Introduction
Monitoring HIV results is a critical step for countries to achie ve the Sustainable Development Goals to 2030 and to end inequalities as stated in the 2021 Political Declaration on HIV and AIDS agreed at the High-Level Meeting on AIDS of the United Nations General Assembly1.
Epidemiological surveillance systems and disease-specific re gistries provide tools that allow this monitoring, however, the data and its results will always rely on the quality of the in formation reported by the healthcare providers or institutions performing the care and reporting of the data2. Awareness of the importance of HIV registries as platforms for research is growing and contributes to knowledge generation. Many reports are the result of government policies, public organi zations such as academia or medical research associations3. Health-information systems offer the potential to improve quality of care and population health by informing clinical decision-making and policy formulation, sector surveillance, health risk management and resource allocation. However, to achieve these goals, key features need to be in place: popula tion definition, temporal elements of data reporting, core data, appropriate terminology, quality and safety in data analysis, and governance, as stated in the framework for high-quality data collection developed by the European Medical Agency4.
A review by Craig et al. aimed to assess the elements that make up a registry and whether they contain research data. This identified 13 HIV registries that include specific-registry infor mation, collaboration processes and research data5. Examples of these registries or observational cohorts were EMBRACE (USA) (6, CFAR Registry (USA) (7, CCR/HIV of the VA Department (USA) (8, San Francisco HIV/AIDS Surveillance Registry9, New York City HIV/AIDS Surveillance Registry10, Stiching HIV Mo nitoring in Netherlands11, HARS in Missouri12, Australian Na tional HIV Registry13, InfCare HIV (Sweden) (14, Minnesota HIV Surveillance Registry15, IeDEA (Rwanda) (16 and others5,17,18.
Colombia has a solidarity-based health insurance system, regulated by the Ministry of Health and Social Protection (MHSP) with two main regimens (Contributive and Subsidi zed, according to payment capacity of their affiliates) that covers nearly 97.64% of the population19, but include the same services, procedures, medicines, and interventions for their users. Insurers are responsible for managing resources, paying to healthcare providers20 and data reporting to most of health information systems.
The MHSP delegated to the High-Cost Diseases Fund (CAC, in Spanish), a technical organization of the health system, the monitoring of this disease through the National HIV/AIDS re gistry. We aimed to describe the data management process of the Colombian registry, its epidemiological results and contributions to research and health risk management.
Methods
1. Background and national policies
The MHSP established the legal framework of the national HIV/AIDS registry (resolutions 4725 of 2011, 0783 of 2012 and 0273 of 2019) (21-23. These norms include the periodicity, form and content of the information to be reported by all insurance companies and healthcare providers. The annual report covers the cases enrolled and cared for by the insu rance companies along a year that starts February 1st of any reporting year to January 31st of the following year. The varia bles are adjusted periodically according to new clinical gui delines, country information needs, and lessons learned from the implementation.
2. Contents of the registry
2.1. Patients included in the HIV/AIDS registry
The national HIV/AIDS registry is a longitudinal database containing data of two types of populations: 1. pregnant women, children born to mothers living with HIV/AIDS and people reported with active tuberculosis (TB) that were ne gative to the HIV test or do not have HIV test results at the time of reporting, and 2. People diagnosed with HIV/AIDS including pregnant women, children born to mothers living with HIV/AIDS and TB patients already diagnosed with HIV. The reason for these two types of reporting is to follow the adherence of the health insurers and healthcare providers to the national HIV/AIDS guidelines and national strategies. The HIV diagnosis is based on the algorithm defined by the na tional clinical guidelines24,25.
2.2. Registry files
Health insurers and healthcare providers collect and sum marize the data from the two types of populations in two different files, one for people living with HIV/AIDS (PLWHA) and the other one for the non-HIV or unknown HIV status patients that are followed. Sociodemographic, clinical, and administrative variables are collected for the reporting pe riod. The CAC provides all entities with reporting instructions and an auditing manual as recommended by the MHSP.
2.3. Structure of the database
Registry structure includes 193 variables, grouped in subsec tions like: identification data, demographics, and adminis trative updates for all patients; clinical information, current ART, AIDS-defining clinical conditions, current clinical status, prevention interventions and prophylaxis for the HIV/AIDS patients; pregnant women, children born to mothers living with HIV/AIDS and people with active TB with unknown HIV diagnosis or negative HIV test have their own registry files, that cover only 43 variables.
The sociodemographic data (personal identification, date of birth, sex, etc.) are crucial for cross-checking with other official sources of information like the “Unique Affiliated Database” (BDUA, in Spanish) to verify the reliability of the patient’s status as vital statistics are updated on the last date of the period.
The clinical section includes variables related with the diag nosis (date of diagnosis, transmission mechanism, reason for testing, clinical stage, CD4 T-cell count and viral load at diag nosis); treatment (initial and current treatment, substitution therapies, reasons for switching ART, among others) and fo llow up (success of ART therapy determined by suppression of viral load, most recent CD4 T-cell count, AIDS-related di seases, opportunistic infections and presence of coinfections developed along the care) are also requested. Administrative variables updates refer to PLWHA status in the health sys tem (insured, disaffiliated, change of healthcare provider, i.e.) and the cost of HIV/AIDS care services (total costs of hospital care and total costs of non-hospital care). In addition, if the patient has died, the cause and date of death are recorded.
3. Software: Interconnection System and Health Information Exchange (SISCAC, in Spanish)
SISCAC is the platform set up by the CAC for healthcare in surers and their providers to upload their reports. It is imple mented on SharePoint; storage is done in Structured Query Language and MongoDB databases as well as cognitive ser vices for artificial intelligence and Power BI are in the Azu re cloud, which are Microsoft® services. This platform has a simple interface and can be adapted to the technological characteristics of each healthcare provider in the country. The portal offers notifications, system, and web services and two main menus: apps and microservices.
4. Data management
4.1. Data collection
Data from clinical records of the reporting populations (des cribed above) is extracted by health care providers to the standardized registry files once a year and reported to the health insurers. These ones collect the data from different providers, review the data and fill the gaps according to the services supplied along the year. On the reporting dates, when data is considered complete, they upload the informa tion into the SISCAC platform established by the CAC and run an automated validation mesh prior to sending the data.
4.2. Data validation
The platform contains an automatic validator mesh to check security, structure, consistency, and confidentiality of regis tered data. This validation is done at the time of reporting. Consolidated data is cross-checked by the CAC professionals with an official database of persons affiliated in the health system to verify the insurer and vital status of the reported patients. After this step, the data management coordinator approves the dataset to be audited.
4.3. Data auditing
As reporting is done based on medical records, this informa tion is sent later to an auditing process, conducted by health professionals with experience in clinical auditing. They com pared the database information against the clinical records. If the information reported is different from the available re gisters, an adjustment is requested based on the registers.
According to the findings, the information is classified in three groups: conforming data (CD) when reported informa tion was the same found in medical records; non-conforming data (NCD) when reported information was different from the one registered on medical records, but adjustments can be done; and original data not available (ODNA), when the information is not found on clinical records. In the last case, the information is not taken into account for the analysis. This classification is considered in the rating process for health insurers.
4.4. Data analysis
After the auditing process, a single database is consolidated. Sometimes a patient could be reported by two insurers that had the patient at different times along the year or that pro vided different services. In those cases, the patient is coun ted only once for epidemiological indicators. Cases that died outside the period are excluded from analysis. Crude and standardized measures are estimated for the epidemiological indicators defined below. Numeric data are presented as me dians and interquartile ranges (IQR) or media and standard deviation (SD) according with distributions. Category data sets are presented as frequencies and proportions.
HIV prevalent cases are all the ones reported in the period; HIV Incident cases are the ones that were diagnosed within the re porting period; and mortality cases are those reported by health insurers within the period and verified with external sources from the MHSP. Finally, 25 HIV risk management indicators are estimated to evaluate the quality of care provided by insurers and health care institutions. These indicators were chosen after a literature review and agreed by expert´s consensus26.
4.5. Data dissemination and use
When information is ready, an annual report is prepared and published through the CAC´s website. Information is also pre sented through technical documents, research publications and conferences to HIV/AIDS partners in the country that can use it for knowledge management and decision-making at the local and national levels and for international reporting.
5. Distribution mechanism
With the results of the indicators, the CAC calculates an ex-post incentive mechanism for the contributive and subsidized insu rers following the provisions of current regulations (Resolution 1912 of 2015 - MHSP27). This mechanism seeks to improve risk management of this disease and is based on four indica tors: screening for HIV in pregnant women, early detection of PLWHA, undetectable viral load and HIV prevalence. Those healthcare insurers that achieve the highest results receive the resources established for the mechanism, while those that get the lowest results are the ones that deliver those resources.
6. Security and confidentiality
All records in the database are numbered with a unique iden tifier number to ensure data anonymization and the follow-up across the years. This database is stored on the SISCAC plat form, which has strict controls for each user to ensure that only those who have authorization can access the information.
Figure 1 illustrates the complete process of building the na tional registry from data collection to data dissemination
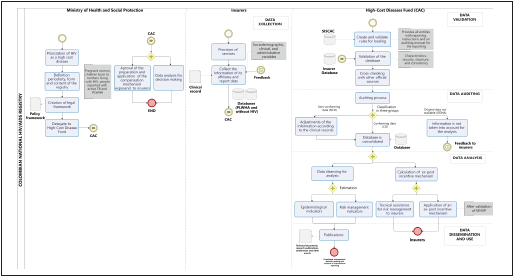
Figure 1. Illustrates the complete process of building the national registry from data collection to data dissemination. Construction processof the HIV/AIDS national registry from the normativity of the MHSP until the publication of the information and use of data for decision-making. Abbreviations: MHSP: Ministry of Health and Social Protection, PLWHA: People living with HIV/AIDS, SISCAC: Interconnection system and health information exchange, TB: Tuberculosis.
Results
1. Epidemiological situation of the HIV/AIDS in Colombia, 2012-2019
From 2012 to 2019, the country has seen a 300% increase in the reported cases to the registry, going from 37,325 to 109,056. Prevalence increased from 0.09% to 0.22%. Mortality went up from 1.1 per 100,000 population to 3.6 per 100,000 population, and the incidence increased from 13.8 per 100,000 population to 21.9 per 100,000 population (Figure 2).
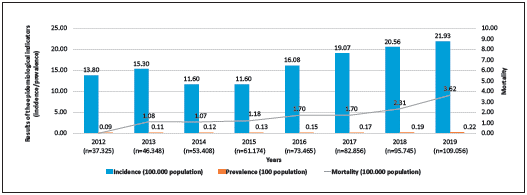
Figura 2 Epidemiological situation of the HIV/AIDS in Colombia, 2012-2019- Epidemiological indicators of the national HIV/AIDS registry in Colombia: incidence, prevalence and mortality from the beginning of the registry in 2012 up to the most recent measurement.
Data showed that in Colombia HIV affects men in a major proportion (75.8%) than women. The male/female ratio was 3.14:1. The mean age of diagnosis for men was 32.6 years (SD ±11.7) while in women was 36.1 years (SD ±13.5). The highest incidence and prevalence by geographical regions were ob served in Bogotá, while the highest mortality was seen in the Caribbean and the Central regions (Figure 3). The main way of transmission was sexual (90.3% of cases) and 56.5% of all reported cases were diagnosed on stage 3 (AIDS) 2014 CDC´s classification28, with AIDS wasting syndrome (13.7%) and TB (7.3%) being the most frequent opportunist infections.
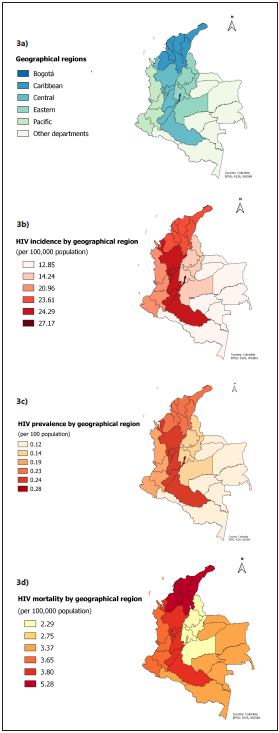
Figure 3 Age-standardized epidemiologic indicators of HIV/AIDS by geographical region. Geographical region distribution (3a), age-standardized incidence (3b), age-standardized prevalence (3c) and age-standardized mortality (3d).
Since 2019, data on key populations is being recorded, finding that 43.1% of new HIV infections in 2019 were diagnosed on men who have sex with men. The median lymphocyte count at diagnosis increased from 249 cells/μl (IQR 106-440) in 2012 to 329 cells/μl. (IQR 309-686) in 2019. The proportion of people with viral suppression (VL < 50 copies/ml) increased from 50.8 % in 2012 to 63.9% in 2019. This data is published annually at https://cuentadealtocosto.org/site/publicaciones/.
2. Risk management indicators
Over the 2012-2019 period, we have measured 25 national level indicators. 18 indicators have shown an increase in per formance compared to the baseline (median 16.9%; IQR 1.7% - 26.5%). The biggest difference was seen with the indicators: proportion of HIV exposed children under 6 months tested for HIV (23.5% versus 98.0%), proportion of PLWHA with annual Tuberculin Skin Test (TST) (18.8% versus 61.4%), and proportion of PLWHA in latent tuberculosis therapy (14.6% versus 48.7%). Instead, two indicators saw a poorer perfor mance: Coverage of antiretroviral therapy in line with Natio nal Clinical Guidelines, decreasing 2.4%, and the proportion of PLWHA, not on antiretroviral therapy, with a CD4 count done in the reporting period, decreasing by 17.8%.
In 2019, 3 risk management indicators met the established tar get: proportion of HIV exposed children under 6 months tested for HIV (>95%), proportion of PLWHA on antiretroviral therapy with a viral load done in the reporting period (≥95) and the pro portion of PLWHA with antiretroviral therapy switch (<30). Table 1 shows the results of the indicators since the establishment of the registry and the percentage difference between the baseline in 2012 and the last measurement in 2019.
Table 1 Risk management indicators of Colombian HIV/AIDS Registry, 2012-2019
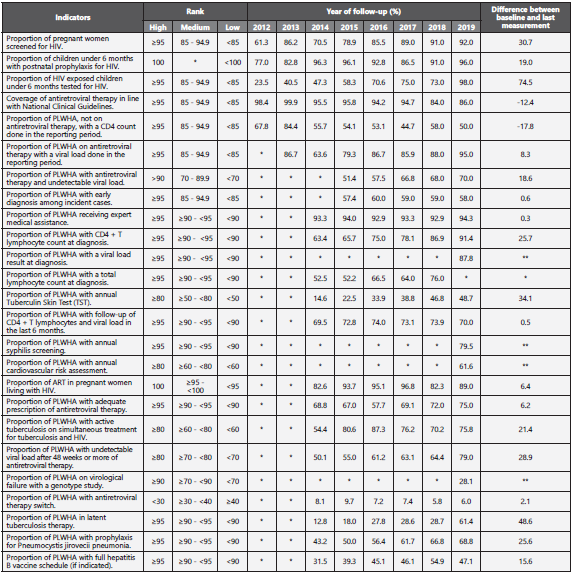
Abbreviations: PLWHA: Persons living with HIV/AIDS. * The indicator was not measured. ** It is not possible to calculate the difference.
Management risk indicators measured in Colombia from 2012.
Likewise, the Colombian registry has made possible to mo nitor Colombia’s progress towards the UNAIDS 90-90-90 targets. Of the estimated 119,000 PLWHA that know their HIV status in 2019 (information given by the MHSP), 84,992 (71.4%) were on ART treatment, and 72,272 of them (85.0%) were virologically suppressed (<1,000 copies/ml).
3. Research
With the HIV/AIDS registry information, three scientific arti cles have been produced describing the situation of PLWHA in Colombia: “Burden and magnitude of risk in HIV/AIDS in the Colombian health system: a real-world data approach” (29, “Epidemiological and clinical characterization in minors under 13 years living with HIV in Colombia. 2018: a cross-sectional study” (30, and “Survival in people living with HIV in the fra mework the Colombian health system: 2011-2018 in the pe diatric population31”, which shows the multiples sub-analyses that can be done with the wide range of variables collected.
4. Distribution mechanism
Implementation of the ex-post incentive mechanism for the contributive and subsidized regimes have increased the perfor mance of the insurers. This is reflected on the key four indicators that are evaluated: screening for HIV in pregnant women, which went up from 61.3% to 92.0%; early detection of PLWHA, that increased mildly from 57.4% to 58.0%; undetectable viral load, from 51.4% to 70.0%; and the HIV prevalence that increased from 0.09% to 0.22%. The best they perform the higher the amount they received. Extra resources are expected to be rein vested in strengthening the insurer´s HIV/AIDS programs.
Discussion
The main results of the National HIV/AIDS registry are: 1. Epidemiological information to follow trends of the HIV epi demic in Colombia; 2. Periodic measurement of risk manage ment indicators; 3. Data for operational research; and 4. In formation for an incentive distribution mechanism.
The registry shows the increase in reported HIV cases in the country and improvement of the main risk management in dicators to reduce the burden of this disease. While, UNAIDS monitoring shows that since 2010, new HIV infections have decreased by about 31%32, Colombia has seen an increase reported cases, probably due to the differences in these es timates33 a better capture of cases by the health information system or major access the PLWHA to care services.
Our registry complies one of the key definitions set by the Agency for Healthcare Research and Quality (AHRQ)3 as an organized system that uses observational methods to collect uniform data on a population defined by a particular disease (in this case HIV/AIDS), followed over time. The purpose of this registry is to describe the impact of the disease on pa tients’ health, estimate the burden of the HIV/AIDS epidemic in the country and the trends over time. In a health care sys tem with limited funds, AIDS care represents an important economic burden34. Therefore, HIV registries contribute to improving outcomes related to healthcare and managing re sources efficiently35. This registry periodically publishes infor mation available and accurate, avoiding expensive data col lection to assess health system results, as indicated by Cylus J et al36. As data infrastructure grows and more suitable data becomes available, the necessity to transform the data into useful information emerges.
Mayer et al4, in a systematic review of the literature, identified 13 HIV registries, of which 61.5% are the product of a col laboration policy and 53.8% have a management process for requesting data collaboration, although there are shortcom ings related to ethical support and research data5. In com parison, our registry contains all the key features for data collection like criteria for inclusion of participants (case defi nitions, data elements collected, etc.) and support for data interoperability between the MHSP, the CAC, health insurers and health care providers.
We have a legal commitment, supported by an epidemiolog ical team, to produce an annual report of the HIV/AIDS situ ation in the country and to propose ideas for future research. In this way, the information generated allows the monitor ing of HIV case management and follow-up of global goals aimed at elimination of the pandemic37, as demonstrated by Zheng et al33, who indicates that National HIV registries are the starting point for this follow up as the requested informa tion is available on the national system and easily accessible from the clinical records. Although, it can contain incomplete data, it can also be strengthened by routine record practices.
Our registry, that started with 37,325 cases reported, captured information from 109,056 PLWHA in 2019. This has allowed us to characterize this population and to provide a useful tool for public health making decisions in Colombia. Other registries have a variable number of PLWHA, like the Singapore one33 with up to 5,000 PLWHA per year; The Swedish National HIV Registry33 with 7,000 cases; Australia in the Kirby institute38 reported 28,000 cases and close to 1,000 new cases per year. These registries are usually part of a wider national health infor mation system. Other registries consolidate information from several countries to have a larger volume of information and to analyze the behavior of the disease between regions. This is the case of IeDEA registry39, which consolidates and analyzes data on almost 2 million PLWHA under care in routine settings from 46 countries around the world, or the Global AIDS Monitoring (GAM) report of UNAIDS40 which is a worldwide HIV monitor ing and evaluation system.
The Colombian HIV/AIDS registry works as a complement to other sources of national information like The Public Health Surveillance System (SIVIGILA in Spanish) (41, which monitors case notification of diseases of public health interest like HIV, but doesn´t do follow up of linking to care, ART initiation or retention on care. In this way, the CAC registry and the SIVIGILA help to show different faces of the epidemic in the country.
Registry’s strengths include data collection of priority groups for screening like pregnant women, children born to HIV-positive mothers and TB patients, with disaggregated data by geo graphic regions and municipalities. Self-reporting of PLWHA within key populations is also important to identify disease bur den among smaller groups like MSM, transgender women or people who inject drugs. Other strength of the data collection process is the inclusion of health services planning information like affiliation, insurers, health care providers and costs, which have standardized definitions and labels for all elements of the dataset. This information also undergoes an auditing process and frequent feedback to the reporting entities.
Our estimates and analysis may be limited due to under-reporting and missing data (estimated on 5%) not found in the medical records. In fact, we only rely on data reported in the medical records of persons affiliated to public or pri vate health insurance regimes, with a coverage of 97.8% in Colombia19. However, as it is a high-cost disease, a low per centage of Colombians would probably look for care with out-of-pocket money. The percentage of PLWHA who are not affiliated to the health system and, therefore, are not re ported in this registry, is unknown. This is the case for PLWHA assisted by non-governmental organizations that look after illegal migrants or vulnerable populations. Although the health insurers report all their affiliates, it´s possible that the cases lost to follow up (PLWHA without care in the last year) have outdated information, however this percentage is less than 2.0%. Other limitations are the heterogeneity in medi cal records among health providers (manual and/or digital) and practices of care in health programs. On the other hand, the process of auditing and analyzing the data highlight the need to continue strengthening the information system and move forward to reporting of HIV data in real time.
In conclusion, the Colombian HIV/AIDS registry has been use ful not only for the clinical follow-up of patients but also to evaluate the quality of care provided by insurers and health institutions and for building public health policies based on evidence that aimed at PLWHA. Although different informa tion sources in Colombia track HIV/AIDS at different points of care, the CAC registry consolidates the most significant number of variables that relate to the continuum of care. However, additional work is needed to integrate data from different sources and follow-up programs within the primary care to have a full comprehension of the HIV situation in the country. Finally, this exercise could be useful for other coun tries in the Latin American region that are interested in the development of specific disease follow-up registries.














