (2.4%) extramural patients.
72 patients (58.1%) were men, and the average age was 63 years. The most frequent comorbidity was hypertension (51.6%), diabetes (50.8%) and obesity/overweight (10.5%).
Among the factors that could be related to the infection, it was found that, in the two weeks prior to the start of the outbreak, 19 (15.3%) patients had been hospitalized, 5 (4.0%) had been referred from another institution to start their dialysis therapy, 4 (3.2%) had received a transfusion of some blood product, 2 (1.6%) had received previous antibiotic therapy and 1 (0.8%) had had surgery.
78 (62%) patients were hospitalized, 6 patients with blood culture positive died but only one of these deaths was attributable to Ralstonia spp. Therefore, the case-fatality rate was 0.8%.
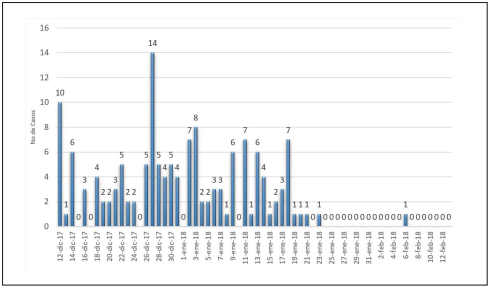
Epidemic curve and Attack rate
For this outbreak, a common origin curve with intermittent exposure was found (Figure 1). An outbreak of Ralstonia spp. was established on December 20, 2017, with the first case having occurred on December 12 of the same year. Attack rates are presented in Table 1. During the outbreak, patients who were on dialysis therapy were kept together and no new patients were admitted.
Lab results
Isolates was analyzed for 97 of the 124 patients. This bacterium showed high percentages of resistance to beta-lactams including carbapenems, however, it displayed very good sensitivity to fluoroquinolones (Figure 2).
A clonality test was performed to 21 isolates of R. mannitolilytica sent by both, south and north units, and 2 non-clinical isolates sent by INVIMA, which belonging to the suspected batch of heparin. The analysis of the patterns obtained con firms that all the isolates were related with a 97.68% of genetic similarity.
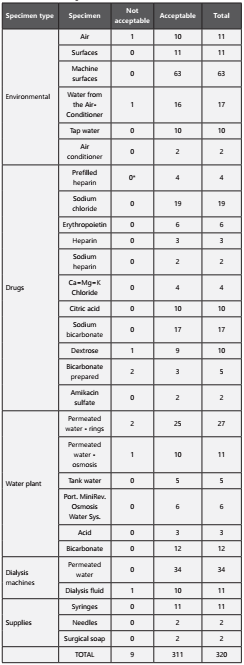
The results of the drugs and environmental cultures are shown in Table 2.
Traceability of drugs, devices
Six drugs were found to be from a batch shared between the 2 sites at the time of the start of the outbreak: amikacin sulfate 100 mg, amikacin sulfate 500 mg, iron (III)-hydroxide in sucrose 100 mg, erythropoietin 4000 UI, prefilled heparin 1000 units/mL and heparin sodium 5000 IU/mL. 4 medical supplies with shared batches were identified: 1mL disposable syringes, 20 mL disposable syringes, No. 16 disposable needles, and macrodrip infusion sets.
Confirmation of hypothesis
On January 11, 2018, the first extramural case was confirmed. This strengthened the hypothesis that the source was a medicine, as the extramural patients had no association with the others, the dialysis water used was independent, the care personnel and protocols were different, and, since they were hospitalized patients, the only medication administered during the procedure was prefilled heparin. The batch of this drug had been changed at the start of the outbreak as part of the control measures, however, on requesting additional information from the supplier, it was found that the preparation batch had been changed, but the batch of raw material was the same.
The prefilled heparin had already been cultured several times with negative results, so recommendations were requested from international experts in microbial isolation techniques. The recommended protocol was the following:
Combine several units of prefilled heparin to a total volume of 100 mL.
Pass the 100 mL of the medication through a 2-micron filter.
Grow a culture of the resulting concentrate on tryptone glucose extract agar (TGEA) and Reasoner Number 2 (R2A) between 17-23°C for 7 days.
If these culture media are not available, trypticase soy agar (TSA) can be used, although the results may not be as good.
Using this method, the microorganism was isolated and, consequently, the source of the outbreak was identified.
Discussion
In the literature, the few reports of Ralstonia spp. Outbreaks have a maximum of 34 affected patients, making this outbreak the largest reported in the literature, and none of them occurred in Latin America3.
Most of the them are due to the use of contaminated solutions, blood products, chlorhexidine, saline, sterile water and colonization of medical devices. In addition, there are cases of outbreaks due to contamination of blisters such as heparins, fluconazole or another commonly used injectable drug contaminated with the bacteria1-3,11. In this outbreak, prefilled heparin was found to be the source of infection. As this was one of the first hypotheses that emerged during field research, it was cultured several times following the method of the current United States Pharmacopeia (USP) which concurs with the International Pharmacopoeia of the World Health Organization8 and is accepted in Colombia9. However, the microbiological cultures and sterility tests were always negative for the growth of microorganisms, so the opinion of international experts was requested. They suggested another method previously recommended by the Spanish Society of Infectious Diseases and Clinical Microbiology10.
This confirms the importance of the microbiology laboratory in the investigation of an outbreak associated with health care, since in addition to identifying the microorganism involved, it provides its resistance profile and helps with patient follow-up, as well as assisting in the identification of potential sources of infection and transmission mechanisms when hypotheses have been derived from the analysis of the epidemiological investigation12,13.
The literature has clearly described the ability of Ralstonia spp. to penetrate the 0.2 and 0.22 μm filters used in sterility tests, in conditions of poor nutrient availability such as drug solutions, where they can survive by decreasing in size. For this reason, several authors suggest using filters of 0.1 μm,14,15however, this recommendation has not been included in the current pharmacopeias.
Despite the low virulence of Ralstonia spp. the literature has described cases resulting in death. In the present outbreak investigation, the fatality rate was 0.8%. Other investigations have shown low fatality rates consistent with those reported in our case3,4.
Regarding treatment, there are no standardized recommendations for the treatment of Ralstonia spp. Differences in sensitivity to antibiotics are found, particularly carbapenems and aminoglycosides16. However, most reported infections have been treated with piperacillin-tazobactam, meropenem, ciprofloxacin, amikacin, and a combination of cephalosporins and aminoglycosides with good results3,17. In this outbreak, most patients received treatment with ciprofloxacin with adequate results.
Emphasis was placed on changing the catheters of all patients in whom the infection was confirmed, given that Ralstonia spp. is able to produce biofilms and cause persistence of infection and rise values suggested internationally18,19.
We suggest for outbreak study to know in detail the medicines received, the coincidences between them and we carry out deeper methods for the study of each one, without forgetting other possible causes of infection such as water, devices or frequently used elements.